Abstract
Molecular chaperones encompass a group of unrelated proteins that facilitate the correct assembly and disassembly of other macromolecular structures of which they themselves do not remain a part. Chaperones associate with a large and diverse set of cofactors termed cochaperones that regulate their function and specificity. Chaperones and cochaperones regulate the activity of several classes of signaling molecules, including steroid receptors. Upon binding ligand, steroid receptors interact with discrete nucleotide sequences within the nucleus to control the expression of diverse physiological and developmental genes. Molecular chaperones and cochaperones are typically known to provide the correct conformation for ligand binding by the steroid receptors. While this contribution is widely accepted, recent studies have reported that they further modulate steroid receptor action outside ligand binding. Specifically, they are thought to contribute to receptor turnover, transport of the receptor to different subcellular localizations, recycling of the receptor on chromatin and stabilization of receptor DNA binding. In addition to these combined effects with molecular chaperones, cochaperones have additional functions that are independent of molecular chaperones, some of which impact steroid receptor action. Two well-studied examples are the cochaperones p23 and Bag-1L, which have been identified as modulators of steroid receptor activity in the nucleus. Understanding details of their regulatory action will provide new therapeutic opportunities for controlling steroid receptor action independent of the widespread effects of molecular chaperones.
Introduction
In the early 1990s, studies with the glucocorticoid receptor (GR; a member of the steroid/nuclear receptor superfamily) showed that immunoprecipitated GR, when incubated with reticulocyte lysate that contains molecular chaperones and a source of adenosine triphosphate (ATP), could be made to bind hormone in vitro [1]. These reconstitutions could also be achieved with purified GR, molecular chaperones (Hsp90, Hsp70) and cochaperones (Hsp60 or Hip, Hsp40 or its yeast equivalent YDJ-1 and p23), which displayed an orderly and dynamic assembly of the receptor. The first step in this assembly was the formation of a molecular complex (Hsp90)2.Hop.Hsp70.Hsp40, termed the foldosome [2,3] (Figure 1, A and B). Two of the major players of the foldosome, Hsp90 and Hsp70, contain nucleotide-binding domains that act as ATP/ADP-binding switches that allow them to assume different properties depending on which form of energy is bound. In its ATP-bound form, Hsp90 interacts with the cochaperone p23 and activates client protein activity through folding, whereas in its ADP-bound form it shows high affinity for hydrophobic proteins [4,5]. In the case of Hsp70, its ATPase activity is enhanced by the cochaperone, Hsp40, as well as a variety of other cochaperones [6,7]. In general, Hsp-interacting cochaperones can be grouped according to the presence or absence of tetratricopeptide repeats (TPR) in their sequence (Figure 2). The TPR domains are typically composed of three tandem repeats of a loosely conserved 34 amino acid sequence motif [8]. Each motif favors formation of two anti-parallel α-helices, and the core TPR domain consists of six total α-helices that form a saddle-like structure. The surface of the domain provides an interaction site that can accommodate specific peptide binding [9]. For example, TPR-containing cochaperones are able to interact with Hsp90 through its EEVD motif at the extreme C-terminus, as well as with Hsp70 via a EEVD-like sequence at its C-terminus [9,10]. The repertoire of the TPR-containing cochaperones known to regulate steroid receptor signaling pathways includes the Hsc70-interacting protein (Hip; p48), the Hsp70/Hsp90-organizing protein (Hop; p60), FK506 binding protein of 51 kDa (FKBP51), FK506 binding protein of 52 kDa (FKBP52), cyclophilin 40 (Cyp40), protein phosphatase 5 (PP5), FK506-binding protein like protein (FKBPL), and general cell UNC-45 (GCUNC-45) [11–14] (Figure 2). More recently, a small glutamine-rich TPR-containing protein alpha (SGTA) has been added to the growing diversity of TPR-containing cochaperones involved in the modulation of steroid receptor action [15,16] (Figure 2). It should be noted that although several cochaperones use TPR motifs to bind to the molecular chaperone [9,21], other cochaperones such as Prostaglandin E synthase 3 (p23), Activator of Hsp90 ATPase homolog 1 (Aha1) and Bcl-2-associated athanogene 1 (Bag-1) lack TPRs (Figure 2) and use their own, unique sequences to associate with Hsp90 and Hsp70. These cochaperones may have other activities of their own that are independent of their interaction with the molecular chaperones. Such actions were referred to in a recent quantitative analysis of the chaperone-cochaperone-client interaction networks in human cells, where the physical interaction landscape of all known Hsp70-and Hsp90-bound cochaperones was analyzed [17].
