Abstract
Mercerization, the treatment of cotton with concentrated sodium hydroxide under tension, is performed to enhance absorption, luster, strength and dimensional stability. After mercerization, the unreacted sodium hydroxide is neutralized with acids. Neutralization with polycarboxylic acids (PCAs) is likely not only to crosslink the cellulose molecules, but to also enhance other functional attributes like crease recovery behavior, fame retardancy, and soil release properties. In this research, four PCAs with different functional groups were used for fabric neutralization to merge mercerization and poly-functional finishing into one continuous process. The PCA treated fabrics were then assessed for their performance. All four PCAs were effective in improving crease recovery and soil release properties, and reducing the flammability, of the treated cotton fabric.
Keywords
Introduction
Mercerization is a common practice in wet processing of cotton materials to modify the microstructure, morphology, and conformation of the cellulose chains. It improves sorption, tensile strength, extensibility, luster, roundness of the fibers, and also affects the handle of the resulting fabrics. In this process, fabrics are treated with a concentrated solution of sodium hydroxide (caustic soda) of 15-20% strength at a relatively low temperature of 16-32 °C for 45-60 s under tension. The magnitude of the changes that occur depends on many variables, such as sodium hydroxide concentration, the processing time and temperature, slack or taut treatment, the amount of applied tension during the treatment, and the source, physical state, and degree of polymerization of cellulose. The fabrics are then rinsed several times and neutralized to remove the remaining sodium hydroxide by using acids, like acetic acid or diluted sulfuric acid, followed by washing to remove the residual acid from the fabric. The process is continuous. Good results are obtained through proper saturation, sufficient tension, and complete washing.1-6
Polyfunctional finishes are gaining interest in the wet processing industry as they provide the advantage of attaining more than one functional finish in a single step. They also offer the benefits of saving water and energy expenses, and minimizing time and labor with increased production, enabling the fabric to attain the desired level of finishing effects.7,8
Cotton occupies an excellent position among textile fibers worldwide. This pre-eminence is due to an excellent combination of properties such as abundance, high strength, and durability, easy to spin and dye, ability to absorb moisture, and good heat conduction, which leads to comfortable wear during hot weather and related benefits. For improved performance of cotton in various areas such as dimensional stability, wrinkle recovery, flexural rigidity, flammability, soil release property, feel, and appearance, chemical treatments might be required. A one-step application process to improve a few of the above properties would be beneficial. 9
To reduce such shortcomings, it is a must to produce what is called “stabilization” of the fiber structure by introducing covalent crosslinking with adjacent cellulose chains within fibers. Crosslinking occurs within the accessible regions (i.e., the amorphous regions) of the fiber providing an increase in the fiber resistance to deformation and improved elastic recovery from distortion. Crosslinking of the cellulose molecules decreases the stress-induced slippage of polymer chains and improves the tendency to recover from any successive deformation, and thus render crease retention properties in cotton fabrics.10-12 Also, physio-chemical and structural modification are brought about by alkaline treatment. Formation of alkali cellulose I takes place from native cellulose when treated with a concentrated alkaline solution. Furthermore, during washing and neutralization, cellulose II is produced. The internal hydrogen bonds are broken and the available hydroxyl groups are increased, leading to an increased uptake in the dye.13,14
A comparatively new approach to formaldehyde-free crease-resistance finishes are based on the ester crosslinking of cotton by PCAs. Using Fourier transform infrared (FTIR) spectroscopy, various researchers have studied the formation of ester crosslinks in cellulose chains of cotton treated with PCAs and found them to be effective as crosslinking agents.15-18 They suggested a two-step anhydride mechanism for the formation of ester crosslinks in cotton by PCAs. The first step is the formation of a five- or six-membered cyclic anhydride intermediate by dehydration of two carboxylic groups, followed by the second step where the ester linkage is formed through the reaction between cellulose and the anhydride intermediate.9,10,16,19 The esterification reaction of PCAs is accelerated when applied with alkali metal salts of a phosphorus-containing acid. Sodium hypophosphite (SHP) is the most effective curing catalyst for the ester crosslinking of cellulose with PCAs as it provides the highest level of durable press (DP) rating and crease recovery angle (CRA), as well as the reduction in cure temperature, while maintaining a satisfactory level of tensile strength retention and whiteness.20,21
