Abstract
Low doses of psychoactive drugs often elicit a behavioral profile opposite to that observed following administration of more substantial doses. Our laboratory has observed that these effects are often age-specific in rats. For instance, whereas moderate to high doses of the dopamine agonist apomorphine increase locomotion, suppressed locomotor activity is seen following low dose exposure, with this low dose effect not emerging consistently until adolescence. A somewhat earlier emergence of a low dose “paradoxical” effect is seen with the 5HT1a receptor agonist, 8-OH-DPAT, with late preweanling, but not neonatal, rats showing increases in ingestive behavior at low doses but suppression at higher doses. In contrast to these ontogenetic increases in expression of low dose drug effects, low dose facilitation of social behavior is seen following ethanol only in adolescent rats and not their mature counterparts, although suppression of social interactions at higher doses is seen at both ages. This hormesis-like low dose stimulation appears related in part to overcompensation, with brief social suppression preceding the subsequent stimulation response, and also bears a number of ontogenetic similarities to acute tolerance, a well characterized, rapidly emerging adaptation to ethanol. Implications of these and other ontogenetic findings for studies of hormesis are discussed.
Hormesis is a dose-response relatifonship characterized by a low-dose response that is in the opposite direction from that seen at higher doses (Calabrese & Baldwin, 2002a). Although best characterized in the toxicology literature, examples of hormesis-like dose-response relationships also abound in pharmacology (Calabrese & Baldwin, 2001). Little attention has been directed thus far to hormesis from a developmental perspective. Several examples of hormesis-like phenomena appearing during ontogeny will be discussed here, with a particular focus on developmental psychopharmacology. Examples will be given of both delayed ontogenetic emergence of low dose effects as well as an ontogenetic decline in a hormetic-like low dose effect with ethanol. Evidence will be presented to show that the low dose stimulatory effect on social behavior seen in adolescent rats following ethanol administration is preceded by a short-lasting initial disruption in homeostasis in the opposite direction, evidence consistent with an overcompensatory hormetic response. Several other ontogenetic examples from the literature will also be presented that raise the broader issue of whether variation in expression of hormesis across different models/challenges might result in part from different starting positions on U-shaped perturbation/response functions reflecting altered levels of baseline adaptation due to prior developmental history and ongoing exposure to related challenges naturally occurring in the environment.
ONTOGENY AND HORMESIS: ONTOGENETIC EMERGENCE OF LOW DOSE EFFECTS IN PSYCHOPHARMACOLOGY
In our ontogenetic investigations, we have seen several examples of the developmental emergence of low dose effects that resemble hormesis. Two examples are given below.
Ontogeny of Biphasic Effects of 8-OH-DPAT on Ingestion-Related Behaviors
In response to the 5HT1a agonist 8-hydroxy-2(di-n-propylamino)tetralin (8-OH-DPAT), adult rats typically exhibit an increase in ingestion across a dose range from approximately 0.03–1.0 mg/kg (e.g., Hutson et al., 1988), with decreases in ingestion seen at higher doses, perhaps in part related to competitive behaviors associated with the “serotonergic syndrome” that emerge at higher doses (Dourish et al., 1985). This biphasic effect of 8-OH-DPAT on ingestion is not evident in young rat pups but appears later during ontogeny. In neonatal rats, only inhibitory effects of 8-OH-DPAT were observed on independent ingestion when assessed across doses ranges from 0.5–10 mg/kg (Kirstein & Spear, 1988; Frambes et al., 1990) and 0.01–1 mg/kg (Spear et al., 1991). As shown in the top panel in Fig. 1, beginning at the lowest effective dose of 8-OH-DPAT, only a suppression in mouthing was seen in neonatal (postnatal day [P] 3–4) rat pups. In contrast, P17–18 preweanlings exhibited both a low dose stimulatory and high dose depressant effect of 8-OH-DPAT on mash-related ingestive behaviors (see Fig. 1, middle panel). In young adolescent (P28–29) animals, an increase in ingestion was seen only at the highest dose (lower panel of Fig. 1); given that none of the doses within this relatively low dose range were sufficient to yield signs of the serotonergic syndrome, it is likely that this dose range was below that necessary to induce the decreases in ingestion expected at sufficiently high doses.
