Abstract
Objective
To determine the effects of hypoxia and hypoxic exercise (HE) on the norepinephrine levels of various tissues in rats.
Methods
Male Wistar rats were randomly assigned to 3 groups: an HE group (n = 6), a hypoxic-sedentary (HS) group (n = 6), and a normoxic-sedentary (NS) group (n = 6). The HE rats had access, ad lib, to an exercise wheel for 8 weeks. HE and HS rats were maintained in a normobaric hypoxic chamber with an F
Results
Liver norepinephrine levels in the HE and HS groups were significantly lower than the levels in the NS group (P < .05). No significant difference was found in liver norepinephrine levels between the HE and the HS groups. The heart norepinephrine levels in the NS group were significantly lower than the levels in the HE (P < .01) and HS groups (P < .01). In contrast, no significant differences were found in the norepinephrine levels for the diaphragm and soleus muscle among the 3 groups. The norepinephrine levels in the gastrocnemius white muscles were significantly higher in the HS group than in the HE (P < .05) and NS groups (P < .01). P < .01 represents a significant difference at the level of 1%.
Conclusions
This study demonstrated that hypoxia and HE both elicit a decreased sympathetic response in the liver tissue of male Wistar rats but cause an increased response in heart tissue. These results suggest that the sympathetic responses to long-term hypoxia and HE training are different in various rat tissues.
Introduction
The catecholamines, both as neurotransmitters and as neurohormones, have very powerful regulatory properties over a number of critical physiological and metabolic functions.1,2 Measurement of tissue catecholamine content has been used as an indicator of the extent of local sympathetic nervous system activity in response to various stimuli, including exercise. 1 Acute exposure to hypoxia has been shown to be associated with a reduction in the level of norepinephrine or dopamine in the entire rat brain.3,4 In various regions of the rat brain, the norepinephrine level has been shown to decrease after hypoxia exposure, lasting from 2 to 24 hours4,5 and may not change for up to 2 hours after hypoxic breathing has begun.6,7 In addition, Roubein et al 5 showed that the levels of monoamines in brain regions return to initial levels after 36 hours in a hypoxic environment.
Few reports have addressed the effects of exercise on monoamine levels in the liver, heart, adrenals, and skeletal muscles. Mazzeo and Grantham 8 reported that strenuous exercise caused norepinephrine to decrease in the heart and adrenals but increase in the liver and soleus muscle. However, it has also been reported that exercise training left the norepinephrine level unchanged in the liver.8,9 There have also been conflicting reports about the effect of exercise training on norepinephrine levels in the heart. Studies have shown that exercise training can increase heart norepinephrine content, 10 decrease the content,11,12 and leave the content unchanged8,13,14 at rest. No data regarding the effects of hypoxia and hypoxic exercise (HE) training on the norepinephrine levels of the liver, heart, diaphragm, and skeletal muscles in Wistar rats have been reported.
We investigated the effects of hypoxia and HE training on the levels of norepinephrine of the liver, heart, diaphragm, soleus, and gastrocnemius white muscles in Wistar rats.
Methods
Animal Care and Equipment
All animals were housed and bred under similar conditions in Nihon Clea (Fuji-gun, Japan). The animal room was maintained at 24°C ± 1°C with a 12:12 hour light-dark cycle. The animals were allowed free access to standard food and water. All experiments were carried out according to ethical principles established by Nihon Clea for the care of animals. In addition, Nihon Clea's Ethical Committee approved the protocols for the animal experiments. Male Wistar rats were randomly assigned to 3 groups: an HE group (n = 6), a hypoxic-sedentary (HS) group (n = 6), and a normoxic-sedentary (NS) group (n = 6). The HE group was allowed 8 weeks of free access to a running wheel (Natsume, Tokyo, Japan) beginning at 4 weeks of age. The number of revolutions of the wheel, which was 1.16 m in circumference, was recorded with a mechanical counter that was read daily. The rats of the sedentary groups were housed individually in small cages (floor, 14 × 20 cm; height, 14 cm) to limit their physical activity. The HE and HS groups were subjected to an F
Tissue Preparation
At the end of the study periods, rats were anesthetized with ether. Their livers, hearts, diaphragms, soleus, and gastrocnemius white muscles (the superficial white portion of the medial head of the gastrocnemius) were then rapidly dissected, washed with chilled saline, and frozen in liquid nitrogen. All samples were weighed and homogenized with 10 volumes of distilled water per wet weight of tissue. The homogenate (450μL) was mixed with a 1M perchloric acid solution (50μL) and centrifuged for 10 minutes at 22 000g. The supernatant was filtered through a Milipore filter (0.45-μm pore size).
