Abstract
The odor-resistant property of socks is often linked with the antibacterial activity, depending on the antimicrobials used in odor-resistant finishing. However, antibacterial activity tests are not suitable for odor-resistant socks finished using non-antibacterial agents. In this study, a new method was developed to evaluate the odor-resistant effects of newly developed undyed, anti-odor cotton socks treated with a non-antibacterial agent. Worn, undyed (treated and untreated) cotton socks were sprayed with silver nitrate solution for color development under UV light. The resulting shade would be from white to brown, depending upon the intensities of the odor. A preliminary positive correlation between K/S values (color strength) and odor intensities with were found with the organoleptic test. The color strength method was more objective and had better reproducibility than the organoleptic test.
Introduction
The odor intensity of worn socks depends on the wearing individual, their physical activities, and the inner atmospheric situation in shoes, where oxygen is lacking. Malodor generated from socks can be very unpleasant. Therefore, socks with odor-resistant properties are being manufactured. Generally, antimicrobials such as quaternary ammonium salts or heavy metal cations were used in odor-resistant socks. 14 The antibacterial activity of socks can be used to evaluate their odor-resistant effects.5-7 However, most antimicrobials are biologically toxic.
It is reported that a new, “green” technology has been used for odor-resistant socks. 8 Socks finished by certain sol-gel techniques can release oxygen-containing negative ions during wear due to the combined effect of humidity from sweat, energy import (from walking & rubbing), and nanometer-sized particles (gel) on the fabric. The oxygen enrichment in shoes from these socks can restrain the propagation of anaerobes to prevent odor from being produced, instead of using antimicrobials to kill bacteria. 9 Therefore, traditional antibacterial activity tests are unsuitable for evaluation of these new odor-resistant socks.
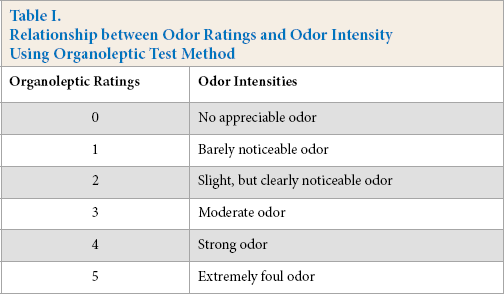
Since an organoleptic test (a test method that relies on a judges sense of smell to determine odor intensity) is a basic sense method, it can be used to evaluate all worn odor-resistant socks. Odor intensity is classified into 6 ratings (Table I).10,11 The organoleptic test requires at least five judges for one sample, with the ultimate result being the average of all the judges’ ratings. The advantages of the organoleptic test are ease of performance and low cost. However, the results of organoleptic test are often not reproducible, as they depend on the subjective assessment of the judges and other influencing factors (e.g., age, gender, and time of day). In addition, using the organoleptic method is unpleasant for the judges.
Relationship between Odor Ratings and Odor Intensity Using Organoleptic Test Method
In this study, a new test method based on color strength evaluation was introduced to reduce the need for organoleptic testing. In this method, a silver nitrate solution was sprayed on the surface of worn, undyed socks and the wet socks were exposed to UV light for color development. As a result, the surface of the undyed worn socks will become brown. The correlation between color strength (K/S value) and odor intensities (organoleptic rating) was investigated and comparison was made.
Materials and Methods
Undyed cotton socks having a plain construct of weft-knitted fabric (18.4 tex, weighing 156.0 g/m2, and pretreated with 10 g/L sodium hydroxide and 2 g/L hydrogen peroxide at 85 °C for 10 min) were used for samples. The white samples were worn by eight male students (20-26 years old) for 12 h, including at least 1 h of playing football or tennis. The samples were collected and sealed in airtight polyester plastic bags.
Organoleptic Measurement
The organoleptic panel consisted of five healthy female judges (20∼25 years old). This demographic is generally considered to have a more sensitive sense of smell than other groups.10,11 For each pair of samples worn and stored in sealed polyester plastic bags, the sealed mouth of the plastic bag was opened at a distance of ∼10 cm from the nose of the organoleptic panel member in random order.10,11 Organoleptic results were recorded independently by each judge on a scale of 0 to 5 (Table I). The averaged value of the judges’ results was used.
Color Strength Method
Color Reaction
A specimen (5 cm × 5 cm) was taken from the sole of the sample in the polyester plastic bag. The specimen was evenly sprayed with at least 20 mL of 0.1 M silver nitrate solution to moisten. The wet specimen was placed in a standard light box for 15 min under UV light for color development, and then taken out and air dried at room temperature. The specimen color ranged from white to brown, according to the odor intensity of the specimen.
Color Strength Measurement
Color strength (K/S) values of the color-developed specimen, with reference to the undyed sample, were measured using a Datacolor SF600 spectrophotometer. Each specimen was tested four times in various places at the maximum absorption wavelength of 550 nm (SWL method in AATCC Evaluation Procedure (EP) 6). 12 The average of the four values was used.
Results and Discussion
Test Results
The eight samples were tested with both organoleptic and color strength tests. The results are shown in Table II. The K/S value increased with increased organoleptic rating (Table II).
Results of the Two Test Methods for the Eight Samples
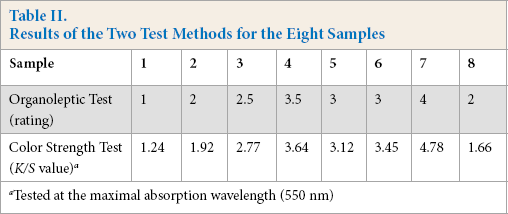
Tested at the maximal absorption wavelength (550 nm)
Correlation between Test Methods
The relationship between organoleptic rating and color strength (K/S value) test results is shown in Fig. 1. A straight line relationship (R = 0.9716) for correlation between the two tests was found. There was a positive correlation between K/S values and organoleptic ratings for the worn socks. Therefore, the instrumental color strength method, providing objective test results, can be used to evaluate the odor of worn socks. Compared with organoleptic testing, the color strength method had better reproducibility, without the need for judges to evaluate unpleasant odors.