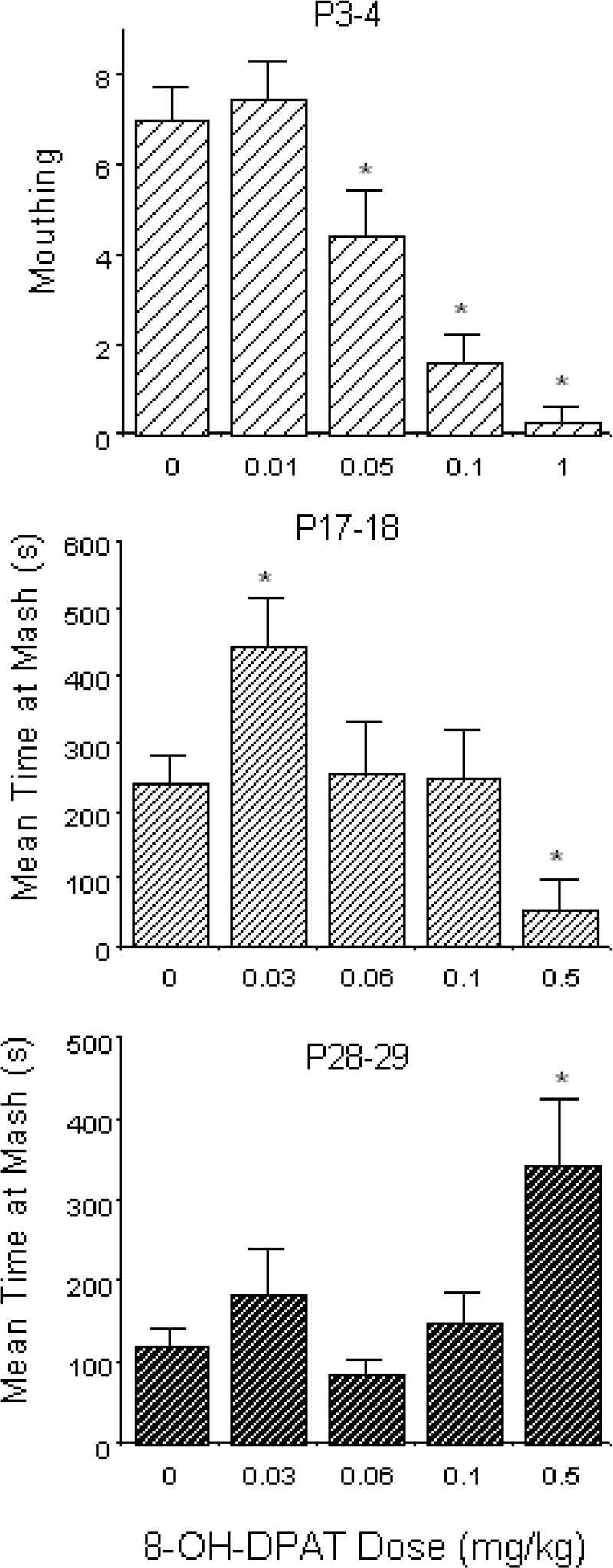
Acute effects of various doses of the 5HT1a agonist 8-OH-DPAT administered subcutaneously on behavioral measures of ingestion of P3–4 (top panel), P17–18 (middle panel) and P28–29 (bottom panel) Sprague-Dawley rat pups. P3–4 pups were injected with 0, 0.01, 0.05, 0.1 or 1 mg/kg 8-OH-DPAT 10 min. prior to a 5 min. ingestion test conducted in a test incubator with warm Half and Half dairy cream puddled on the terry cloth covered floor; mouthing exhibited by each pup during the test was determined using time sampling. Preweanling (P17–18) and post-weanling (P28–29) animals were injected with 0, 0.03, 0.05, 0.1 or 0.5 mg/kg of the test drug immediately prior to a 25 min. ingestion test, with time spent at dishes containing wet mash and milk determined (note: time spent at the milk dish was low in all groups at both ages, and hence only mash data are summarized here). Data shown here were derived from Spear et al. (1991).
