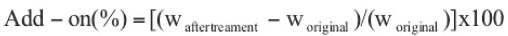Abstract
Conventional pad-dry-cure (non-scCO2) and supercritical carbon dioxide (scCO2) application methods were used to study the effectiveness of a newly synthesized phosphazene derivative as a flame retardant on cotton fabric. The 1,1′,4,5-tetrahydrotrispiro[1,3,2-diazaphosphole-2,2′-[1,3,5,2,4,6]triazatriphosphinine-4′,6”-dibenzo[d, f][1,3,2] dioxaphosphepine-6′,6”′-dibenzo[d, f][1,3,2]dioxaphosphepine] (2), was synthesized in two steps, in high overall yield, and applied onto cotton fabrics. The flame retardant properties of the treated fabrics were examined by 45° angle flammability test, limiting oxygen index test (LOI), microscale combustion calorimetry (MCC), thermogravimetric analysis (TGA), and scanning electron microscopy (SEM). The results indicate that fabrics treated with 2 are flame resistant; however, non-scCO2 fabrics performed better than scCO2 fabrics in a preliminary durability study.
Keywords
Introduction
Flame retardants (FRs) used in textiles, furniture, electronics, and insulation can be divided into two main categories: halogenated and non-halogenated FR.1,2 Halogenated FRs typically contain bromine or chlorine, but due to environmental and health concerns, they are becoming restricted in the marketplace.3–5 Current research is shifting towards non-halogenated FRs due to their non-toxic byproducts and lower environmental impact.3,4 Non-halogenated FR, often contain phosphorus, nitrogen, sulfur, and silicon. Phosphorus and nitrogen containing FRs are gaining more attention due to their combined effect of reducing flammable gases generated near the cotton surface and retarding fires. 3 Molecules that contain both phosphorus and nitrogen exhibit better FR properties and higher char yields.2,3,6,7 For example, phosphazene derivatives impart high thermo-oxidative stability and excellent flame retardancy. 8 Additionally, phosphazene is easily modified through nucleophilic substitution, offering various synthetic routes that can provide different functionalities, properties, and novel FRs. 8
Different methods, including pad-dry-cure and inclusion, have been used to incorporate FRs into cotton textiles.9,10Conventional pad-dry-cure is extensively used to incorporate FRs into cotton fabric by dipping the fabric into the FR solution, padding to remove excess solvent, and drying and curing the fabric.9–11 Inclusion is an impregnation process achieved when one molecule is non-covalently enclosed within another molecular structure; however, a drawback of this method is the release of molecules due to the re-swelling of cotton fabric with water.12,13 Given its limitations, inclusion is not widely used to incorporate FRs. An alternative method to enclose molecules into fabrics involves the use of a supercritical fluid (SCF).
SCFs are solvents that exist above the specific SCF's critical pressure and temperature. 14 SCFs are inexpensive, environmentally friendly, non-flammable, sustainable, and are known for their high diffusion in organic matter. Furthermore, SCFs can eliminate or reduce the use of organic solvents and water when dyeing or impregnating hydro-phobic or hydrophilic compounds into cotton textiles.15,16 A commonly used SCF is supercritical carbon dioxide (scCO2), which has been used for extractions, impregnations, drying, and dyeing fabrics or polymers.14,17–19 Since 2012, Nike and Adidas have endorsed the use of scCO2 as a water-free process to dye fabrics.20–22 These dyes contain functional groups that react with the cotton fabric to produce a permanent attachment. Not only does the use of scCO2 eliminate the consumption of billions of gallons of water each year, but it also saves time and energy. 22 The supercritical process has also been used to impregnate hydrophilic and hydrophobic molecules such as D-panthenol, thymol, polymers, and nanoparticles into cotton systems.15,23,24
Current scCO2 impregnation studies have not yielded the levels of durability obtained when cotton fabric is treated with molecules having no reactive functional groups, such as the FR reported in this study.23,25,26 Herein, we investigate the use of scCO2 to impregnate FRs into cotton fabric as another avenue to achieve durability. The current research focus includes (1) the synthesis of a phosphazene-based FR that is reacted with 2,2′-dihydroxybiphenyl and 1,2-diamino-ethane, (2) the determination of flammability when cotton fabric is treated with the FR via conventional pad-dry-cure (non-scCO2) and scCO2 methods, and (3) the investigation of whether the scCO2 method can provide durability. The flammability, thermal properties, char, and durability of the fabrics treated by each method were explored.
