Abstract

To the Editor:
Intermittent hypoxia (IH), or episodic exposure to hypoxia with intervening normoxic intervals, occurs under a variety of conditions that individuals may encounter during recreational activities, athletic training, or work. Individuals with cardiopulmonary pathology also experience hypoxia with varying exposure profiles. For example, patients with obstructive sleep apnea-hypopnea (OSAH) have brief periods of hypoxia (eg, seconds) in the context of a cyclic pattern of desaturation-resaturation during sleep. 1 Those with chronic obstructive pulmonary disease (COPD) may experience intermittent intervals of oxyhemoglobin desaturation, each lasting several minutes throughout a night's sleep due to reduced ventilation usually accompanying rapid eye movement sleep2,3 and in conjunction with perturbed pulmonary mechanics.
The intent of this letter is to describe the method of hypoxic gas delivery used in a study that examined hemodynamic and molecular response, in normal participants, to intermittent and continuous periods of lowered blood oxygenation. A variety of techniques have been used in an attempt to recreate IH exposure profiles (in healthy participants) similar to those seen in patients with cardiopulmonary disease to study relevant physiological and biological factors. Examples of IH paradigms utilized in healthy participants include inhalation of asphyxic gas (hypercarbic and low fractional inspired oxygen tension, FIO2) and induction of hypoxia by voluntary breath-holding. 4 –6
Normobaric hypoxic gas mixtures can also be delivered via nitrogen dilution (eg, where zeolite crystals are used as a “molecular sieve”) via devices that are commercially available worldwide. Such devices have been widely and safely used for athletic training and preacclimatization applications during sleep and exercise.7,8
The same commercially available normobaric hypoxic gas delivery system used for athletic training can be used to induce oxyhemoglobin desaturation in healthy humans during physiological experimentation, a clinically relevant utilization of this technology. In a study we conducted at the University of Pittsburgh, conditions of IH and continuous hypoxia (CH) were created, during which hemodynamic responses were recorded in healthy participants. We compared these responses obtained during comparable total durations of hypoxic exposure provided with IH and CH profiles on each of 3 consecutive days using a crossover design (Figure 1). 9 We also examined the relationship between hemodynamic responses to IH/ CH and inducible nitric oxide synthase mRNA expression during this study. The decision to use comparable cumulative duration of hypoxic exposure under each of the IH and CH profiles resulted from difficulty interpreting data from prior studies that used dissimilar durations during each condition.
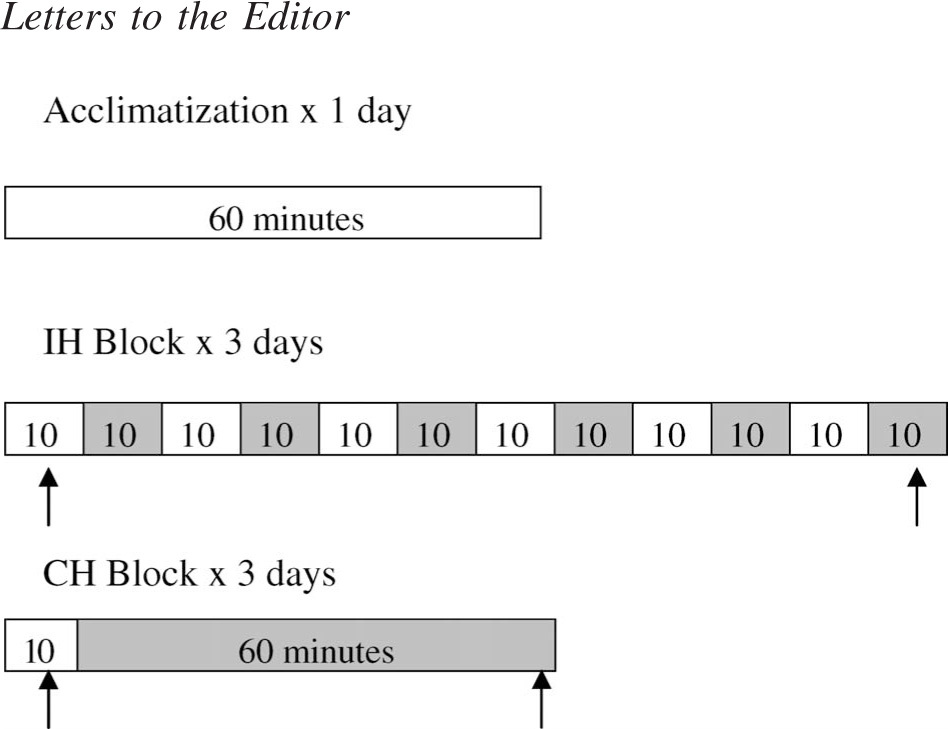
Scheme of normoxic (□) and hypoxic (▪) exposures for the habituation trial (60 minutes room air), intermittent hypoxia (IH) session (6 hypoxic [FIO2 approximately 0.135] exposures for 10 minutes each), and continuous hypoxia (CH) session (one 60-minute hypoxic [FIO2 approximately 0.135] exposure). Arrows indicate data collection periods during last 5 minutes of normoxia prior to the day's initial hypoxia exposure, as well as during the last 5 minutes of daily hypoxia exposure. Reprinted with permission from Rodway et al. 9
The equipment used for this study provided a means to assure that, regardless of exposure profile, participant experienced comparable total duration (60 minutes) and intensity of hypoxic exposure, supported by the arterial blood oxygen saturation (SpO2) similarity during the last 5 minutes of all trials (Figure 2). The target SpO2 value was 85% for the entire exposure time. Consequently, we were able to minimize the impact of the potentially confounding factors of varying durations and intensity of total exposure when comparing participant response during IH and CH. Our methodology contrasts with that used by Tamisier et al 6 who compared the hemodynamic response to IH and CH in humans using a crossover design in which CH was achieved by adjusting the inhaled gas mixture using a valved system with nitrogen added as necessary to maintain the SpO2 at approximately 85%. To create IH, the inhaled gas source was a bag filled with 100% nitrogen with the participant alternately breathing from the bag and room air to produce a desaturation frequency of 30 to 40 dips in SpO2 per hour over a 2-hour period. This method produced SpO2 values that differed significantly during IH and CH (92.00 ± 1.08% vs 85.06 ± 0.51%, respectively; P < .05). A notable limitation of the system we employed was that while it permitted exposure to comparable durations and intensities of desaturation, it did not lend itself to the higher frequency of desaturation (of the nature utilized by Tamisier et al 6 ) during IH exposure.
