Abstract
Nanofiber substrates have been used for various biomedical applications, including tissue regeneration, drug delivery, and in-vitro cell culture. However, despite the high volume of studies in this field, current clinical applications remain minimal. Innovations for their applications continuously generate exciting prospects. In this review, we discuss some of these novel innovations and identify critical factors to consider before their adoption for biomedical applications.
Introduction
The use of nanofiber (NF) materials1-6 for bioengineering has generated some exciting results and achievements, particularly with the development of artificial organs over the past 30 years. They have numerous potentials in biomedical applications, for instance, in-situ drug and toxicity studies, tissue grafts, and organ replacement.
To date, numerous studies have been conducted on innovative techniques for tissue engineering using various substrates as bio-scaffolds. However, current clinical applications using bio-artificial organs remains minimal.2,7 This could be attributed to a number of reasons, from the complexity of tissue and organs to the intricate cellular arrangement in organ systems. In addition to this, the low rate of translational applications in even simpler organ systems, such as intestines, pancreas, and bone, also indicates the difficulty of biomedical applications of bio-scaffolds.
One of the promising tools in tissue engineering has been adopting nanofibrous (NF) substrates as scaffolds for cellular attachment and tissue replacement. These applications have been investigated,1,8,9 and have demonstrated great potential to be adopted for numerous studies, including tissue engineering. 10
The popularity of NFs are due to a number of advantages for use as a bio-artificial skeleton for tissues and organs. Firstly, a majority of them do not trigger a profound immune response to relevant sites post-transplant. 11 Meanwhile, NF scaffolds can be designed to be degraded in some instances, leaving the transplanted cells to form a natural biological matrix. 12 This is particularly important for transplants with aesthetic considerations or with high possibility of immunological responses. Another significant advantage is that NF-based scaffolds, especially for those created by electro-spinning (Fig. 1), have uniform space consistency, which enables the seeded cells to be dispersed on the scaffolding material uniformly. This is especially essential in designing tissues and organs with high uniform consistency, such as the intestines or pancreas. 13 Furthermore, NF materials could be combined with numerous beneficial substances such as growth factors, which could promote the differentiation of stem cells, have anti-inflammatory activity, and release immune-suppressive agents to reduce foreign body reaction. They could also be combined with therapeutic agents such as antitumor agents 14 and antiviral agents. 15 An additional feature that could enhance the adaptation of nanofibers for organ systems is the property of shape memory. This was recently highlighted in a review by Yao et al., 16 in which this shape memory property was greatly enhanced via use of NF for tissue transplants.
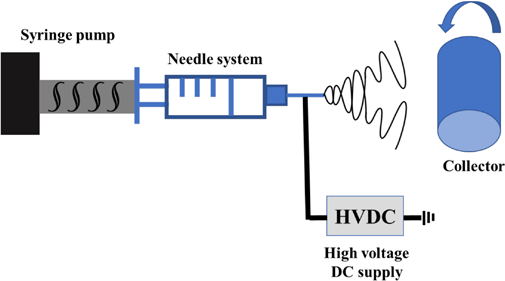
Single-needle electrospinning system with rotating drum as collector.
Numerous reviews have been written over the years about tissue engineering. A number of them focused on various aspects of electrospun NFs, including optimization of the process and niche concentrations for various primary studies.12,17,18 This review article is different, as it portrays some crucial considerations for nanofiber-based scaffolds to be applied for tissue engineering. Due to the current poor translational application, more reviews are necessary to provide a continuous conversation on the use of NF substrate for biomedical engineering. We also discuss some current innovations that could be relevant in future applications of nanofibers.