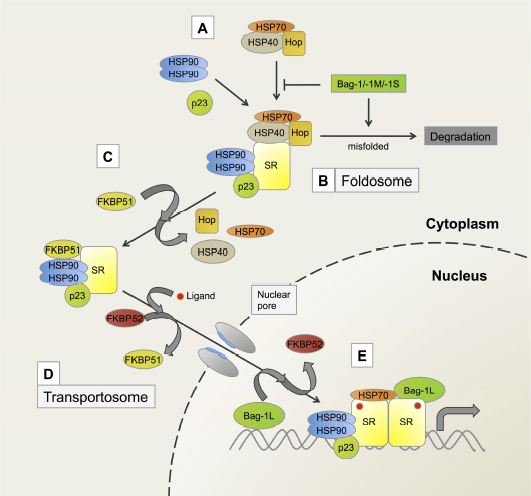
A model depicting some of the key steps of the maturation pathway of steroid receptors.
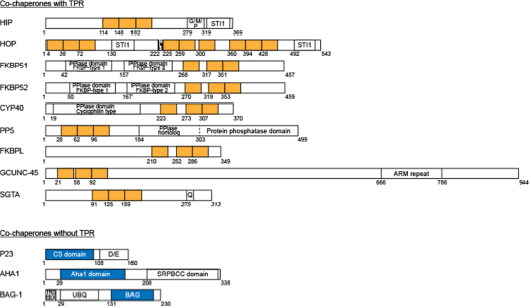
The structure of TPR-containing and TPR-lacking cochaperones.
General perceptions of the role of Hsp70 and Hsp90 in steroid receptor action have changed in recent times. For example, cryoelectron microscopy studies have demonstrated that Hsp70, known to facilitate GR delivery to Hsp90, actually inactivates GR through partial unfolding of the receptor [18]. Conversely, Hsp90 is able to reverse this function and promote GR activation. Although this unfolding/inactivation by Hsp70 and refolding/reactivation by Hsp90 might seem contradictory, this combination could in fact be complementary; constant rounds of Hsp70-mediated unfolding/ligand release and Hsp90-mediated refolding/ligand binding could allow for the non-liganded GR to remain in a non-aggregating, high-affinity state poised for a rapid response to changing hormone levels. In another cryoelectron microscopy study, Hop, previously thought of as an adaptor for Hsp90 and Hsp70 binding which coordinated their actions on folding protein substrates [19], was shown to have additional functions in a reconstruction of the Hsp90/Hop complex. Here Hop formed extensive interactions with Hsp90, preorganizing its N-terminal domain for ATP hydrolysis and client protein binding [20]. In the classical model, Hop is eventually released from the complex and is replaced by one of the other TPR cochaperones such as FKBP51, FKBP52 or Cyp40 [21] (Figure 1C). This dynamic exchange occurs on Hsp90 dimers of the foldosome. Although any one of the TPR proteins can in principle replace Hop, only one TPR protein is found bound to the Hsp90 dimers at any one time [22], implying that Hsp90 might survey the local environment for available TPR proteins with which to interact. There is some evidence suggesting that the type of ligand that ultimately associates with the receptor complex influences which TPR protein is recruited. For example, aldosterone binding to the mineralocorticoid receptor (MR) favors the exchange of FKBP51 for FKPB52, while binding of 11,19-oxidoprogesterone favors the association with the immunophilin-like PP5 [23].
In previous studies it was though that steroid receptors remain in the cytoplasm in complex with molecular chaperones and cochaperones in the absence of hormone [24]. Upon ligand binding, it was thought that the complex dissociates and the receptors are transported into the nucleus, where they bind chromatin and regulate the expression of multiple target genes. However, this classic model of molecular chaperones action is perhaps a bit too simple. Steroid receptors are known to shuttle between the cytoplasm and the nucleus [25] and some, such as the estrogen (ER) and progesterone (PR) receptors, remain in the nucleus bound to molecular chaperones in the absence of hormone [26,27]. Several questions therefore arise concerning how molecular chaperones and cochaperones are transported into the nucleus. Are they transported on their own or in complex with the steroid receptors and what role do they play in the nucleus? More importantly, in view of the recent data on chaperone-independent functions of cochaperones, one might ask whether cochaperones exert specific effects on steroid hormone action. In this review, we will focus on two cochaperones, p23 and Bag-1L, that are present in the nucleus, where they reportedly influence steroid receptor action through receptor recycling or via modulation of receptor binding to chromatin. We will describe how these cochaperones exert their action and suggest how cochaperone/ steroid receptor action could be targeted for therapeutic purposes.
