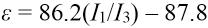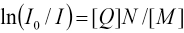Abstract
Coomassie brilliant blue G250 (CBBG250) is used in textile, printing, and biotechnology applications. Fluorescence probe, UV-Vis spectrophotometric, and electrical conductivity methods were used to study the interactions between CBBG250 and the surfactant Tween 20 in aqueous solution. The fluorescence intensity ratio of pyrene (I3/I1) in the mixed system showed that I3/I1 increased significantly when the concentration of Tween 20 was greater than the critical micelle concentration (cmc, 5.25 x 10−5 mol/L). The micropolarity of the association system decreased as the concentration of CBBG250 in the solution increased. All results show that an interaction between CBBG250 and Tween 20 existed. Thermodynamic parameters were then determined, showing that the association was a spontaneous process that was driven by both enthalpy and entropy.
Introduction
Surfactants are widely used in textile, printing, photosensitization, 1 ion detection, 2 dyeing, chemical production, and oil extraction. 3 Many researchers focus on the interactions between surfactants and macromolecules.4–6 These interactions can affect the properties of the system; the rheological properties can significantly change.7,8 There are many different methods for studying these interactions,9–12 including surface tension, conductance, viscosity, light scattering, solid state membrane electrodes, dye solubilization, and computer simulations.
The study of interactions between surfactants and dyes in aqueous solutions is important for textile dyeing processes as well as for chemical research. 13 UV-Vis spectroscopy is a common method of analysis and detection, and is also an effective technique for characterization. 14 Most previous studies used typical methods to study the interaction between dyes and surfactants. However, the use of fluorescent probe methods used in this research has rarely been reported. Steady-state fluorescence probe technology is simple and convenient;15–19 the substances are labeled by adding a fluorescent dye (probe) to the system. 20 In general, macromolecules, such as proteins, can be used directly due to the endogenous fluorescence of their molecules; for systems where the molecule itself does not fluoresce, such as surfactants, a fluorescent probe is needed.21–24
The hydrophobic probes commonly used in aqueous media are pyrene and its derivatives. The solubility of pyrene in water is very small, and the fluorescence spectrum of pyrene monomer shows five peaks. 25 The ratio (I3/I1) of the fluorescence intensity of the third peak (384 nm) to the first peak (373 nm) strongly depends on the polarity of the environment the pyrene molecules are in.26–28 The fluorescence intensity of Coomassie brilliant blue G250 (CBBG250) is weak. Its presence alone is not enough to clearly indicate its interaction with surfactants. Therefore, pyrene can be used as a fluorescent probe to study the interactions between CBBG250 and Tween 20.
CBBG250 is commonly used in the determination of protein. 29 Tween 20 is widely used as an emulsifier, a dispersant, a solubilizer, and a stabilizer. Tween 20 is a nonionic surfactant, and CBBG250 is an anionic dye. Studying the interaction between Tween 20 and CBBG250 provides considerable value for exploring the interaction of dyes with surfactants in industrial production and textile printing and dyeing, especially in the process of dyeing, decolorizing, and dye wastewater treatment.30–32 In this research, we studied the interactions between the nonionic surfactant Tween 20 and CBBG250 and determined the critical micelle concentration (cmc). A series of thermodynamic properties of the mixed dye-surfactant system were obtained by the fluorescence probe, UV-Vis spectrophotometric, and electrical conductivity methods.
Experimental
Materials
CBBG250 dye (C.I. 42655, 98%) was obtained from Sinopharm Chemical Reagent Co. Ltd. (Fig. 1). Tween 20 surfactant (CP, 99.5%) was obtained from Yantai Chemical Industry Co. Ltd. The fluorescent probe pyrene (95%) was purchased from Saen Chemical Technology Co. Ltd. The quencher acetophenone (99.5%) was obtained from Tianjin Kaixin Chemical Industry Co. Ltd. All solutions were prepared with high-purity deionized water.
