Abstract
Coloration of cotton fabric in one step via in situ deposition of manganese oxide or cobalt oxide nanoparticles, using microwave techniques, was performed. The structure and surface morphology of the obtained nanoparticles were studied by x-ray diffraction analysis and transmission electron microscopy (TEM). The cobalt oxide nanoparticles had a nanotube-like shape, with a length of 100–200 nm and a diameter of ∼58 nm, whereas manganese oxide nanoparticles had a linear structure of nanorods with a diameter of 50–55 nm and a length of 70-80 nm. Treated cotton fabrics were characterized by scanning electron microscopy (SEM) and the amount of nanoparticles deposited was quantitatively analyzed by SEM coupled with high energy dispersive x-ray (XRD) analysis. The fastness properties of dyed cotton fabrics containing these nanoparticles were evaluated.
Introduction
Over the last few years, the development of nanotechnology has attracted increasing interest from both industrial and governmental institutions in many parts of the world. Nano-technology has potential applications in a very wide range of industries and products, such as medical appliances, communication technologies, and biotechnology. Also, in the textile sector, nanotechnology is expected to have considerable potential for the development of new materials as well as for improving their functionality.
Microwave chemistry is the science of applying microwave radiation to promote chemical reactions.1–5 Microwave irradiation as an unconventional reaction condition 6 has been applied in various areas of chemistry and technology to produce or destroy diverse materials and chemical compounds, and to accelerate chemical processess. 7
In addition, microwave heating is widely used to prepare various refractory inorganic compounds and materials (e.g., double oxides, nitrides, carbides, semiconductors, glasses, and ceramics), 8 as well as in organic chemistry (e.g., for pyrolysis, esterification, and condensation reactions).9,10Recent excellent reviews describe microwave-assisted synthesis of various types of compounds and materials, in particular organic9,11,12 and organometallic 13 compounds, polymers, and applications in analytical chemistry. 14
Microwave heating occurs through the interaction of elec-tromagnetic radiation with the dipole moment of molecules. As required by green chemistry principles, water as an inex-pensive, non-toxic, non-flammable, and abundant material in nature, is a safe and ideal solvent for microwave-activated reactions.15,16 Direct transformation of the energy into the components present in the reaction vessel not only reduces synthesis time, but also affords the desired nanoparticles with a narrow particle size distribution.17–23 Furthermore, microwave-assisted oxide synthesis is gaining popularity due to its high reaction rate, efficient heat transfer, and environmentally friendly nature. Microwave heating is an energy conversion method in which electromagnetic radiation is converted into heat energy rather than heat transfer by convection as in conventional heating.24,25
Pigments are particulate inorganic or organic substances that are insoluble or substantially insoluble in water or in the organic medium in which they are used as dispersions. They impart color, opacity, mechanical rigidity, and reinforcement to the continuous phase in which they are dispersed. Pigments alter the appearance of the coating by selective absorption or by light scattering. Pigments are classified on the basis of their performance as decorative pigments or protective pigments. Pigments also impart certain special properties to the coatings, such as reflection of infrared radiation. 26
Pigments may be white, black, colored, or fluorescent, and do not react chemically or physically with the substrate.27,28The most important pigment characteristic is its capacity to develop color (pigmentary capacity). 29 The most common method of obtaining pigments is the conventional mixing of oxides, although several other chemical methods are used to obtain nanometric material with highly-controlled purity and chemical homogeneity. Some of the most well-known methods are sol-gel, 30 micro-emulsion, 31 co-precipitation, 32 supersonic radiation, 33 Pechini's method, 34 hydrothermal synthesis, 35 freeze drying 36 , and combustion synthesis.37–39In connection with textile dyeing, nano-particulate pigments can be directly used as dyes, 40 textile surfaces can be treated with nano-structured coatings to render them hydrophilic and improve their colorability, 41 and dyes can be included into nano-thin coatings to improve bleaching resistance and leaching stability. 42 Dye immobilization in the nano-sol matrix significantly reduces the leaching fastness. 42 Ultra-fine modified pigment dispersions usually show relative stability and higher color strength (K/S values), which closely approaches that of dyes.43–48
The objective of the present study was to examine the use of colored nanoparticles as an alternative to traditional dyes to dye cellulosic fabrics, without salt and binders and with only a short washing-off procedure at the end of the dyeing process. Coloration of cotton fabric in one-step via in situ deposition of manganese oxide or cobalt oxide nanoparticles using microwave techniques was performed. The structure and surface morphology of the obtained nanoparticles were studied by x-ray diffraction (XRD) analysis and transmission electron microscopy (TEM). These nanoparticle-treated cotton fabrics were characterized by scanning electron microscopy (SEM) and the amount of nanoparticles deposited in situ on the cotton fabrics was quantitatively analyzed by SEM coupled with high-energy dispersive XRD analysis. The fastness and color strength of dyed cotton fabrics containing these nanoparticles were evaluated.