NF Substrate for Tissue Engineering
Characteristics of Nanofiber Polymers for Tissue Engineering
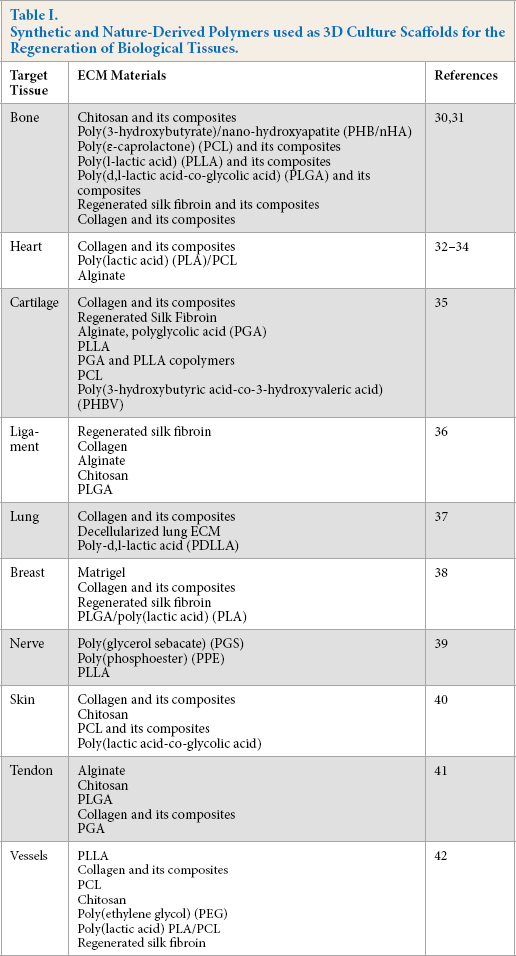
Neurons within the central nervous system have unique and various structural components/arrangements, which enable them to have extraordinary functions. Properties of scaffold materials for tissue engineering have been previously reviewed. 19 NF substrates used as a scaffold should possess some unique characteristics, which include biocompatibility, uniform surface area, and biodegradability. Meanwhile, the scaffold should be degradable over time and leave behind the cells with the extracellular matrix. It should also possess a vast system of interconnectivity that can encourage cell differentiation and attachment, as well as nutrient exchange, and a waste removal system via competent pre/post-formed vessels. To ensure proper vascularization, pore connectivity and spacing should not be greater than 200 micrometers. 19 Lastly, it should possess suitable functional characteristics to the target tissue for transplantation. This is particularly important for tissues within the central nervous system. 20 Bowlin et al. 21 reviewed and outlined various synthetic and nature-derived polymers used as 3D culture scaffolds that were successfully used for the regeneration of biological tissues. It suggested that nano-additives, particularly aliphatic polycarbonates (APCs), could be used to modify the scaffolds to enhance biocompatibility and control degradation when the scaffolds were embedded in the tissues. 22 The commonly-used biocompatible materials for tissue engineering include chitosan and its composites, poly(3-hydroxybutyrate)/nano-hydroxyapatite (PHB/nHA), 23 poly(∊-caprolactone) (PCL) and its composites, poly(L-lactic acid) (PLLA) and its composites, poly(D,L-lactic acid-co-glycolic acid), (PLGA) and its derivatives, regenerated silk fibroin and its derivatives, and collagen and its derivatives, as well as composited materials (containing more than one polymer system).21,24 The detailed polymer list for bio-scaffold designing using electrospinning is summarized in Table I.
Synthetic and Nature-Derived Polymers used as 3D Culture Scaffolds for the Regeneration of Biological Tissues.
The morphology of electrospun fibers can be adjusted according to the tissue/organ types. In Fig. 2, randomly distributed P(LLA-CL) nanofiber patches have been used for cell culture, where cells were randomly oriented on the nanofiber surface. The macro and micro structures of fiber substrates are dependent on material types, processing parameters, and the regulation of interrelated variables (i.e., spinneret design, electric field intensity, auxiliary electric/ magnetic field, applied voltage, flow rate, collection distance, solution conductivity, and solution viscosity). 25 Fiber morphology needs to be controlled26,27 especially for bone and tissue scaffolds. This is because pore size and biomechani-cal properties of bone tissue play a role in ensuring bone rigidity and adequate mineral contents. 28 In some tissue types, such as mammary gland tissue and liver tissues, there is a need to form a complex ductal system. The NF-based scaffolds should be designed to address organs with complex ductal systems, 29 because previous studies have shown this ductal system can self-proliferate breast tissue grown on a scaffold. 21 Lastly, the proliferation of a vascular structure in tissue structures for mass transfer of nutrients are an important consideration in the development of tissue engineering.