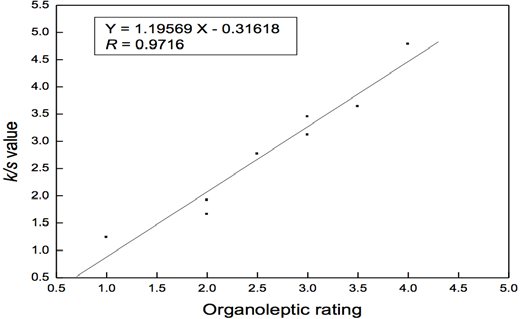
Relationship between K/S values and organoleptic ratings.
To confirm the positive correlation between organoleptic color strength testing, 22 undyed samples were worn by 22 volunteers and collected in polyester plastic bags and tested. The results are shown in Fig. 2. These results confirmed the relationship between K/S values and organoleptic ratings in Fig. 1, where K/S values increased with the increase in organoleptic ratings. The quantitative relationship of the results from both tests was simplified (Table III). There was a semi-quantitative relationship between K/S values and organoleptic ratings of worn socks, providing a new test method for the odor-resistant effect of socks against odor-producing anaerobic organisms. By measuring the K/S values of worn socks after treatment with silver nitrate solution for color development, the odor intensity can be evaluated. It must be stressed that this color strength method is only suitable for undyed socks.
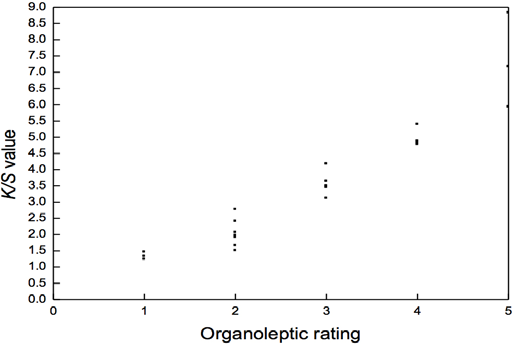
Comparison of test results from organoleptic and color strength methods.
Correlation between K/S Value Range and Organoleptic Rating

Tested at the maximal absorption wavelength (550 nm)
Color Reaction
Foot odor originates from secretions such as sebum, sweat, and proteins. Sweat consists of materials such as water, salts, urea, and lactic acid.1,13,14 Sweat and protein themselves are odorless, but they create an unpleasant odor under the action of anaerobes, which are easily propagated in an environment that lacks oxygen and has high humidity1,9 Foot secretion anaerobes and their metabolic products exist, containing odor ingredient residues on worn socks.
Color development with silver was complicated because odor ingredients contain many substances. Among others, urea was taken as the representative model to imitate the color reaction. An experiment confirmed that color development of urea with silver nitrate was similar to that of the specimen with silver nitrate (Fig. 3). The ultimate color became brown when both the specimen and urea reacted with silver nitrate solution for 15 min under UV light. Thus, one of the main color reactions probably occurred as shown in Scheme 1. Urea hydrolysis under UV light produced ammonia, and the ammonia reacted with silver nitrate to produce brown silver oxide. The amount of urea determined the amount of silver oxide formed. And this, in turn, determined the color strength of the specimen of sock sample. Previous experiments (Figs. 1 and 2) indicated that the color strength was directly related to the odor intensity (organoleptic rating) of the sample. This is because sweat containing urea is beneficial to the propagation of anaerobes in foot secretion under conditions of high humidity and lack of oxygen in shoes, resulting in odor being more easily produced and released.

Color comparison after color reaction of specimen (left) and 0.08 M urea (right) with 0.1 M silver nitrate under UV light.
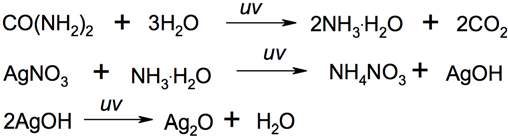
Probable color reaction mechanism of urea hydrolysis with silver nitrate.
Proteins in foot secretion may also play an important role in silver color development. Proteins are composed of amino acids. It is reported that the color of a test solution changed from colorless to orange-red then to brown-red when silver (I) ion reacted with an amino acid under certain conditions due to the new species generated. 15
It should be emphasized that these color reactions may be complex and further research needs to be conducted.
Conclusions
To evaluate the odor-resistant effect on worn socks using a new technology that releases oxygen-contained negative ions, a new test method based on color strength evaluation of undyed worn socks treated with silver ions was introduced to evaluate the odor intensity, minimizing the need for organoleptic testing. By spraying silver nitrate on worn test socks and then developing to give color change under UV light, the resulting K/S value was determined with a spectrophotometer. This exploratory study shows that there was a positive correlation between color strength and odor intensities. Compared with organoleptic test results, instrumental color strength testing using color strength was objective and had better reproducibility. Hence, the odor intensity of worn, undyed socks and its odor-resistant effect can be evaluated by measuring K/S values of silver ion treated samples.
For comparison with the SWL method, future work will include using the K/S Summed method in AATCC EP6, and its correlation with organoleptic rating results will be made.
Footnotes
Acknowledgments
This work was supported by the General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China [grant number 2009IK105].