Experimental
Materials
Phosphazene and anhydrous sodium sulfate (Na2SO4) were purchased from Sigma Aldrich. Dichloromethane (DCM), methanol (MeOH), acetone, and hexanes were purchased from Fisher Scientific. Anhydrous potassium carbonate (K2CO3) and 1,2-diaminoethane were purchased from Alfa Aesar. Deuterated dichloromethane-d2 and acetone-d6 with 1% trimethylsilane (TMS) were purchased from Cambridge Isotopes Laboratories. All chemicals were used as received without further purification. Cotton fabric (100%) was purchased as twill fabric style 423, with a weight of 258 g/ m2, from Test Fabrics, Inc. The fabric was desized, bleached, and was free of all resins and finishes. Nuclear magnetic resonance (NMR) spectroscopy was performed on a Varian 400 MHz spectrometer. 31 P-NMR spectra were given in δ (ppm) relative to an external 85% aqueous H3PO4 standard. Liquid chromatograph mass spectrometry (LC-MS/MS) data was collected on an Agilent 6520 QTOF LC-MS/MS instrument. Mathis dryers (W. Mathis AG LTF and Mathis Labdryer LTE) were purchased from Mathis U.S.A. Inc. The supercritical reactor was purchased from Supercritical Fluid Technologies Inc.
Synthesis
FR Precursor
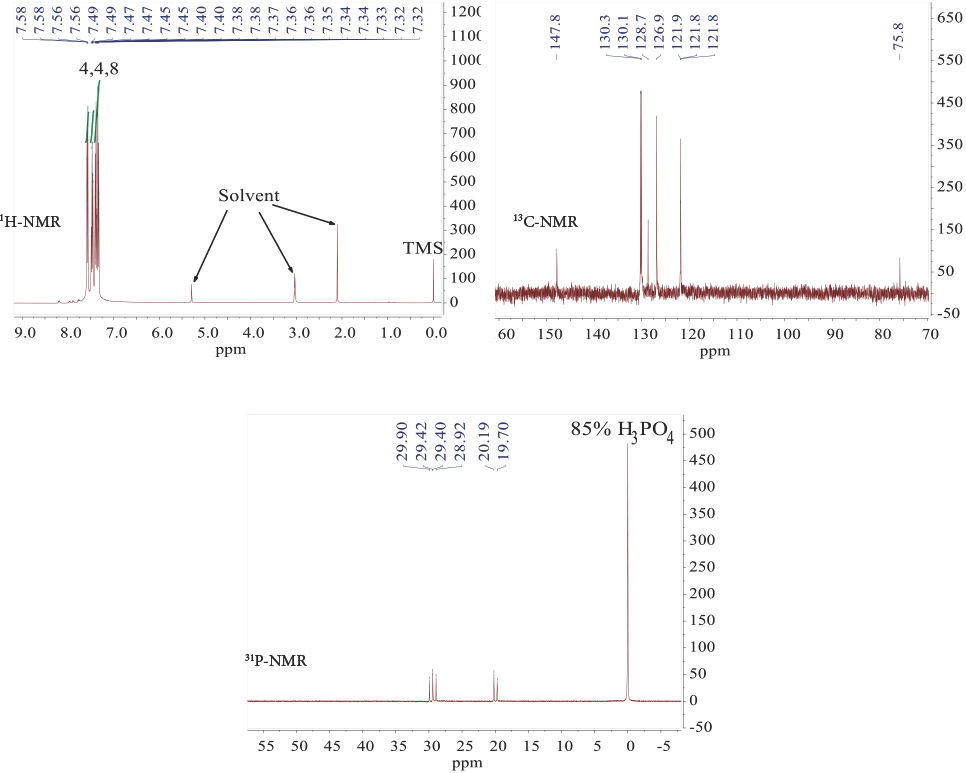
6′,6′-Dichloro-4′l5,6l5,6′l5-dispiro[dibenzo[d, f][1,3,2] dioxaphosphepine-6,2′-[1,3,5,2,4,6]triazatriphosphinine-4′,6″-dibenzo[d, f][1,3,2]dioxaphosphepine] (
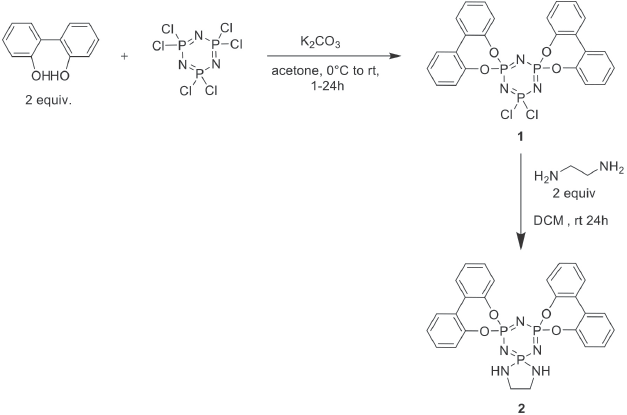
Synthesis of compound 2.