Analytical Methods
The filtered tissue samples were analyzed by high-performance liquid chromatography with electrochemical detection. For the analysis of norepinephrine, the apparatus consisted of a Shimadzu model LC-9A pump (Kyoto, Japan), an autosampler AS-8010 (Toso, Tokyo, Japan), and a chromatopac Chromatocorder 12 (Sic Instruments, Tokyo, Japan). Norepinephrine was quantitatively determined with an LC-4C amperometric detector (Bioanalytical Systems, West Lafayette, IN). The mobile phase consisted of 100 mM of tartaric acid and 100 mM of sodium acetate (pH 3.2) containing 500 μM of EDTA-2Na, 650 μM of sodium 1-octanesulfonate, and 5% acetonitrile.
The flow was 0.7 mL min−1. A capcellpac C18 analytical column (5 μm, 4.6 mm × 150 mm, Shiseido, Tokyo, Japan) was used. The injection volume was 50 μL. Quantification of the norepinephrine was carried out by comparing each peak area with that of the corresponding standard. The working electrode was a WE-3G grassy carbon electrode (detector potential against Ag/AgCl reference electrode; +700 mV).
Statistics
All measurements were carried out in duplicate for each tissue sample. Statistical analysis was a 1-way analysis of variance followed by a Scheffé post hoc test. Differences of P < .05 were considered to be statistically significant. All values were represented as the mean ± SEM.
Results
The daily running distance of the HE group increased from the second to fifth weeks of voluntary running, where it reached a plateau. The distance decreased after the seventh week. During the 8 weeks, the average distance in this group was 2276.9 ± 423.0 m·−1. Figure 1 shows the differences in the norepinephrine levels in the liver for all 3 groups. The norepinephrine levels in the HE and HS groups were significantly lower than the levels of the NS group (P < .05). However, no significant differences were found in the liver norepinephrine levels for the HE and HS groups. Figure 2 shows the differences in the norepinephrine levels in the hearts for the 3 groups. The norepinephrine levels in the NS group were significantly lower than the levels in the HE (P < .01) and NS groups (P < .01). However, no significant differences were found in the heart norepinephrine levels for the HE and HS groups. As for the diaphragm norepinephrine levels, no significant differences were found among the 3 groups (Figure 3). This was true for soleus norepinephrine levels as well (Figure 4). Figure 5 shows the differences in the norepinephrine levels in the gastrocnemius white muscles for the 3 groups. The norepinephrine levels in the gastrocnemius white muscles were significantly higher in the HS group than in the HE (P < .05) and NS groups (P < .01). P < .01 represents significantly difference at the level of 1%. However, no significant difference was found in the norepinephrine levels for the HS and NS groups.