Ontogenetic differences in the effective doses necessary for inducing specific drug effects—such as the notable shift in effective stimulatory dose seen between P17–18 and P28–29—may reflect potential age differences in drug distribution/metabolism. It is difficult, however, for pharmacodynamic considerations to contribute to the delayed ontogenetic emergence of biphasic effects, assuming that appropriate dose ranges to detect biphasic effects were examined at each age, as we attempted to do in our work with 8-OH-DPAT in neonatal rat pups (Kirstein & Spear, 1988; Frambes et al., 1990; Spear et al., 1991). Given that low doses of 8-OH-DPAT have been suggested to increase feeding via preferential stimulation of 5HT1a autoreceptors (e.g., see Dourish et al., 1988), these data were hypothesized to support the delayed ontogenetic emergence of functional 5HT1a autoreceptors, with low dose stimulation of ingestion evident in late preweanling, but not neonatal, rat pups (see Spear, et al., 1991, for discussion). Interpretation of these data within a hormesis context is given below, following a discussion of another study that also yielded a delayed ontogenetic emergence of a low dose, potentially autoreceptor-mediated drug effect.
Ontogeny of Low Dose Effects of Apomorphine
Reminiscent of the 8-OH-DPAT findings, a delayed ontogenetic emergence of a low dose effect was also observed in the dopaminergic system. At high doses, the dopamine agonist apomorphine stimulates locomotor activity, whereas a suppression in activity is typically seen within a narrow low dose range (Shalaby & Spear, 1980). This low dose suppressant effect emerges only slowly during ontogeny. For instance, as shown in Fig. 2, whereas stimulant effects of apomorphine on locomotor activity were evident at all ages, a low dose suppression of locomotor movements emerged only in adolescents at the oldest age examined in this study (P35) (Shalaby & Spear, 1980). Although this study was conducted using fewer doses than desirable to fully characterize this effect, other laboratories have likewise reported the emergence of the low dose suppressant effect of DA receptor agonists only around the time of adolescence in rats (Hedner & Lundborg, 1985; Van Hartesveldt et al., 1994). Reminiscent of the 8-OH-DPAT data, these low doses suppressant effects have been postulated to reflect delayed maturation of inhibitory DA autoreceptors in mesolimbic brain regions, although data supporting this hypothesis (Shalaby et al., 1981; Spear & Brake, 1983) are not ubiquitous, with some recent work not supporting this suggestion (Franz & Van Hartesveldt, 1995; Andersen et al., 1997).

Acute effects of the dopamine agonist apomorphine on number of locomotor movements in the open field in preweanling (P7, P14), weanling (P21), early adolescent (P28), and mid-adolescent (P35) Sprague-Dawley rats. Animals were given the drugs subcutaneously and were tested beginning 1 min. following injection for 20 min. in size-adjusted open field chambers. The number of 5 sec. time bouts (sampled every 15 sec.) where locomotor movements were exhibited by each animal was determined. Data are derived from Shalaby and Spear (1980).
Do These Low Dose Effects Reflect Hormesis?
In a number of respects, the low dose response that emerges gradually during ontogeny with apomorphine and 8-OH-DPAT resembles hormesis. In both instances, typical (albeit reciprocal) versions of hormetic dose-response curves (see Calabrese & Baldwin, 2002a) eventually emerge developmentally, with the low dose effect in the opposite direction from that seen at higher doses. With both drugs, the low dose effects that emerged ontogenetically were reliable and of a magnitude (55–85% change from control) typical of hormetic responses. Whether or not these low dose effects meet the “adaptive benefit” component sometimes linked to hormesis (e.g., Calabrese & Baldwin, 2001) is doubtful, and indeed considerations of adaptive significance of low dose effects would appear to have more applicability in toxicology than pharmacology.
With both apomorphine and 8-OH-DPAT, the ontogenetic emergence of low dose effects of these drugs seem to provide an example of the delayed ontogeny of hormesis or a hormesis-like response relative to effects evident at higher doses. From these two examples, however, it should not be concluded that hormetic-like responses are necessarily less likely to be expressed in younger than more mature animals. Indeed, the data discussed in the following section document a hormetic-like low dose effect present in younger animals but not apparent in adults.
ETHANOL AND SOCIAL FACILITATION IN ADOLESCENCE: AGE-SPECIFIC HORMESIS OF THE OVERCOMPENSATION TYPE?