Experimental
Materials
Mill desized, half-bleached cotton fabrics, were supplied by El-Mehalla Spinning and Weaving Company (100 g/m2). Cobalt nitrate and manganese nitrate were purchased from Sigma-Aldrich Co. Sodium hydroxide pellets, L-histidine monohydrochloride monohydrate, and sodium dihydrogen orthophosphate were purchased from Merck Co. All chemicals used were analytical grade.
Pigment Preparation and Cotton Fabric Coloration
Metal nitrates (manganese nitrate or cobalt nitrate) were dissolved in ion-exchanged water to prepare metal solutions at a concentration of 0.5 M. Cotton fabric was padded in the prepared metal nitrate solution bath at a wet pickup of 100% and padded cotton fabric was padded in the second bath containing sodium hydroxide (pH 10.5). Determination of the pH values was based on the pH value at which metal hydroxides precipitated from metal ions in the solution. The prepared fabrics containing in situ precipitated metal hydroxides were then transferred to the microwave-hydro-thermal synthesis process equipment. They were placed in a PTFE-lined vessel and maintained at 180 °C for 2 min in a microwave sample preparation system (Multi-wave 3000, Perkin Elmer Japan Co. Ltd.). The system was a 2.45-GHz MW unit and the power was controlled from 0 to 1400 W for accurate temperature and/or pressure control. The reaction products were collected, washed at least five times with ion-exchanged water to remove Na+ and NO3− ions and other impurities, and then dried at 100 °C for 30 min. Yellow shades were obtained for cotton fabrics containing manganese oxide nanoparticles and violet shades were obtained for cotton fabrics containing cobalt oxide nanoparticles.
Analysis
Prepared nano-Mn oxide and nano-Co oxide colloidal solutions were characterized by ultraviolet-visible (UV-Vis) spectroscopy, transmission electron microscopy (TEM, Zeiss EM-10), and high-energy dispersive x-ray (XRD) analysis. The crystalline phase on cotton fabrics containing nano-Mn oxide or nano-Co oxide were determined using XRD diffraction with a D/max-IV diffractometer and Cu Kα radiation (λ = 1.5418 Å). The sample was scanned from 10° to 80° (2θ). The crystallite domain size (D) was examined from the XRD peak based on the Scherrer equation. 49 In addition, the surface morphology of cotton fabrics containing nano-Mn oxide or nano-Co oxide was characterized using SEM (JXA-840 with an electron probe microanalyzer, JEOL). Nano-Mn oxide and nano-Co oxide deposited in situ into cotton fabrics were examined qualitatively and quantitatively by SEM-energy dispersive X-ray spectrum (SEM-EDX), along with SEM. The distribution and size of the nano-metal oxide inside the cotton fabrics were char-acterized using TEM on a cross-sectional area of prepared colored cotton fabric.
Fastness Properties
Washfastness
Washfastness was determined according to AATCC Test Method (TM) 61-1975 using a Launder-Ometer (SDL Atlas LLC). Specimens (5 × 10 cm) were sewn between two similar pieces of bleached cotton fabric. The composite specimens were immersed into an aqueous solution containing 5 g/L soap and 2 g/L sodium carbonate at a liquor ratio (LR) of 1:50. The bath was thermostatically adjusted to either 60 °C or 95 °C. The test was run for 45 min at 42 rpm. The samples were then removed, rinsed twice in a 100-mL water bath at 40 °C for 1 min with occasional stirring or hand squeezing, scoured in 100 mL of a 0.014% solution of acetic acid for 1 min at 27 °C, and rinsed again for 1 min in 100 mL water at 27 °C, followed by drying.58 Washfastness was evaluated using the Gray Scale reference for color change.