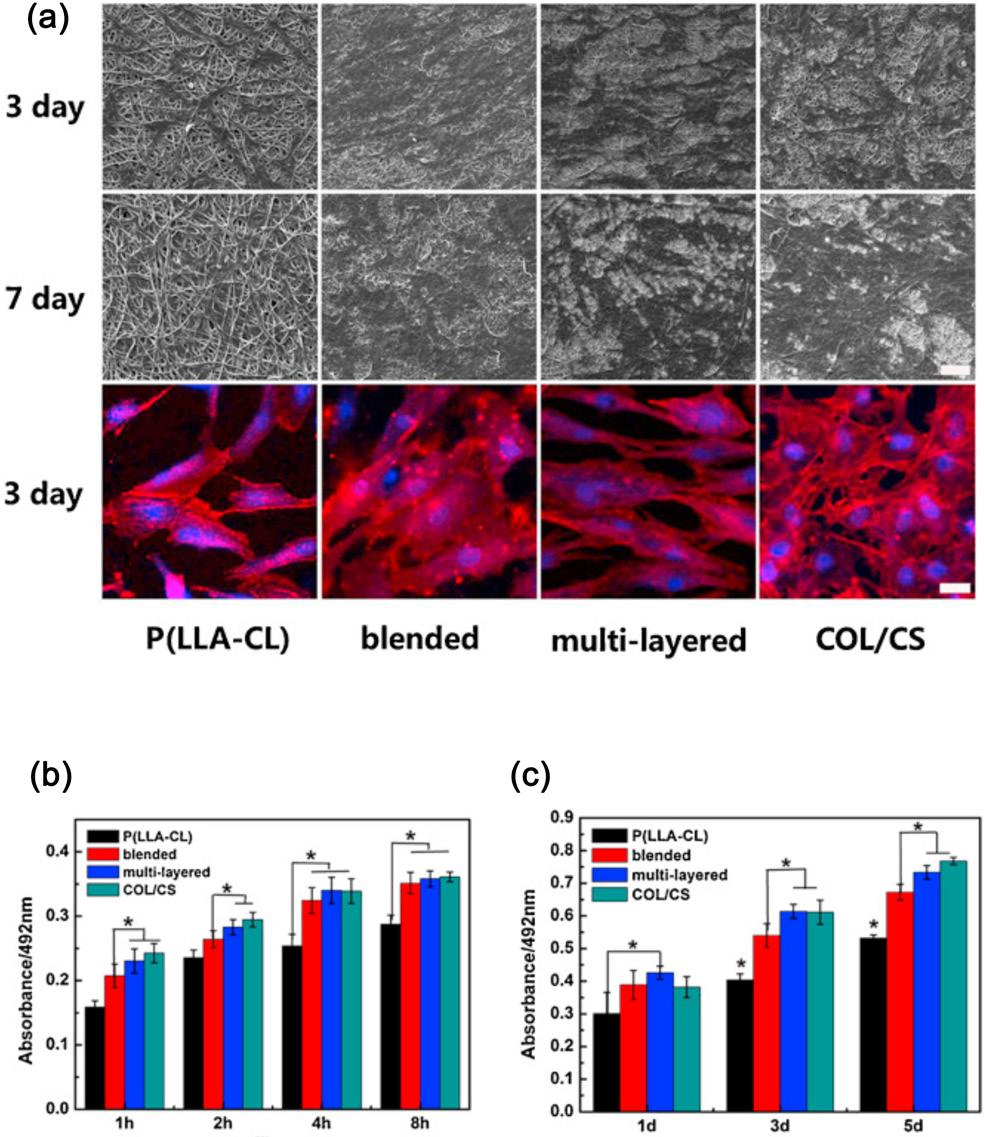
(a) Scanning electron micrographs of human vascular smooth muscle cells (hSMCs) on different scaffolds after culturing for 3 and 7 days, and confocal images of hSMCs on different scaffolds after culturing for 3 days; adhesion and proliferation of hSMCs after culturing in 8 h (b) and 5 days (c); bar = 25 μm. P(LLA-CL), standing for poly(L-lactide-co-epsilon-caprolactone), is the raw polymer for the nanofiber scaffold that hSMCs are to be seeded on, and COL/CS stands for collagen/chitosan (9:1, v:v). IMAGE CREDIT: Elsevier. 26
Suitability of Electrospun Nanofibers for Cell Scaffolding
The importance of scaffold porosity in tissue engineering includes the provision of anchorage points for cell attachment, as well as facilitation of the mass transfer of nutrients between the scaffold and the environment.43,44 Scaffolds act as a 3D template for initial cell attachment before the formation of an extra cellular matrix. 45 Therefore, the porosity of a scaffold, regardless of its application in cell or tissue culture, requires careful selection. Also, the size of the pores in the scaffold need to be considered. If the pores are too small, they could hinder communication between cells. If they are too large, the scaffold no longer mimics the conditions in-vivo and can disrupt cellular attachment. 46 With modification of the amount of chemical blowing agent (BA) incorporated in PCL nanofiber webs, an increased pore size and porosity, from 10 to 130 µm and from 2% to 8%, respectively, were noticed from Kim et al.'s research. 43 They also found that the electrospun mat, with increased porosity and pore size of 50-70 µm, contributed to high human dermal fibroblast cell adhesion. Similarly, Frey-man et al. 46 summarized the importance of a porous scaffold, with a porosity greater than 95% and a pore size of 20-120 µm in tissue engineering. From these research results, a two times greater average pore size than the cell diameter would be good for cell attachment.