More recently, a major focus of our laboratory has been on ontogeny of ethanol responsiveness, with particular attention paid to the adolescent period. Ethanol has been shown to exhibit evidence for hormesis using a variety of dependent measures ranging from locomotion to effects on liver, kidney, and cardiac function (Calabrese & Baldwin, 2003). We have also observed a striking example of a hormesis-like effect in our work examining the effects of ethanol on social facilitation in adolescent rats (Varlinskaya et al., 2001; Varlinskaya & Spear, 2002). For testing of social behavior, each experimental rat is placed in a familiar test situation in the presence of a non-manipulated, unfamiliar age mate. Social interactions of the experimental animal over the 10 min. period are videotaped and later analyzed for incidence of a variety of social behaviors, including social investigation, contact behavior, and play behavior. When P30 adolescent rats are given a variety of different doses of ethanol 30 min. pre-test, a biphasic dose-response curve is revealed, with low doses of ethanol increasing overall social activity, and higher doses suppressing social behavior (Fig. 3) (Varlinskaya et al., 2001). The low dose facilitation of social behavior by ethanol is specific to social interactions; as shown in Fig. 4, no facilitation is evident when adolescents are given the opportunity to interact with an inanimate object (an equivalent sized cotton ball) (Varlinskaya et al., 2001). As illustrated in Fig. 5, this low dose facilitation is age specific, and is not evident in adult (P70) rats, despite similarities in blood alcohol levels across age (Fig. 6) (Varlinskaya & Spear, 2002). The ontogenetic decline of this low dose facilitation occurs late in the adolescent period, an age range conservatively defined as the interval from P28 to P42 in rats, based on timing of neurobehavioral discontinuities and pubertal-associated changes (see Spear, 2000, for discussion). As shown in Fig. 7, a low dose facilitation of ethanol-induced social interactions is significant at doses of 0.5 and 0.75 g/kg in early (P28) adolescents and at a dose of 0.5 g/kg in mid (P35) adolescents, but does not reach significance at P42, although social suppression is seen at higher doses at all ages (Varlinskaya & Spear, 2004).

Acute effects of ethanol on overall social activity of P30 adolescent rats. Overall social activity was scored as the sum of social investigation, contact, and play behavior frequencies. Animals were tested 30 min. following intragastric administration of various doses of ethanol or water (0 dose control). Data are derived from Varlinskaya et al. (2001).