Crockfastness
Crockfastness was determined according to AATCC TM 8-97. This test is designed to determine the degree of color that may be transferred from the surface of the colored fabric to another surface by rubbing. A colored test specimen fastened to the base of a crockmeter was rubbed with a white crock test cloth under controlled conditions. 50
For dry crocking, the test specimen was placed fat on the base of the crockmeter. A white testing cloth was mounted on the finger of the crockmeter. The covered finger was lowered onto the test specimen and slid back and forth 20 times by making 10 complete turns at a rate of one turn per second. The evaluation was performed using the Gray Scale for staining.
For wet crocking, the white test sample was thoroughly wetted in distilled water to a 65% wet pickup. The procedure was run as before. The white test samples were then air-dried before evaluation.
Colorfastness to Perspiration
Two artificial perspiration solutions were made up as follows:
Acidic solution: L-histidine monohydrochloride mono-hydrate (0.5 g), sodium chloride (5.0 g), and sodium dihydrogen orthophosphate (2.2 g), were dissolved in 1 L distilled water to make a final 0.1 N solution.
Alkaline solution: L-histidine monohydrochloride mono-hydrate (0.5 g), sodium chloride (5.0 g), and disodium hydrogen orthophosphate (2.5 g), were dissolved in 1 L of distilled water. The pH was adjusted to pH 8 by a sodium hydroxide solution to make a final 0.1 N solution.
A colored specimen (5 × 4 cm) was sewn between two pieces of uncolored specimens (so that a 5 × 1-cm area of the colored cloth was not in contact with the specimen) to form a composite specimen. The composite sample was then immersed (for 15–30 min) in each of the solutions with occasional agitation and squeezing to insure complete wetting. The test specimen was placed between two glass plates under a ∼4.5-kg force. The plates containing the composite specimens were then held vertical in an oven at 37 °C for 4 h. The effect on the color of the test specimens was determined by reference to the Gray Scale for color change.58
Results and Discussion
Characterization of Prepared Nanoparticles TEM
The TEM image of nano-Co oxide (Fig. 1a) shows that the materials had a nanotube-like shape. The length of the nanotube was 100–200 nm (mean length was ∼130 nm) and the diameter was ∼58 nm. The synthesized nano-Co oxide had a good nanotube structure and was stable. TEM images of the nano-Mn oxide formed under deposition conditions are shown in Fig. 1b. Each particle had a linear nanorod structure with a diameter of ∼50–55 nm and a length of ∼70–80 nm.

TEM images of (a) nano-Co oxide and (b) nano-Mn oxide.
XRD
The XRD patterns of the nano-Co oxide nanotube products are shown in Fig. 2a. According to the standard Co3O4 XRD pattern (JCPDS card No. 43-1003), all the peaks of cobalt oxide can be indexed to the cubic phase (Fd 3m).
Fig. 2b shows XRD patterns for a nano-Mn oxide sample. The presence of a few broad peaks was related to the poor crystallinity and nanoscale nanoparticles. The profiles of the few broad peaks around 2θ = 33.1°, 37.1°, 45.5°, and 65.6° seemed to be consistent with the peaks of γ-MnO2 (JCPDS No. 14-0644). The peak around 2θ = 21° revealed the existence of γ-MnO2.
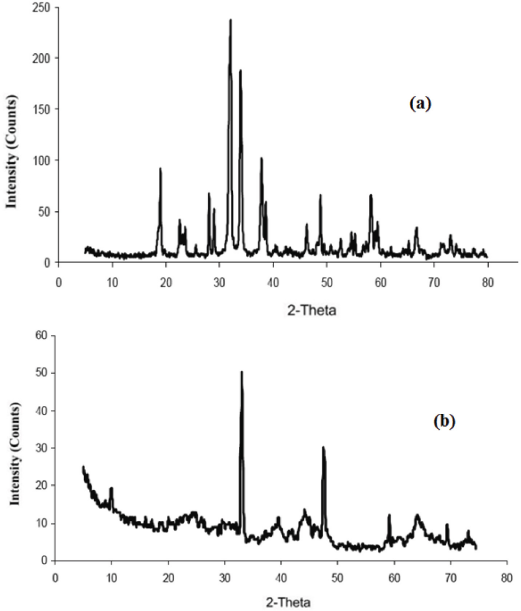
XRD patterns of the (a) nano-Co oxide and (b) nano-Mn oxide.