The biomechanical properties of scaffolds are dependent on the tissue type to be made. Structural concerns include rigidity versus flexibility, as well as the degradation rate of the scaffolds. Burst pressure has been a considerable factor in the failure of tissue engineered vascular grafts (TEVG). Biodegradable viscoelastic polymers that can be reabsorbed in-vivo were proposed due to their ability to survive contraction from muscle forming tissues. 47
Electrospinning has provided a unique advantage, because the structure, chemical, mechanical stability, and functionality, as well as other properties of the electrospun products, can be modified to specific applications. One of those properties includes the pore size. 48 This is because the proliferation of cells on the scaffold could be related to the pore size, which can be modulated by control of the electrospinning process. This is evidenced in a study by Maghdouri-White et al., 37 who evaluated the effects of pore size and fiber diameter on the surface area and cellular viability. They observed that optimal cellular infiltration and viability were 7% and 12% for silk scaffolds, with air flow at 100 and 400 KPa, respectively, for 14 days. This was due to the large average pore size of 13.4 and 26.03 µm, respectively. Meanwhile, the use of 7% silk fibroin electrospun at 100 kPa and 12% silk fibroin (SF) at 400 kPa showed some promise for adaptation to other cell types.
A second important factor is the degradation time of the scaffolds. This is important because of the time necessary for the cells to develop their own extracellular matrix (ECM). It has been advocated that polymers with tunable degradabil-ity characteristics can be used as grafts in tissue scaffolds. In 2016, a tissue-engineered vascular graft was developed using a modified amino acid based polyester urea (PEUs) with a tunable degradation rate and non-toxic byproducts. 49 They were able to successfully implant this graft into a mouse model for one year without failure of the graft. Ultrasound and micro-computed tomography (CT) studies of the graft showed that the graft lumen was patent and functional. Histological observations revealed CD31 (representative of endothelial cells) and smooth muscle cells after 12 months post-implant.