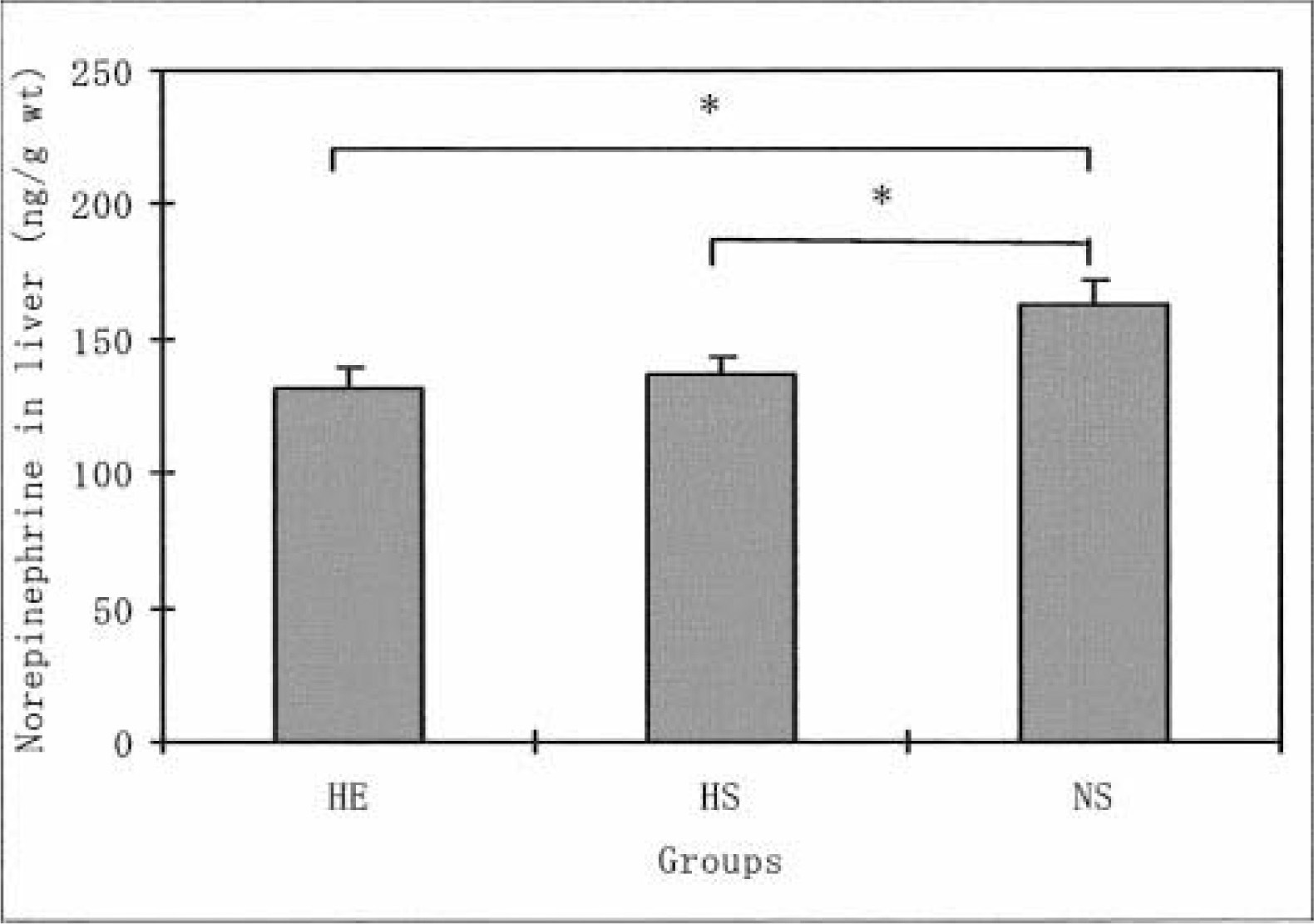
The effects of hypoxia and HE training on norepinephrine levels in the rat liver. The values are means ± SEM. *P < .005 indicates significant difference between both groups. HE indicates hypoxic-exercise training group; HS, hypoxic-sedentary group; NS, normoxic-sedentary group.