FR
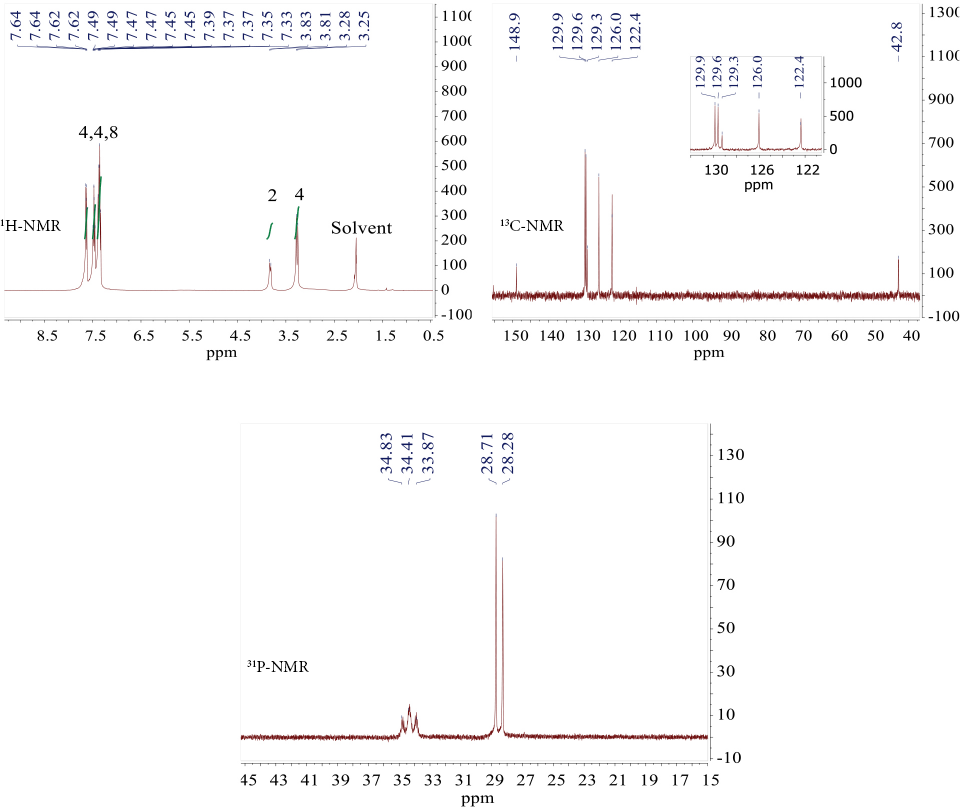
1,1′,4,5-Tetrahydrotrispiro[1,3,2-diazaphospho-le-2,2′-[1,3,5,2,4,6]triazatriphosphinine-4′,6″-dibenzo[d, f] [1,3,2]dioxaphosphepine-6′,6″′-dibenzo[d, f][1,3,2]dioxa-phosphepine] (
Fabric Treatments
Pad-Dry-Cure Method
Cotton twill test fabric 423 was bleached, mercerized, and scoured prior to use. Cotton twill was immersed in a shallow pan containing the solution of
Waftertreatment is the fabric weight after treatment and Woriginal is the original fabric weight.