Nanofibers from synthetic and natural biopolymers show the most promise, with a long term duration of about 44 days using poly(∊-caprolactone), poly(ethylene terephthalate), and polyglycolic acid scaffolds. A review by Abbott et al. 50 showed that silk, as a biomaterial, can support long term 3D tissue cultures for six months. This could be attributed to its bio-compatibility and a modifiable degradation rate from a few weeks to years. However, a disadvantage of silk is that it does not have proper binding sites for cell-scaffold attachment. However, this can be countered by the addition of collagen or other polymers to the silk scaffolds. He also illustrated previous studies using silk in various tissues such as adipose, brain, intestinal, and bone.
As discussed previously, NF applications possess great potential for tissues such as bone, blood vessels, and simple tissue systems. Numerous in-vivo studies have been conducted using scaffolds, which have been reviewed extensively.7,14,51 A study conducted by Shin'oka et al. successfully extracted and differentiated mesenchymal stem cells from rat bone marrow and seeded them on a NF-based scaffold. The constructs were cultured in a bioreactor before transfer to the rat omentum for a month. After incubation in-vivo, the constructs were harvested, and their histological morphology revealed miner-alization. 52 Kurosawa et al. were able to conduct a clinical trial in which tissue-engineered grafts were seeded with autologous bone marrow cells using polyglycolic acid scaffolds. 52 After a six-year follow-up, the grafts were still in good condition with native ECM growth complementing the scaffold.
ECM Substitution with Electrospun Nanofibers
The ECM provides biochemical and structural support to cells in tissues; they also regulate intercellular communica-tion. 53 ECM formation is essential for processes like wound healing, fibrosis, and cell growth. ECMs are finely organized, with nanoscale structures, where NFs exhibit similar structure properties. 54 In addition, the ECM has important implications in cell migration, gene expression, and differentiation due to its stiffness, elasticity, and secretory capability of various cytokines that are vital to cellular differentiation and communication.55,56
The ECM is composed of carbohydrates, polysaccharides, proteins, and other substances. NF membranes have a high surface-to-volume ratio, high porosity, and adjustable pore size; the small diameter can provide an excellent substitute for the ECM. The ECM greatly influences the survival, proliferation, and differentiation of stem cells. 57 NFs, which provide structural support similar to the ECM, can be functionalized to provide similar functions as the ECM, including differentiation of human embryonic stem cells (hESCs). 58 This further indicated the role of the extracellular environment in neural lineage specification.
Cell Growth on Scaffolds
Cells form an essential factor for tissue engineering, along with the NF matrix and growth factors. They are the singular units of tissue and organ systems. 59 Depending on the tissue type to be regenerated, various cell sources can be selected for tissue engineering applications. Some of these include autologous sources, where the cells are derived from the patient; allogenic sources, where cells are derived from a different human source other than the receiving patient; and xenogenic cells from animal origin. Both allogenic and xenogenic sources have not been positively adapted for clinical applications due to immunogenic reactions and disease transmission from animal sources. Autologous cell sources remain the most viable method of generating tissues/organs for clinical adaptation. However, technological innovations to enable the development of an organ from growing cells for organ replacement are still in its infancy.
Viability of Cells on Scaffolds
Optimization of cells to scaffolds could either be for in-vitro applications—including drug screening, toxicity testing, and disease modelling—or for in-vivo transplantation. Long term viability of cells seeded on scaffolds in-vitro are essential factors that determine the potential of mass transfer of nutrients and excretion of waste products. They are important in providing optimal conditions for growing cells before or after cells are seeded on the scaffold. In addition to provision of nutrients and release of waste products from the cells, they control the conditions of the cells, such as oxygen content, temperature, and osmolality. Cell bioreactors are special culture vessels that can control in-vitro cell variables, such as mass transfer of nutrients and mobility in culture systems. They can provide the biochemical and physical regulatory signals required for cells to proliferate, differentiate, and produce. 60 The use of bioreactors to develop a cell culture version of a physiologically-based pharmacokinetic (Pk-PB) model, with various cell types in bioreactors interconnected to mimic a biological organism, has also been proposed.91 This is of considerable importance in pharmacological and toxicological studies. Bioreactors include rotating wall vessels, direct perfusion systems, and spinner flasks. A detailed explanation of these bioreactor types can be found in a previous review.61,62
Bioreactors have been applied that demonstrated better results compared to static systems. 63 However, for large volume in-vitro tissue engineered scaffolds, challenges include oversupply of nutrients at the periphery, and poor supply of nutrients at the core or or at depths greater than 200 micrometers from the scaffold surface. Ball et al. 64 was able to develop a 3D vascular network to address this problem. It was made of an inert biopolymer, EShell 300 (Envision Tec), which is used for 3D printing. It was subsequently implanted into a hydrogel-based scaffold with a porous structure before seeding with mesenchymal stem cells. This structure was subsequently attached to a specialized tubular perfusion system (TPS) bioreactor media supply. The TPS model is a perfusion bioreactor that utilizes a bottom-up approach, which helps ensure that all cells have adequate nutrient supply and waste removal. These results indicated great promise and high potential of using bioreactors to seed bio-scaffolds.