Acute effects of ethanol on overall activity of adolescent rats directed towards an age mate or towards an inanimate object (cotton ball) 30 min. following intragastric administration of the challenge substance. Data are derived from Varlinskaya et al. (2001).

Acute effects of ethanol on overall social activity of P35 adolescent and P70 adult rats. Animals were tested under familiar test circumstances in the social interaction test 30 min. following intraperitoneal administration of saline or the test doses of ethanol. Data are derived from Varlinskaya & Spear (2002).
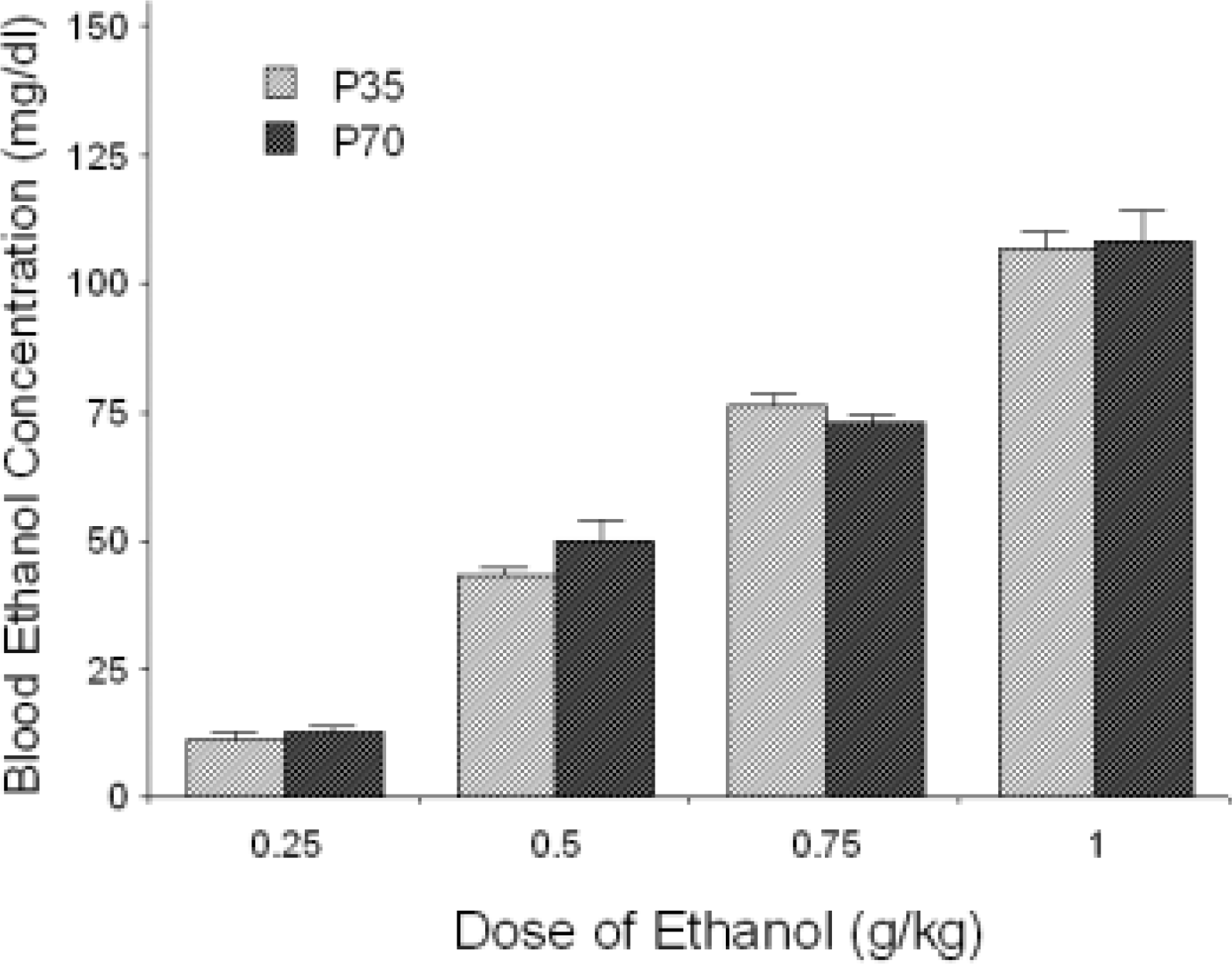
Blood alcohol levels of P35 adolescent and P70 adult rats determined 30 min. following intraperitoneal administration of 0.25, 0.5, 0.75 or 1.0 g/kg ethanol. Data are derived from Varlinskaya & Spear (2002). For the interpretation of these data, it may be useful to consider that 1 drink in humans has been estimated to produce peak blood alcohol levels in the range of 9–25 mg/dl (see Eckardt et al., 1998).

Acute effects of ethanol on overall social activity of early (P28), mid (P35) and late (P42) adolescent rats. Animals were tested under familiar test circumstances 30 min. following intraperitoneal administration of the challenge doses of ethanol or saline. Data are derived from Valinskaya & Spear, in press.
Does this behavior reflect hormesis? The magnitude of the response (about a 30–50% increase over control values) is within the range typical of hormetic responses, and this low dose response is in the expected, opposite direction from the acute ethanol response evident at higher doses. Whether this low dose effect reflects a direct stimulatory response (direct stimulation hormesis) or is the result of an overcompensatory response following an initial homeostatic disruption (overcompensation stimulation hormesis) (see Calabrese & Baldwin, 2002b) is uncertain, although we have some preliminary data that begin to address this question. To the extent that a particular instance of hormesis reflects an overcompensatory response to a disruption in homeostasis, we reasoned that it might be possible to detect a short-lasting precipitating event in the opposite direction from the subsequently appearing overcompensatory response. That is, to the extent that the test circumstances support detection of a brief initiating homeostatic disturbance, a transient initial response should be evident shortly after administration in the opposite direction of the typical low dose effect.
