Abstract
The aim of this study is to research the effects of degree of deacetylation (DD) of chitosan on the antimicrobial properties of nylon 6,6 fabrics. Chitosan (70% DD) was deacetylated to 98% DD. Subsequently, nylon 6,6 fabrics with a warp-knitted powernet structure were treated with 1% chitosan (70% or 98% DD) by separately crosslinking with 0.1% dimethylol dihydroxyethyleneurea (DMDHEU). Treated samples were characterized using X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM), and Fourier transform infrared spectroscopy (FTIR) analyses. Antimicrobial tests showed that all treated samples had very good antimicrobial activity even after 10 washes. Fabrics treated with lower DD (70%) chitosan exhibited better antimicrobial activity.
Introduction
Chitosan and its parent compound, chitin, are naturally occurring β-(1,4)-linked linear aminopolysaccharides. Chitosan, though less prevalent in nature, is a useful and easily accessible derivative of chitin. Chitosan is the deacetylated derivative of chitin, the second most abundant polysaccharide found on earth next to cellulose. Both polymers are biodegradable, renewable resources with versatile chemical and physical properties. As such, they are the subject of active scientific and commercial scrutiny1 Chitosan is a high molecular weight heteropolysaccharide composed mainly of β-(1,4)-2-deoxy-2-amino-D-glucopyranose units, and partially of β-(1,4)-2-deoxy-2-acetamido-D-glucopyranose (Fig. 1). 2 – 4
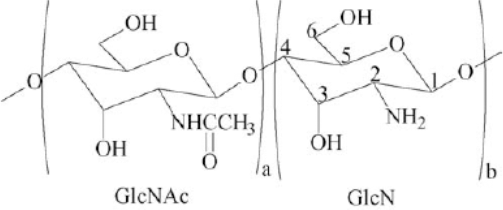
Chemical structure of 2-acetamido-2-deoxy-D-glucopyranose (a, GlcNAc) and 2-amino-2-deoxy-D-glucopyranose (b, GlcN) units joined by β-(1,4) glycosidic bond. The a and b units distinguish chitin from chitosan.
Chitosan is an N-deacetylated product of chitin found as the main component in the shells of crustaceans (e.g., crabs, lobsters, and shrimp). It is also found in exoskeletons of mollusks, insects, and in the cell walls of some fungi.5-6 Structurally, chitin is an insoluble, linear mucopolysaccharide consisting of
The only chemical difference between chitin and chitosan are the acetyl groups. The presence of the acetyl groups causes more high occupancy H-bonds along the inter-sheet direction of chitin model. 12 , 13 In contrast, as there are few acetyl groups within the chitosan model, the H-bond occupancy along two directions is similar. 14 Degree of deacetylation (DD) is a percentage measurement of free amine groups along the chitosan backbone. 15 , 16 There are several methods available to determine the DD of chitosan. Czechowska-Biskup et al. used H-nuclear magnetic resonance (NMR) spectroscopy, titration methods, UV-Vis spectroscopy, and infrared spectroscopy 17
In the present work, 70% DD chitosan was further deacetylated to 98% DD. Next, nylon 6,6 materials were treated with 98% or 70% DD chitosan. Finally, the resulting chitosan-nylon 6,6 fabrics were characterized using X-ray photoelectron spectroscopy (XPS), scanning electron microscopy (SEM), and Fourier transform infrared (FTIR) spectroscopy analyses. Antimicrobial tests were then conducted.
Experimental
Materials
Several pieces of nylon 6,6 raschel warp knit fabric with inlaid elastane yarns (average weight, 3.00 g; BSN Medical Inc.), chitosan (viscosity average MW = 1.0 × 106, 70% DD, Sigma-Aldrich), and the other reagents used were analytically pure.
Fabric Treatment
Chitosan Preparation
Commercial chitosan particles were first rinsed with methanol/deionized (DI) water (50:50) and the excess water was filtered using a nylon fabric to prevent contamination. The particles were dried at 80 °C for 30 min. Hydrochloric acid was used as the solvent for chitosan during DD determinations.
Chitosan DD Determination
The DD of the rinsed commercial chitosan, which was calculated using Eq. 1, was measured by acid-base titration of chitosan in HC1 solution.
