Abstract
Various pre-treated cotton yarns were oxidized with potassium periodate. The effects of pre-treatments on oxidation were studied by quantitatively assessing functional groups, while the extent of cotton yarn degradation was determined by investigating pre-treated yarn mechanical properties and the degree of polymerization. Pre-treatments included various scouring procedures and process sequences. The type of scouring has a significant influence on the aldehyde and carboxyl groups before oxidation, while the pre-treatment sequence did not. The number of aldehyde groups was significantly influenced by oxidation and strongly dependent on the pre-treatment sequence applied. Alkaline-scoured yarns had the least number of aldehyde groups both before and after oxidation, followed by alkaline pectinase and acid pectinase scoured, regardless of the pre-treatment sequence.
Introduction
Chemical modification of polysaccharides is an interesting route for preparing new polymers with specific properties. Modification by selective or non-selective cellulose oxidation, under mild aqueous conditions, allows control of aldehyde and carboxyl group formation. 1 Selective oxidation on secondary hydroxyl groups (C-2 and C-3) is achieved using potassium or sodium periodate, while the C-6 hydroxyl group can be oxidized with (2,2,6,6-tetramethylpiperidin-1-yl)oxyl (TEMPO).2–4 Non-selective oxidation can be achieved using permanganate, sodium hypochlorite, or hydrogen peroxide.5,6
Oxycellulose serves as a useful intermediate for the manufacture of cellulose-based materials, which can adsorb certain chemicals, and are widely used for pharmaceutical applications, drug delivery, cosmetic products, polymer composites, and as metal absorbents. 7
Di-aldehyde celluloses can be used to immobilize proteins, drugs, 8 or amino polysaccharides by reaction with their amino functional groups. Periodate oxidation leads to the greatest amount of cellulose depolymerization and causes significant degradation. 9
Hydrogen peroxide, sodium hypochlorite, and sodium periodate have been used as oxidizing agents for different kinds of textile materials. The greatest number of aldehyde groups was achieved on cellulose oxidized by sodium periodate, followed by those treated by hypochlorite and peroxide. 5
Cotton oxidation is commonly applied after conventional pre-treatment processes, such as scouring, bleaching, or mercerization.5,9,10 In traditional alkaline scouring, the cuticle is completely removed. Enzymatic scouring with pectinase, through its selective action, breaks down the pectin in the cuticle and thereby assists the removal of waxes, proteins, and other non-cellulosic components.11,12Mild working conditions and the selective action of enzymes avoid degradation and aid cotton surface modification. 13
Previous research was mainly focused on the analysis of charge on oxidized cotton prepared under more aggressive conditions. Less attention has been paid to the oxidation of cotton pre-treated under milder, “green” treatment, conditions that protects both the fiber structure and the environment.
Our previous research has been devoted to evaluating the effects of various “green” pre-treatments on cotton's surface and on its non-cellulosic components. 14 The pre-treatments studied strongly influenced the surface charge of cotton fiber and reduced the non-cellulosic components, although they could be still detected on the fiber surface. 14
The objective of the present paper was to investigate the influence of various pre-treatment processes on the selective oxidation of cotton yarns with potassium periodate, including various scouring procedures (such as alkali, alkaline, or acid pectinase), and different pre-treatment sequences (mercerization-scouring or scouring-mercerization). The number of aldehyde and carboxyl groups and the amount of oxidized cotton yarn degradation were determined and compared to values obtained for un-oxidized samples.
Experimental
Materials
Plied ring-spun worsted cotton yarn, with a linear density of 30 × 2 tex and 330 twists/m, was supplied by VETEX AD Veles. Unless otherwise noted, all chemicals used were purchased from Sigma-Aldrich.
Treatment Methods
Mercerization was done on Jäegli hank mercerization equipment by rolling the hanks in a 23.5% NaOH solution and 1 g/dm3 of Subitol MEZ-N (CHT) wetting agent at 18 °C. Next, the hanks were extended to the original length by applying tension, rinsing with hot (80 °C) and cold (20 °C) water for 1 min under tension, neutralizing, rinsing again, and drying at 20 °C.