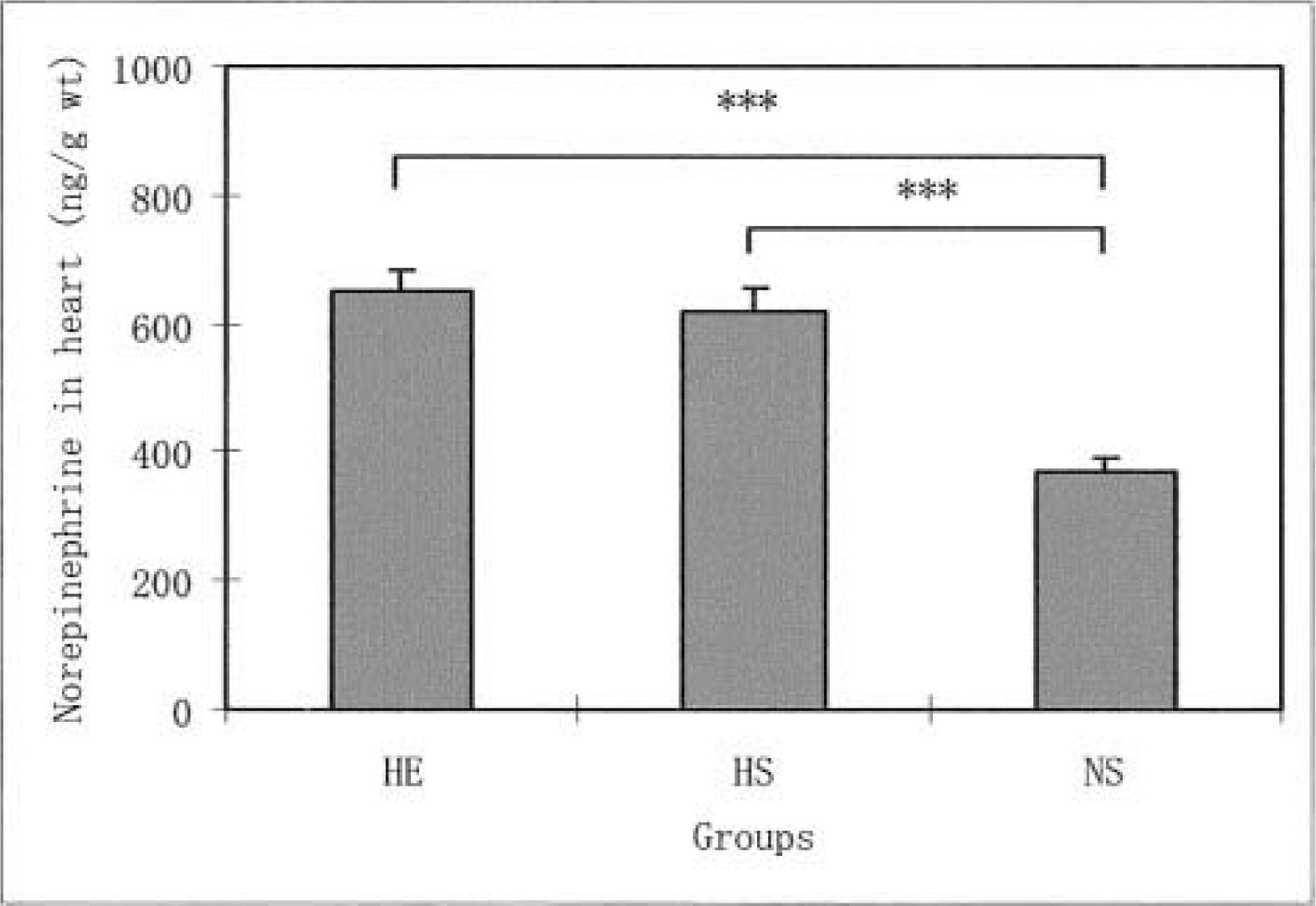
The effects of hypoxia and HE training on norepinephrine levels in the rat heart. The values are means ± SEM. ***P < .001 indicates significant difference between both groups. HE indicates hypoxic-exercise training group; HS, hypoxic-sedentary group; NS, normoxic-sedentary group.
