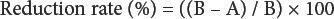Abstract
Ozone (O3) was reacted with unsaturated fatty acids in vegetable oils to yield products with antibacterial activity. In this paper, olive oil, containing large amounts of unsaturated fatty acids, was ozonated and characterized with gas chromatography (GC), Fourier transform infrared (FTIR), and thermogravimetric (TGA) analyses. The ozonated oils were sequestered in β-cyclodextrin (β-CD) inclusion complexes and applied onto 100% cotton fabric. The inclusion complex was analyzed with scanning electron microscopy (SEM), FTIR, and TGA, and treated fabric with FTIR and SEM. The antibacterial activity of the ozonated oil and treated fabric was assessed using ASTM E2149-01. The results showed that the ozonated oil had high antibacterial activity and the resulting β-CD/ozonated olive oil inclusion complex also had antibacterial activity when applied to cotton fabric.
Introduction
Ozone (O3) is widely recognized as one of the best bactericidal, antiviral, and antifungal agents, and is used as a clinical therapeutic agent for chronic wounds. The beneficial effects of ozone on wound healing may be due to decreased bacterial infection, ameliorated dermal wound healing, or increased oxygen tension by ozone exposure in the wound area. Recently, it has been suggested that ozonated oil could be a way to slowly deliver ozone to the target tissue, namely skin. 1
Vegetable oils contain 97–98% triglycerides. Depending on their origin and nature, they have a variable composition of saturated and unsaturated fatty acids (e.g., oleic, stearic, palmitic, linoleic, lauric, myristic, and linolenic acids) all bonded to the glycerol backbone. 2 Unsaturated triglycerides in vegetable oils give them many favorable properties. The double bonds are often chemically modified (e.g., hydrogenation). The reaction between ozone and unsaturated triglycerides occurs by the Criegee mechanism. 3 During the ozonation of unsaturated triglycerides, it is expected that aldehydes and carboxylic acids with three, six, and nine carbon atoms would be obtained. In this reaction, hydroperoxides, ozonides, and some other peroxidic or polyperoxidic species can also be obtained. The peroxidic and hydroperoxidic species partially decompose forming aldehydes and carboxylic acids with different numbers of carbon atoms in their structures.2–11
Ozonated vegetable oils have been proposed for use in a variety of applications (e.g., food, cosmetics, and pharmaceuticals). 4 They have been used topically for the treatment of wounds, anaerobic infections, herpetic infections (HHV I and II), trophic ulcers and burns, cellulitis, abscesses, anal fis-sures, decubitus ulcers, fistulae, fungal diseases, furunculosis, gingivitis, and vulvovaginitis. 12 It was hypothesized that the mechanism of action of ozonated oil on wound healing is due to its antibacterial effect, ability to promote the generation of growth factors, activation of local antioxidant mechanisms, and promotion of tissue repair. 12 The antibacterial properties of ozonated oils have been studied.5,6,11,13,14 Ozonated oils are good candidates to produce antibacterial medical textile surfaces, where the antibacterial oils are sequestered as β-cyclodextrin (β-CD) inclusion complexes before being applied to the textile. β-CD inclusion complex preparation is a facile process, with improved masking of malodor. 15