From foldosomes to transportosomes
The Jensen two step hypothesis of cytoplasmic/nuclear transportation of steroid receptors [28], together with the finding that molecular chaperones and cochaperones bind to nonliganded receptors, generated the concept that molecular chaperones confine steroid receptors in an inactive, cytoplasmic state. However nonliganded receptor/molecular chaperone complexes have been found to constantly shuttle between the cytoplasm and the nucleus [29,30]. Nevertheless, shuttling by the PR and ER, which are largely nuclear, may be mechanistically different from shuttling by MR and the androgen receptors (AR). This is in turn distinct from shuttling of GR, which is mainly localized in the cytoplasm, but can also be found at the nuclear periphery as part of a (non-liganded) GR/molecular chaperone complex in association with the integral nuclear pore glycoprotein Nup62 and importin β [31]. The Hsp90 cochaperone Aha 1 may also be involved in this complex as it was shown to contribute to the nucleocytoplasmic transport of the GR; cells lacking Aha 1 showed a reduced and impartial translocation of the receptor into the nucleus [32]. Whether Aha 1 is transported into the nucleus together with the GR is however not known. Several studies now show that molecular chaperones and cochaperones are transported along with the liganded steroid receptors into the nucleus [33,34]. In fact more recent studies have shown a constitutive requirement of Hsp90 throughout the functional lifetime of the GR and not just during the initial folding phase [18].
Ligand-dependent translocation of GR to the nucleus has been found to be considerably reduced using the Hsp90 inhibitor geldanamycin (GA) [35,36]. Furthermore, microinjection of an antibody against the cochaperone FKBP52, but not an isotype control, inhibited ligand-induced nuclear transport of the GR [37], suggesting that FKBP52 contributes to nuclear translocation of the GR. Additional evidence that FKBP52 plays a role in the nuclear transport of the GR is the finding that it directly binds to the motor protein dynein via dynamitin [38,39]. Through this interaction, the receptor/chaperone complex is thought to move along the cytoskeleton to the nucleus on what has been described as the transportosome [40] (Figure 1D). The affinity of the FKBP52-receptor complex for dynein possibly determines the rate of transportation of the steroid receptors into the nucleus [39]. While GR has a high affinity for FKBP52, MR has a preferred affinity for FKBP51. Experiments using cross-linked MR-Hsp90 or GR-Hsp90 heterocomplexes showed that these large heterocomplexes can be found in the nucleus in the presence of hormone, demonstrating that they can pass undissociated through the nuclear pore to the nucleus [31,41]. Once in the nucleus, the steroid receptor/molecular chaperone complex dissociates and the steroid receptor is converted into a DNA-binding form as has been shown by Davies et al. (2002) [42].
Given that FKBP51 and FKBP52 have been implicated in steroid hormone action in vitro, mouse knockout models of FKBP51 and FKBP52 have been generated to determine the impact of these cochaperones on steroid hormone action in vivo [43]. FKBP1 knockout mice failed to exhibit any disruption in endocrine activity, whereas no overt defects in GR-regulated physiology were observed in FKBP52 KO mice. Subsequently, GR function was compared in mouse embryonic fibroblasts (MEFs) from wild-type (WT) and FKBP52-deficient animals [44]. Contrary to expectation, loss of FKBP52 had no effect on the composition of hormone-free GR heterocomplexes, GR hormone-binding function or nuclear translocation, nor on global GR regulation of gene expression. Rather, loss of FKBP52 was found to result in selective loss of expression of specific genes, such as glucocorticoid-inducible leucine zipper (GILZ) and FKBP51, but not the serum- and glucocorticoid-kinase (SGK) and p21 genes [44]. The FKBP52 knockout mice however demonstrated the importance of this cochaperone for reproductive tissue development. Female knockout mice showed defects in uterine receptivity for embryo implantation and male knockouts displayed ambiguous external genitalia and dysgenic prostates [45,46]. Furthermore, in the female knockout mice FKBP52 was shown to be an essential regulator of PR action in the uterus, while being a non-essential but contributory regulator of steroid receptors in the mammary gland and ovaries [47]. These data may now provide the basis for selective targeting steroid-regulated physiology through co-chaperones.