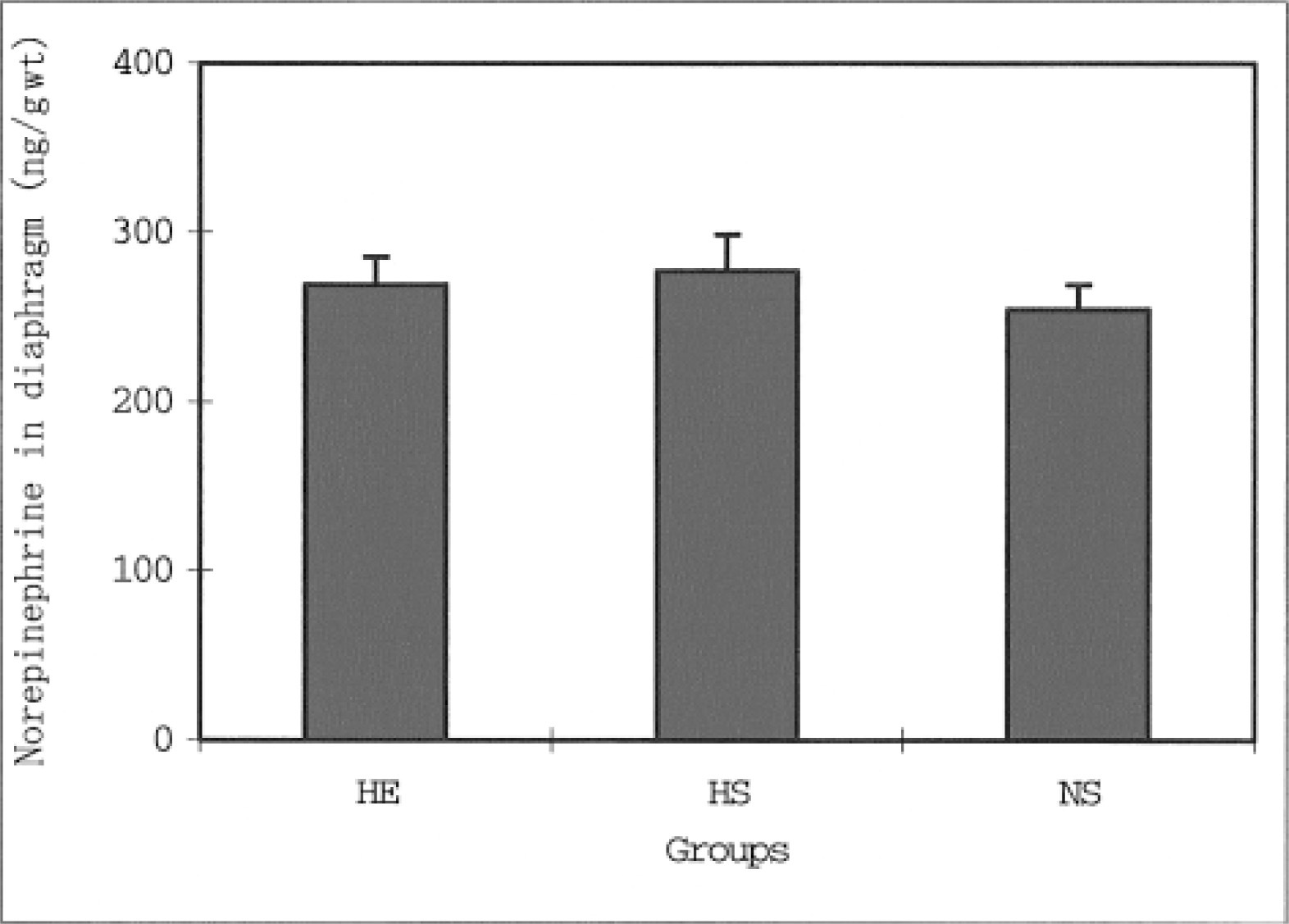
The effects of hypoxia and HE training on norepinephrine levels in the rat diaphragm. The values are means ± SEM. HE indicates hypoxic-exercise training group; HS, hypoxic-sedentary group; NS, normoxic-sedentary group.