Functionalization of Scaffolds for Tissue Engineering Applications
Growth factors have gained considerable attention in promoting sustained development of cells on scaffolds. The extracellular matrix (ECM) not only functions as a mechanical support, but also plays an active role in modulation and development of cells through various growth factors and locally-acting cytokines. Growth factor efficacy in regeneration consists of a symphony of various regulatory activities in the regenerative cycle. 65 The application of growth factors to scaffolds was successfully conducted through various techniques. One way was the coating of the scaffold polymer matrix with the growth factor, which can improve cell differentiation. A second method was the addition of the growth factor into the NF substrate solution, which ensured that the growth factor is incorporated into the scaffold. Growth factors were released with the NF substrate degradation and ECM replacement. 66 NFs can also be modified with cell chemo-attractants to enhance recruitment of stem cells to sites where injury has occurred. 67 Addition of growth factors to nanofiber polymers were met with some challenges including high cost, poor availability to cells, and high rate of metabolism. 68
3D Cell Culture using Nanofibers for Drug and Toxicity Testing
It is imperative to reduce the large numbers of animals used for in-vivo studies with more innovative approaches. The current trend is to use in-vitro cell-based studies in monolay-er cultures. However, these studies exhibit a poor correlative output with in-vivo data. A new method using the same in-vitro cell cultures can be applied using 3D cultures. They have been widely acknowledged as a more efficacious replacement of in-vivo data compared to 2D cultures. This is especially true for target organ toxicity studies such as liver and kidney, and those involving immune and nervous systems.
Numerous studies showed the potential of using a bio-artificial extracellular matrix as a 3D environment for cell growth.69,70 However, the majority of in-vitro studies were conducted using cell culture well plates. Using 2D monolayer culture models had some advantages, particularly the availability of well-defined and well-established protocols.71,72 However, the major disadvantage of 2D cell culture is that it does not have cell-matrix interactions. This is because in a complex biological organism, cells not only connect to each other, but also to the extracellular matrix. 73 The culturing of adherent cells on a fat surface, as is the case in 2D monolayers, subjects the cells to severe geometric and mechanical constraints due to the adherence of the cell surface to only one layer, which is not the ideal scenario in biological organisms. These problems contribute to the poor extrapolative capability of in-vitro studies.74-76
Culturing cells as monolayers on fat surfaces may force them to make some morphological adjustments. It may also necessitate changes in oxygen, nutrient, and ECM interactions. As they have been isolated from a complex 3D network initially, this may result in alteration of cellular metabolism and gene expression patterns in the 2D cell culture.77,78 It was also observed that not all epithelial cell types have the ability to adhere and grow on artificial substrates. 72 The signal transduction patterns of integrins and the cell-matrix adhesions were found to be strongly influenced by the fat, rigid geometry of tissue culture dishes. 79
Also, the absence of a matrix-like environment translates to poor growth and differentiation of cells. 80 3D cell culture practices are known to mimic in-vivo conditions compared to routine well plate culture. The gene expression patterns/ profiles and other biological activities of cells grown in 3D cell culture are close to what happens in-vivo. 73 In the case of modeling tumors, 3D cell cultures consist of a variety of phenotypes, such as proliferating, non-proliferating, and necrotic cells. The presence of such cellular heterogeneity in 3D cell culture models enables their use in drug testing/ drug development for tumors. 3D cell culture models also facilitate the culture of multiple cell models (co-culturing of cells). Such a model assists in studying the interactions between various cells, such as the epithelial and stromal cells. 72 With regards to drug testing, skin cells tested in 3D culture were found to be more resistant to cytotoxic agents like hydrogen peroxide and silver, compared to those grown in 2D cultures. 75 Similarly, breast cancer cells (MCF-7) were found to be more resistant to tamoxifen (a drug used in breast cancer therapy), when grown in 3D cultures. 81 This suggests that the use of 2D cultures for drug testing overestimates the effect of those drugs, often leading to false conclusions.