Various PCAs—succinic acid (SUA), malic acid (MLA), maleic acid (MA), tartaric acid (TTA), itaconic acid (ITA), citric acid (CA), and 1,2,3,4-butane tetracarboxylic acid (BTCA) have been studied as crease resistance finishes by various researchers.19,22-30 Except for BTCA, the other mentioned PCAs have some drawbacks in practice. Due to the presence of only two carboxylic groups SUA, MLA, MA, TTA, and ITA did not exhibit effective crosslinking, and when used alone, showed only slight improvements in crease resistance. Consequently, two dicarboxylic acids are used together or in conjunction with CA or BTCA. CA, having three adjacent carboxylic acid available for crosslinking, is a suitable candidate for crease resistance finishing, but causes yellowing due to the formation of α,β-unsaturated acids.30,31 BTCA is the most effective formaldehyde-free crosslinking agent in terms of crease-resistant properties achieved, speed of curing, laundering durability, retention of fabric whiteness, and lack of odor. But it is relatively expensive, requires a large amount of sodium hypophosphite, and may cause loss of mechanical strength.20,32-35
Cotton is one of the most flammable types of textile materials. It undergoes thermal degradation when ignited by forming combustible volatile compounds, which further disseminate fire.11,36 To ensure the safety of the end user from unsafe apparel, flame resistance is a critical property for textile products. The most common approach to obtain durable flame-retardant finishes for cotton is to apply products based on tetrakis(hydroxymethyl)phosphonium (THPX), and N-methylol dimethyl-phosphonopropionamide (MDPA). 37 These products contain formaldehyde and are expensive, requiring multiple alkaline washes after curing. They are also responsible for acid hydrolysis of cotton during storage, causing deterioration of mechanical strength.38,39
Researchers apply various PCAs in the presence of sodium hypophosphite (SHP) to enhance the flame resistance of cotton. SHP deposits phosphorus during the curing reaction; the phosphorus concentration on treated fabric depends on the concentration of SHP and curing temperature. Here, SHP acts as a co-reactant on flame resistant cotton fabric. The mechanism of phosphorus-containing flame retardants is quite versatile. They generally act in the condensed phase and can react or crosslink with hydroxyl-containing polymers like cellulose. These compounds prevent the formation of levoglucosan (an extremely flammable material and the main contributor to cellulose combustion), as well as flammable volatiles, and increase the formation of char, which acts as a barrier between flame and cotton resulting in quenching combustion. Moreover, phosphorus-containing flame retardants emit less toxic combustion products among other halogen-free flame retardants.40,41
SHP is retained on cellulose first by forming a five or six-member cyclic anhydride intermediate by dehydration of the two carboxylic groups of PCAs. Later, SHP reacts with this cyclic anhydride intermediate and then forms an ester linkage with the hydroxyl group of cellulose fibers (Fig. 1).9,10 Wu and Yang applied three bifunctional carboxylic acids, including SUA, MLA, and TTA, as low-cost, non-formaldehyde flame retardant agents for cotton fleece fabric. 42 They also investigated the combination of MLA and SHP in flame retardant finishing for the same fabric. 43 The effectiveness of four polycarboxylic acids, BTCA, CA, SUA, and MLA, were compared as durable flame retardants for cotton fleece in another study. 44 Binary combinations of SUA, ITA, MLA, and CA were applied on cotton fabric and found more effective as flame retardants than when applied alone. 7 MLA, ITA, and CA were found to improve many functional properties like crease recovery behavior, soil release property, and flame resistance, in addition to antimicrobial properties, when they are mixed with chitosan. 45 Similarly, the efficacy of the low cost, eco-friendly crosslinking agent CA was shown for the flame retardant and DP finishing of cotton and lyocell.39,46 On the other hand, the formaldehyde-free alternative BTCA was also examined to enhance the flame resistance and durability of cotton. 47 In another study, significant improvement of the flame resistance of cotton fabrics were found when BTCA was applied with a hydroxy-functional organophosphorus oligomer in the presence of SHP. 48
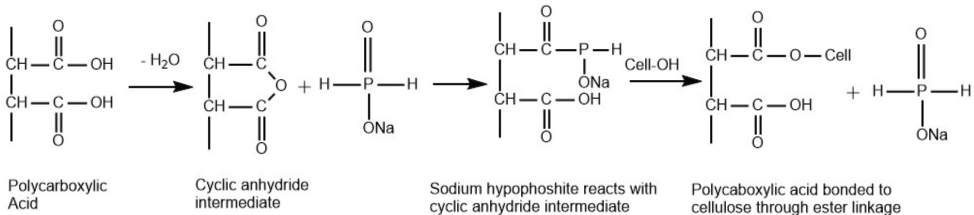
Reaction of PCAs with cellulose.