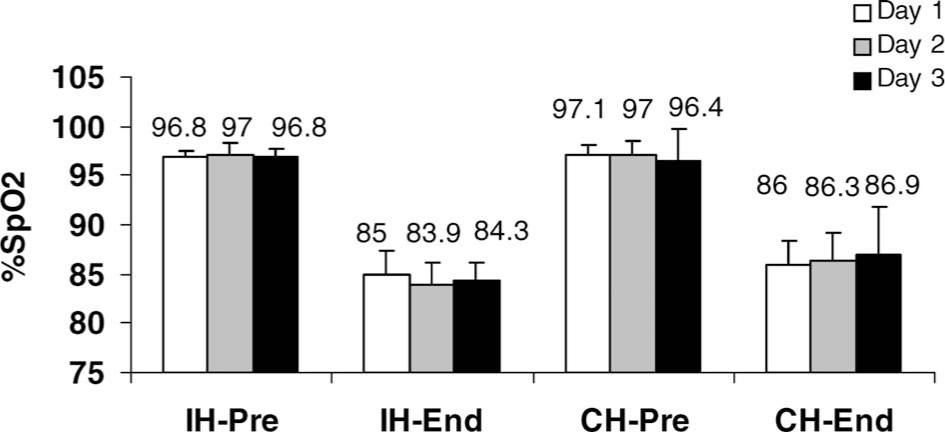
SpO2: Mean of pre-exposure and last 5 minutes of exposure by hypoxia profile and day (IH indicates intermittent hypoxia; CH, continuous hypoxia). Values are means ± SE.
The device used in our study, the HYP-100 hypoxic gas generator (Hypoxico, Inc., New York, NY), did not generate an exceptionally strong airflow. As a result, we found that a reservoir with a limited capacity was inadequate for meeting the ventilatory demand of several participants. This problem was resolved by fitting a 15-liter Douglas-type collection bag to the airline running from the gas generator. With this modification, a relatively large reservoir of air with a low fraction of inspired oxygen (FIO2), and/or medical grade air (MGA), was available to the participants whenever the face mask was worn. On two separate occasions, we found that a 20-liter collection bag was required to accommodate the ventilatory response to the hypoxia. We therefore recommend using an approximately 20-liter collection bag in future studies.
Pedlar et al 7 reported that the HYP-100 hypoxic gas generator (Hypoxico, Inc., New York, NY) is designed to deliver 100 L/min−1 of hypoxic gas. During an investigation examining acute sleep responses in a normobaric hypoxic tent, 10 concerns were noted regarding the ability of the gas-generating unit to provide adequate air flow in order to flush carbon dioxide from the tent. In an attempt to accurately quantify the gas generator flow over time, the investigators measured the output volume from 8 HYP-100 hypoxic gas-generating units (Hypoxico, Inc.) over an 8-hour period. They found that while the consistency of the units over time was satisfactory, the output volume was, on average, 30.4% less than the 100 L/min−1 advertised by the manufacturer. Pedlar and colleagues concluded that this factor should be taken into consideration when calculating the output volume required from any given gas generator. 7
Our hypoxic gas generator (Hypoxico, Inc.) was fitted with an adapter device supplied by the manufacturer that allowed for an FIO2 of approximately 0.135 (equivalent altitude, approximately 3760 m). Therefore, it was often necessary to bleed MGA into the reservoir bag in order to adjust the SpO2 to the desired percentage. This method of titration proved to be a quick, effective way of controlling participant's SpO2. Although an oxygen analyzer was not utilized during the procedure, it would have allowed verification of the FIO2 produced by the generator.
There are at present at least 2 other manufacturers of normobaric hypoxic gas generators that market their devices for purposes similar to that of the machine used in this study (Colorado Altitude Training, Boulder, CO; GO2Altitude, Melbourne, Australia). The manufacturers of these generators offer an increasingly sophisticated array of options and accessories for their machines.
Although the intent of the protocol in our study was not to explicitly mimic the type of hypoxic exposure characteristics of pulmonary disorders, our methodology may provide a platform for future studies that might more closely reflect the pathophysiology associated with human diseases. With the advent of escalating levels of technological sophistication of hypoxic gas generators, the ease in which the frequency, duration, and intensity of normobaric hypoxia can be manipulated for investigational purposes has become increasingly apparent. This will hopefully stimulate the development of further, more complex studies that are reliant (methodologically) on gas generators to closely simulate the pattern of hypoxia found in various disease entities.
Footnotes
Acknowledgments
This work was supported primarily by American Lung Association Lung Health Award LH-12-N, NRSA Fellowship F31 NR 08458-01, Sigma Theta Tau International (Eta Chapter), and the Leslie A. Hoffman Acute Care Endowed Fund (University of Pittsburgh).
Funding
Additional support was also obtained from the NIH (R01 AG023977, Dr Sanders; R01 HL 70301, Dr Sanders; R01-HL60234, R01-AI42365, R01-HL55330, Dr Choi); and the American Heart Association (grant, Dr Ryter).