Raw and mercerized cotton yarns (as hanks) were scoured by sodium hydroxide, alkaline pectinase, or acid pectinase.
Alkaline scouring was done using 3.2 g/L of NaOH in the presence of 2 mL/L of Cotoblanc HTD-N anionic surfactant (CHT) and 1 mL/L Kemonecer NI nonionic surfactant (Kemo), at a material to liquor ratio (LR) of 1:16.6, at 100 °C for 60 min.
Alkaline pectinase scouring was carried out using 0.666 g of BioPrep 3000L per kg of material, at an LR of 1:16.6, containing 0.15 g/L of Na3PO4 (pH 9) in the presence of 1 mL/L of Kemonecer NI at 55 °C for 30 min. Then, 0.4 g/L of EDTA was added to the scouring bath and the temperature was raised to 90 °C for 15 min to stop enzymatic activity.
Acid pectinase scouring was performed with 0.625 g of NS 29048 (Novozymes) per kg material, at an LR of 1:16.6, in acetate buffer (pH 4), in the presence of 1 mL/L of Kemonecer NI, at 45 °C for 30 min. After that, 0.8 g/L of EDTA was added and the temperature was raised to 90 °C for 15 min to stop enzymatic activity.
All scouring procedures were followed by rinsing the yarns at 90 °C for 10 min, at 70 °C for 10 min, and then once with cold water to fully neutralize.
Demineralization was achieved with 0.01 M HCl, at a material to liquor ratio of 1:35, at 20 °C for 120 min. The yarns were rinsed five times with cold water for 15 min. After demineralization, the yarns were oxidized without drying. Oxidation was done with 1 g/L KIO4, at an LR of 1:50, at 60 °C for 1 h. The yarns were rinsed five times with cold water for 15 min and dried at room temperature (RT).
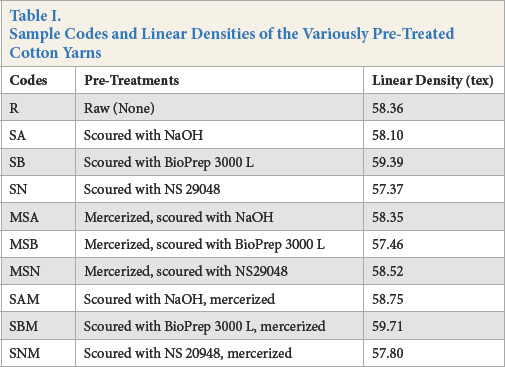
Sample codes and linear densities of the yarns used are given in Table I.
Sample Codes and Linear Densities of the Variously Pre-Treated Cotton Yarns
Testing Methods
Cellulose purity was determined by dyeing with direct Siri-uslichtblau FGG 200 % (C.I. 225) and reactive Bezactiv Blue HE-RM 133 (CHT Group) dyes. Determination of waxes, pectins, and proteins present were done by staining with Oil Red (C.I. 26125), cationic Methylene Blue (C.I. 52015) and acid Bemacide Blue GLF 200 % (CHT Group) dyes, respectively.15,16 After dyeing, values of the lightness color coordinate (
The
Determination of aldehyde groups by the tetrazolium method (TTC) was carried out according to the literature method. 10
Determination of aldehyde groups by the modified iodine method (I) was as follows. A 0.08 g cotton sample was immersed into 30 mL of 0.008 M NaOH and 10 mL of 0.001 M iodine solution. After a 3 h treatment at RT and under frequent shaking, a 25-mL portion of the liquid and 15 mL of 0.3 M HCl were titrated with 0.0075 M Na2S2O3•5H2O using a starch indicator.