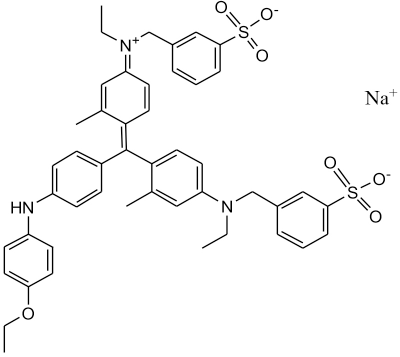
Structure of Coomassie brilliant blue G250.
Apparatus and Methods
UV-Vis Spectral Measurements
A LabTech UV-2100 UV-Vis spectrophotometer (Shanghai Yuanxi Instrument Co. Ltd) was used to measure the absorption spectra, with a wavelength accuracy of ±0.3 nm. A series of aqueous solutions with various concentrations of Tween 20 ranging from 0 to 1.8 × 10–4 mol/L and a dye concentration of 2 × 10–5 mol/L were prepared for spectral measurements.
Electrical Conductivity Measurements
A electrical conductivity meter (DDS-307, Shanghai Tianmei Scientific Instruments Co. Ltd., cell volume of 1.0 cm3) was used, which was equipped with a DJS-1T plastic electrode (measuring error: ±1.0% full scale). Dye-surfactant solutions containing 2.0 × 10–5 mol/L of dye and 1.0 × 10–5 to 1.2 × 10–4 mol/L Tween 20 were prepared.
Fluorescence Measurements
A stock solution (5 × 10–6 mol/L) of the fluorescence probe, pyrene, was prepared. The quencher, acetophenone, was dissolved in absolute ethyl alcohol at a concentration of 1 × 10–3 mol/L.
Fluorescence intensities were determined using a F-7000 fluorescence spectrophotometer (Tianmei Scientific Instrument Co. Ltd.) equipped with a thermostated cell unit (25 °C). Excitation and emission wavelengths used were 335 and 345 nm, respectively, and the excitation and emission slits used were 5 and 2.5 nm, respectively. The scan speed was 1200 nm/min and each sample was measured three times.
Results and Discussion
Surfactant and Dye Interactions
UV-Vis Spectra
As shown in Fig. 2, successive additions of Tween 20 into a CBBG250 solution produced a bathochromic shift and the intensity initially fell and then rose. Moreover, as the Tween 20 concentration increased, the maximum absorption value of CBBG250 increased. This could be because the binding degree of Tween 20 to CBBG250 through hydrogen bonding increased with increased Tween 20 concentrations, which lead to increased delocalization degree of the system and decreased electron transition energy. This is also the main reason for the bathochromic shift in the spectra.
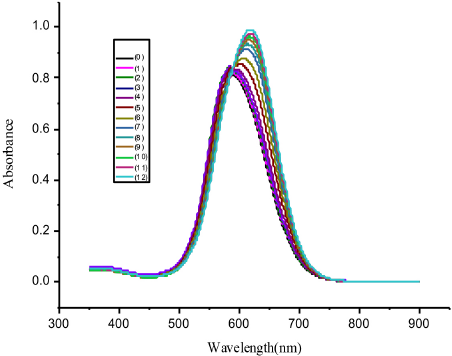
The spectra of CBBG250 in Tween 20 solutions of various concentrations. (0) 0, (1) 4.0 × 10–6, (2) 6.0 × 10–6, (3) 1.0 × 10–5, (4) 2.0 × 10–5, (5) 4.0 × 10–5, (6) 6.0 × 10–5, (7) 8.0 × 10–5, (8) 1.0 × 10–4, (9) 1.2 × 10–4, (10) 1.4 × 10–4, (11) 1.6 × 10–4, and (12) 1.8 × 10–4 mol/L.
Fig. 3 shows the relationship between the maximum absorption peak intensity and the corresponding wavelength for various concentrations of the Tween 20 aqueous solution. The change in absorbance value behavior differed on both sides of the concentration range from 2.0 × 10–5 mol/L to 6.0 × 10–5 mol/L. This phenomenon can be attributed to the micellar formation.
