Abstract
Chitosan is a poly-β-(1-4)-D-glucosamine obtained from the deacetylation of chitin. Its chemical properties and biological properties provide a broad potential application in the textile industry. In the current review, versatile properties of chitosan related to textile finishing are presented and briefly discussed. The application of chitosan in the textile industry including dyeing, printing, and functional finishing is summarized. Meanwhile, the advantages, disadvantages, and improved application methods are discussed. It is hoped that the review will be a good resource for those interested in antimicrobials and biocidal textile materials, and will help stimulate further interest in this area.
Keywords
Introduction
Chitosan is a renewable polymer derived from deacetylation of the polysaccharide chitin. Chitin, poly-(1,4)-2-acetoamido-2-deoxy-β-D-glucose, is extracted from crustaceans (e.g., crab and shrimp shells) by acid treatment to dissolve calcium carbonate followed by alkaline extraction to solubilize proteins. Chitin is the second most omnipresent natural material after cellulose on earth, which means that the sources of chitosan are of widespread occurrence.1-7 As a natural material, chitosan has always appealed to researchers, owing to its unique combination of properties, such as biodegradability, biocompatibility, non-toxicity, moisture permeability, reactivity, adsorption, and antimicrobial activity.1,3-5,8-12 It has been extensively investigated for its applications in the biomedical, engineering, pharmaceutical fields, food industry, and so forth.13-19 In recent years, chitosan also has been applied to textile materials and fabrics, and significant progress has been obtained in this field because of various functions for clothing, especially for its antibacterial function, high absorption, and ability to staunch bleeding.4,11-12,20-27
Chitosan is a linear polyamine consisting of poly-(1-4)-2-amido-2-deoxy-β-D-glucose. Its molecular structure (Fig. 1) is similar to cellulose and chitin, with the only differences being that the amino group (-NH2) replaces the hydroxyl group (-OH) on the C-2 position.5,8-9 Due to the presence of -NH2 groups, chitosan can be converted to a polyelectrolyte in aqueous acidic media by protonation. Thus, chitosan is soluble in aqueous acidic media and nearly all characteristics of chitosan are dependent on the pH value of the solution.1,8,9,28 Tat is also the reason why chitosan possesses antimicrobial properties and inhibits the growth of a wide variety of fungi, yeasts, and bacteria. 5 Because of the existence of abundant reactive amino group and hydroxyl groups, the pseudonatural cationic polymer, chitosan, can participate in versatile reactions including graft, ionic, and chelation reactions.9,29,30 The characteristics of chitosan make it suitable for different functional finishing of textiles to improve antimicrobial activity, crease recovery, colorfastness, and so forth.
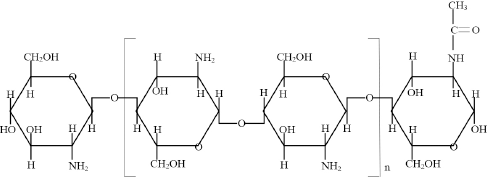
Molecular structure of chitosan.
Chitosan already has numerous applications in the textile industry, including chitosan nonwoven fabrics, chitosan fibers and yarn, pretreatment of textiles, dyeing, printing, functional finishing of fabric, and wastewater treatment.2,4,10,11,14,21-23,26,30-44 This review aims to present versatile properties of chitosan related to textile finishing and summarize applications of chitosan in the textile industry. Meanwhile, the advantages, disadvantages, and improved methods of chitosan application will be discussed briefly.