Determination of carboxyl groups by Methylene Blue adsorption (MB) was as follows. 17 A 0.5 g cotton sample was treated in 25 mL of Methylene Blue solution (300 mg/L) and 25 mL of 0.2 M borate buffer (pH 8.5) for 1 h at 20 °C. Then, 25 mL of the solution was transferred to a 100-mL volumetric flask and water was added up to 100 mL. The methylene blue quantity of the liquid was determined photometrically using a calibration plot. The number of carboxyl groups was obtained using Eq. 1.
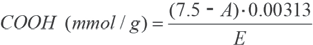
Determination of carboxyl groups by the calcium-acetate method was as follows. 18 A 1-g cotton sample was immersed into 100 mL of distilled water and 60 mL of 0.25 M calcium acetate solution for 12 h at RT with frequent shaking, which facilitated interchange reactions. Then, the solution was titrated with:
•EDTA. The calcium acetate solution (25 mL) was titrated with 0.1 M EDTA at pH 10 (NH4Cl + NH4OH buffer) using 0.1 g Eriochrome Black T (C.I. 14645) as the indicator. The quantity of carboxyl groups was calculated using Eq. 2.
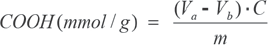
•NaOH. The calcium-acetate solution (25 mL) was titrated with 0.01 M NaOH using 2-3 drops of Thymol Blue as the indicator. The quantity of carboxyl groups was calculated using Eq. 3.
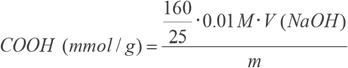
0.01 M is the concentration of NaOH used,
The results presented for aldehyde and carboxyl groups were mean values of three measurements at a confidence level of 95%.
Mechanical properties, such as tensile strength
Degree of polymerization (DP) was determined viscosimet-rically after dissolving the sample in EWNN (sodium salt of ferric tartaric acid). 19 The results presented were the mean values of three measurements at a confidence level of 95%.
Statistical Analysis
The influence of various pre-treatment processes on the selective oxidation, including various pre-treatment sequences (only scouring, mercerization-scouring, or scouring-mercer-ization) and various scouring type (alkali, alkaline pectinase, or acid pectinase) were analyzed using ANOVA analysis of variance (STATISTICA 6 program). A three-factor (2 × 3 × 3) design was used to structure the experiment, wherein the factors (independent) variables were oxidation in 2 levels (un-oxidized and oxidized), pre-treatment sequences in 3 levels (only scouring, mercerization-scouring, or scouring-mercerization), and scouring type in 3 levels (alkali, alkaline pectinase, and acid pectinase). Response (dependent) variables were the number of aldehyde and carboxylic groups determined by the method used.
Significant levels (p-values) of the ANOVA main effect are reported in this paper. ANOVA main factor and interaction plots, as well as the complete ANOVA tables, are available from the author upon request.
Results and Discussion
Functional Groups of Pre-Treated Un-Oxidized Cotton Yarns
Pre-treatment processes of cotton could have a crucial effect on its further oxidation. Therefore, in our previous work, 14 we studied changes occurring on the cotton surface, and on its non-cellulosic components, after various “green” pre-treatments. After treatments, various amounts of non-cellulosic residues caused the treated cotton samples to turn different colors, when stained with appropriate dyes. Changes were analyzed by dyeing with the two cellulose purity testing dyes, and with the three dyes used to separately determine the amounts of waxes, pectins, and proteins present.
The results showed that the greatest degree of cellulose purity and the least amount of waxes were obtained for the yarns subjected to alkaline scouring, while greater amounts of waxes and pectin residues were noted for alkaline pectinase scoured yarns, followed by those scoured by acid pectinase (Table II).