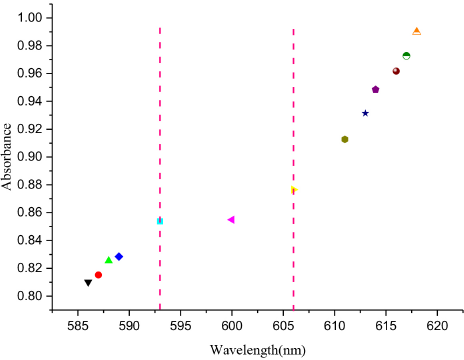
The absorbance of CBBG250 at the maximum absorption peak for various concentrations of Tween 20. (From left to right: 0, 4.0 × 106, 6.0 × 106, 1.0 × 105, 2.0 × 105, 4.0 × 105, 6.0 × 105, 8.0 × 105, 1.0 × 104, 1.2 × 104, 1.4 × 104, 1.6 × 10–4, and 1.8 × 10–4 mol/L).
Electrical Conductivity
To study the role of the cmc value in this surfactant-dye system, the electrical conductivity of Tween 20 in the presence of CBBG250 was measured at 298.15 K. Because nonionic surfactants have little electrical conductivity, the cmc value of nonionic surfactants cannot be directly measured. By adding the anionic dye CBBG250, the cmc of the nonionic surfactant Tween 20 can now be measured by the electrical conductivity method. The data are shown in Fig. 4.
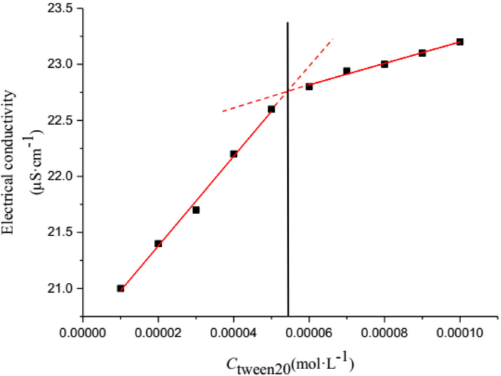
Electrical conductivity of CBBG250/Tween 20 aqueous solution at 298.15 K.
According to the electrical conductivity fitting method, 33 cmc values can be obtained from linearly-fitted electrical conductivity plots before and after the infection point. We determined that the cmc value of Tween 20 in the mixed system was 5.25 × 10–5 mol/L, which was consistent with literature results. 34 This value also falls within the range of cmc values determined by us using the UV-Vis spectrophotometric method.
Fluorescence Spectroscopy
The position of the probe in single surfactant micelles is dependent upon the probe's polarity. Probes with different polarities will solubilize in different areas of the micelle, as shown in Fig. 5. 35 Because of the low solubility of hydrophobic probes in water, the fluorescence intensity of the system is very low, but its solubility in the hydrophobic environment of the micelle is significantly greater, and the fluorescence intensity of the system is accordingly greater.
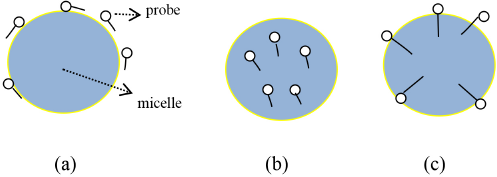
The position of the probe with the micelle. (a) Adsorbed on the interface; (b) into the micelle core; and (c) insert the fence layer.
Using the fluorescent probe, the formation of the hydro-phobic micelle area was verified by measuring the change of the fluorescence intensity of the system (i.e., the increase of the fluorescence intensity indicates that the aggregates were formed in the solution, and the probe molecules were transferred from the aqueous phase into the hydrophobic micellular domains, so the aggregation behavior of the amphiphilic dye molecules can be determined.