Chitosan Properties
The performance of chitosan is related to its properties such as degree of deacetylation (DDA), average molecular weight (MW), polydispersity (MW/MN), crystallinity, and the pattern of acetylation. Chitosan varies greatly in its MW, DDA, and crystallinity in terms of the chitin source and the methods of hydrolysis. The MW of chitosan can vary from 30 kDa to well above 1000 kDa. The typical DDA of chitosan is over 70%, making it soluble in aqueous acidic solutions. Higher crystallinity leads to the poor solubility of chitosan in common diluted acids, limiting methods to process chi-tosan. In general, reduced MW and lowered crystallinity by random deacetylation can improve its solubility in solvents. Accordingly, the properties of chitosan are dependent upon the extraction process of chitosan.1,4,45,46
The factors affecting the properties of chitosan in the extraction process are the concentration of alkali, temperature, reaction time, and structure of chitin. 47 In the treatment process, alkali plays a part in removing the protein in chitin and works as a catalyst in deacetylation as shown in Fig. 2. Generally, chitin is deacetylated with 40% aqueous alkali, sodium hydroxide in most cases, at 100∼120 °C for 1-3 h, and 70∼85% deacetylated chitosan can be obtained.2,47,48 Deacety-lation proceeds rapidly to ∼70% within the first hour of alkali treatment in 50% NaOH solution at 100 °C, but its progress decreases gradually after this, reaching 80% in 5 h, and further alkali treatment does not deacetylate significantly and only degrades the molecular chain.49,50
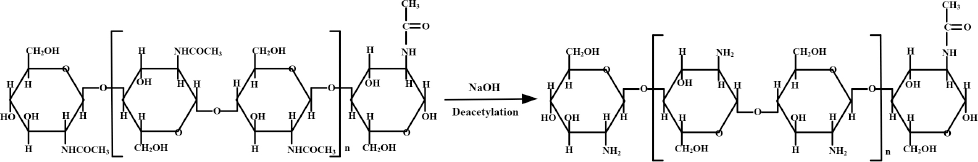
Conversion of chitin to chitosan.
After extraction treatment, nitrogen in chitosan, the deacetylated form of chitin, is
in the form of primary aliphatic amino groups. Therefore, chitosan is soluble in
acid solution and its solubility in water and organic solvents can be controlled by
manipulating solution conditions including temperature, pH, ionic strength,
concentration, and solvent.
45
Because chitosan amino groups carry a positive charge at pH
values below 6.5, materials bearing negative charges are potentially capable of
interacting with chito-san.51,52 As
a reactive copolymer, reaction active sites of chitosan are concentrated in
-NH2 and -OH.
53
Due to the presence of amino groups, reactions that
chitosan always undergo are typical of amines, of which
Textile Applications of Chitosan
Fibers and Yarn
Chitosan is a tractable material for processing into fibers, which are primarily prepared with a chitosan solution through wet spinning and electrospinning.7,11,12,19,46,48,54-59 Generally, spinning proceeds after dissolution of the chito-san in acidic aqueous solution, deaeration, and filtration of the solution. After spinning, the chitosan fibers also undergo refining and drying, as well as relative post-treatment.2,11,56,60
Due to the limitations of the chitosan fibers in terms of their high electrostaticity, poor mechanical properties, and high cost, chitosan fibers are usually used with the assistance of secondary fibers in the textile industry. This can enhance the commercial value of the chitosan fibers and achieve the best performance of chitosan fibers by maintaining the scientifically-proven properties of chitosan in the fabrics.10,11,56 On the other hand, 100% chitosan also has disadvantages, such as being unwashable, having bad elasticity and softness, and being too thin to be stabilized in place. 12
Chitosan and cellulose possess quite similar chemical structures, so they are easy to blend together to produce fibers. These blended fibers keep the excellent properties of chitosan and the physical properties of cellulosic fiber permanently over time. Due to the similar chemical structure of chito-san and cellulose, these fibers possess excellent dyeability with reactive and direct dyes, as well as soft hand. Crabyon (Swicofil) fiber, a composite of chitosan with cellulose viscose, is manufactured by uniformly blending chitosan and cellulose viscose molecules and extruding the blended viscose into the spin bath, while Chitopoly (Fuji Spinning Co. Ltd.) fiber is made by kneading an emulsion of chitosan into a ripening polynosic fiber via the mechanical method. Both of these fibers have good dyeability and performance.31,61-63