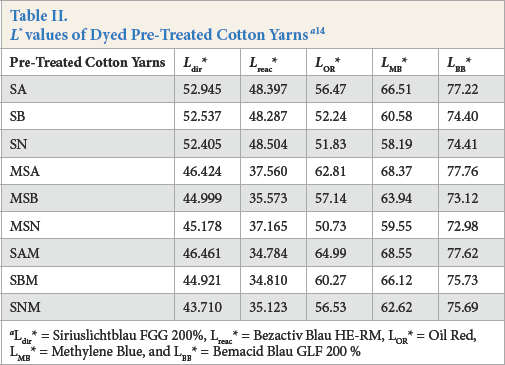
Ldir* = Siriuslichtblau FGG 200%, Lreac * = Bezactiv Blau HE-RM, LOR * = Oil Red, LMB * = Methylene Blue, and LMB * = Bemacid Blau GLF 200 %
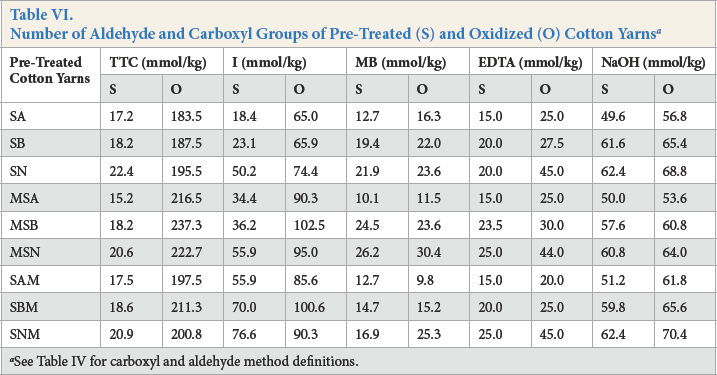
Results for the number of aldehyde and carboxyl groups on un-oxidized yarns are given in Table III. The least amount of aldehyde and carboxyl groups was obtained for SA, followed by SB and SN pre-treated cotton yarns. This trend was maintained regardless of the pre-treatment sequence. The ANOVA analysis of the main effects results confirmed that the type of scouring had a significant influence on the number of aldehyde groups obtained by TTC (p = 0.004) and I (p = 0.013) methods and carboxyl groups obtained by EDTA (p = 0.010) and NaOH (p = 0.000) methods, while the sequence of the pre-treatment was not significant (Table IV).
Number of Carboxyl and Aldehyde Groups of Pre-Treated Cotton Yarns a
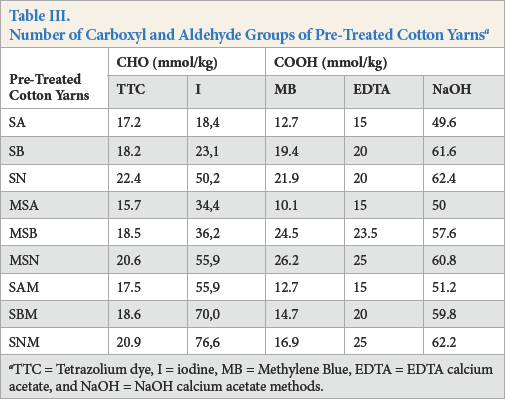
TTC = Tetrazolium dye, I = iodine, MB = Methylene Blue, EDTA = EDTA calcium acetate, and NaOH = NaOH calcium acetate methods.
In traditional alkaline scouring, proteins are hydrolyzed to lower molecular weight components, while waxes are saponified and emulsified. On the other hand, during enzymatic scouring, pectinases break down the pectin from the cuticle and thereby assist in removing waxes, proteins, and other non-cellulosic components. As a result of the mild working conditions and selective action of the enzymes, part of the non-cellulosic residues, such as waxes, pectin, and proteins, remained on the cotton surface, thus decreasing
Irrespective of the pre-treatment sequence, mercerized and scoured cotton yarns presented the same evolving trend of aldehyde and carboxyl groups as the scoured yarns. Alkaline-scoured samples showed the least number of aldehyde and carboxyl groups, followed by the enzymatically-scoured yarns using alkaline pectinase and acid pectinase (Table III). The results obtained from the ANOVA analysis of the main effects confirmed that the sequence of pre-treatment had no significant influence on the number of aldehyde and carboxyl groups (Table IV).