We investigated the fluorescence intensity of pyrene in a single surfactant solution at room temperature and in mixtures containing CBBG250 (Fig. 6a). The color of the CBBG250 solution changed after the addition of Tween 20 (Fig. 6b). Initially, the value of I3/I1 for the two solutions increased with increasing concentrations of Tween 20, after which an infection point appeared, corresponding to the cmc value of the two systems. The ranges for the corresponding solutions in the absence of CBBG250 and in the presence of CBBG250 were 3 × 10–5 to 5 × 10–5 mol/L and 5 × 10–5 to 7 × 10–5mol/L, which agreed with the results of the UV-Vis spectrophotometric and electrical conductivity methods. With increased concentrations of the Tween 20, the I3/I1 values of two solutions all increased. But the value of I3/I1 with CBBG250 was significantly less than that without CBBG250. This demonstrated that CBBG250 could interact with Tween 20 and weaken the interaction between Tween 20 and pyrene, which resulted in a significant decrease of I3/I1. When the concentration reached 5 × 10–4 mol/L, no additional CBBG250 bound to the Tween 20. The influence of CBBG250 on the interaction between Tween 20 and pyrene in the solution can be ignored, so the I3/I1 of two systems are basically consistent.
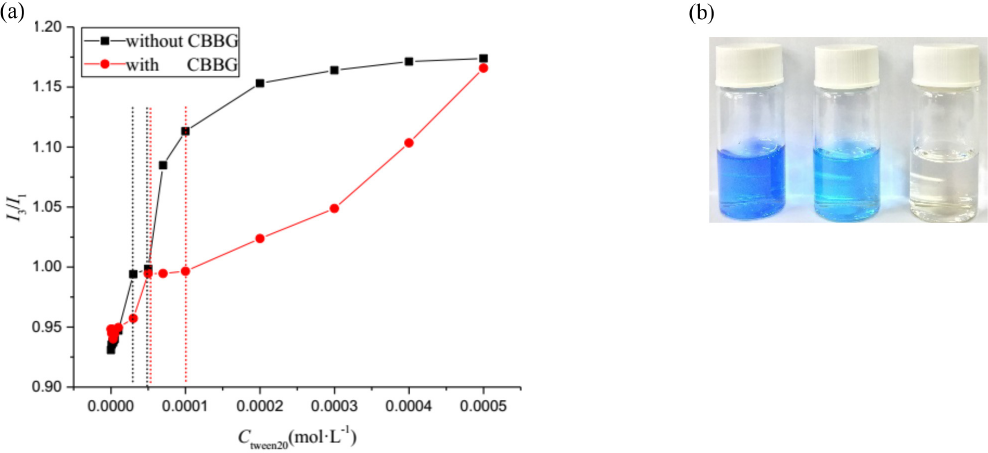
(a) The I3/I1 of pyrene at various concentrations of Tween 20. (b) From left to right: appearance of single CBBG250 solution, CBBG250 and Tween 20 mixture, and aqueous solution, respectively.
From the above, compared with the single Tween 20 aqueous solution, the association of CBBG250 with Tween 20 influenced the formation of micelles and significantly affected the fluorescence intensity of pyrene.
Micropolarity
The ratio of I1/I3 in the fluorescence spectrum of pyrene has a linear relationship with the micropolarity ε of the environment (Eq. 1) in which pyrene is located. 36
The larger the I1/I3 value, the greater the ε value, and the stronger the polarity of the pyrene molecule in the micro-environment. We studied the interactions of different types of surfactants with CBBG250. Besides the non-ionic Tween 20, the other surfactant tested was the cationic surfactant cetrimonium bromide (CTAB) (Fig. 7). The fluorescence intensities of pyrene containing CTAB and Tween 20 in the CBBG250 solutions were measured. The ε value of CBBG250-CTAB solution was greater than that of the CBBG250-Tween 20 solution. The difference in the micropolarity between the two systems became even greater as the CBBG250 concentration used was increased.