To avoid the limitations of chitosan fibers and enhance the performance of the blended yarns, numerous researchers investigated the influence of the properties of chitosan fibers, blending ratio, blending methods, and so forth, on the performance of the yarn samples.10,11 Lam et al. reported that yarn tenacity deteriorates with the increase in the length of the chitosan fiber and chitosan fibers in lengths of 30 and 38 mm are compatible for blending with long cotton fibers. The sliver-blending method offers better tensile properties of yarn samples, while the fiber-blending method offers higher uniformity of fiber distribution. 11 Lam et al. also suggested that increases in the concentration of chitosan (50% or above) in the blend ratio reduces the rigidity of the fabric and provides a softer handle for the inner surface of the chi-tosan/cotton blend fabric. This facilitated the development of the chitosan fibers in medical textiles. 10 Ramie/chitosan yarn blended using the air-jet spinning method shows good yarn structure and distinct antimicrobial properties. 64 Besides cellulosic fibers, chitosan is also blended with other fibers to obtain yarns of high quality. Duan et al. obtain ultrafine chitosan fibers with the aid of poly(ethylene oxide) (PEO) in aqueous acetic acid solutions by electrospinning. The ultrafine fibers could not be electrospun from chitosan solution in aqueous acetic acid, but formed when a certain amount of PEO was added. 56 Hirano blended chitosan with collagen by wet spinning to improve its biocompatibility, because chitosan is not a structural component in human tissues.56,65 Zheng et al. blended chitosan with 10-50% of poly(vinyl alcohol) (PVA) forming fibers by spinning the solutions into a coagulation bath containing aqueous NaOH and ethanol. The mechanical properties of the blended fibers improved significantly due to the presence of PVA.56,66 Liu et al. blended chitosan fibers with polyacrylonitrile (PAN) and got a chitosan/PAN yarn with superior tenacity, because of the complementary effect of positive and negative charges. 12 Cheng et al. optimized the blending ratio and the strength of chitosan/acrylic yarns, and verified it in test production. 67 Alginate-chitosan (Alchite) fibers are developed to be used as wound dressings because of the synergism between alginate and chitosan on cell attachment, proliferation, antimicrobial effect, hemostasis, and biocompatibility. Alchite fibers are composite fibers with calcium alginate as core material and hydrolyzed chitosan on the outer surface. Usually, it is produced through extrusion of sodium alginate into a calcium chlorite bath, followed by passing through a hydrolyzed chito-san bath, as well as drawing and rinsing in water baths before pickup.51,68,69 These fibers have superior tensile and absorption properties in different liquid media compared to pure alginate or chitosan fibers for wound care applications. 70
Nonwoven Fabrics
Chitosan fibers are significantly more expensive than other fibers. The price ranges from US$50,000 to US$100,000 per ton depending on the degree of deacetylation of the chitosan.11,12 Although many researchers try to improve the performance of the chitosan fibers through various methods, they still possess some key limitations including high electrostaticity and poor mechanical properties, which inhibit chitosan fiber applications.11,12,71 Use of nonwoven technology can facilitate chitosan application in the textile industry, lowering the production costs and facilitating prototype production.10,11 Generally, the structure of nonwoven fabrics are highly branched and irregular and the fibers in the fabrics are unmanageably brittle. Therefore, it is necessary to include in the process a plasticizer and a wet-laid mat. 72 On the other hand, the limited solubility of chitosan in aqueous acids and a high degree of inter- and intra-chain hydrogen bonding hinder the spinning of pure chitosan. Also, 100% chitosan material has poor elasticity, is unwashable, possesses poor softness, and is too thin to be stabilized in place when chitosan is applied in wound healing. Chi-tosan's textile application are valuable for wound healing due to their antibacterial properties and resistance to attack from bile, urine, and pancreatic juice as absorbable sutures. 12 Therefore, almost all of nonwoven chitosan fabrics also need the assistance of secondary materials.5,14,56,58,73,74 Ohkawa et al. tried to prepare a pure chitosan nonwoven fabric through electrospinning, and the optimal pure chitosan nonwoven fabrics produced by selection of electrospinning solvent and adjustment of the chitosan concentration possess the same characteristics as chitosan/PVA blended nonwoven fabrics. 73 This is the first time pure chitosan nonwoven fabrics were prepared, providing a new method for it.