Furthermore, there is a need for studying a multitude of cell types and the effects of various cellular interactive processes on the contaminant studied. While this could be challenging in 2D studies, it is quite feasible in a 3D matrix. 79
Currently, many techniques can be applied to produce 3D cultures. The first is the formation of spheroids through the aggregation of cell types to form small clusters on their own without any supporting intercellular structures. 74 The spheroids can form on a matrix-free ECM. The second method is to use hydrogels to suspend cells; these hydrogels are made of natural and/or synthetic polymers. The third method uses solid scaffolds from either electrospinning or from melt blown NFs. Adaptation of these kinds of models could ensure a result more representative of native human tissues as compared to animal models.
Considerations for Adaptation of 3D Cell Cultures
New possibilities in terms of in-vitro 3D cultures have indeed become mainstream; the formation of new testing methods as well as end-point evaluations have become a necessity. The first consideration is how to optimize nutrient transfer and mass transfer of waste products in the medium. One way to approach this is to replace the static medium with a pre-programmed mass transfer bioreactor in a unidirectional flow-through format with either a gravitational pull or a pump style mode. This could require a redesign of traditional well plate static systems to a flow-through mechanism, which is more realistic compared to the standard well plate in-vitro approaches.
A second consideration is the assessment of toxicity endpoints, which could gravitate towards a more tissue pathology-like evaluation. This can also include assessment of biomarkers as well as morphological damage to the cells growing in the extracellular matrix. Traditional cytotoxicity assays, such as MTT, could be used after the cells are mobilized. Molecular evaluation techniques, such as proteomics, western blot, and RNA expression, can provide more realistic in-vivo data compared to results from 2D monolayer cell cultures.
Pharmaco-Terapeutic Applications of Electrospun Nanofibers
NFs have been promoted for numerous pharmaco-therapeutic applications due to their customizable physicochemical characteristics, ability to combine compounds of interest in a physical mixture, and biodegradability (which can also be modified). 82 These properties make nanomaterials suitable candidates as vehicles for drug delivery and gene therapy. Potential pharma-co-therapeutic applications of NFs were illustrated in a review by Sill et al., 17 who discussed the numerous advantages of using electrospun NFs for drug delivery and their potential advantages in terms of drug loading. The advantages were dependent on the type of NF polymer used for the drug loading and the method of production and incorporation. 83
In addition, control release techniques are a possibility and long term therapeutic agents, such as antimicrobials or chemotherapeutic agents, could be used in an NF-based drug delivery system.84,85 This is especially important for surgical procedures and after excision of a malignant tumor. Impregnation of the therapeutic agent in the fiber ensures a slow release with a modifiable release rate. 86 A second advantage of this method is that the medication-loaded fiber could be applied at the site of action, which reduces the dose amount that would be administered systematically due to the elimination of metabolic and excretory mechanisms. 87 A third advantage is it could reduce the frequency of medication administration, especially for long term medications. This approach was evident in a study by Gagandeep et al., 88 which demonstrated a biodegradable polymeric NF as a drug delivery system for timol maleate and dorzolamide hydrochloride. The results showed slow release lasting for 72 h due to the slow release of the medication along with the scaffold degradation. Another study further confirmed this result by using a nano-carrier, which exhibited extended release for a period of 120 days, 89 as shown in Fig. 3. Furthermore, the pharma-cotherapeutic applications of NF scaffolds can be used as a prophylactic agent. In a study by Grooms et al., 15 a griffthsin-based NF scaffold was effective in preventing infection with the HIV-1 virus. A nanofiber-based controlled release system could also be achieved by combining drugs with inorganic nanoparticles. As shown in Fig. 4, PVA/VB2/TiO2 composite nanofiber webs were designed, 2 which showed controlled release potential for as long as ten days.