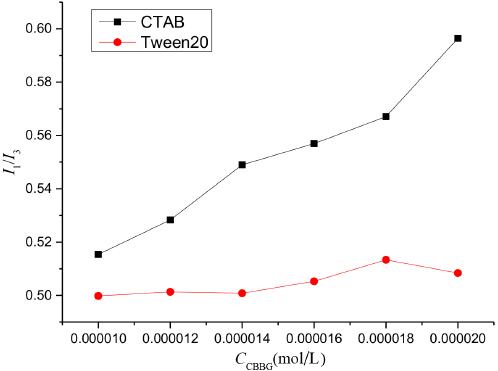
Effect of various concentrations of CBBG250 on the polarity of different mixed systems.
The resulting increased micropolarity of the association system as the CBBG250 solution concentration was increased indicated greater interaction between CBBG250 and the surfactant. The interaction between CBBG250 and CTAB was therefore stronger than that of CBBG250 and Tween 20.
Steady-state fluorescence quenching is a reliable technique for the determination of the average aggregation number (N) of surfactant micelles.37-39 Quencher (Q) is added to a solution containing a known amount of surfactant and a small amount of fluorescent probe (F); F and Q have a very high selectivity to micelles. N can be calculated based on Eq. 2.
I and I0 are the fluorescence intensities with and without quencher, respectively, [M] is the concentration of the micellarized surfactant ([M] = ct – cmc, where ct is the total concentration of the surfactant), and [Q] is the concentration of the quencher. Researchers have shown that the interaction between surfactant and polymer leads to a decrease of N, and that the interaction between anionic surfactant and water-soluble polymer is stronger than that of cationic surfactant.38,39 The stronger the hydrophobicity of the polymer, the stronger its interaction with the surfactant. 40
To study the interaction between Tween 20 with CBBG250, we used pyrene as the fluorescent probe and chose acetophenone as a quencher to measure the changes in N values for the Tween 20 system with various concentrations of CBBG250. Fluorescence spectroscopy data shows that the fluorescence intensity of pyrene dropped significantly after addition of quencher, but the ratio of fluorescence intensity I3/I1 changed little at each CBBG250 concentration tested, as can be seen by comparing lines 1 and 2 in Fig. 8. By comparing lines 3 with 1 and 2, it can be seen that as the concentration of CBBG250 increased, the slope of the I3/ I1 line containing Tween 20 was much less than that for the solution system without Tween 20, indicating that significant interaction occurred between CBBG250 and Tween 20.
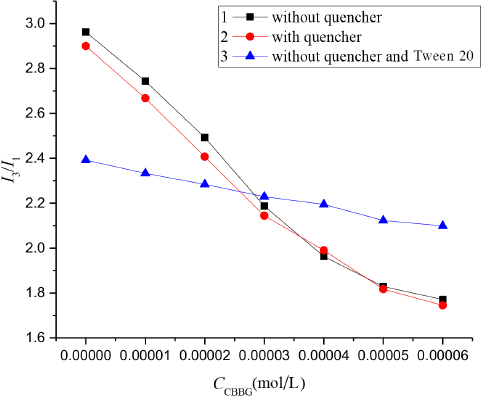
Effect of quencher on I3/ I1 of CBBG250 solutions.
We defined I3 as I0 and I, before and after adding the quencher, respectively. As shown in Fig. 9, the fluorescence intensities I0 and I decreased at increased CBBG250 concentration, then leveled of at higher CBBG250 concentrations.
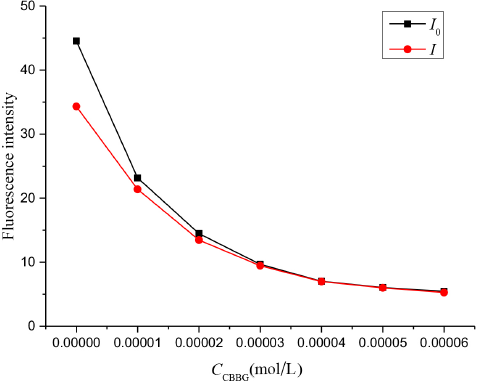
The fluorescence intensity I0 and I of CBBG250 solutions.