The first important application of chitosan nonwoven fabric is in wound dressings because of chitosan's properties. Nakajima et al. reported that fibers made of chitin and chi-tosan are useful as absorbable sutures and wound dressing materials. Also, Nara et al. have patented a wound dressing comprising a nonwoven fabric composed of chitin fibers made by the wet-spinning technique, which enables a better wound healing process.2,12,72,75,76 Wang et al. modified one nonwoven fabric by coating chitosan on both internal and external surfaces, and found that the composite membranes exhibited high flux, high effluent quality, and excellent anti-fouling properties through characterization of the chemical structure and morphological variations. 77 It has been claimed that chitosan nonwoven fabrics have applications in wastewater treatment. The removal of heavy metal ions by chitosan through chelation has received much attention.2,12 Chitosan nonwoven fiber mats produced through electrospinning have a high specific surface area and small pore size, which offer a distinctly high surface area to mass ratio (typically ranging from 40 to 100 m2/g). This property of chitosan nonwoven fabric makes it beneficial in filter applications.5,56,78
Dyeing and Printing
Chitosan contains a high proportion of amino groups, which provide more dye sites for anionic dyes (e.g., direct, acid, and reactive dyes) to be absorbed by fabrics with chitosan pretreatment through van der Waals forces and electrostatic attraction.32,33 Therefore, cotton and wool fabrics with chito-san pretreatment have good affinity to anionic dyes, high dye uptake, and color strength.21,32-34,79,80 Due to similarities in structure between chitosan and cellulose, chitosan has good adsorbability to cotton fabrics through van der Waals forces. And chitosan can bind to cellulose through Schiff base reaction between cellulose's reducing end (-CHO) and the amino group of chitosan. Meanwhile, hydrogen bonding also plays an important role in the absorption of chitosan to cellulose. Consequently, chitosan has good fastness when it is used for functional finishing of cotton fabrics. Cotton fabrics coated with chitosan have uniform depth of shade after dyeing because chitosan can cover the neps formed in immature cotton fibers.22,32 The disadvantages of chitosan applied in fabric pretreatment are poor colorfastness and ring dyeing, and stiffness. 33 Srisuk et al. treated the chitosan-cotton fabrics with sodium nitrite to partially remove chitosan film present on the fabric surface under mild conditions. This enhances the fastness properties and stiffness of fabrics as well as overcome ring dyeing issues simultaneously. 33 Srisuk's research suggests that chitosan with low molecular weight can reduce these dyeing defects. 33 For wool with chitosan pretreatment, the dye uptake is high and color strength is strong, but the fastness is not good enough and the distribution is uneven. 81 Addition of nonionic surfactant, octaethyleneglycolmono-n-dodecyl ether, can improve the low affinity of chitosan for wool and the conformation of chitosan in solution and on the application of chitosan to wool. The color strength and color fixation of fabrics are also enhanced. 81 To practice the use of safe, biodegradable, and eco-friendly natural dyes, wool fabrics pretreated with chitosan have a high dye uptake and good colorfastness after dyeing when dyed with natural dyes, such as henna, Thian King, natural dyes extracted from the valves of capsules of the cotton plant, and so forth.34,35,82
Application of chitosan in printing can also improve dye uptake and impart comparable colorfastness to textiles. Chito-san possesses water-binding capacities and reactive properties due to the presence of -NH2 and -OH groups; therefore, it can be used as thickener and binder in textile printing.21,83-85 After chitosan is blended with other appropriate auxiliaries, the hand of printing textiles is improved.21-23 Abdou et al. blended chitosan with gelatinized starch in textile screen printing. 23 Both color strength and colorfastness of printed fabrics are enhanced. This is because the homogeneous molecular structure of chitosan and starch exhibits pseudoplastic behavior in printing pastes. Interactions between the hydroxyl groups of starch and the amino groups of chitosan result in the formation of an edible coating with a good film forming and mechanical properties. 23 Chitosan also can be used in digital inkjet printing to replace the conventional thickener, sodium alginate, for pretreatment of fabrics, and to improve the color-fastness and outline sharpness of printed fabrics.36,86,87
Functional Finishing
Antimicrobial Finishing
Chitosan possesses a positive charge due to the presence of amino groups, which is advantageous for growth and attachment of cells. Bacteria are generally considered to be negatively charged, which can result in a positive inhibition effect when the bacteria and chitosan are in contact with each other. 12 Antimicrobial mechanisms of chitosan consist of 1) the cationic amino groups of chitosan binding to anionic groups of microorganisms, thus disturbing the physiological activities of the bacteria and resulting in growth inhibition; 2) the chitosan on the surface of the cell forming a polymer membrane, which prevents nutrients from entering the cell; and 3) chitosan of lower molecular weight entering the cell through pervasion.2,12,22,37-39 The antimicrobial activity of fabric finished with chitosan is not proportional to the degree of deacetylation of chitosan. 88 However, one limitation of chitosan used in textiles is that the fabric hand is adversely affected by chitosan treatment, especially by high molecular weight chitosan.