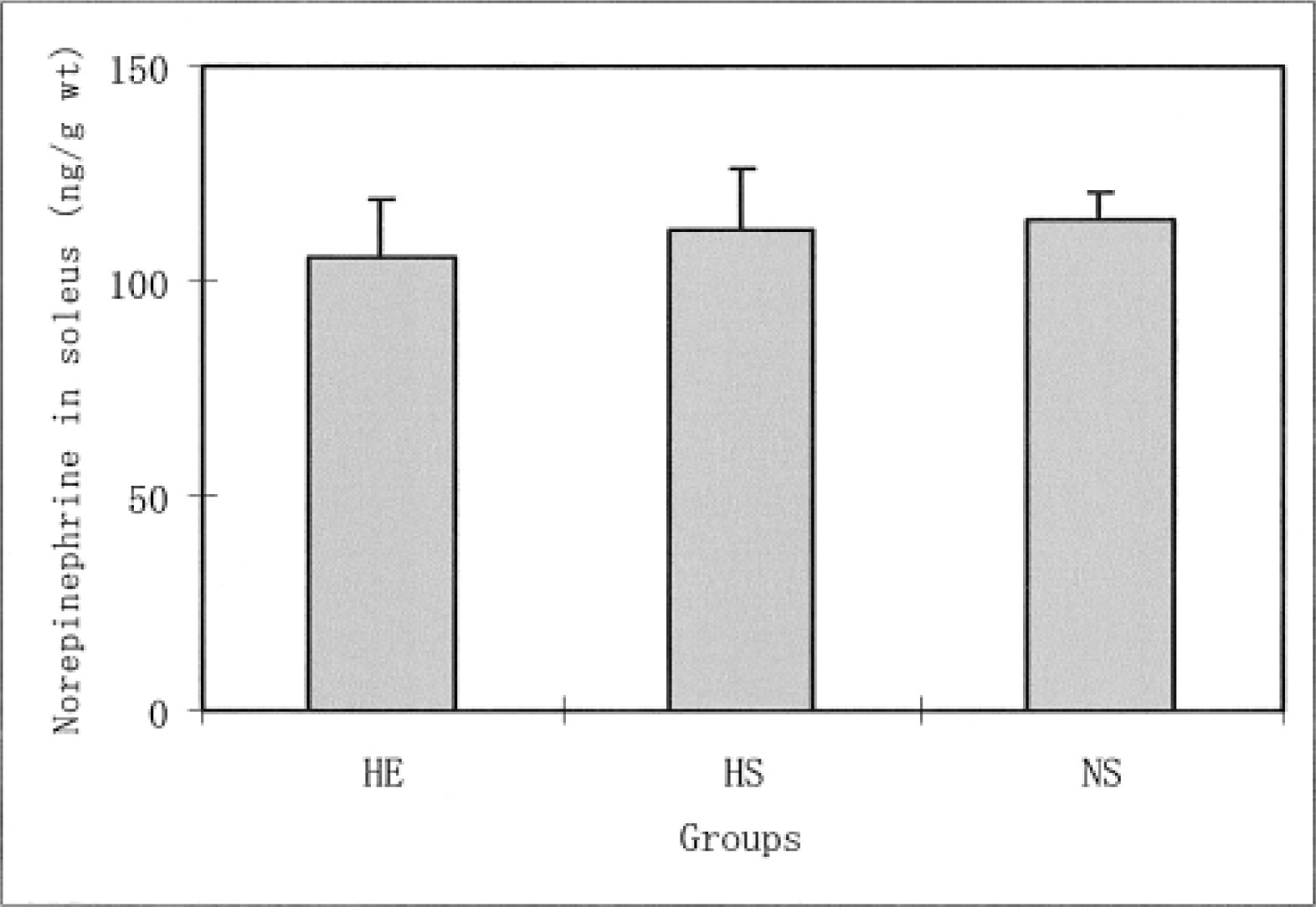
The effects of hypoxia and HE training on norepinephrine levels in the rat soleus muscle. The values are means ± SEM. HE indicates hypoxic-exercise training group; HS, hypoxic-sedentary group; NS, normoxic-sedentary group.