Due to the smearing of soil during use, fabrics tend to develop an overall grey and dingy look, and in the worst case may cause undesirable stains. Soil release is a chemical finish that facilitates relatively easy removal of soiling matter during laundering. The hydrophilic surface characteristics of fabrics are improved by this finish, which hinder soil penetration deep into the fabric during laundering, and its distinct functional groups remove dust and dirt from the fabric and shift it to the detergents.5,11 Improvement of soil release properties of cotton by PCAs have also been reported in the literature.7,45
Most of the research carried out on mercerization emphasizes the structural change of cotton cellulose due to mercerization and its impact on physical and chemical properties. Similarly, PCAs have been reported mainly as an alternative crosslinking agent to improve crease recovery. Improvement in one or two functional properties (e.g., flammability, antimicrobial activity, strength retention, dimensional stability, and whiteness retention) as additional benefits have also been reported by a few researchers.
No research has been reported yet combining the two process (i.e., mercerization and polyfunctional finishing) into one continuous process. This present work concentrates on the application of PCAs to neutralize mercerized fabric and simultaneously impart functional finishing effects on cotton fabric, combining them into a single step. Mercerization is known to enhance the water absorption of cotton, thereby facilitating greater penetration of agents such as PCAs, that can lead to crosslinking. As a consequence, the amount of unreacted PCAs, which cause fabric yellowness and degradation, should decrease.
Experimental
Materials
Undyed 100% cotton plain weave fabric (180 g/m2) was desized, scoured, and bleached by the supplier (Baximco Textile Ltd., Bangladesh). Before use, it was fully washed and dried. Sample fabric size for each application was 12 × 12 in. A wooden frame having a dimension of 13.5 × 13.5 in. was fabricated. Fixed pins were provided on sides of the frame to maintain the same dimensions and tension on the fabric. Mercerizing oil (Mercerol QW-LF) and a bifunctional reactive dye, Covazol Red 3BSN (C.I. Reactive Red 195, CAS Registry No. 93050-79-4) were supplied by Imperial Dye-Chem Co. Ltd. Bangladesh. BTCA was purchased from Sigma-Aldrich (Merck, India) with a purity of 99%. Sodium hydroxide, acetic acid, MLA, SUA, CA, BTCA, and SHP (Merck, India) used in this study were of laboratory grade. The chemical structures of SUA, MLA, CA, and BTCA are shown in Fig. 2. The sample codes of the treated fabric are given in Table I.
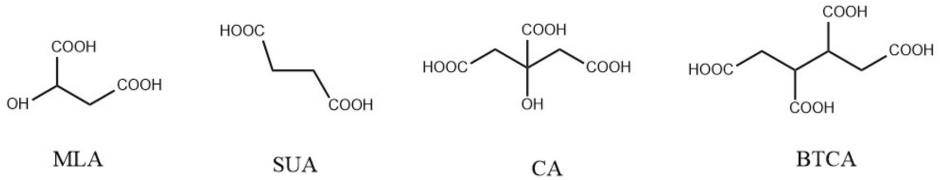
PCAs used in this study.