As shown in Fig. 10, the value of N in the surfactant system without CBBG250 was 2.5, then gradually decreased at increased CBBG250 concentrations in the surfactant-dye mixture. N represents the aggregation number between CBBG250 and surfactant and not the number of micelles formed. The changes in the N value in the mixture can also reflect the interaction between Tween 20 and CBBG250.
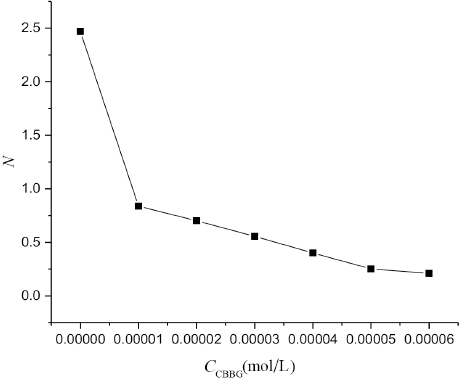
Effect of CBBG250 on micelle aggregation number N.
Thermodynamics of Interaction
We further studied the thermodynamic interaction parameters between CBBG250 and Tween 20 by using the surfactant-dye association interaction (SDAI) model.29,41,42
Molar Absorptivity of CBBG250
Various concentrations of CBBG250 were prepared and the measured absorbance at 635 nm was determined by UV-Vis spectrophotometer at various temperatures. It can be seen from Fig. 11 that the absorbance of CBBG250 increased with increased temperature at the same concentration, and the absorbance of CBBG250 increased with increased CBBG250 concentration at the same temperature.
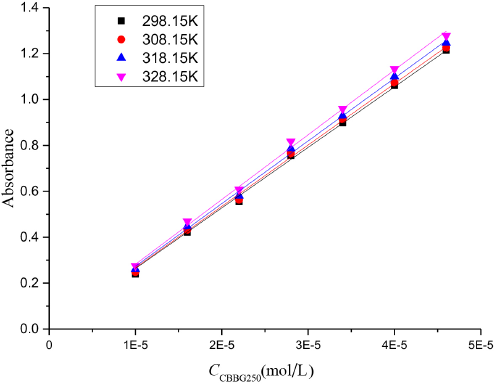
The absorbance of various concentration of CBBG250 in aqueous solutions at various temperatures.
According to the Beer-Lambert law, the measured data were linear. The molar absorptivity of CBBG250 aqueous solutions εCBBG250 was obtained from the slope of lines at different temperatures, which are listed in Table I.
Molar Absorptivity of CBBG250 at Various Temperatures

Thermodynamic Interaction Parameters
Based on the SDAI thermodynamic model, we prepared a series of CBBG250/Tween 20 aqueous solutions in seven 25-mL volumetric flasks, with a CBBG250 concentration of 2.0 × 10–5 mol/L and concentrations of Tween 20 varying from 1 × 10–3 to 7 × 10–3 mol/L. By measuring the absorbances of these solutions, linear results were obtained as shown in Fig. 12.
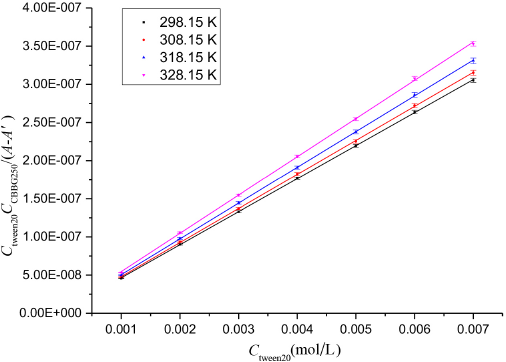
Plot of CTWEEN20 CCBB250/(A - A′) versus CTWEEN20 at various temperatures.
According to Eq. 3 of the SDAI model, the molar absorptivity εc of the association compound and the association equilibrium constant K of the reaction were calculated from the slope and intercept of these straight lines (Table II).