Results of a preliminary study where P28 adolescents were tested only 5 min. following intraperitoneal injection of ethanol rather than 30 min. post-challenge as in the studies discussed above revealed significant social suppression after 0.75 g/kg (Fig. 8), a dose that produces social facilitation at 30 min. post-challenge (see Fig.7). These data showing that the low dose social facilitation seen at this age is preceded by a brief excursion from homeostasis in the opposite direction provide evidence consistent with what we would expect with an overcompensatory hormetic response. Data from animals given 0.5 g/kg are more challenging to interpret. The 0.5 g/kg dose was just as effective at stimulating social interactions at the 30 min. test as the 0.75 g/kg dose in P28 animals (see Fig.6), yet no evidence of social suppression was seen at this dose when testing began 5 min. following injection. Thus, whereas the temporal pattern of findings following the 0.75 g/kg dose in these animals supports an overcompensatory interpretation, the data obtained in response to the 0.5 g/kg do not. The latter negative finding is difficult to interpret, of course. It could potentially reflect a direct, rather than overcompensatory, hormetic effect. Alternatively, it is possible that the initial perturbation seen at this lower dose was so short-lived as to escape detection in a 10 min. social interaction test beginning 5 min. following ethanol challenge. These preliminary findings should be interpreted cautiously until data are confirmed and perhaps extended to a briefer post-injection-test interval and shorter test duration. Traditionally we have avoided using shorter tests of social interactions in adolescents given that initial levels of social interaction during this test are sufficiently high as to approach ceiling levels. Yet, use of shorter test periods might prove useful for detecting transient social suppression relatively close to the time of injection.

Acute effects of ethanol on overall social activity of P28 adolescent rats. Animals were tested under familiar test circumstances 5 min. following intraperitoneal administration of 0, 0.25, 0.5, 0.75 or 1.0 g/kg ethanol.
If ethanol-induced social facilitation during early and mid adolescence reflects (at least partially) an overcompensatory hormetic response, why might this response decline ontogenetically? Neural mechanisms underlying the ontogenetic decline in expression of ethanol-induced social facilitation during the adolescent period remain to be determined, although work is planned to explore the contribution of mu-opioid receptor systems, given the importance of these receptors in the regulation of social behavior in adolescence (Vanderschuren et al., 1997). One potential clue regarding the apparent ontogenetic decline of this hormetic-like social facilitation following low doses of ethanol is provided by considering the ontogeny of a form of rapid neural adaptation to ethanol that has been termed “acute tolerance”. Acute tolerance is a rapid decline in sensitivity to ethanol that occurs within a single bout of ethanol exposure (see Kalant, 1993, for review). This form of tolerance can be detected in a number of different ways (for review, see Silveri & Spear, 2004), including via an attenuated response to a subsequent high dose challenge shortly following clearance of a lower dose (Ponomarev & Crabbe, 2002). The latter approach is virtually identical to one of the adaptive pharmacological properties cited for hormesis—i.e., that “… a low dose … administered prior to a higher and more threatening dose, often reduces the toxic potential of the subsequent exposure” (Calabrese & Baldwin, 2001, p. 355). Given these similarities, it is of particular interest that the ontogenetic decline in ethanol-induced social facilitation resembles the ontogenetic decline observed in acute tolerance to ethanol, with evidence for acute tolerance (at least to ethanol's sedative effects) seen in adolescent and even younger animals under test circumstances that do not support the emergence of acute tolerance in adults (e.g., Silveri & Spear, 1998; Silveri & Spear, in press). Expression of acute tolerance in young but not adult rats appears related in part to transient developmental overexpression of NMDA receptors, with the NMDA antagonist (+)MK-801 blocking expression of acute tolerance in young animals (Silveri & Spear, 2004).
Of course, further work is needed to determine whether ethanol-induced social facilitation in young adolescents represents a hormetic compensatory response, and whether neural substrates underlying this potentially adaptive process and its ontogenetic decline are related in part to those associated with the rapid adaptations termed “acute tolerance”. Yet, even at this early stage of examining hormesis within a developmental context, we hope that the utility of ontogenetic considerations for the study of hormesis has been demonstrated.
OTHER ONTOGENETIC EXAMPLES OF HORMESIS-LIKE PHENOMENA
Moving beyond the fields of toxicology and pharmacology and the work of our laboratory, other examples of hormesis-like phenomena in the developmental literature emerge. Two examples will be given here as a basis for further discussion.