Fabric Sample Codes
Combined Mercerization and Polyfunctional Finishing
The scoured and bleached fabric sample was mercerized using 25% w/w of sodium hydroxide and 1% mercerizing oil at room temperature (RT) without tension using a laboratory padding machine (Electronic & Engineering Company, India) with a pressure of 3 kg/cm2. After padding, the fabric was placed in a frame under tension for 15 min, followed by washing in hot water at 90-95 °C for 3 min, and then rinsing at RT for the same duration. The washed samples were then neutralized with acetic, SUA, MLA, CA, and BTCA with SHP catalyst in padding mangle maintaining the same pressure. Based on preliminary trials, the concentrations of PCAs were fixed to 6%, 8%, and 10%, with 6% SHP, in each case. Weight add-on was measured after every padding and 100% wet pickup was achieved maintaining a 3 kg/cm2 padder pressure to ensure a uniform add-on percentage and pad liquor concentration. To formulate the optimal curing condition with maximum crosslinking and minimum yellowing, we performed separate trials using the Box-Behnken experimental design taking three factors into account: curing temperature (140, 160, and 180 °C), curing time (3, 4, and 5 min), and catalyst (SHP) concentration (4%, 6%, and 8%). The optimal curing conditions were 160 °C for 3 min at a 6% SHP concentration. These samples were dried at 80 °C for 5 min in a hot air oven (Binder, USA) and curing was carried out at 160 °C in a high temperature curing oven (Binder, Germany). The samples were then washed and dried.
Dyeing
The acid neutralized samples were dyed with the bifunctional reactive dye to a 0.5% shade in an Ahiba IR (Datacolor, USA), keeping a liquor ratio (LR) of 1:20 at 60 °C for 60 min. Salt (15 g/L) and soda ash (5 g/L) were added for exhaustion and fixation of the dye respectively. After dyeing, the dyed samples were washed at 80 °C for 10 min using 2 g/L soaping agent (Ladipur RSK, Clarichem) to remove unfixed dye from the fabric.
Test Methods
All samples produced were tested for their functional properties after conditioning for 12 h at 20 ± 2 °C and 65 ± 2% relative humidity (RH) in a conditioning chamber. The performance properties were evaluated as per the following international standards: barium activity number (AATCC TM89-2003), whiteness index (AATCC TM110-1995), brightness index (ISO 2470-1977), crease recovery angle (AATCC TM66-2008), tensile strength (ASTM D5035-06-2008), tearing strength (ASTM D1424-09), stiffness (BS 3356-1991), dimensional properties (BSEN 25077-1994), and soil release properties (AATCC TM130-2000). The vertical flammability, limiting oxygen index (LOI), and 45° flammability test of the fabrics were assessed according to ASTM D6413-99, D2863-97, and D1230-94, respectively. Phosphorus concentration on the PCA treated cotton fabrics after three home laundering (washing/drying) cycles was measured according to the stannous chloride method for phosphate determination using a UV-Vis spectrophotometer (Varian Cary 5000).41,49 Home laundering was done according to AATCC TM124-2018 (smoothness appearance of the fabrics after home laundering) using the 1993 AATCC Standard Reference Detergent WOB (powder form). The absorption and scattering value (K/S) of the colored samples was also measured using a computerized color matching system (Datacolor 650, USA) at the chosen maximum wavelength (680 nm). K/S values were measured using the built-in software of the color matching system.
Results and Discussion
Mercerization of Cotton Fabric
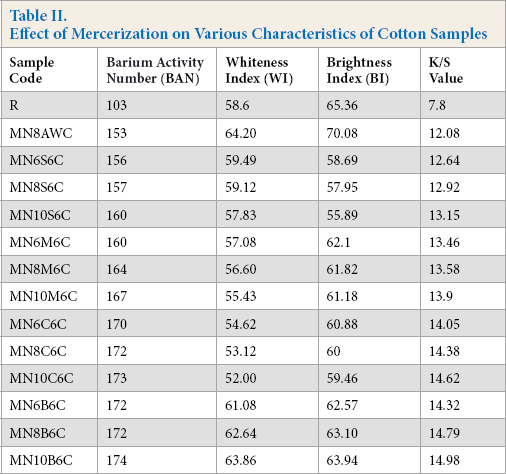