Cyclodextrins are torus-shaped cyclic maltooligosaccha-rides composed of glucose units linked by α(1-4) glucosidic bonds. There are three main types of cyclodextrins, α-CD, β-CD, and γ-CD, having 6, 7, and 8 glucose units, respectively. 16 β-CD (Fig. 1) is preferred for use in textile applications, such as essential oil, odor, and dye sequestration. The cyclic structure of cyclodextrins presents characteristic 3D structural features, thus having the appearance of a truncated cone. 15 The cyclic glucopyranose units form an apolar cavity, and the hydroxyl groups of the glucopyranose units are oriented outside of the cone. Thus cyclodextrins have the capacity to sequester substances in their apolar cavity.15,17,18Substances that contain apolar groups and have suitable dimensions are molecularly sequestered by cyclodextrins. Molecular sequestration with cyclodextrin forms an inclusion complex, and cyclodextrins screen some of the physical and chemical features of the sequestered substances. 15
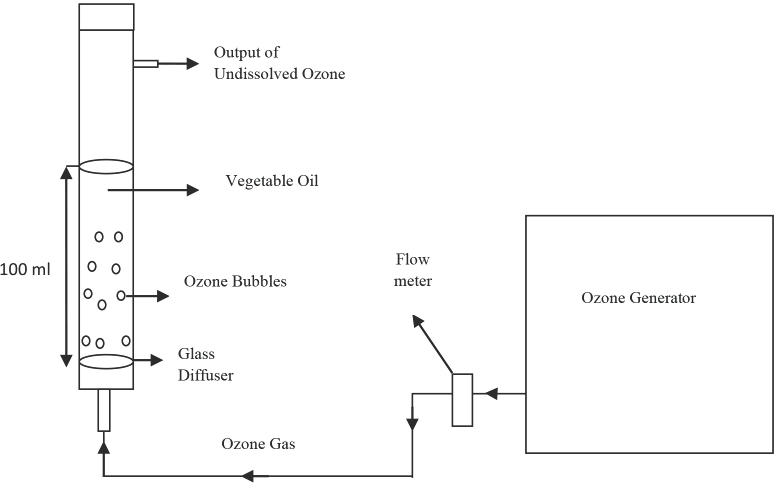
Ozonation process experimental setup.
The aim of this study was to apply antibacterial finishing to cotton fabrics using a β-CD inclusion complex with ozonated olive oil to produce a disposable surface without a characteristic ozone odor. The olive oil was ozonated for 3 h using a laboratory-scale ozone generator and the β-CD inclusion complex was prepared. The prepared inclusion complex was applied to 100% cotton fabric by the padding method. The ozonated oil was characterized with GC, FTIR, and TGA, and the inclusion complex-based ozonated olive oil and the cotton fabric treated with the inclusion complex were characterized by SEM and FTIR analyses. In addition, the antibacterial activity of the ozonated olive oil and treated fabric were assessed according to ASTM E2149-01 against Escherichia coli (ATCC 35218).
Experimental
Material
In this study, olive oil was purchased from Aksu Vital, and β-CD and ethanol purchased from Sigma Aldrich. Cotton (100%) woven fabrics (46 thread/cm warp, 22 thread/ cm weft, 270 g/m2) were used. All chemicals were used as received without any further purification. The fabric was desized, scoured, and bleached by the supplier (Matesa). To produce ozonated oil, an ozone generator with the capacity of 25 g/h (Teknozone TKZ) and a glass bubbling column (2-cm diameter, 90-cm height, 200-mL volume) with a glass diffuser placed at the bottom were used.
Method
Ozonation
The ozone gas produced by the generator was directed through the olive oil using a diffuser placed at the bottom of the bubbling column. Ozonation was carried out in 100 mL of oil during 3 h at room temperature (RT) with a flow rate of 4 L/min. The ozonation experimental setup is given in Fig. 1. After ozonation, the ozonated oil was stored in the refrigerator at 4 °C for further analysis and use.
Preparation of β-CD Inclusion Complex with Ozonated Olive Oil
For inclusion complex preparation, the ratio of ozonated oil: β-CD was kept at 1:6 (v/v). Solutions of olive oil (10%) in ethanol and β-CD (10%) in ethanol:water (1:2) were prepared separately and stirred by magnetic stirrer. The oil solution (20 mL) was added dropwise into the β-CD solution (120 mL) while stirring at 1000 rpm and stirred for 4 h in the laboratory environment. Ten, the solution was kept in the refrigerator for 20 h. The inclusion complex precipitate was then filtered and dried at RT.
Application of the Inclusion Complex to Fabric
A 20% ozonated olive oil/β-CD inclusion complex solution and a β-CD only solution (20%, without the ozonated oil) were prepared with distilled water and the solutions were applied to the 10 × 15 cm fabric samples by a laboratory-type padder. The fabrics were squeezed up to 65% pickup and dried at RT.