Role of molecular chaperones in the nucleus
Studies using in vitro receptor/DNA interaction techniques and in vitro transcription experiments have provided hints that, in the nucleus, molecular chaperones function as modulators of the DNA-binding and transcriptional activities of steroid receptors [48,49]. One example is the work of Etienne Baulieu and colleagues in 1996 which showed that the binding of ERα to the estrogen response element from the vitellogenin A2 gene is inversely dependent on the relative concentration of Hsp90 [48]. In another assay, recombinant (ligand-free) PR was only able to bind to and induce transcription on (hormone-containing) chromatin templates in the presence of rabbit reticulocyte lysate rich in molecular chaperones [49]. The use of the Hsp90-specific inhibitor GA blocked the transcriptional activity of this receptor on chromatin [49], demonstrating a crucial role of Hsp90 in the nuclear function of the PR.
Additional experiments have provided more proof for a regulatory role of molecular chaperones on steroid receptor action at the chromatin level. Using live cell imaging, the groups of Gordon Hager, David Toft and Don DeFranco demonstrated that molecular chaperones contribute to the rapid mobility and dynamic exchange of steroid receptors at transcriptionally active chromatin sites [36]. They showed an impairment of nuclear mobility of GR and PR using transcriptionally active nuclei depleted in soluble factors. Receptor mobility was regained upon incubation of the nuclei with an ATP-dependent regenerating system and combinations of purified chaperones and cochaperones. A mixture of seven components (Hsp90, Hsp70, p23, Hop, Ydj-1, FKBP51) and CHIP was the most effective, but a mixture of five proteins (Hsp90, Hsp70, p23, Hop, and Ydj-1) or even three (Hsp90, Hsp70, and Hop) restored steroid receptor movement, albeit to a lesser extent [36]. Together these findings led to the conclusion that the molecular chaperones and cochaperones are involved in recycling of the receptors on chromatin.
Nuclear action of p23
Although the experiments described above argue that molecular chaperones and cochaperones enter the nucleus bound to the steroid receptors, some of the cochaperones appear to have a nuclear function of their own, independent of their regulation of receptor action. Two examples are the cochaperones p23 and Bag-1L. Mammalian p23 (or Sba1 in yeast) is composed of a simple molecular structure consisting of a compact eight β-strand antiparallel sandwich (the CS domain in Figure 2) followed by an acidic C-terminal tail [50,51]. This structure is conserved from humans to yeast with orthologs in both plants and protozoa. The main role of p23/Sba1 is to bind the ATP-engaged N-terminal domain of Hsp90, thereby stabilizing a high-affinity client binding conformation. At the same time this slows down the hydrolysis of ATP and increases the dwell time of client proteins in the Hsp90 chaperone complex [52]. However, it appears that p23 also has a chaperone activity of its own, independent of Hsp90. A large portion (approximately 69%–75%) of the p23/Sba1 interacting proteins (as determined by genetic and proteomic high throughput approaches in yeast) is not shared with Hsp90 [53]. Further analysis of the effect of Sba/p23 on chromatin events showed that deletion of the yeast p23 (Sba1Δ) reduced the number of DNase I hypersensitive sites in chromatin. The number of sites was decreased from 3260 in wild-type to 2439 in Sba1Δ cells. However, this reduction in the total number of DNase I hypersensitive sites in Sba1Δ cells (approximately a 25% loss), was not a mere reduction but was associated with the appearance of novel sites within chromatin. p23 does not harbor any obvious DNA-binding domains and it appears that its effect on transcription factors and chromatin is therefore mediated through protein-protein interactions with chromatin remodelers, such as the histone acetyltransferase GCN5 [54].