ANOVA of Pre-Treatment Sequence and Scouring Type on the Number of Aldehyde and Carboxyl Groups a
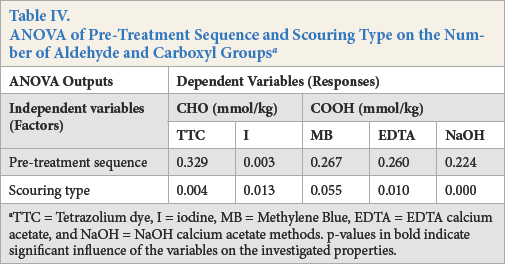
TTC = Tetrazolium dye, I = iodine, MB = Methylene Blue, EDTA = EDTA calcium acetate, and NaOH = NaOH calcium acetate methods. p-values in bold indicate significant influence of the variables on the investigated properties.
The only exception was when the aldehyde groups were determined by the I method (p = 0.003). In this case, the pre-treatment sequence was found to be of great importance. The I method is usually used for determining the number of aldehyde groups and the fine structure of cotton.22,23Solution compositions in both cases are the same, but the reaction conditions applied are different. When measuring the amount of aldehyde groups, iodine reacts with aldehyde groups and transforms them to carboxyl groups. 24 When observing the fine structure (accessibility) of cotton, three-iodine ions form a monolayer film, and therefore can be used for specific internal surface area determination. 22 In our case, iodine reacted with aldehyde groups, but probably, also made a monolayer film inside the cotton fiber surface.
High coefficients of correlation were found between
Coefficients of Correlation between
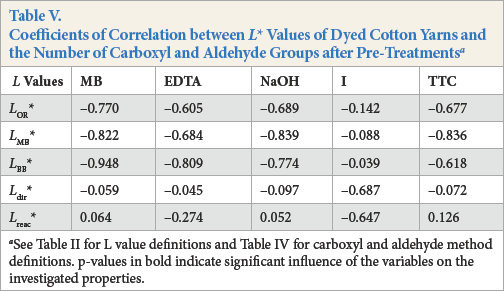
See Table II for L value definitions and Table IV for carboxyl and aldehyde method definitions. p-values in bold indicate significant influence of the variables on the investigated properties.
Functional Groups of Pre-Treated Oxidized Cotton Yarns
Aldehydes
Differently pre-treated yarns were further oxidized with potassium periodate, which quite selectively cleaves the C2-C3 bond of the anhydroglucose units, introducing aldehyde groups at those two positions.
After oxidation, the number of aldehyde groups were determined by the TTC and I methods (Table VI). Although the two methods provided different values, the increasing or decreasing trends were maintained throughout all results. TTC proved to be a more sensitive detection method, which was confirmed by the up to 9× greater values of aldehyde groups detected after oxidation. On the other hand, the increase in the number of aldehyde groups after oxidation was only up by 3× when determined by the I method.
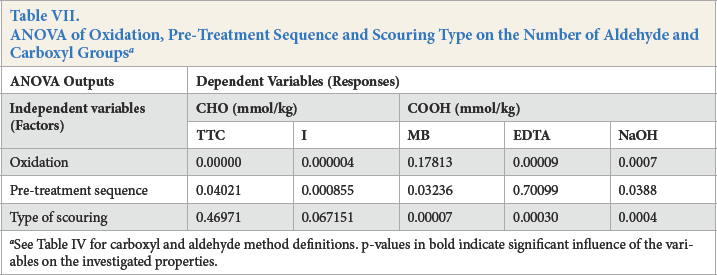
ANOVA analysis of the main effects confirmed that oxidation had a significant influence on the number of aldehyde groups, as assessed by both TTC (p = 0.000) and I (p = 0.000) methods (Table VII).