Characterization
Gas chromatography (GC) analysis was carried out for both ozonated and raw oils. The oil samples were methylated prior to analysis. 19 GC analysis was carried out using fame ionization detection (FID) via Shimadzu 2010 Plus GC. The FID temperature was set to 240 °C. Auto-injection was performed on the column (60 m × 0.25 mm, 0.25-mm film thickness), applying identical operational conditions to each sample. The column was used with helium as the carrier gas (0.8-mL/min gas flow). GC oven temperature was kept at 80 °C and adjusted to 240 °C at a rate of 3 °C/min. Relative percentage amounts of the separated compounds were calculated from FID chromatograms.
Fourier transform infrared spectroscopy (FTIR) with attenuated total reflectance (ATR) studies of the ozonated oil, raw oil, inclusion complex, and the treated fabric were carried out by the transmission method using a Spectrum 400 spec-trophotometer (PerkinElmer) scanning between 400 and 4000 cm−1. Resolution for the infrared spectra was 4 cm−1, and there were four scans for each spectrum.
Thermogravimetric analysis (TGA) was performed on the ozonated oil, raw oil, and inclusion complex using an SII TG/TDA 6300 analyzer to investigate the changes of thermal stability of the oils and inclusion complex. TGA analyses were carried out within a 30 to 600 °C heating range (at a 20 °C/min heating rate) under nitrogen gas.
The surface morphology of the inclusion complex and inclusion complex treated fabrics were characterized via SEM (Zeiss Evo LS10) and the antibacterial activity of the treated and raw fabrics, along with oil samples, against E. coli (ATCC 35218) was tested using ASTM E2149-1. In this antibacterial assessment method, the circular fabric swatches (38 mm in diameter) were challenged with 1.0 ± 0.1 mL (optical density at the 1.0 McFarland standard) of the bacterial inoculum in a 250-mL container. The inoculum was a nutrient broth culture containing 1.0 × 104 to 1.0 × 106 colony forming units (CFUs) of the bacteria/mL. Sterilized and distilled water (100 mL) was poured into a vessel and was vigorously shaken and incubated for 24 h at 37 °C. After the test and control swatches were in contact with the bacteria for 24 h, the diluted solutions were plated on Muller-Hinton II agar and incubated for 24 h at 37 °C. The viable colonies of the bacteria on the agar plate were counted, and the reduction in the number of the bacteria was calculated using Eq. 1.
A is the number of bacteria recovered from the inoculated treated test specimen swatches in the jar incubated over the desired contact period (24 h) and B is the number of bacteria recovered from the inoculated treated test specimen swatches in the jar immediately after inoculation (at zero contact time).
Results and Discussion
Ozonated Olive Oil Characterization
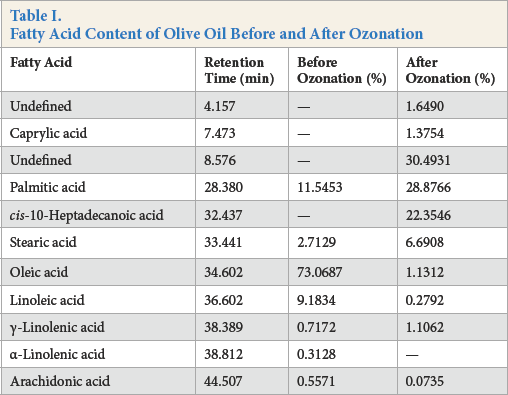
The fatty acid contents determined by GC analysis of the ozonated and raw olive oils are given in Table I. The reaction of ozone occurs at the carbon-carbon double bonds of unsaturat-ed fatty acids. 6 Therefore, the amount of unsaturated fatty acids in the oil decreases after ozonation. The oleic and linoleic acid component amounts, which are the most important unsaturated fatty acids in olive oil, 2 were dramatically reduced after ozonation, with two new peaks appearing at retention times around 4 and 8 min after ozonation. These results strongly support the synthesis of ozonized olive oil during this process.
Fatty Acid Content of Olive Oil Before and After Ozonation
The FTIR spectra also helped to confirm the synthesis of ozonized olive oil (Fig. 2). In the FTIR assessment of olive oil, C=O stretch (1742 cm−1), C=C stretch (1654 cm−1), C–H stretch (3009 cm−1), and C-H vibration (721 cm−1) bands were present. In addition, the ozonide C-O stretch (1105 cm−1) band of ozonized unsaturated fatty acid moieties are crucial for confirming ozonation.3,20,21 Fig. 2 showed that the C-O stretch (1105 cm−1) band increased and the C–H stretch (3009 cm−1) band decreased significantly after ozonation. Thus, the presence of the ozonide structure formed after the reaction of ozone with the unsaturated fatty acids of the olive oil was confirmed.