UV-Visible Spectrophotometry
Presence of the nano-Co oxide was determined by the UV-visible (UV-Vis) absorption spectroscopy. The peak positions and shapes of the absorption spectra for the nanoparticles are shown in Fig. 3a.
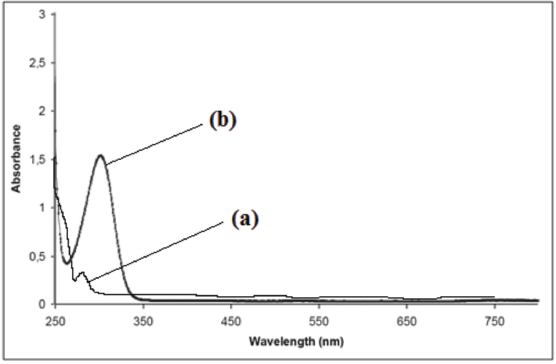
UV-visible absorption spectra of (a) nano-Co oxide and (b) nano-Mn oxide.
The optical absorption spectra of nano-Mn oxide, obtained in the 250–750 nm range, are presented in Fig. 3b. The strongest absorption peak of the prepared sample appeared at ∼300 nm. nano-Mn oxide had good absorption in the 250–350 nm range.
Characterization of Treated Fabrics XRD
Fig. 4a–c show XRD patterns of (a) untreated cotton fabric, (b) cotton fabric containing nano-Mn oxide, and (c) cotton fabric containing nano-Co oxide. The peaks obtained at different crystal planes of nano-Mn oxide and nano-Co oxide were very similar to the data shown in Fig. 4. Moreover, the peaks observed for cotton reflected its B-type structure.
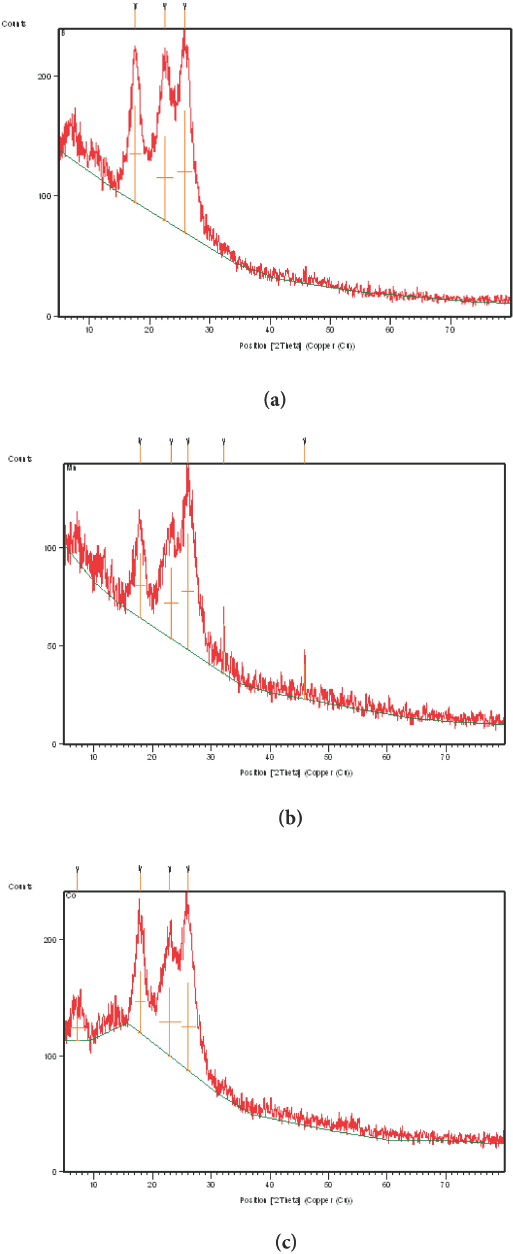
X-ray diffraction of (a) untreated, (b) nano-Mn oxide treated, and (c) nano-CO oxide treated cotton fabrics.