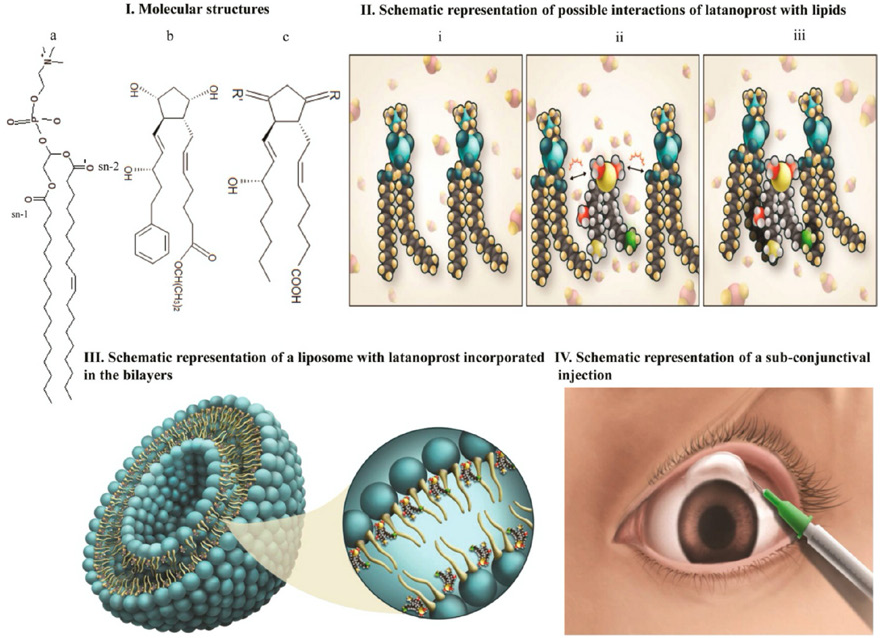
Latanoprost incorporated nanocarrier with sustained drug delivery properties. IMAGE CREDIT: American Chemical Society. 89

Optical micrographs of PVA/VB2/TiO2 nanofiber: (a) PVA nanofibers; PVA/VB2/TiO2 nanofiber with TiO2:VB2 ratio of 18:1; PVA/VB2/TiO2 nanofiber with TiO2:VB2 ratio of 9:1; PVA/VB2/TiO2 nanofiber with TiO2:VB2 ratio of 5:1; PVA/VB2/TiO2 nanofiber with TiO2:VB2 ratio of 25:1; PVA/VB2/TiO2 nanofiber with TiO2:VB2 ratio of 1:1; TiO2/VB2 nanoparticles in DI IMAGE CREDIT: American Chemical Society. 2
A second potential mode for pharmacotherapeutic applications of NF substrate is the addition of biological molecules such as DNA or siRNA, which can act as effective modulators of gene expression, particularly for small interfering RNAs. Nguyen et al. 90 regenerated axons using NF scaffolds in the form of a hydrogel for delivery of proteins and nucleic acid therapeutics using small non-coding RNAs for nerve injury treatment. This is one of the current potential applications of the electrospinning process.
Conclusions
The application of NF continues to expand, which can be attributed to the high volume of research output, especially through the process of electrospinning. This is in spite of the slow rate of end-user adoption of these innovative processes. With further development, the conversation on the adoption of the electrospinning process needs to continue to remove the bottlenecks and improve NF's applications.