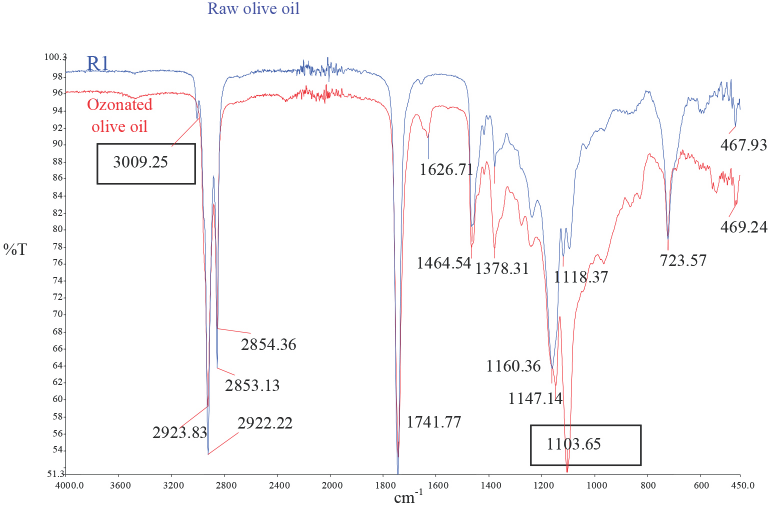
The IR spectra of the ozonated and raw olive oils.
TGA analysis results of raw and ozonated olive oils are given in Fig. 3. Ozonation decreased the thermal stability of olive oil (Fig. 3). Mass loss began at 386.4 °C for the raw oil and at 154.6 °C for the ozonated olive oil. It was thought that the fatty acids of olive oil decayed to smaller fatty acids after ozo-nation and the smaller fatty acids had lower thermal stability.
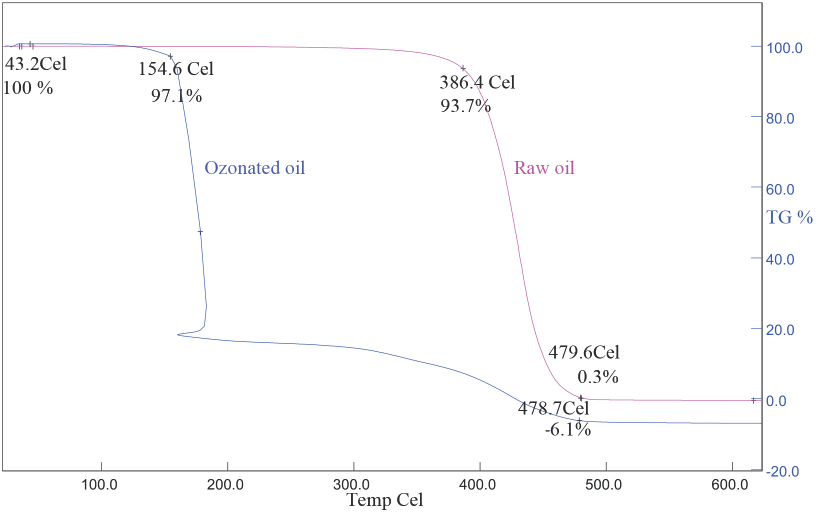
TGA of raw and ozonated olive oils.
Table II shows the antibacterial activity test results of oil olive before and after ozonation. Positive (+) results in the table indicated an increase in the bacterial population, while negative (-) results indicated a decrease in bacterial population. The results show that the ozonated oil had strong antibacterial activity, with 100% of E. coli killed within 24 h after incubation.
Percent Reduction of Bacteria After 24 Hours a

The inoculum concentration was 3.79 × 108 CFUs.