Supercritical Carbon Dioxide Method
The supercritical reactor consisted of ultra high purity carbon dioxide, a series II Prime/Purge, and a high pressure series temperature control reactor. Supercritical treatments were performed by first preparing a 5% co-solvent solution of
45° Flammability Test
The 45° flammability test was performed according to ASTM D1230-10 using a 45° angle chamber (Dwyer TC-45). 27 The flame exposure time was modified from 1 s, which is standard protocol, to 10 s. Fabrics were cut to 6.4 × 15.2 cm, treated with FR, and conditioned at 21 °C with 65% relative humidity (RH) for 48 h. All specimens were classified as Class I or Class III textiles that do not have a raised fiber surface.
Limiting Oxygen Index (LOI)
LOI was performed according to ASTM D2863-13 using a Dynisco Polymer Test Limiting Oxygen Index chamber. 28 Treated fabrics cut to 6.4 × 12.7 cm (W × L) were conditioned at 21 °C with 65% RH for 48 h prior to use. The average LOI values of four consecutive measurements were reported.
Microscale Combustion Calorimetry (MCC)
MCC was performed according to ASTM D7309-13 using a Govmark FAA Micro calorimeter. 29 The samples were investigated in an 80% nitrogen and 20% oxygen environment from 75-500 °C at a heating rate of 1 °C /s. Tree replicate measurements were conducted and averaged for each sample with fabric weight ranging from 4.5-5.5 mg.
Thermogravimetric Analysis (TGA)
TGA was performed on a TA Instruments Q500 under nitrogen. Sample masses of 4.0-8.0 mg were heated from 0–600 °C at a rate of 10 °C/min. Tree replicate measurements were conducted and their average thermal parameters were reported. Char yield and onset temperature was analyzed using Universal Analysis 2000 software.
Scanning Electron Microscopy (SEM)
The control and treated fabrics were examined by SEM (Phillips, XL 30 ESEM) at 1000 and 2000X magnifications. Samples were coated with a gold-palladium alloy using a vacuum sputter coater, and an acceleration voltage of 10 keV and a beam current of 0.5 nA were used.
Preliminary Study of Durability
Preliminary data for home laundering was obtained by following a modified version of AATCC Test Method (TM) 124-2011 for washing. 30 Briefly, a beaker containing 500 mL of deionized water was heated to 40 °C. The fabric was then added to the beaker and allowed to stir at 500 rpm for 10 min. This process was repeated 3 to 10 times. After each washing, the fabrics were dried at 110 °C for 10 min, re-equilibrated, and reweighed to determine the remaining percent add-on. Eq. 1 was used to calculate the percent addon of treated fabrics.
Results and Discussion
FRs are designed to incorporate moieties that reduce textile flammability. These systems typically include phosphorus, nitrogen, sulfur, silicon, and boron atoms combined in various ways to produce FRs. The composition of
Synthesis
The synthetic route of the FR herein is shown in Scheme 1. Phosphazene was reacted with 2,2′-dihydroxybiphenyl in the presence of acetone and K2CO3 at 0 °C under inert conditions according to the protocol published by Carriedo et al.
32
The purified precursor
Compound
Fabric Treatments
Cotton twill fabric was treated with
Flammability
The 45° flammability test, limiting oxygen index test (LOI), and microscale combustion calorimetry (MCC) test were performed to determine the flammability of
Flammability Test Results of Control and Treated Fabrics
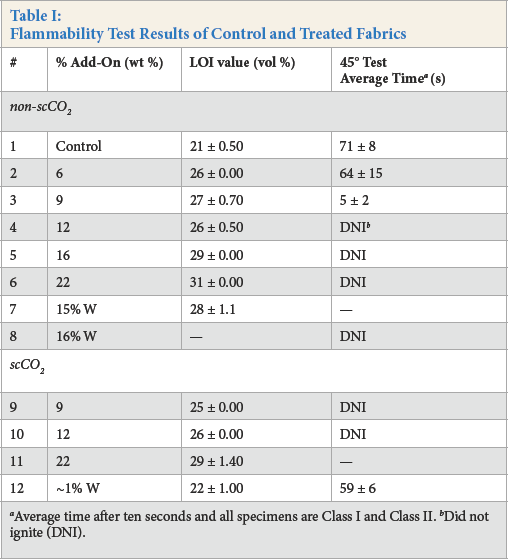
Average time after ten seconds and all specimens are Class I and Class II.
Did not ignite (DNI).

45° flammability test of control and treated fabrics.