The relationship between antimicrobial activity and molecular weight of
chitosan has been studied.37,39 Studies show that the antimicrobial activity of
chitosan is related to both the molecular weight of chitosan and the strains
of bacteria tested. The antimicrobial activity of chitosan against
Besides molecular weight, the antimicrobial activity also depends on the concentration of chitosan and degree of deacetylation. That is, antimicrobial activity strengthens as the chitosan concentration increases, when its molecular weight is below 305 kDa and when the degree of deacety-lation increases.37,39,89 Another disadvantage of chitosan applied to textiles is poor laundering duration. Addition of binder or crosslinker can increase percent add-on. However, the binder may mask the amino groups in chitosan or the crosslinker may react with the amino groups in chitosan, which result in a decrease of antimicrobial activity. For the same reason, the poor hand of fabric after chitosan treatment can be improved by the addition of other auxiliaries. But the addition of chemicals must not affect the antimicrobial activity of chitosan. 22 Ferrero et al. exposed fabrics coated with chitosan to UV radiation, which resulted in high antimicrobial properties and good hand properties. Fastness can be improved by diluting chitosan with an acetic acid solution before spreading on fabrics and giving enough time for the chitosan to penetrate into fibers before curing (e.g., an impregnation time of 12 h at ambient temperature or 1 h at 50 °C).24,90
Due to its antimicrobial, biodegradability, and non-toxic properties, chitosan can be used as a preservative in sizing of yarns before weaving, antimicrobial agents for antimicrobial finishing, and deodorant agents for textiles. 91 The properties of chitosan-treated textiles, including stiffness, moisture regain, and tensile strength, can be improved by adjustment of pH, selection of chitosan with the desirable molecular weight and degree of deacetylation, addition of other suitable chemicals, and modification of chitosan through suitable methods. 22
Shrink-Proof Finishing Agent
Due to structural characteristics, wool fibers felt and shrink easily under mechanical action in the presence of water. Felting shrinkage results from the interlocking and hooking of contingent fibers due to the scales on the wool fibers. Chitosan has good film-forming properties. Accordingly, when chitosan is deposited on wool fibers, forming a layer of protective thin membrane, the interlocking and hooking between wool fibers are prevented and the directional friction effect of wool is decreased.22,25 Under acidic conditions, both wool fibers and chitosan possess a positive charge, resulting in a difficult sorption between chitosan and wool. The increased wettability and anionic character of wool fibers can enhance chitosan's sorption on wool fibers and the uniformity of its distribution. Oxidative (i.e., oxygen or air) plasma treatments can create sulfonate and carboxylate groups on wool fibers, which provide anionic groups and improve its wettability. 40 Therefore, plasma pretreatment can promote the spreading and adhesion of chitosan on wool fibers and enhance the level of shrink resistance of wool fabrics. Pretreatment of wool fibers with hydrogen peroxide under alkaline conditions can remove fatty acids on the epicuticle of wool fibers and increase the hydrophilicity of wool fibers. Moreover, addition of anionic surfactant and crosslinker, as well as application of low-molecular weight chitosan, especially nano-chitosan, can promote the sorption and affinity between wool fibers and chitosan and increase the shrink resistance of wool fabrics.22,41,42 In addition, pretreatment with protease enzymes can partially destroy the wool fiber scales and subsequent application of chitosan greatly improves wool shrink-proofing.22,92