p23-mediated regulation of steroid receptor action on chromatin
Brian Freeman and Keith Yamamoto have previously suggested a genomic action of the cochaperone p23 in the dissociation of receptors from coactivators and response elements on chromatin [55]. Using chromatin immunoprecipitation (ChIP), they observed that molecular chaperones together with cochaperones are recruited to the response elements of the liver-specific, glucocorticoid-inducible tyrosine aminotransferase (TAT) and tryptophan oxygenase (TO) genes in rat hepatoma HTC cells. They also showed that p23 and Hsp90, but not Hsp70, were localized to the same response elements in a hormone-dependent manner [56] (Figure 1E). In an unrelated immunofluorescence experiment, Hsp90, Hsp70 and p23 were visualized together with GR on an integrated array of glucocorticoid response elements (GRE) from the mouse mammary tumor virus (MMTV) DNA in the genome of a mouse adenocarcinoma cell line [57]. In this assay, the molecular chaperones and particularly Hsp90 and p23, were shown to stabilize GR binding. Inhibition of Hsp90 activity simultaneously reduced the binding of the molecular chaperones, p23 and GR to the response elements [57]. Accordingly, two contrasting explanations of the action of molecular chaperones and cochaperones have been presented, namely, their involvement in the dissociation of the receptor complex [55] on the one hand, and on the other, their ability to stabilize the receptor complex as outlined above. This discrepancy could be due to a key difference in the experimental setup of the two studies. In one study, Hsp90 function was disrupted by GA treatment and since this drug prevents p23 binding to Hsp90 [58], it was thought it would also affect p23 action [57]. In the other study, altered p23 activity was achieved by the addition of purified p23 to in vitro transcription assays [56]. Although p23 and Hsp90 normally act in concert within the chaperone complex, they may not act together on DNA. Inhibiting the interaction of Hsp90 with p23 may therefore not have the same effect as the addition of p23. However, both studies agree that Hsp90 and p23 modulate the action of the GR at the chromatin level.
The physiological function of GR requires that ligand be presented to target cells in discrete pulses consistent with the pulsatile production of glucocorticoid by the adrenal gland [59]. Using ChIP, a connection between the function and cyclical action of GR and intranuclear molecular chaperones and cochaperones at glucocorticoid target genes was demonstrated [60]. When the chaperone activity was disrupted by GA treatment, pulsatile GR transcriptional activity was abrogated [60]. Furthermore it was shown that the ligand-bound GR complexes exchange rapidly and continuously with response elements in chromatin (in the time scale of seconds). During each exchange, the receptor may lose its ligand and require entry into the “chaperone cycle” (possibly through the foldosome activity) to re-acquire its ligand. Alternatively, the receptor may retain its ligand, but may need to return to the chromatin template with the help of the chaperone/cochaperone complexes [59]. These cycling reactions of the GR are different from those of the other steroid receptors, such as the AR or ER. Although cycling of AR and ER on chromatin have previously been described [61,62], these cycling events are intrinsic properties of these receptors and they depend on proteasome function and the degradation of the receptors. Furthermore, these processes occur in the presence of constant hormone levels, which is different from the oscillations of the GR.
Increased expression of p23 achieved by transfection in the estrogen-dependent breast cancer cell line MCF-7 increased ERα recruitment and activity at select regulatory elements of ER target genes [63]. Utilizing ChIP-sequencing (ChIP-seq), a 230% increase in the number of estrogen-induced ER-binding sites, compared with control cells, could be demonstrated in response to the increased expression of p23. Interestingly, motif analysis indicated that ERα bound to the same DNA sequences, regardless of p23 status. The increase in ER binding sites was therefore not due to enhanced ER binding but most likely due to p23-mediated changes in histone modification, a consequence of which would be increased chromatin accessibility and binding by ERα [63]. It has been shown more recently that p23 enhances AR transactivation function in both the cytoplasm, by increasing AR ligand-binding capacity, possibly via direct interaction, and in the nucleus, by enhancing AR occupancy at target promoters [64]. An interaction between AR and p23 was demonstrated even after treatment with the geldanamycin analog, 17-N-Allylamino-17-demethoxygeldanamycin (17-AAG), a Hsp90 inhibitor that displaces p23 from the complex with Hsp90 [64]. The effects of p23 on AR activity were, at least in part, Hsp90-independent, since a mutant form of p23 that was unable to bind Hsp90 increased AR activity nevertheless [64]. Collectively, these studies suggest that p23 has other functions in addition to its action as an Hsp90 cochaperone.
Knockout mouse model of p23
A knockout mouse model has been generated for studies on the role of p23 in steroid hormone action. p23 was found absolutely necessary for perinatal survival, but it was inessential for overall prenatal development and morphogenesis [65]. The skin barriers at the final fetal stages of development were incompletely formed and the lungs of p23 null embryos displayed underdeveloped airspaces and substantially reduced expression of surfactant genes [65]. The defects in skin and surfactant gene expression correlate with defects in glucocorticoid function in promoting lung maturation, and the development of epithelial barriers [66–68]. Accordingly, embryonic fibroblasts from p23 null mice displayed a defective glucocorticoid response [65]. The perinatal phenotype of the p23 null mice has prevented analysis of the contributions of this cochaperone to other endocrine functions in adult mice.