Molar Absorptivity εc and the Association Equilibrium Constant K at Various Temperatures
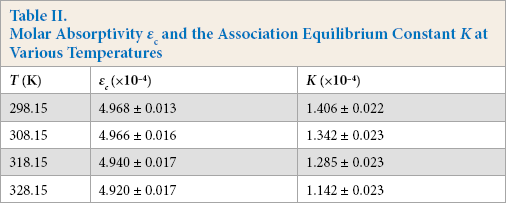
The data show that as the temperature increased, the molar absorptivity εc and the equilibrium constant K of association products all decreased. This indicates that the interaction between Tween 20 and CBBG250 will be inhibited by temperature increases. Based on Le Chatelier's principle, 43 it was concluded that an exothermic reaction occurred between them.
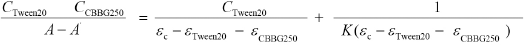
The relationship between the Gibbs energy change ΔG and the equilibrium constant K is based on Eq. 4.
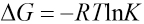
ΔG values at various temperatures were calculated and shown in Fig. 13. These ΔG values were used in Eq. 5 to obtain ΔH and ΔS values from the intercept and slope. The enthalpy change ΔH and the entropy change ΔS for the association process were calculated as -5.463 kJ/mol and 61.22 J/mol/K, respectively.
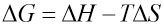
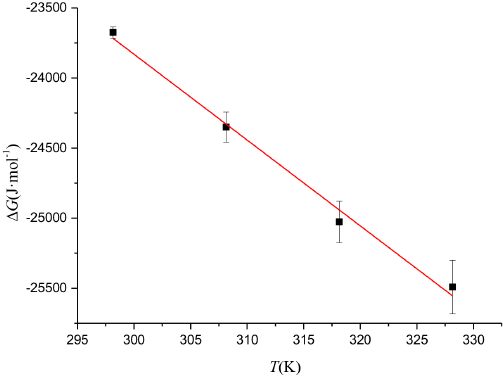
The relationship between ΔG and T for the CBBG250/Tween 20 aqueous solution.
The thermodynamic parameters at various temperatures are listed in Table III.
Thermodynamic Parameters for the Dye-Surfactant Interaction
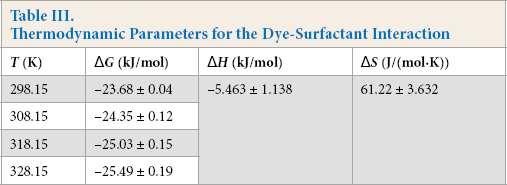
All ΔG changes were less than zero, indicating that the association reaction was spontaneous. The enthalpy change ΔH < 0 and the entropy change ΔS > 0 show that this association process was both enthalpy and entropy driven.
Conclusions
Spectra of CBBG250 were obtained that changed significantly at various concentrations of Tween 20. The cmc range of the dye-surfactant system was basically the same as determined by the fluorescence probe, UV-Vis spectrophotometric, and electrical conductivity methods. The fluorescence intensity ratio of pyrene (I3/I1) in the mixed system showed an increase in I3/I1 when the concentration of Tween 20 was greater than the cmc value. This indicated that the association of CBBG250 with Tween 20 influenced the formation of micelles and significantly affected the fluorescence intensity of the pyrene. By studying the polarities of the mixed systems, it was observed that various interactions between dye and surfactants occurred. The aggregation number N gradually decreased with increased CBBG250 concentration in the mixed system, indicating that changes of the N value in the mixed system also reflected the interaction between Tween 20 and CBBG250. The interaction parameters between Tween 20 and CBBG250 were obtained by use of the SDAI model. From these thermodynamic results, the association process was observed to be spontaneous and was enthalpy and entropy driven. Since the study of a one dye-surfactant system is a complex process, we will continue to study other systems to provide a richer background for textile dyeing and printing and dye polluted wastewater treatment.