Abstract
Fungal laccase-catalyzed in situ polymerization of the naturally-occurring phenol syringic acid was used as an environmentally-friendly alternative for wool coloration at room temperature. Through the incorporation of amino acid small molecules in the polymerization process, the polymeric chemistry of syringic acid was changed, and the color appearance obtained was controllably-tuned in a simple way. By using this method, wool fabrics could be dyed with natural yellowish brown color appearances and different color hues.
Introduction
Several hundreds of thousand tons of dyes are consumed annually worldwide. Some of them are discharged together with dyeing auxiliaries as polluting effluents.1,2 Dye waste-water often poses a threat to the environment, including drinking water and agricultural production, and can lead to human health issues.3,4 The mechanism of mussel adhesive proteins (MAPs) provided an alternative as a potential bio-mimetic means for wool dyeing. MAPs contain high levels of 3,4-dihydroxy-L-phenylalanine (DOPA), which contains catechol and ethylamine functional groups. 5 Natural phenols derived from plants contain similar structural units of catechol. 6 Furthermore, red and black hairs consist of 44% and 15% amino acids by mass, respectively. 7 The addition of amino acids to phenolic precursors may alter the chemistry of polymers derived from them and thus allow tuning the colors of the obtained polymer colorant. In this study, the dyeing performance of syringic acid and the tunability of different amino acids on the obtained color hues were investigated in the presence of laccase enzyme. 8
Experimental
Materials
All wool fabrics (274 g/m2) used in this study were available in the market. The fabrics were washed in 5% sodium dodecyl sulfate solution at 80 °C for 20 min and then rinsed with deionized water. Laccase (Sigma-Aldrich), syringic acid (Alfa Aesar), and amino acids (J&K) were commercially available and used as received (Fig. 1).
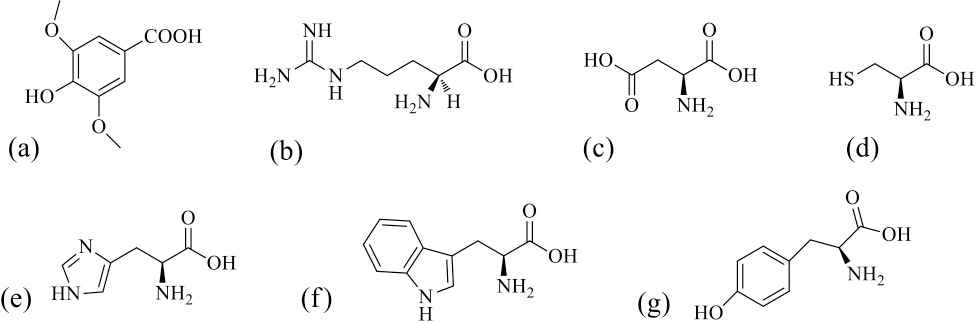
The chemical structures of syringic acid (a, SA), arginine (b, Arg), aspartic acid (c, Asp), cysteine (d, Cys), histidine (e, His), tryptophan (f, Trp), and tyrosine (g, Tyr).
Coloration Procedures
Syringic acid (100 mg) and amino acid (10 mg) were dissolved in 20 mL of Tris buffer (pH 5.0) in 3 mL of ethanol. The clean wool fabric (7.5 × 9.0 cm2) was immersed into the solution and kept for 1 h at 70 °C. It was then taken out for spraying with laccase solution (7.5 mg of laccase in 2 mL of deionized (DI) water) after the temperature was decreased to room temperature. The sprayed fabric was kept overnight. Finally, the obtained fabric was rinsed with tap water and air dried for the measurements.
Characterizations
The colorimetric assay of the dyed wool fabrics was evaluated using a Datacolor
650 spectrophotometer having an illuminant that includes the UV component and
excludes the specular component. CIELAB data of the dyed fabrics were recorded.
Result and Discussion
In the presence of laccase, syringic acid was first oxidized, followed by coupling polymerization to form polymeric colourants.10,11 Conjugated double bonds created by aryl-aryl coupling contribute to chromophore formation. 6 At the same time, it was found that phenolic groups could be easily oxidized to form quinones, which can react with nucleophilic species, such as amino acids and heterocyclic compounds. Thus, amino acid molecules were introduced into the aryl-aryl chromophores of syringic acid, which would alter the polymerization chemistry and the physical and chemical properties of the polymerized syringic acid. Therefore, different color hues could be produced.10,12 Based on this, the dyeability of wool fabrics by polymerized syringic acid and its color tunability in the presence of amino acids was investigated. Using this method, wool fabrics could be dyed a natural yellowish-brown color (Fig. 2). After the addition of amino acids, the obtained color hues of the dyed wool fabrics changed slightly. Especially, the addition of cisteine (Cys) resulted in a browner color hue compared to that of syringic acid only, which was due to the high reactivity of the thiol group in Cys.