Nuclear action of Bag-1
The Bag-1 isoforms
Another cochaperone that has been shown to function in the nucleus in addition to p23 is Bag-1L, a member of the Bag-1 family of proteins. In humans, the Bag-1 family is made up of polypeptides translated from one mRNA by a leaky scanning mechanism [69,70]. This generates four differentially-sized isoforms; Bag-1L, Bag-1M, Bag-1 and Bag-1S (Figure 3A). These proteins differ at their N-terminal sequences, but have a conserved C-terminal Hsp70-binding domain (otherwise known as BAG domain) [71,72]. Additionally, Bag-1 proteins contain a ubiquitin-like domain (UBQ) through which they can be connected to the proteasome [73]. The UBQ was shown to be important in CHIP (carboxyl terminus of Hsp70-interacting protein)/E3 ligase-dependent degradation of the GR [74]. Bag-1 is therefore a coupling factor, which can link the chaperones and the proteolytic complex together and thereby plays a role in steroid receptor turnover (Figure 1A) [75].
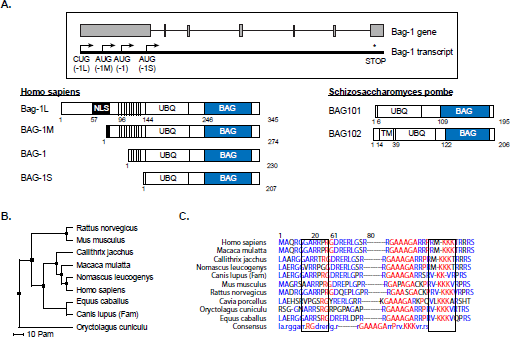
The Bag-1 protein family members and their structural domains.
The Bag-1 proteins do not contain a TPR motif but instead use their BAG domain to bind the Hsp70 ATPase. The BAG domain is approximately 100 amino acids in length and is made up of three antiparallel alpha helices, which serve as a protein-protein interaction surface for a number of cellular proteins [76]. In a 1.9 Å crystal structure in complex with the ATPase of Hsc70 (a homologous protein of Hsp70), the BAG domain was shown to induce a conformational switch in the ATPase that is incompatible with further nucleotide binding. A similar switch was observed in the bacterial Hsp70 homolog DnaK, when bound by the structurally unrelated nucleotide exchange factor GrpE. The Bag-1 proteins and in particular Bag-1 are therefore often described as mammalian nucleotide exchange factors of Hsp70 [72]. Furthermore, the interaction of Bag-1 with Hsp70 and with the unliganded GR has made it a member of the foldosome complex [77]. In fact, early studies showed that Bag-1 is involved in the release of Hop from the foldosome complex [77] (Figure 1A and B) but later analysis demonstrated that it actually competes with Hip for binding to the Hsp70 ATPase domain [6].
Bag-1L, the largest member of the family, possesses a N-terminal nuclear localization sequence (NLS) and is therefore exclusively localized to the nucleus. The other Bag-1 isoforms are mainly cytoplasmic [70], although under stress conditions these members and notably Bag-1M, are reported to also localize to the nucleus [78]. Intriguingly, two orthologs of Bag-1, Bag101 and Bag102 (Figure 3A), have been identified in the fission yeast
Nuclear action of human Bag-1L
The human Bag-1 proteins were shown to either positively or negatively regulate the action of many steroid/nuclear receptors ranging from GR, PR, MR, AR, ER and vitamin D receptor (VDR) to retinoid X receptor (RXR) [76,80]. In particular ERα, ERβ, AR and VDR were reported to be positively regulated by Bag-1L, and to a lesser extend by Bag-1M [81–83]. This suggests that the (NLS-containing) N-terminus of Bag-1L, which is absent in Bag-1M, contributes to the positive effect of Bag-1L on steroid receptor action. To confirm this experimentally, the cytoplasmic Bag-1 proteins (Bag-1M and Bag-1S) were tagged with a SV40 NLS and their effect on AR regulation was monitored. Although NLS-Bag-1M and NLS-Bag-1S translocated to the nucleus and were able to exert positive regulation on AR transactivation, they did not achieve the regulatory activity of Bag-1L [84,85]. This suggests that the N-terminus of Bag-1L has a function distinct from the cochaperone activity of its C-terminal BAG domain.