Cotton fabric containing nano-Mn oxide or nano-Co oxide showed no change in the diffraction planes of the cotton, which might be due to the relatively lower inclusion of metal oxide in the planes of the cotton. This finding could be further explained by an increase in both the intermolecular hydrogen bonding between the sheets and the crystallinity index of the metal oxide composite that appeared in the intensity pattern of the metal oxide rather than in the cotton itself.
SEM
The surfaces of the treated fabrics were observed by SEM. SEM micrographs in Fig. 5a and b show the nano-Co oxide and nano-Mn oxide on the cotton fabric samples. The nanoparticles were well dispersed on the surface of the cotton fabric samples.

SEM micrograph of cotton fabrics containing (a) nano-Mn oxide and (b) nano-Co oxide.
EDX
Characterization of the treated fabric samples by SEM coupled with EDX was used to qualitatively and quan-titatively confirm the presence of nano-metal oxides internally and on the surface of the prepared fabric samples. The EDX of a cotton fabric containing nano-Mn oxide and one containing nano-Co oxide are shown in Fig. 6a and b, respectively. This figure shows that in addition to the manganese and cobalt, two new strong peaks emerged: one for carbon and the other for oxygen in the EDX pattern. The strong peaks for carbon and oxygen were caused by the fabrics, while the peaks for Mn and Co were very weak because the Co content in the fabric was only 16.7% and the Mn content was 13.37%. Co and Mn existed both on the fabric surface and internally (Figs. 5 and 6). These findings indicated that nano-Co oxide and nano-Mn oxide were successfully immobilized on the cotton fabrics. The surface area of particles and their surface energy increased linearly with a decrease in particle size. When the particle size was reduced to nanometer scale, the surface energy of the particles became very large, and it was difficult to disperse aggregated nanoparticles or to remove nanopar-ticles adhering to solid surfaces.
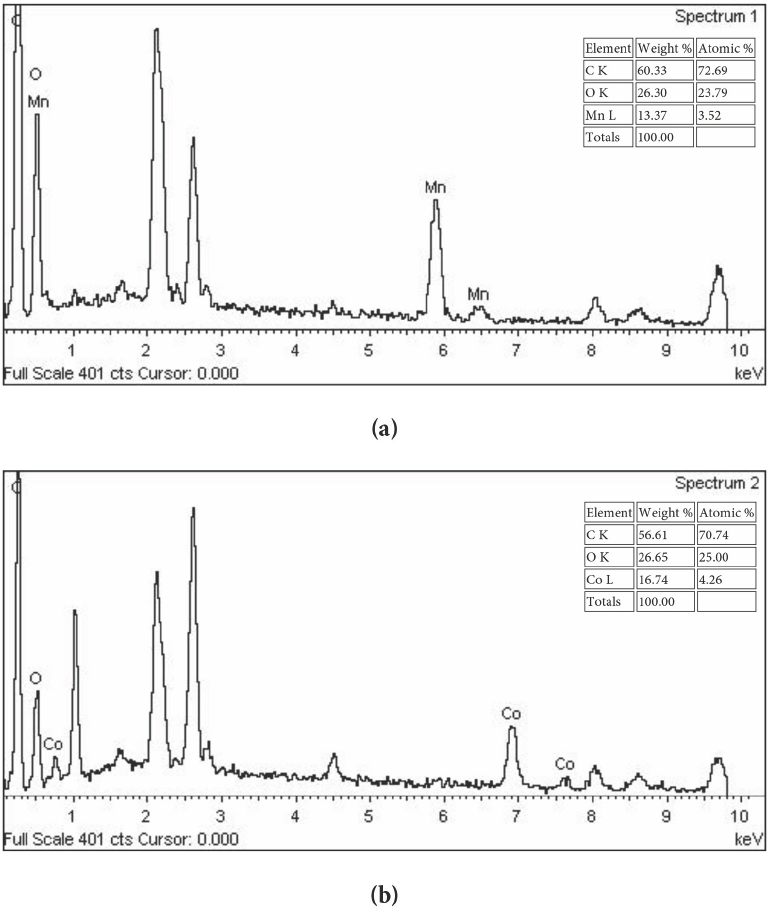
EDX of (a) nano-Mn oxide treated and (b) nano-Co oxide treated cotton fabrics.