The number of aldehyde groups in the oxidized yarns followed the same evolving trend as that for the un-oxidized yarns. SA yarns presented the least number of aldehyde groups, followed by SB and SN. This trend could be observed in all sequences of pre-treatment. Although enzymatically-scoured and oxidized yarns contained a greater number of aldehyde groups than the alkaline-scoured and oxidized yarns, the latter exhibited a more significant increase in the number of aldehyde groups (in all sequences of pre-treatment) when compared to un-oxidized yarns. The greater increase in the number of aldehyde groups in alkaline-scoured and oxidized yarns (for all pre-treatment sequences) was due to a completely removed cuticle and a fully available cellulose surface for oxidation.
ANOVA analysis of the main effects confirmed that the type of scouring did not have a significant influence on the number of aldehyde groups using TTC (p = 0.46971) and I (p = 0.067151) methods, while the sequence of pre-treatment appeared to have a major effect on their number when assessed by TTC (p = 0.04021) and I (p = 0.000855) methods (Table VII).
The greatest number of aldehyde groups was recorded for mercerized-scoured yarns, followed by scoured-mercerized yarns, and the least number was reported for scoured yarns. Our previous results indicated that significant changes occurred in cuticle structure and composition during mercerization. 14 The altered cuticle allowed a different behavior of mercerized vs. non-mercerized cotton during scouring and oxidation, thus explaining the differences between the number of aldehyde groups in mercerized-scoured and scoured-mercerized yarns after oxidation.
Carboxyl Groups
Carboxyl groups on pre-treated and oxidized cotton yarns were measured by MB and calcium acetate ion-exchange methods (Table VI). While aldehyde groups significantly increased after oxidation, as determined by TTC, carboxyl groups increased only slightly. Although periodate has specific action during oxidation, the formation of carboxyl groups is still possible during this process. Oxidized cotton yarns maintain the same behavior related to the number of carboxyl groups as un-oxidized cotton yarns (Table VI). Alkaline-scoured samples presented the least number of carboxyl groups, followed by cotton yarns scoured with alkaline pectinase and acid pectinase. This trend could be observed in all pre-treatment sequences. ANOVA analysis confirmed that the type of scouring and the pre-treatment sequence had a significant influence on the number of carboxyl groups after oxidation (Table VII).
Degradation of Scoured and Oxidized Cotton Yarns
Mechanical Properties
The mechanical properties of oxidized and un-oxidized cotton yarns were determined to establish the relationship between chemical modification and the degree of degradation. The effects of oxidation on the tensile strength, elongation, and work of rupture are shown in Table VIII.
Tensile Strength, Elongation, and Work of Rupture of Pre-Treated Cotton Yarns before (S) and after Oxidation (O) a
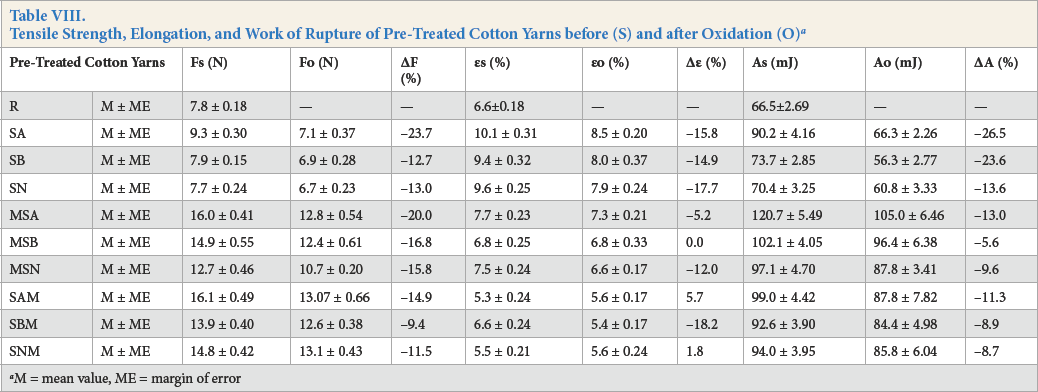
M = mean value, ME = margin of error
The increased tensile strength of alkaline-scoured cotton yarns, in all pre-treatment sequences, was the result of increased inter-fiber friction. Non-cellulosic components remaining on the enzymatically-scoured cotton decreased inter-fiber friction; therefore, these cotton yarns showed lower tensile strength than the alkaline-scoured yarns. 25