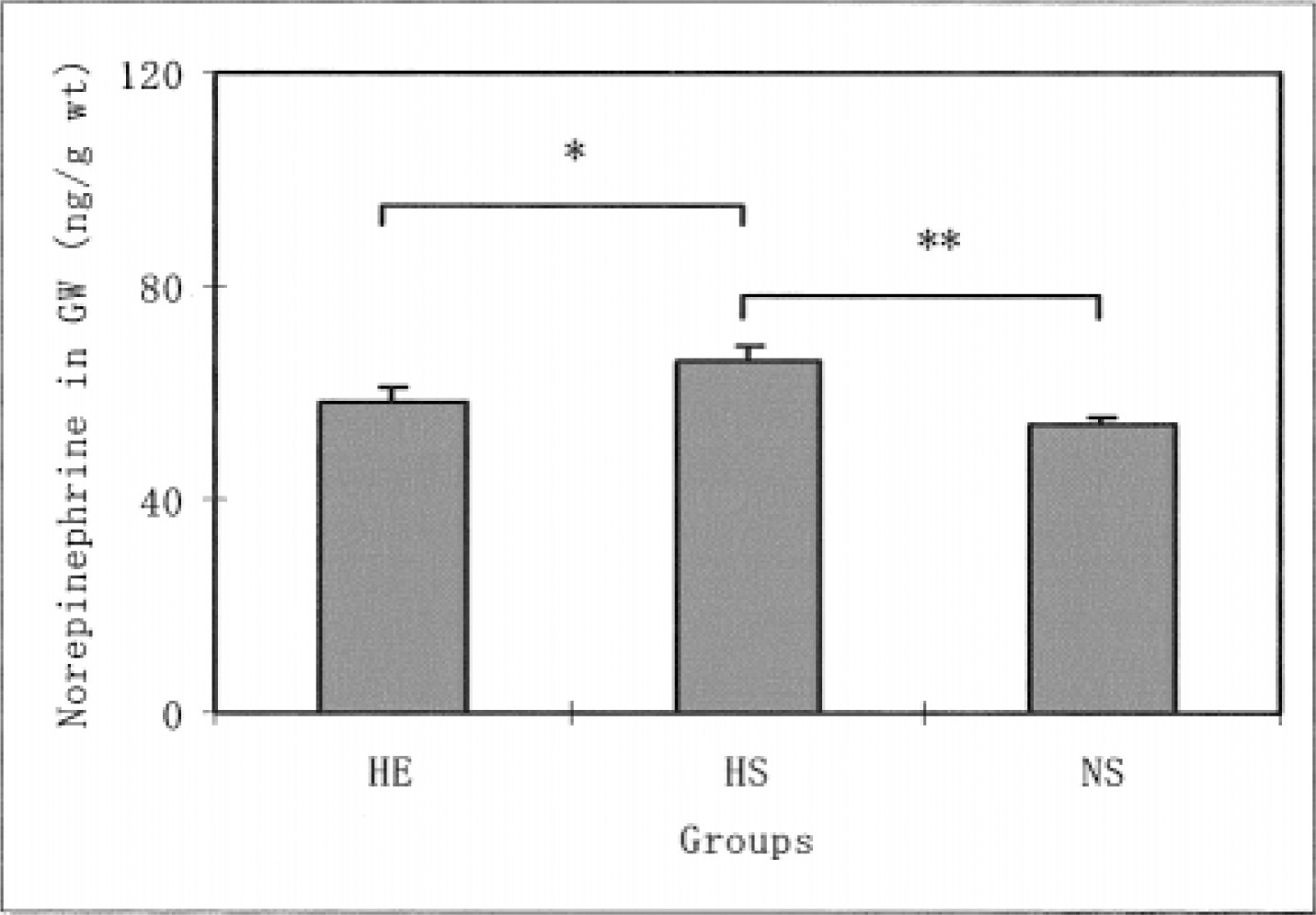
The effects of hypoxia and HE training on norepinephrine levels in the rat gastrocnemius white muscle. The values are means ± SEM. *P < .05, **P < .01 indicates significant difference between both groups. HE indicates hypoxic-exercise training group; HS, hypoxic-sedentary group; NS, normoxic-sedentary group.