In addition to the presence of a NLS at the N-terminus, Bag-1L binds the AR, ERα and VDR [82,85,86] and harbors two additional, unique functions. First, the region between amino acids 72 to 79 is reported to bind non-specifically to DNA. This region contains positively charged sequences of three consecutive lysine and three arginine residues, separated by a centrally located neutral residue. Mutational analysis has identified both trimeric blocks as essential for DNA binding [87,88]. Second, sequences between amino acids 17–50 have been described as important for the nuclear retention of Bag-1L. It is thought that this region holds the cochaperone anchored to structures in the nucleus, possibly histone proteins [84]. Together, these regulatory elements keep Bag-1L in the nucleus and contribute to its effect as a modulator of steroid receptor action. In ChIP experiments, Bag-1L was bound to chromatin along with the AR at androgen-regulated target genes [85,89].
The GARRPR motif of Bag-1L
More recently a duplicated sequence “GARRPR” at positions 6–11 and 66–71 at the N-terminus of Bag-1L was shown to interact with the AR and the ERα [89]. Mutation of these motifs destroyed binding of Bag-1L to the AR but did not impair the chromatin binding potential of the mutant Bag-1L [89]. This indicates that chromatin and receptor binding are not linked for this protein. While the C-terminal BAG domain shows a high degree of sequence homology among the Bag-1 proteins found throughout evolution (in yeast, invertebrates, amphibians, mammals and plants), the first 128 N-terminal amino acids containing the duplicated GARRPR motif are less well conserved [90].
Nevertheless, a high sequence homology exists among the GARRPR motifs of human and monkey (M. mulatta, N. leucogenys and C. jacchus) (Figures 3B and C). Protein binding studies using peptides encompassing the GARRPR motifs showed that they bind to the ligand-binding domain (LBD) of the AR to a region termed binding function-3, which allosterically modulates the activity of the receptor [89]. Structure-based sequence alignments of the LBD of multiple steroid receptors show that the BF-3 pocket is highly conserved among steroid receptors as well as being present in other major nuclear receptors [91]. It is therefore expected that the GARRPR motif would also bind to other steroid/nuclear receptors. However, in vitro experiments showed that the GARRPR motif bound only to the AR and ERα and not the PR or GR [89]. This suggests that other receptor sequences contribute to the binding to the GARRPR motif. So far the only other protein that has been found to bind the BF-3 region is the cochaperone FKBP52 that has a GARRPR-like sequence [89,92].
It is interesting to note that for both Bag-1L and FKBP52 the sequences that interact with the BF-3 domain are different from those reported to bind Hsp70 and Hsp90. Another interesting observation is that mutation of the GARRPR motifs in both proteins results in a gain-of-function phenotype [89,93]. In Bag-1L the mutations do not completely inhibit AR-mediated gene expression but rather increase the expression of a subset of androgen-regulated genes involved in metabolic processes [89].
The BAG domain of Bag-1L
In addition to the regulation of steroid receptor action through the GARRPR motif, several lines of evidence suggest that Bag-1L may indirectly regulate the activity of the steroid receptors through its BAG domain. First, deletion of the BAG domain destroys the ability of Bag-1L to enhance the activity of the AR [85,94]. Second, mutation of amino acids in the BAG domain involved in the interaction with the ATPase domain of Hsc70, not only destroyed the interaction between Bag-1L and Hsc70, but simultaneously obliterate the ability of Bag-1L to enhance the transactivation function of the AR [94]. Collectively, these results show that the BAG domain of Bag-1L, which also acts as a nucleotide exchange factor for Hsp70, contributes to the regulation of steroid receptor action, at least for AR. Intriguingly, protein-protein interaction studies showed that the BAG domain of Bag-1L does not interact with the AR-LBD, but rather with its N-terminal transactivation domain [85]. This region of the AR is intrinsically disordered, which means that it exists without a stable tertiary structure. However, the lack of structure has several advantages. For example, it provides a large interaction surface compared with other globular proteins of the same size. Secondly, intrinsically disordered proteins have short linear motifs (SLiMs) that allow them to recognize binding partners by undergoing coupled folding and binding processes. SLiMs have extremely compact protein interaction interfaces that are generally encoded by less than four major (affinity- and specificity-determining) residues within a stretch of 2–10 amino acids [95]. The occurrence of SLiMs in intrinsically disordered regions gives way to specific, yet transient, interactions that enable them to play central roles in signaling pathways and allow them to act as hubs for protein interaction networks [96]. It is likely that the ability to be involved in such protein-protein interactions allows the BAG domain of Bag-1L to exert a great impact on AR function. Since several steroid receptors have SLiMs-containing regions at their intrinsically disordered N-termini [97], it seems plausible that they could also be targeted by the BAG domain of Bag-1L. It is likely that they already account for the reported effects of Bag-1L on steroid receptor action [81–83].