Flame retardants with an LOI value of 27–28% are desirable for cotton textiles. LOI measures the minimum amount of oxygen required for fabric to burn in the presence of flowing oxygen and nitrogen gas, while maintaining a downward candle-like combustion.
8
Flame retardants that have an LOI value of less than 21% are flammable, those with an LOI value of 21–26% are slow burning, and those with an LOI value greater than 27% are self-extinguishing.37,38 However, higher LOI values indicate a stronger flame retardancy effect.
4
The LOI value of the control was 21%; this indicates the flammability of cotton fabric. Compound
MCC measures the rate at which the heat of fuel gasses are released from a solid under inert conditions.10,39,40 MCC profiles indicate the peak heat release rate (pHRR), total heat release (THR), and the maximum temperature (TMAX) achieved by treated fabrics (Figs. 4 and 5). The HRR profiles of the control and treated fabrics had one main stage between 300–375 °C. Treated fabrics had sharp heat release peaks that contrasted with the broad heat release peak observed for the control. The control exhibited a significantly lower pHRR value of 221 W/g, whereas treated fabrics pHRR values ranged between 367–577 W/g. Higher pHRR values indicated an increased amount of heat released during the burning process, which may result from rapid pyrolysis and combustion of treated fabrics.
39
This may account for the sharp HRR profiles of treated fabrics. Generally, as the add-on levels of
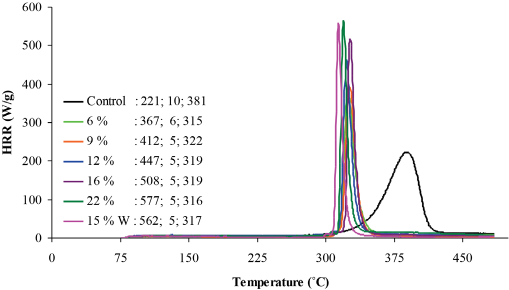
Heat release rate profiles of control and non-scCO2 treated fabrics. Data in the graph corresponds to pHHR, THR, and TMAX respectively.
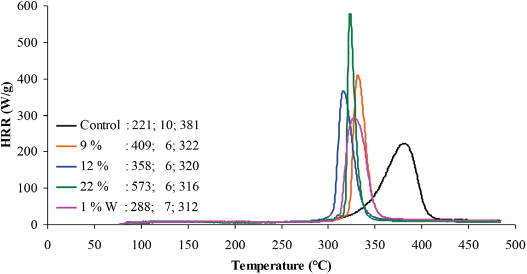
Heat release rate profiles of control and scCO2 treated fabrics. Data in the graph corresponds to pHHR, THR, and TMAX respectively.
The results of 45° flammability test, LOI, and MCC indicated that add-ons greater than 12 wt % had comparable results to those with 9 wt % add-ons. These findings imply that the threshold of
Thermal Behavior
TGA measures the change in mass with respect to temperature and was performed to investigate the thermal stability of
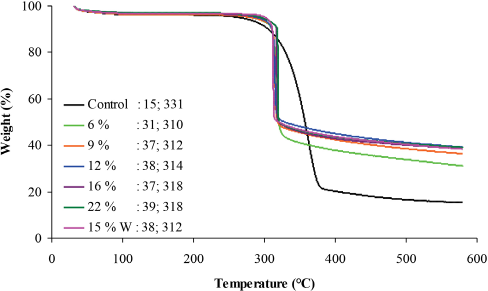
TGA thermogram of control and non-scCO2 treated fabrics with char yield and onset temperature values.
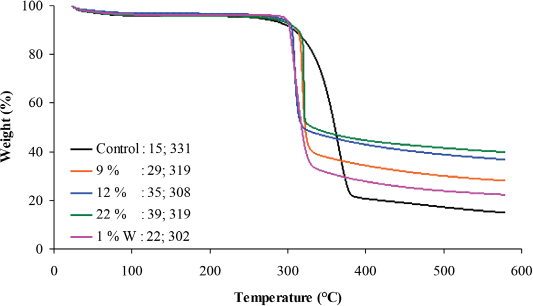
TGA of control and scCO2 treated fabrics with char yield and onset temperature values.