Textile Finishing Carriers
Due to its unique polymeric cationic character, and gel and film forming
properties, chitosan can be used as carriers for other chemicals that do not
easily adhere to textiles or are bioactive, such as drugs, proteins,
antimicrobial agents, rmones, dyes, flame retardants, phase change
materials, and fragrances. This involves microencapsulation or
nanoencapsulation in which chitosan is a shell material acting as barrier
walls for solids or liquids. These encapsulated solid or liquid materials
can be imparted to textiles in the form of tiny solid particles.26,43,44,54,93-103 Encapsulation offers a
controlled and sustained release system for drugs, fragrances, and other
functional finishing agents. Meanwhile, fabrics finished with encapsulation
technology avoid the cytotoxicity of some finishing agents. El-Feky finished
wound dressings with silver sulfadiazine-loaded chitosan nanocapsules to
prevent injured skin from infection, which could extend to over 24
h.
44
Yip et al. prepared chitosan microcapsules loaded with clotrimazole or
miconazole, which can be applied onto bandages or socks to provide an
antifungal drug continuous-release system for
Besides imparting functional properties to fabrics, chitosan can be used for wastewater treatment as carriers of metal ions, dyes, and auxiliaries.2,5,12,30,96,104,105 Chitosan, as a naturally occurring material, has distinct advantages for use in filtration system over other current technologies, because it is environmentally friendly and can filter out or neutralize microbes and toxic metal ions, such as Cu (II), Cd (II), Ag (I), Zn (II), Pb (II), Fe (III), and Mn (II), in wastewater through chela-tion.30,106 Chitosan nonwoven mats possess a large surface area to mass ratio, which is beneficial in filtration applications. 5 Chitosan also has a high affinity for many dyes, including disperse, direct, reactive, acid, vat, sulfur, and naphthol dyes, due to the presence of cationic amino and hydroxyl groups, as well as having a similar structure to cellulose.2,107 The cationic amino groups of chitosan can absorb anionic dyes, auxiliaries, and other chemicals under acidic conditions, while the amino and hydroxyl groups are main receptor sites for dye binding through chelation.2,30,104,108
Conclusion
Chitosan's use in the textile industry includes spinning, weaving, dyeing, printing, and finishing. For spinning, chitosan can be mixed with other polymer-forming spinning solutions. Chitosan-based fibers possess antimicrobial and hemostatic properties, which makes it popular in the medical industry for wound dressings. For weaving, addition of chitosan in sizing makes the size solution stable due to its antimicrobial properties. For dyeing and printing, pretreatment of fabrics with chitosan increases the uptake of dyes and enhance the color strength as well as colorfastness. As a thickener in printing, chitosan enhances pattern sharpness. For functional finishing, chitosan coatings and use with encapsulation technology imparts numerous special functional properties to fabrics, including antimicrobial properties, shrink resistance, fame resistance, and fragrance. In addition, chitosan can be used in wastewater treatment to remove metal ions, dyes, and auxiliaries.
Chitosan is a natural, biodegradable polymer. Therefore, it has long-term potential for application in the textile industry due to its environmentally-friendly properties.
Footnotes
Acknowledgement
The authors would like to thank the financial support of the Hong Kong Polytechnic University, Natural Science Foundation of China (Grant No. 51703084), Natural Science Foundation of Jiangsu Province of China (Grant No. BK20150134), National First-class Discipline Program of Light Industry Technology and Engineering (Grant No. LITE2018-21), and 111 Project (Grant No. B17021).