The photographic images of (a) blank fabric, and fabrics dyed with (b) SA, (c) SA+Arg, (d) SA+Asp, (e) SA+Cys, (f) SA+His, (g) SA+Trp, and (h) SA+Tyr.
The UV-Vis absorption spectra of fabrics were also investigated. From Fig. 3, the blank wool fabric had no obvious absorption peak, while the dyed samples showed absorption peaks at ∼460 nm. Moreover, the dyed fabrics had ca. 2-3 times greater absorbance than that of the blank fabric. This absorption pattern resulted from the yellowish brown color of the dyed wool fabrics. Addition of amino acids during the coloration process produced no clear influence on the absorption bands, but it did produce an obvious influence on the absorbance level.
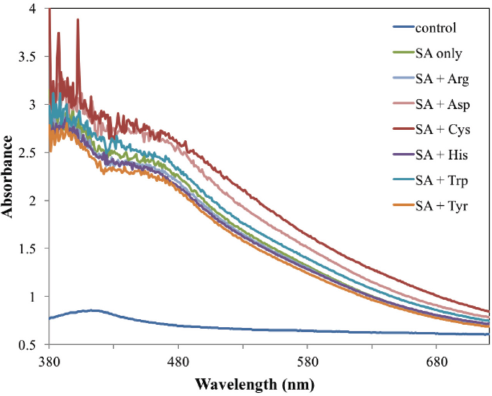
Visible absorption spectra of the dyed wool fabrics.
The influence of the addition of amino acids on the obtained color hues was further
investigated with CIELAB data (Table
I). The blank wool fabric had an
Colorimetric and
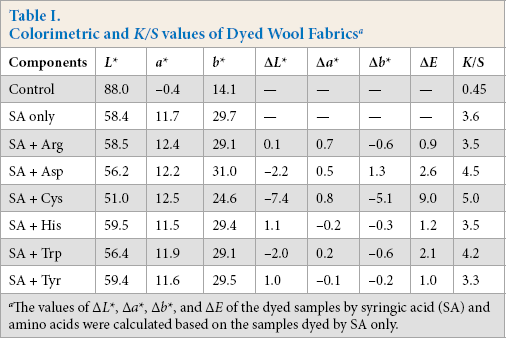
The values of Δ
The microscopic analysis of the dyed fibers was further investigated (Fig. 4). From their SEM images, the surface morphology of the dyed fibers was much rougher and the scales on the fiber surface were less sharp when compared to the blank wool fibers. These findings indicate that polymeric colorant layers or particles were deposited on the fiber surfaces after dyeing. In the cross section images, the blank fibers were their original white color, while the dyed fibers were a brown color. These results indicated that wool fibers could be successfully dyed using this method. Moreover, the inner and exterior parts of the fibers could be dyed simultaneously.

SEM micrographs and cross-sectional images of wool fibers before dyeing (a,c) and after dyeing (b,d).
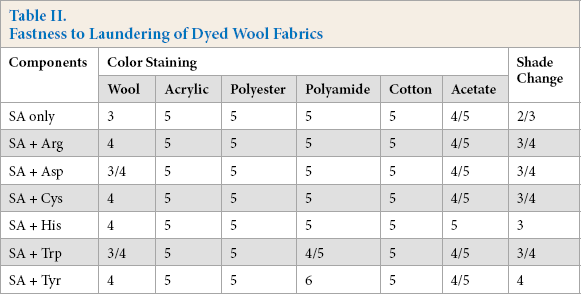
The washfastness of the dyed wool fabrics were tested (Table II). All dyed fabrics had excellent colorfastness to staining, rating at 4/5-5, except on wool fabrics. However, the fastness to shade change was moderate, which meant some colorants were washed away from the fabrics during laundering. The addition of amino acids during the dyeing step improved the fastness to shade change. This was probably because the incorporated amino acids had functional groups similar to protein fibers and thus could enhance the interactions with wool fibers.
Fastness to Laundering of Dyed Wool Fabrics
Conclusion
Laccase-catalyzed in situ polymerization of plant phenols is a potential alternative as an environmentally-friendly wool dyeing method at room temperature. The addition of amino acids during the polymerization process altered the polymeric chemistry of phenols and thus allowed tunable color hues. At the same time, the dyeing properties of the dyed fabrics were also improved.
Footnotes
Acknowledgement
The authors acknowledge the funding by the National Natural Science Foundation of China (No. 21376197).