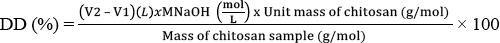
To determine DD, 0.0976 g of the rinsed commercial chitosan was completely dissolved in 10 mL of 0.1N HC1 solution and was titrated with a 0.1N NaOH solution. The initial volume was 16.7 mL. After titration with 0.1 N NaOH, the final volume was 20.9 mL and the resulting DD was 70%.
Further Deacetylated Chitosan Preparation
Commercial chitosan (2 g) was dissolved in 150 mL of 50% w/w NaOH under nitrogen. The solution temperature was kept between 40∼120 °C using a water bath. The nitrogen pressure was adjusted to 6 kPa (6 psi). A three-neck round-bottom glass flask was used during the experiment. One of the necks of the glass flask was fitted with a nitrogen bubbler to maintain an inert atmosphere, another neck was closed with a stopper, and the third neck was used to measure and control the temperature of the solution during the experiments. After reaching 120 °C, the solution was cooled down to 50 °C. The samples were then washed with DI water and then with methanol, until the pH reached 7. Finally, all samples were left to air dry for two days.
Further Deacetylated Chitosan DD Determination
The further deacetylated chitosan (0.0940 g) was dissolved in 10 mL of 0.1N HC1 solution. Next, 10 mL of DI water was added. The temperature was kept constant at around 20.4-20.5 °C using a water bath. A few drops of phenolphthalein were added into the chitosan HC1 solution. Then the solution was titrated with 4 mL of 0.1N NaOH.
After titration, the final volume was measured as 8.7 mL. Using Eq. 1, the final DD of the further deacetylated chitosan was 98%.
Chitosan Application to Nylon 6,6 Fabrics
Greige nylon 6,6 (150 g) fabrics were weighed and scoured (with 0.5 g/L sequestering agent, 2 g/L nonionic, octylphenol ethoxylate surfactant penetrating agent, and 2 g/L sodium carbonate) in a 1:20 liquor ratio (LR) for 60 min at 80 °C. The cloth was then rinsed in water at 38 °C for 90 min and dried in a convection oven at 50 °C for 30 min.
Chitosans (1.0 wt %, 70% and 98% DD) were each separately added to 2.0% lactic acid solutions, and were constantly stirred at 20.5 °C until each chitosan was fully dissolved. Dimethylol dihydroxyethyleneurea (DMDHEU, 0.1% w/v) was then reacted with each 1% w/v chitosan (70% and 98% DD) solution separately, followed by stirring for 24 h at 20.5 °C.
Each solution prepared above was then used for binding chitosan onto the nylon 6,6 fabrics. For each experiment, 3.0 g of fabric was weighed and a 1:16 LR was used. The samples were placed into each solution separately. The treatment lasted for 20 min at 20.5 °C. The samples were then dried in a convection oven for 30 min at 50 °C and subsequently cured in an oven for 2 min at 130 °C. Finally, the samples were weighed using an electronic balance.
Test and Analytical Methods
Antimicrobial Activity
Antimicrobial activity was determined using AATCC Test Method (TM) 100: 2004 “Assessment of Antibacterial Finishes on Textiles” Standards, using
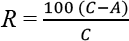
XPS
The surface properties of nylon 6,6 bound with chitosan were characterized though XPS using an XPS Specs system with a PHOIBOS 150 analyzer. XPS spectra are obtained by irradiating a material with a beam of X-rays while simultaneously measuring the kinetic energy and number of electrons that escape from the top 1 to 10 nm of the material being analyzed.
SEM
SEM analyses were performed to observe the surface changes on treated and untreated fibers. A JEOL JSM 5900-LV SEM was used with an accelerating voltage of 15 kV. Specimens were mounted on aluminum stubs using conductive carbon tape. They were then coated with gold/palladium using a Hummer 6.2 Sputter Coating System (Anatech) to obtain a conductive coating around 100 Å thick. Untreated control samples were scanned for comparison.
FTIR
FTIR was performed on the samples using a Nicolet Nexus 470 FTIR spectrophotometer with an AVATAR Omni Sampler in attenuated total reflectance (ATR) mode. The specimen was mounted onto the surface of the germanium crystal in the ATR assembly. A total of 64 scans were aggregated between 1000 and 4000 cm–1 with each spectrum having a 4 cm–1 resolution. The aggregated scans showing the absorbance across the infrared spectrum was acquired using OMNIC software.