The barium activity number (BAN) is widely used to determine the degree of mercerization. 5 According to AATCC TM89-1998, a BAN in the range of 100-105 indicates no mercerization, and a value above 150 indicates a substantially complete reaction between the cotton and the mercerizing bath. The un-mercerized fabric had a BAN of 103 (Table II). On the other hand, all of the mercerized and PCAs neutralized samples gave values greater than 150, indicating complete mercerization. Among the four PCA neutralized samples, BTCA neutralized samples showed a slightly greater BAN. The mercerization process improved the whiteness index (WI), brightness index (BI), and dye uptake (higher K/S value). The increase in dye uptake of the mercerized fabric crosslinked with PCAs is possibly due to a decrease in crystallinity. Physio-chemical and structural modification is brought about by the alkaline treatment. Formation of alkali cellulose I occurs from the native cellulose when treated with a concentrated alkaline solution. Furthermore, during washing and neutralization, cellulose II is produced. The internal hydrogen bonds are broken and the available hydroxyl groups in the amorphous region are increased leading to an increased uptake in dye.13,14 However a slight effect on the WI and BI occurred as it caused the fabrics to become a little yellowish. The hydroxy-functional PCAs (CA and MLA) caused slightly more yellowing on the mercerized white cotton than their unsubstituted counterparts (BTCA and SUA). Among the PCAs, CA caused more yellowness (less WI) of the cotton fabric. Yellowness of the fabric caused by CA can probably be attributed to the formation of α, β-unsaturated acids on the fabric. 30
Effect of Mercerization on Various Characteristics of Cotton Samples
Crease Recovery
Crease recovery is a fabric property enabling it to recover from folding deformation and can usually be expressed as the crease recovery angle (CRA). The larger the CRA value, the greater the resistance of a fabric is to crease. CRA values of the treated samples were measured after three home laundry cycles to ensure the durability of the crosslinked fabric (Table III). The crease recovery behaviors of all the PCA treated samples were found to be very good. The CRA of the acetic acid neutralized sample was 198, but for the PCA treated fabrics, it reached a maximum of 291. The crosslinking imparted by the esterification reaction of the PCAs with cellulose gave enhanced crease recovery behavior. Moreover, structural changes (cellulose I into cellulose II) in mercerized cotton cellulose also improved the effect of crosslinking resulting in higher CRA values. 4 BTCA, having four carboxylic acid groups per molecule, in the presence of the catalyst, was found to be more effective than PCAs having three or two carboxylic acid groups.
Effect of Neutralization with PCAs on CRA and Strength Properties of Test Fabrics a
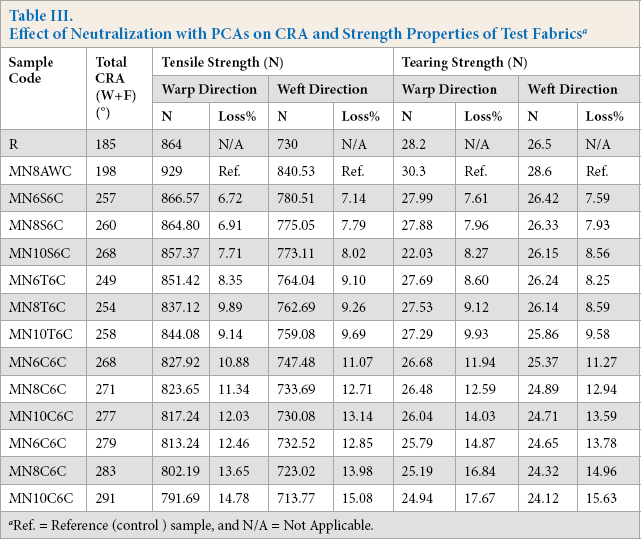
Ref. = Reference (control) sample, and N/A = Not Applicable.
Flammability
The limiting oxygen index (LOI), char length, and 45° flammability testing results of the cotton woven fabrics, treated with the four acids at various concentration levels in the presence of SHP, that were cured and subjected to the combined refurbishing procedure of dry-cleaning and home laundering, are presented in Table IV. The LOI value of fabrics is an important property that determines fiber flammability. Fabrics with LOI values above 21 burn slowly and values of 26-28 are adequate to pass small burning tests. According to ASTM D1230-94, the flammability of a textile fabric can be characterized into three classes: “Class 1” (normal fam-mability)—fabrics having a burning time of greater than 7 s or having surface fash without base burn (regardless of burning time), these textiles are generally considered to be acceptable for apparel; “Class 2” (intermediate fammability)—fabrics having a burning time of 4-7 s with base burn; and “Class 3” (high flammability with rapid and intense burning)—fabrics having a burning time of less than 4 s or having base burn, which is generally considered to be unacceptable for apparel.
