To the Editor:
Today, supplemental oxygen is widely used by mountaineers on the world's highest peaks. Up to the end of 2003, only 121 out of 1909 ascents of Mt. Everest had been made without supplemental oxygen. Although mountaineers rely upon a wide range of devices to deliver oxygen, most receive their supply from a small number of lightweight cylinders designed specifically for mountaineering purposes. However, our recent experience on the 2007 Caudwell Xtreme Everest Expedition has identified an important limitation in their design.
Cylinders used during the final stages of an 8000-m or higher climb are filled with oxygen at a given temperature. The 4-L Poisk BK(4) cylinder (St. Petersburg, Russia), commonly used on Mt. Everest, is filled with oxygen to a pressure of 280 bars at 20°C. However, at a high altitude, the ambient temperature is often considerably colder. Members of the first expedition to climb Mt. Everest successfully in 1953 recorded temperatures as low as −30°C on the South Col (7930 m).
1
The difference between this and the temperature at which the cylinder is filled has 2 important practical implications for the mountaineer. Although the number of molecules of oxygen inside the cylinder does not change with a fall in temperature, a reduction in the number of collisions between each molecule per second does occur, resulting in a lower gauge pressure. Inside a Poisk cylinder, a fall in temperature from 20°C to, say, −20°C corresponds with a reduction in gauge pressure from 280 to approximately 240 bars (15%), if a linear relationship is assumed over this temperature range. From our experience, a number of mountaineers on Mt. Everest have rejected cylinders on the mistaken assumption that oxygen has leaked, when in fact the number of oxygen molecules has remained constant and only the ambient temperature has fallen. As the reducing valves used in mountaineering systems are not temperature compensated, a given flow rate in terms of volume per second will deliver more oxygen molecules at a lower temperature. As this increase is inversely proportional to the fall in gauge pressure, a fall from 20°C to −20°C results in the oxygen molecules being released 15% faster. In 2007, members of the Caudwell Xtreme Everest Expedition climbed the South East Ridge of the mountain using flow rates of between 1.5 and 3 l/Minute. Using a flow rate of 2 l/Minute as an example, the duration of a 4-L Poisk cylinder will be 560 minutes at 20°C (280 × 4 L/2 l/Minute) and only 480 minutes (240 × 4 L/2 l/Minute) at −20°C. Typically, mountaineers use up to 3 cylinders to complete the round trip from the South Col to the summit of Mt. Everest. Therefore, over the course of this journey, it is possible to overestimate the duration of the 3 cylinders by 240 minutes when 2 l/minute of oxygen is used if the difference between ambient (−20°C) and filled temperature (20°C) is not taken into consideration.
An understanding of the relationship between pressure, volume, and temperature leads to 2 potential solutions to the issues highlighted above. First, in order to check for leaks, cylinders can either be tested by measuring both gauge pressure and ambient temperature and checking these values against a simple chart (Figure) or weighing them with accurate lightweight scales instead. Second, any calculations that are made to predict the lifespan of a cylinder in cold conditions must take into account the change in temperature and the increase in the number of oxygen molecules that escape for any given flow rate. As a rule of thumb, it seems reasonable to underestimate the contents of the cylinder by at least 10% in order to avoid a situation where oxygen supplies run out early. By adopting these practical measures, it should be possible to take full advantage of the benefits supplemental oxygen clearly provide at altitude and ensure a safe return from the “death zone.”
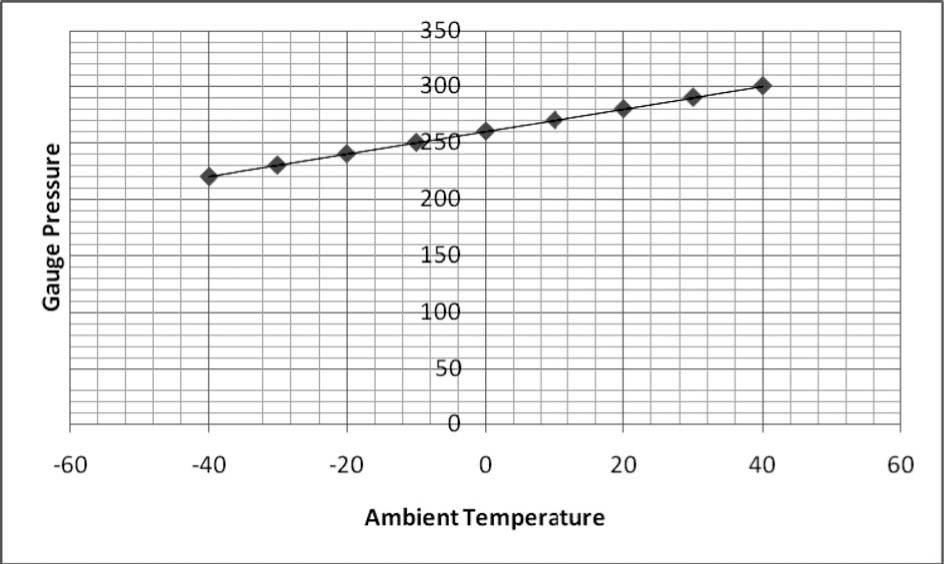
The changes seen in gauge pressure over a range of different ambient temperatures (−40°C to 40°C) in oxygen cylinders filled to 280 bars at 20°C.