As the add-on of the FR increased from 6 to 9 wt %, increases in char yield and onset temperature were observed from 31–37% and 310–312 °C, respectively. These results support an add-on threshold of 9 wt % for
Studies have shown that an increase in the number of P-N bonds leads to higher char yield, a decrease in fuel formation, and a lower additive loading of the FR.7,41 Greater add-ons of 12, 16, 22 wt %, and 15 wt % W, had a char yield of 37–39%, and the on-set temperature ranged from 312–318 °C. The decrease in on-set temperature from 333 °C to 310 °C, when treated, was due to the interference of the FR in the pyrolysis of cellulose to levoglucosan. 43
Supercritical-treated fabrics with a 9, 12, and 22 wt % add-on had char yields of 29, 35, and 39% as compared to non-scCO2 treated fabrics with a char yield of 37, 38, and 39%, respectively. The onset temperatures for scCO2 fabrics were 308–319 °C, which were comparable to the onset temperatures of 310–318 °C for non-scCO2 fabrics. The char yields of scCO2 fabrics with an add-on of 9 and 12 wt % were slightly lower than the char yields obtained for non-scCO2 fabrics. The washed fabrics with an add-on of 1 wt % W had a char yield of 22% and an onset temperature of 302 °C, which was below the 9 wt % threshold of
Between the lowest add-on of 1–6 wt % and 9 wt %, an average 1.5 fold increase was observed compared to the that between the highest add-on of 12–22 wt % and 9 wt %, which resulted in a 1 fold increase in char yield. This data further supports that the threshold of
Compound
Analysis of Char Layer
Generally, burned cotton fabrics treated with an effective FR will generate char that acts as a protective barrier. The formation of a char layer is desirable to reduce heat transfer and the amount of volatile gases generated at the flame zone.45,46 Thehigher the char yield, the better the flame retardant properties, but the amount of char produced is dependent on the composition of the FR.
SEM was used to image control and treated fabrics to examine the unburned and burned sections of the fabrics (Fig. 8). The image of the control fabric (Fig. 8A) showed smooth elongated fibers, and the image of the burned control (Fig. 8B) showed that the fibers were mainly burned to ash. The highest add-on of non-scCO2 (Fig. 8C) and non-scCO2 washed fabrics (Fig. 8E), 22 wt % and 15 wt % W, respectively, were examined by SEM. The images of unburned non-scCO2 (Fig. 8C) and non-scCO2 washed fabric (Fig. 8E) displayed diamond shape crystallites of

SEM micrographs of unburned and burned fabrics. Unburned fabrics include (A) control, (C) 22% add-on of non-scCO2, (E) 15% W add-on of non-scCO2, (G) 12% add-on of scCO2, and (I) 1% W add-on scCO2. Burned fabrics include (B) control, (D) 22% add-on of non-scCO2, (F) 15% W non-scCO2, (H) 12% add-on of scCO2, and (J) 1% W add-on of scCO2.
The scCO2 fabrics with add-ons of 12 wt % and 1 wt % W were examined by SEM. The micrographs of scCO2 fabric (Fig. 8G) and scCO2 washed fabric (Fig. 8I) also showed diamond shaped crystallites of
The washed fabric of non-scCO2 (Fig. 8E) and scCO2 (Fig. 8I) appeared to form clusters between the elongated fibers, which may have resulted from washing the fabric and the hydrophobic nature of the compound to resist water molecules.47,48 The fibers of non-scCO2 and scCO2 fabrics remained intact and were thermally stable. A higher additive loading level of
Preliminary Durability Study
Fabrics treated using the non-scCO2 (conventional pad-dry-cure) method were washfast, had better durability properties than scCO2 treated fabrics, and gave an average 1.5 fold decrease in the loss of
Conclusions
The newly synthesized phosphazene-based FR,
Treated fabrics had comparable thermal and flammability results. LOI, MCC, and 45° flammability test results of treated fabrics established that
Compound
The mechanism of pyrolysis of the treated fabrics will be further examined by thermogravimetric analysis-Fourier transform infrared spectroscopy (TGA–FTIR), attenuated total reflectance-infrared spectroscopy (ATR-IR), and computational studies. These results will be reported at a later date.
Footnotes
Acknowledgements
This project was financed by the US Department of Agriculture. We thank Dr. Casey Grimm for ESI-MS and we thank Drs. Marsha R. Cole and Michael Santiago Citron for their assistance in proofreading.