Flammability and Phosphorus Content (%) of Control and PCA Treated Samples
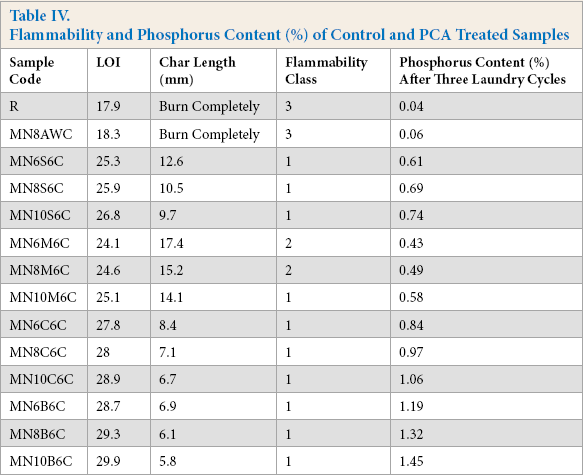
The results revealed that cotton fabric without any treatment could not pass the flammability test and was completely burnt, having an LOI value of only 17.9 (“Class 3” flammability). The performed treatments were able to decrease the flammability of the treated samples. Their performance depended on the number of functional groups present and the concentration of the PCA. The greater the number of functional groups and concentration of the PCA, the greater the resulting flame resistance. Cotton fabric neutralized with 10% BTCA in presence of SHP was the most effective in reducing the flammability of cotton from “Class 3” to “Class 1” with a char length and an LOI of 6.5 and 29.9 mm, respectively. Among the four PCAs, MLA was comparatively less effective in reducing the flammability of cotton fabric, and at a low concentration, reaching only “Class 2” in the 45° flammability test. A similar trend of improved flammability for PCA treated samples was reported in the literature. 44
The percentage of phosphorus present on PCA treated cotton woven fabrics in the presence of 6% SPH measured after three laundering cycles is presented in Table IV. The maximum amount of phosphorus was retained on fabric treated with BTCA, followed by CA and SUA, and then MA. Mercerized samples neutralized with 10% BTCA retained 1.45% phosphorus, whereas the same concentration of MA retained only 0.58% phosphorus, the minimum amount required to obtain “Class 3” flammability. The percentage of phosphorus retained by PCA treated samples followed the experimental trend of the treated sample flammability results. The major difference in phosphorus concentration between BTCA and the other acids is the presence of four carboxylic acids in the BTCA molecule and their interaction of SHP, leading to more effective crosslinking with cellulose. As a result, PCAs with a greater number of carboxylic acid groups (BTCA and CA) are able to form more crosslinks on cotton and retain more phosphorus from SHP with cellulose. Among the bifunctional acids (SUA and MA), the α-hydroxyl group of MA hinders esterification and facilitates hydrolysis for the MA bound to cotton, resulting in the least fabric flame resistance.
Soil Release
Soil release characteristics are essential for creating an easy-care fabric. The untreated control sample had a grade of 3, while all samples neutralized with PCAs were graded 5, which represents the best soil release property. Surface properties of the treated cellulose became more hydrophobic due to crosslinking, which altered its soiling characteristics. 7 Moreover, due to the swelling of cotton fiber during mercerization, crenulation, and lumen of the fiber decreases, resulting in a smoother fiber surface. It increases the pore volume of cotton and hence the chemical accessibility of the fibrillar structure. Due to this structural change, the extent of soil, especially lipid soil, deposition in the lumen is reduced. 50
Strength Properties
It was found in the present study that crosslinking PCAs with cellulose generally results in excellent crease recovery behavior, but at the same time, leads to a strength loss of 7-15%. For all PCAs, both tensile and tearing strength showed similar tendencies, but strength loss was very much dependent on the concentration of PCA. Strength loss mainly arises due to the formation of inter-and intra-molecular crosslinks, which decreases the possibility of equalizing the stress distribution, resulting in the reduction of load-bearing capacity. 7 The higher the concentration of PCA, the greater the percent strength loss in both tensile and tearing strength for all PCA neutralized samples. This can mainly be attributed to the increase of both acid-catalyzed depolymerization and crosslinking of the cellulose molecules, with increased PCA concentration.51-53
Strength loss was considerably less in the case of all PCA neutralized samples. This is because the mercerization process increases the tenacity of cotton fiber by strengthening weak points along the fiber and by increasing the orientation of the cellulose chain. 5 The PCAs with a greater number of functional groups (BTCA and CA) form more ester linkages on cotton and suffers greater fabric strength loss than their bifunctional counterparts (SUA and MLA). Only the acids having carboxylic groups of three or more are capable of crosslinking to cellulose. Bifunctional acids form single ester linkages with cotton.