Results and Discussion
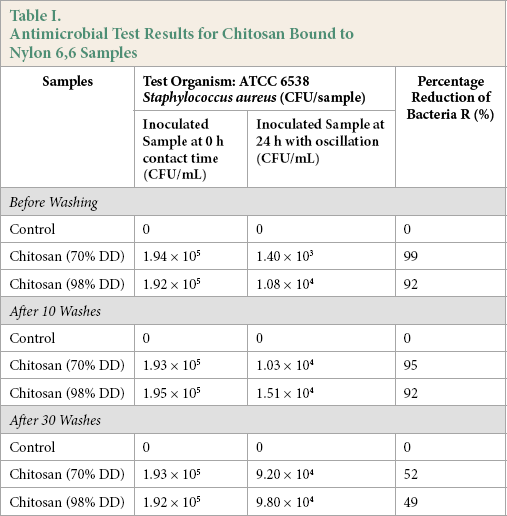
Antimicrobial Activity
The antimicrobial properties of the control and chitosan samples bound to nylon 6,6 samples are presented in Table I. The total population of
Antimicrobial Test Results for Chitosan Bound to Nylon 6,6 Samples
XPS Analysis
High-resolution XPS spectra for the C1s, O1s, and N1s regions were obtained, using a pass energy of 20 eV. Due to their insulating nature, chitosan surfaces became positively charged after the emission of photoelectrons, resulting in a broadening of the spectral lines and drift toward higher binding energies. Thus, the binding energies of the photoelectron peaks were calibrated, assigning a binding energy of 285.0 eV to the aliphatic carbon (-CH2-) C1s peak, present as a carbon surface contaminant. Element atomic percentages were calculated from the integrated intensities of the XPS peaks, taking into account the atomic sensitivity factors of the instrument data system.
The survey spectra of the two chitosans confirmed the presence of carbon, oxygen, and nitrogen (Figs. 2 and 3). The C peak at 285.0 eV was mainly assigned to the carbon surface -CH-contaminant, but also to -C-NH2 chemical bindings, given that amines are reported to induce small chemical shifts, namely of 0.6 eV. 1 The peaks at 284.9 eV was assigned to -C-O, -C-OH, and -C-N-C=O, and the peak at 285.0 eV to -O-C-O and -N-C=O chemical bindings.20–22
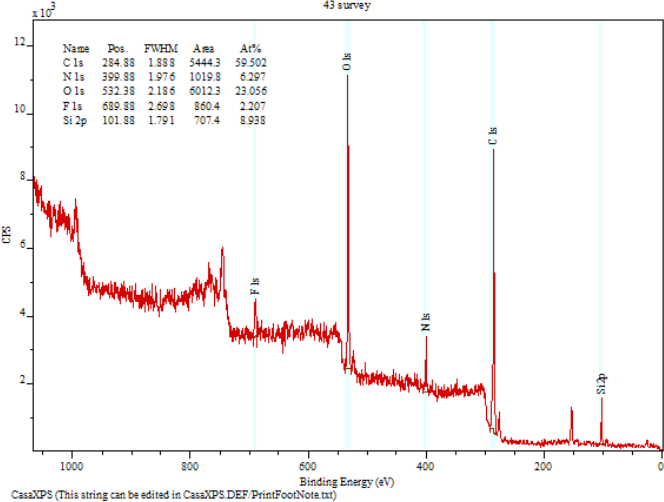
Chitosan (70% DD) bound onto nylon 6,6 fibers.
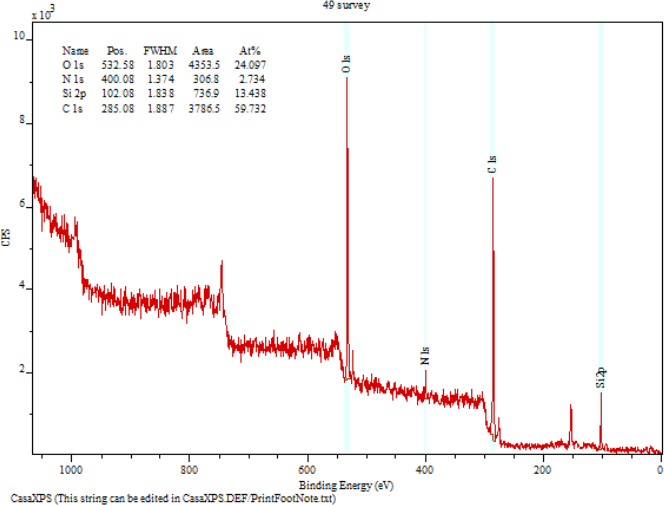
Chitosan (98% DD) bound onto nylon 6,6 fibers.