Discussion
It has been suggested that the norepinephrine concentration in the liver would provide evidence of activation of the liver's sympathetic neurons. 15 Winder et al 15 demonstrated that treadmill and swimming exercises caused a reduction in norepinephrine concentrations of untrained rat liver (P < .05). A few reports in the literature discuss the effects of physical training and acute exercise on changes in liver norepinephrine.9,15 However, less is known about the influence of a hypoxic condition and HE training on norepinephrine in the liver. In our experiments, the liver norepinephrine concentrations in HS and HE groups were significantly lower when compared with concentrations for the NS group. However, no significant difference was found in liver norepinephrine levels for the exercised and unexercised rats exposed to hypoxic conditions. It has also been reported elsewhere that no significant difference was found in liver norepinephrine content between trained and untrained rats when at rest.8,9,15 These previous investigations support the point that physical training in normoxia does not induce changes in liver norepinephrine concentrations. The present study goes further to make it clear that hypoxia and HE training induces a reduction in the degree of activation of the liver's sympathetic neurons.
The mammalian heart is rich in sympathetic nerve terminals involved in the synthesis, uptake, and release of the neurotransmitter norepinephrine. 16 However, no studies have made reference to the effect of hypoxia in a sedentary model or hypoxia during exercise training on the norepinephrine levels in the heart. In contrast to the response in the liver, in this study, the norepinephrine levels in the heart were significantly higher in the HE training and HS groups compared with the NS group. The reason for the increase in heart norepinephrine caused by hypoxia and HE training can at present be only a matter of speculation. It is conceivable, however, that the number of nerve terminals increases and the norepinephrine content in each teminal is greater. 10 In this study, no significant differences were found in the heart norepinephrine levels for the trained and untrained rats exposed to hypoxic conditions. This is in agreement with previous experiments showing that physical training did not change the norepinephrine levels of a heart at rest.1,8,13,14 The present study shows that hypoxia and HE training cause an increase in the degree of activation of the heart's sympathetic neurons.
Although greater norepinephrine concentrations may be in the heart of HE and HS groups, this may not translate to parallel rises in heart rate and contractility because there may be simultaneous beta receptor downregulation, as demonstrated by Richalet et al. 17
The norepinephrine concentrations of gastrocnemius white muscles in HS rats were significantly higher than the concentrations in the HE training and NS groups. No significant differences were found for the soleus muscles. These differing observations for the sympathetic activity of the skeletal muscles in response to hypoxia and HE training might be due to differences in muscle-fiber types.
We concluded that hypoxia and HE training elicit a decreased sympathetic response in the liver but cause an increased response in the heart. These results suggest that the sympathetic responses to long-term hypoxia and HE training differ among various Wistar rat tissues.
Footnotes
Acknowledgment
This work was supported by a Grant-In-Aid Tetsuo Ohkuwa from the Ministry of Education, Science, and Technology of Japan (12558002 and 15500439).