Knockout mouse model of Bag-1
Although Bag-1 knockout mice have been generated to study the effect of loss of this molecule on steroid hormone, these mice die between embryonic days 12.5 and 13.5 (E12.5 and E13.5). Consistent with the essential role of Bag-1 in the survival of differentiating neurons and hematopoietic cells [98], Bag-1 knockout mice display massive apoptosis in cells of the fetal liver and developing nervous system [98]. In contrast, Bag-1 heterozygous mice are viable and show no difference in development, growth and body size compared to their wild-type counterparts, nor do they have obvious defects in the endocrine system (our unpublished observations). The Bag-1 gene is separated by only 414 bp from Chmp5, a gene that codes for charged multivesicular body protein 5. These two genes are encoded by different DNA strands and their 5’ ends are positioned head-to-head [99]. Targeted disruption of Bag-1 resulted in a double knockout, ablating the expression of both Bag-1 and Chmp5 (our unpublished data). The Chmp5 knockout, like the Bag-1 knockout, is embryonic [99]. This double knockout has hampered analysis of the contribution of Bag-1 to steroid receptor action in the mouse. A new strategy for Bag-1 knockout mice (Bag1tm1a(EUCOMM)Hmgu/Ics) has been generated by the European conditional mouse mutagenesis (EUCOMM) program, and future characterization of these mice will determine the contribution of Bag-1 to steroid receptor action.
Conclusion
Although the molecular chaperones Hsp70 and Hsp90 are present in both the cytoplasm and the nucleus, a clear role of these proteins in the regulation of steroid receptor function at the chromatin level has not been identified. Instead unambiguous evidence exists that cochaperones that regulate the activity of these molecular chaperones, and are themselves recruited into the nucleus, modulate the transcriptional activities of the steroid receptors on chromatin. To date, two cochaperones have been characterized in this respect: p23 and Bag-1L. Both proteins belong to the family of cochaperones that do not interact with Hsp70 and Hsp90 through the classical TPR domains. Instead these proteins employ domains that are multifunctional and are also involved in chaperone-independent activities. This is particularly the case for Bag-1L, where in fact two domains have been identified for regulating cellular processes; the BAG domain that serves as a nucleotide exchange factor for Hsp70 and steroid receptor binding, and the GARRPR motif at its N-terminus that is essential for AR and ERα▪binding. It is currently unclear whether the two domains functionally cooperate in regulating steroid receptor action or if they act independent from one another. Similarly, p23 mutational and inhibitor studies have shown that the sequences it uses to bind Hsp90 are different from those for interaction with the AR. Combined, these findings therefore offer great opportunities for the discovery of small molecular-weight chemicals that can specifically target these interaction surfaces and thereby inhibit the steroid receptor action in a novel way without affecting the overall chaperone activity. Some interesting candidate compounds have already emerged. For example, 2-((2-(2,6-dimethylphenoxy)ethyl)thio)-1H-benzo[d]imidazole competes with the binding of the Bag-1L GARRPR motif to the AR [89]. Thio-2 binds to the BAG domain of Bag-1 [100] and Gedunin binds to the N-terminus of p23 [101]. In the latter two cases, the inhibitors are only “partially dissociated” compounds and therefore still inhibit Hsp70 and Hsp90 action, albeit weakly. Future detailed studies on how these cochaperones regulate steroid receptor activity independent of their activity through the molecular chaperones will open new avenues to the identification of more specific compounds that can produce a new generation of steroid receptor therapeutics.
Footnotes
Acknowledgements
This work was supported, in part, by grants from the Deutsche Krebshilfe (110237) to A. C. B. C. and from the National Cancer Institute (1P01CA163227) and (P50CA090381) to M. B.