Ozonated Olive Oil/β-CD Inclusion Complex Characterization
The β-CD inclusion complex produced with ozonated oil was investigated by FTIR analysis (Fig. 4). The inclusion complex had peaks at similar frequencies as the ozonated olive oil; the exception was an O-H stretch (3271 cm−1) band, which is assigned to α-D-glucopyranoside units of β-CD. 15 Thus, it was concluded that a β-CD inclusion complex with ozonated olive oil was formed.
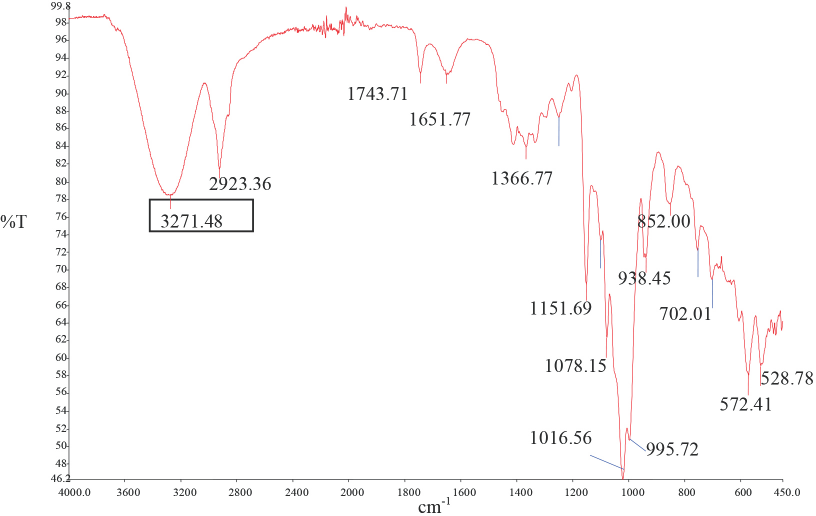
IR spectra of the inclusion complex-based ozonated olive oil.
TGA analysis results of ozonated olive oils and olive oil/β-CD inclusion complexes are given in Fig. 5. Mass loss began at 154.6 °C for ozonated oil and at 294.6 °C for olive oil/β-CD inclusion complex. This indicates that when the ozonated oil was sequestered in the inclusion complex, the thermal stability increased.
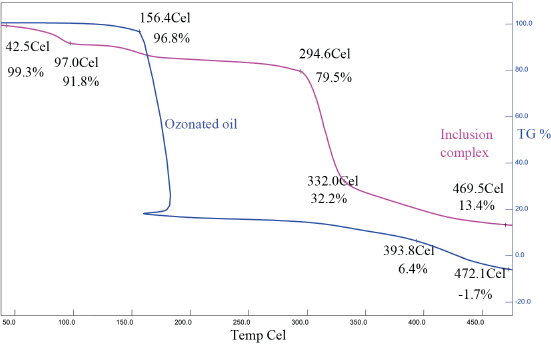
TGA of ozonated olive oil and inclusion complex.
The SEM image of the inclusion complex obtained is shown in Fig. 6. The inclusion complexes had a stable and rough contour, and a certain degree of agglomeration on the fiber surface.
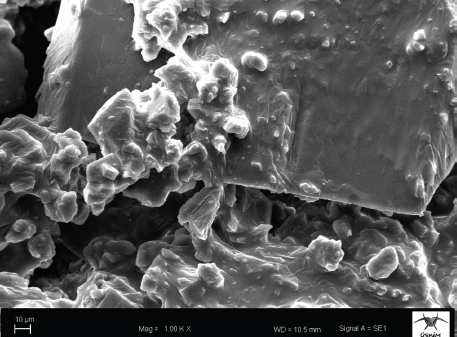
SEM image of inclusion complex-based ozonated olive oil.
Treated Fabric Characterization
Fig. 7 shows the FTIR spectra of treated and raw fabric samples. The treated samples exhibited sharp C=O stretch (1743 cm−1) and strong C-O stretch (1105 cm−1) bands, which indicated bonding of the inclusion complexes to the cotton fiber. SEM images (Fig. 8) also show the presence of ozonated olive oil/β-CD inclusion complex on the fiber surface. These two findings indicated successful treatment of the fabric with the inclusion complex.