The resolved N1s spectra of chitosan samples are also shown in Figs. 2 and 3. Two peaks were identified. The peak at 399.9 eV was assigned to -N-C=O and -NH2 chemical bindings, 20 while the peak at 400.0 eV was assigned to amino groups in the ammonium form (NH3+). 23 Both forms, -NH2 and -NH3+, were likely to be present in chitosan samples, taking into account the pKa of chitosan amine groups (ca. 6.5). 24 The crosslinking of chitosan onto nylon 6,6 was identified to be through the formation of the salt structure (-NH3+ …−OOC-) between the surfaces of hydrolyzed nylon 6,6 and the chitosan layer.
SEM Analysis
SEM analyses are shown in Fig. 4. These revealed that the fibers on chitosan-treated nylon 6,6 samples appeared more compact and more aligned when compared to the control group. Surface morphology for the treated samples was not significantly different.

Morphology of the nylon 6,6 fiber samples. (a) untreated nylon 6,6, (b) after bonding to chitosan (70% DD) with DMDHEU, and (c) after bonding to chitosan (98% DD) with DMDHEU.
FTIR Analysis
The total reflection FTIR spectra of the chitosan-treated nylon 6,6 samples are shown in Fig. 5. Chitosan showed characteristic bands at 3350 cm–1 (O-H stretch and N-H stretch (primary amide), and 2929 cm–1 and 2874 cm– 1 (aliphatic C-H stretch). Chitosans saccharide structure is indicated by glycosidic linkages appearing as bands between 1152 cm-1 and 1156 cm–1 (C-H stretch).
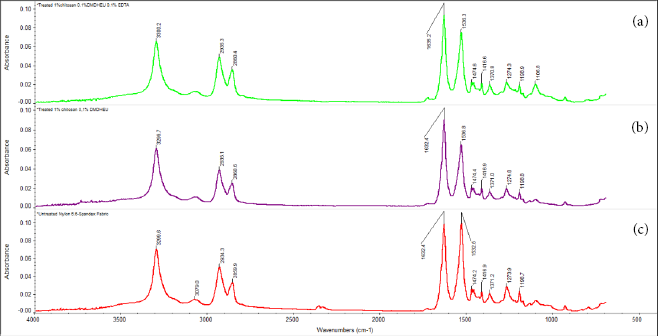
FTIR spectroscopy of (a) nylon 6,6 treated with chitosan (70% DD), (b) nylon 6,6 treated with chitosan (98% DD), and (c) untreated nylon 6,6 fabric.
Two absorption peaks at 1650 cm–1 (acetyl C=O group) and 1595 cm–1 (secondary amide and primary amine NH2) were observed. Peaks at 1425 cm–1 and 1385 cm–1 were attributed to C-H bending. Peaks at 1261 cm–1 corresponded to the acetyl amide III group. Absorption peaks at 1149-1150 cm–1were assigned to amides I and II. Characteristic chitosan peaks in the FTIR spectrum confirmed its saccharide structure. The peak at 1419 cm–1 was the joint contribution of O-H and C-H bend vibrations at the C-3 carbon.
Conclusion
Partially deacetylated chitosans (70% and 98% DD) were used to treat nylon 6,6 fabrics and the resulting samples were characterized using XPS, SEM, and FTIR spectroscopy analyses. According to XPS results, both forms, -NH2 and -NH3+, were likely to be present in chitosan samples, taking into account the pKa of chitosan amine groups (ca. 6.5). The existence of the bonded chitosan in the structures is apparent in SEM images. All treated samples showed a very good antimicrobial activity even after 10 washes. Fabrics treated with lower DD (70%) of chitosan exhibited better antimicrobial activity.