The first example is almost a post-script to a relatively long story examining the consequences of early environmental manipulations, work conducted in the laboratories of Meaney, Plotsky and others (see Ladd et al., 2000, for review). In this work, rodent models were used to examine the effects of early environmental manipulations on later sensitivity to stressors, measured behaviorally as well as in terms of functioning of a stress-sensitive hormonal system called the hypothalamo-pituitary-adrenal (HPA) axis. The question raised by the Meaney and Plotsky groups was whether daily environmental manipulations of rat pups during much of the preweanling period would alter their later stress responsiveness. These daily manipulations were of two types: 15 min. of maternal separation (typically referred to as “handling” for historical reasons—Denenberg, 1964), or a more extended 3 hr. period of separation. Controls typically consisted of animals that were normally colony-reared, receiving only the amount of stimulation associated with normal colony maintenance (cage cleaning and so on). In some experiments, a non-handled group was also included that was explicitly not manipulated in any way during the treatment period. The basic findings from these experiments were clear: as adults, animals that had received early “handling” exhibited more modulated HPA and behavioral responses to stressors than colony-reared controls, whereas the “separated” animals as adults exhibited intensified responses to stressors when indexed both behaviorally and hormonally (Ladd et al., 2000). Of particular importance for the story here, outcomes evident in the non-handled group were very similar to those of the “separated” group (see Ladd et al., 2000). In other words, the most developmentally perturbed group and the least manipulated group exhibited similar outcomes—a pattern that strikingly resembles hormesis.
The second example is drawn from a very different developmental literature, data on human adolescents and risk taking. Although adolescents of a variety of species including humans exhibit more risk taking than is seen at other ages (e.g., Moffitt, 1993), there is variability among adolescents in the amount of risk-taking behaviors. Some human adolescents engage in very little risk taking, many exhibit moderate amounts, and some are extreme risk takers. Evidence suggests that risk taking is not inversely related to adaptiveness, but rather that moderate levels of risk taking may be associated with adaptive competence. For instance, Shedler and Block (1990) found that individuals engaging in occasional drug use during adolescence were more socially competent than either frequent users or abstainers, with the frequent users often characterized as alienated and distressed and the abstainers, anxious, emotionally constricted, and lacking in social skills. This U-shaped relationship between levels of risk taking and social competence provides another example of a hormesis-like response early in development.
It is arguable whether these non-pharmacological examples drawn from the developmental literature reflect hormesis per se, or merely hormesis-like patterns. Yet, they raise an interesting issue. Do prototypic controls always—or even normally—start from the zero point on U-shaped hormesis or hormesis-like curves? It has been postulated that hormesis reactions of the overcompensation type function to counter insults that induce even slight perturbations from homeostasis, with the rebound overcompensation seen at low doses serving to protect against continued insult (Calabrese & Baldwin, 2002b). According to this view, it is the overcompensation response, and not the baseline state, that often yields the most beneficial outcome. Yet, it would seem unusual for evolution to have favored so ubiquitously the retention of systems that maximize function not under normal baseline circumstances, but only following modest perturbations from homeostasis. From a slightly different perspective, perhaps the null level exposure to environmental perturbations and stressors is not the normal baseline state, but rather reflects a zero exposure level that can be produced in the laboratory but may not always be realized in a world full of toxins, infections, and stressors in which we and other species live and have evolved. According to this view, hormesis-like responses may have been highly conserved during evolution because of the challenging, unclean, and natural toxin-filled environments in which species have evolved. Animals may not have evolved for life in a vacuum, but rather may have evolved to respond most adaptively to the typically modest levels of ongoing perturbations of their environment associated with exposure to minor toxins, infections, and other stressors. Variation in findings of hormesis across test organism, tissue type, and challenge may reflect in part different levels of baseline adaptation due to developmental history and prior exposure to related challenges naturally occurring in the environment, and hence different starting points on the hypothesized U-shaped (or inverted U-shaped) dose/response function. Consequently, when assessing the prevalence of hormesis-like phenomena and exploring their underlying mechanisms, it may become of increasing importance to consider the ontogeny of these forms of adaptation and the role of developmental history in their expression.
Footnotes
ACKNOWLEDGMENTS
This work was supported in part by grants from the National Institute of Alcohol Abuse and Alcoholism R37 AA12525 and R01 AA12150 to Linda P. Spear and R01 AA12453 to Elena I. Varlinskaya.