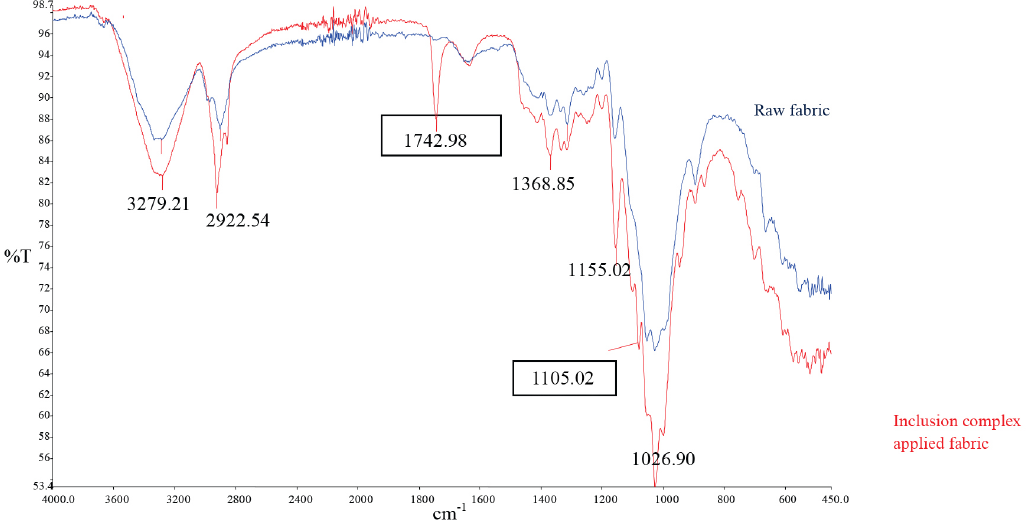
IR spectra of raw and inclusion complex treated fabric.

SEM images of (a) raw fabric and (b) inclusion complex treated fabric.
Antibacterial activity test results of the raw fabric and fabric treated with olive oil/β-CD inclusion complex solution and β-CD solution (without the ozonated oil) after 3 h incubations are given in Table III. The results showed strong antibacterial performance after application of the inclusion complex to the fabric. The treated fabric also did not have the malodor of ozonated oil due to odor sequestration in β-CD.
Percent Reduction of Bacteria After 3 Hours a
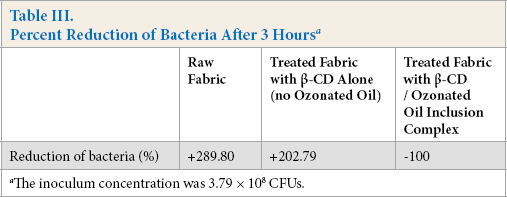
The inoculum concentration was 3.79 × 108 CFUs.
Conclusion
Ozone, which has high antibacterial activity, cannot be stored, but it can be carried by materials having double bonds. Ozone can be reacted with unsaturated fatty acids of certain oils, imparting antibacterial activity to them. In this paper, we investigated the possible antibacterial finishing of cotton fabric with ozonated oil. Since the ozonated oil cannot be directly applied to textiles, the β-CD inclusion complex with the oil was prepared. The malodor of the ozonated oil could be masked due to odor sequestration in β-CD.
For this purpose, the olive oil was ozonated and the β-CD inclusion complex based on ozonated olive oil was prepared. The resulting β-CD/ozonated olive oil inclusion complex was applied to 100% cotton fabric by padding. The ozonated and raw oils were investigated by GC, FTIR, and TGA; the inclusion complex and treated fabrics were characterized using FTIR, TGA, and SEM analyses. In addition, antibacterial activities of the ozon-ated oil and fabric including the inclusion complex were tested. The results showed that ozonated olive oil, the inclusion complex, and the inclusion complex treated fabric all had antibacterial activity. The β-CD inclusion complex with ozonated oil could be simply prepared and imparted antibacterial activity to the cotton fabric without the malodor of ozonated oil.
Footnotes
Acknowledgment
This study was supported by the Scientific Research Project Department of Kahramanmaraş Sütçü İmam University (KSU-BAP) (project number: 2015/1-83D and 2014/3-48M).