The oxidation of variously pre-treated cotton yarns decreased their tensile strength. Alkaline-scoured cotton underwent degradation to a greater extent (resulting in a greater decrease in tensile strength) than enzymatically-scoured cotton. As the cuticle of the alkaline-scoured cotton was completely removed, the cellulose was exposed to periodate attack, which destroyed cellulose chains and resulted in decreased tensile strength. Values for the work of rupture of un-oxidized and oxidized cotton yarns are listed in Table VIII. The work of rupture values for alkaline-scoured cotton yarns were, again, greater than those for the enzymatically-scoured samples. The decrease in the work of rupture after oxidation was more significant than that noted for tensile strength. Thus, the 23% lower tensile strength and 26% lower work of rupture after oxidation confirmed a medium degradation level of the oxidized cotton yarns. Previously published results reported a greater level of cotton damage, with a tensile strength loss of up to 60% for oxidized (alkaline scoured and bleached, followed by KIO4 treatment at 60 °C for 60 min) cotton. 2
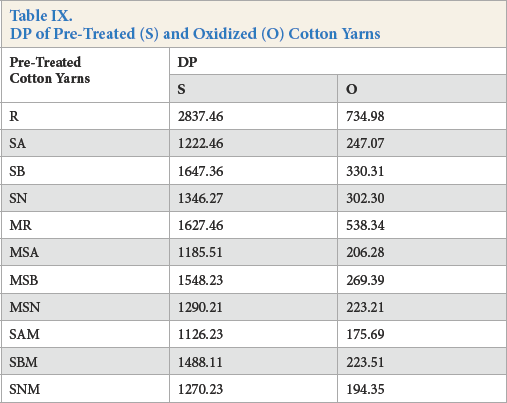
DP
Oxidative degradation of cellulose was also analyzed by assessing DP values before and after oxidation. The DP values are given in Table IX. The periodate oxidation process led to an increased number of aldehyde groups, as well as to C2-C3 bond cleavage, giving weaker and shorter cellulose chains. Oxidized cotton yarns had significantly-decreased DP values compared to the un-oxidized yarns, but followed the same trend as the latter (Table IX). The greater DP values of enzymatically-scoured yarns, compared to values for the alkaline-scoured yarns, were due to the mild scouring conditions. The remaining non-cellulosic components served as a protective barrier during periodate oxidation. This trend could be observed in all sequences of pre-treatment.
DP of Pre-Treated (S) and Oxidized (O) Cotton Yarns
Conclusions
The results obtained in the present study allowed us to draw the following conclusions. Selected scouring processes had a significant influence on the number of carboxyl and aldehyde groups found on cotton yarns before selective oxidation with periodate, while the pre-treatment sequence was of virtually no importance in this instance. Alkaline-scoured cotton yarns (in all pre-treatment sequences) presented the least number of aldehyde and carboxyl groups, followed by the alkaline pectinase and acid pectinase scoured yarns.
The number of aldehyde groups of variously pre-treated and oxidized cotton yarns depended on the oxidation and the pre-treatment sequence applied.
Results, such as the 23% lower tensile strength and 26% lower work of rupture after oxidation, confirmed that periodate oxidation was a suitable method for improving the functionality of cotton, while causing only moderate fiber damage.
Our results emphasized the influence of scouring type and pre-treatment sequence on the periodate oxidation of cotton yarns for attaining different degrees of modification without seriously decreasing the mechanical properties of the cotton yarn.