Stiffness Properties
The stiffness of the fabric was somewhat increased by crosslinking with PCAs when compared to the untreated sample as crosslinking restricted the movement of the fiber elements. Bending length is the measure of fabric stiffness. Stiffer fabric has a higher bending length. Table V shows that bending length of acetic acid neutralized fabric was around 2.13 cm, whereas PCA treated fabrics had a 3.4 cm bending length, which indicates that PCAs treated samples were slightly stiffer than the acetic acid treated sample due to crosslinking between cotton cellulose and PCA.
Stiffness and Shrinkage Properties of Control and PCA Treated Samples
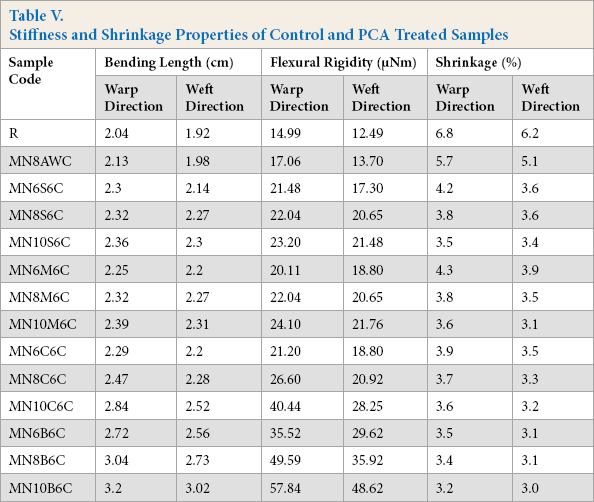
Flexural rigidity, which is a measure of stiffness associated with handle, was also less for the PCA treated fabrics. As flexural rigidity is proportional to the cube of the bending length, the values increased proportionally with bending length. However, since the increase was very small, it did not have significant effect on handle.
Dimensional Stability
Dimensional stability is the property of a fabric that sustains its original dimension when subjected to certain processes. The dimensional stability can be determined by the dimensional change (i.e., growth or shrinkage). The standard testing method for dimensional change after laundering is assessed by BSEN 25077-1994. This process focuses on measuring changes in length and width of the fabric. PCA treatment did not significantly affect the dimensional stability of the fabric. The shrinkage was less than 6% in almost all cases (Table V). Minimum shrinkage was found when BTCA was used due to the maximum amount of crosslinking of cellulose molecules.
Conclusions
In this study, combined mercerization and polyfunctional finishing were performed on cotton fabric using various PCAs having different functional groups along with SHP catalyst/ co-reagent. Results demonstrate that mercerization improved some of the cotton fabric properties and a combined poly-functional finish can be applied during the neutralization process imparting some of the functional properties. Due to mercerization, PCA neutralized samples exhibited a good degree of mercerization with a high BI, WI, and dye absorbency (K/S value). The ester crosslinks made by the PCAs during neutralization in the presence of SHP catalyst/co-reagent provides the fabric with many functional properties, including increased crease recovery angle, flame resistance, and low soiling properties, with a minimum effect on handle properties (reduced bending length and flexural rigidity) and dimensional stability. A corresponding loss of tensile and tearing strength occurred because of acid-catalyzed depolymerization and crosslinking of the cellulose molecules. However, this was less for the PCA treated samples when compared to the value obtained from the acetic acid-treated sample. The PCAs with a greater number of functional group (BTCA and CA) can impart higher crease recovery, flame resistance, and dimensional stability. but at the same time, exhibit greater strength loss of the treated cotton woven fabric than their bifunctional counterparts (SUA and MLA). With a gradual increase in acid concentration, the crease recovery angle and flame resistance also increased, whereas tensile and tearing strength decreased with a minimal negative effect on whiteness. Neutralization with a PCA with a catalyst after mercerization is a cost-effective and eco-friendly way to improve many functional properties of cotton. It is also an alternative formaldehyde-free approach to treat cotton fabric.