Nanoparticle Distribution
To confirm the creation of nano-Co oxide and nano-Mn oxide on and in the cotton fabrics, TEM analysis of cross-sectional samples was carried out for the prepared fabric samples. It is apparent from Fig. 7 that the nano-metal oxides did not appear to form agglomerates and had a good distribution inside the fabric owing to the stability of synthesized nanometal oxides within the cellulosic network. TEM images for cross sections of fabric samples containing nano-Co oxide are shown in Fig. 7a, which demonstrates that the nanoparticles had a nanotube-like shape. The length of the nanotube was 100–150 nm (mean length ∼130 nm) and the diameter was ∼58 nm. The synthesized nano-Co oxide had a good nanotube structure and was stable. TEM images for cross sections of fabric samples containing nano-Mn oxide formed are shown in Fig. 7b. Each particle was a linear nanorod structure with diameter of 50∼55 nm and a length of 70∼80 nm.

TEM images of cross section of cotton fabrics containing (a) nano-Co oxide and (b) nano-Mn oxide.
Fastness Properties
Inorganic pigment dyeing usually involves a binder to fix the pigment color on the fabric. In the present study, inor-ganic pigment nanoparticles were used to color cotton fabric without the use of binder. Table I shows the detailed K/S data corresponding to undyed cotton fabrics and cotton fabric samples containing nano-metal oxides. The K/S values of cotton fabric containing nano-Co oxide (violet shade) were greater than those containing nano-Mn oxide (yellow shade).
Dyeing of Cotton Fabric with Nano-sized Inorganic Pigment Particles a
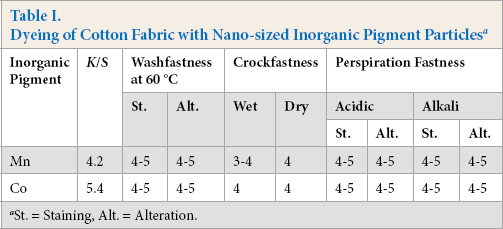
St. = Staining, Alt. = Alteration.
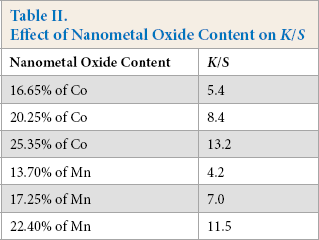
Increasing the metal salt concentrations in the padding baths led to an increase in the nano-metal oxide content on the fabric samples, leading to increased K/S values (Table II). metal salts dyed fabrics were durable to washing and there was no decrease in K/S values for any of the samples after washing. This may be due to easy diffusion of the metal salts into the fabric and more difficult diffusion of nano-metal oxides out of the fabric when washed. Crockfastness and wash-fastness were both improved, whereas fastness to perspiration remained the same.
Effect of Nanometal Oxide Content on K/S
Conclusions
Microwave irradiation offers a very simple and repro-ducible way for the rapid and energy-saving synthesis of metal oxide nanoparticles from their hydroxide form. Cobalt oxide nanoparticles (nano-Co oxide) and manganese oxide nanoparticles (nano-Mn oxide) were prepared using a microwave irradiation process. TEM, UV-Visible, and XRD analysis confirmed that nano-Co oxide had a nanotube-like shape with a length of 100–200 nm and a diameter of ∼58 nm, while nano-Mn oxide had a linear structure forming a nanorod with a diameter of ∼50∼55 nm and length of ∼70–80 nm. The prepared nano-metal oxides can be used for cotton fabric coloration in a single process by in situ deposition of nanoparticles into the cotton fabric without the use of dispersing agents and binders. Cotton fabrics containing nano-metal oxides were confirmed using SEM coupled with energy dispersive XRD. Cotton fabric containing nano-Co oxide had higher K/S values than the cotton fabric containing nano-Mn oxide. The nano-metal oxide-dyed fabrics were durable to washing, and no decrease in K/S was observed in any of the samples after washing. Although the crockfastness and washfastness were improved, the effects of perspiration remained the same.
Footnotes
Acknowledgment
The authors are grateful to the Dean of Scientific Research at King Faisal University for financial support for this work through project number 130190.
