Abstract
Medical science benefits from models that predict antimicrobial activity, as well as the mechanism of antimicrobial agent activity effective in treatment of infectious diseases. A new method to determine the charge density of quaternary nitrogen in 3-(trimethoxysilyl)-propyldimethyloctadecyl ammonium chloride on the surface of polyester (PET) fabric was developed using C.I. Direct Red 47. The properties K/S (color strength), charge density, antibacterial efficiency, hydrophilicity, and surface potentials were examined. The experimental charge density value of 1.33 x 1016 units/cm2 was close to the calculated value of 1.35 x 1016 units/cm2 for 0.05% load quat-silane assumed by the amount of quat applied. In addition, correlation between K/S values and charge density had a linear correlation coefficient close to unity.
Keywords
Introduction
The world population is gradually increasing along with a growing number of bacteria resistant to antibiotics. This alarming trend can lead to serious health problems and some world regions might even require periods of quarantine to control the most severe cases. Furthermore, widespread use of high amounts of biocides as additives in commercial products, especially in textiles, can be harmful to the environment and living organisms because of the “releasing” mechanism.1,2 One method to obtain permanent antimicrobial efficiency is to covalently attach biocidal molecules to the material surface. Hence, a promising mechanism, which is based on physical interactions between the material surface and microorganism, can be observed with a reusable and durable surface against harmful pathogens.
Certain quaternary ammonium (quat) compounds are some of the most important and widely used antimicrobial agents. One such compound consists of a positively-charged nitrogen atom covalently bonded to four carbon atoms and a halogen counter ion.3,4
Natural quat salts such as choline or N, N,N-trimethylethanolammonium salts are water soluble and are essential nutrients occurring in living cells. An alkyl chain in a quat compound should have 8 to 22 even numbers of carbon atoms with a counter ion to show antimicrobial activity.3,5 Fig. 1 shows the chemical structures of common quat chloride compounds that demonstrate antimicrobial activity.
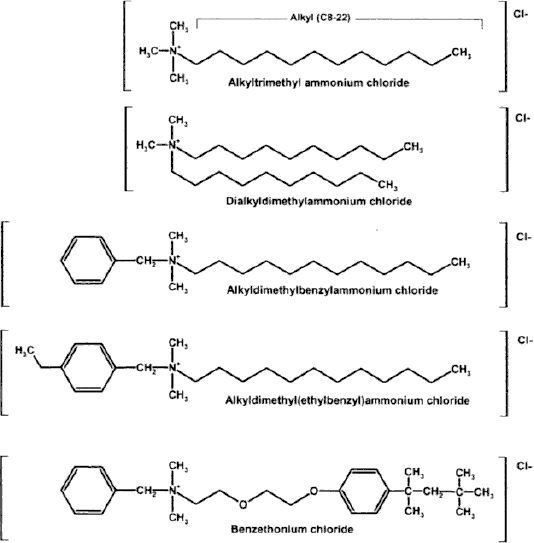
Chemical structures of common quaternary ammonium chloride compounds. Reprinted with permission from Taylor & Francis. 3
A number of organosilicone quat compounds are used as antimicrobial agents against a broad range of microorganisms.
6
Positively-charged quat groups are able to kill bacteria upon contact.
7
The charge density, which is usually expressed in number of moles of quat ions per square centimeter, is a critical factor in designing a surface of maximal antimicrobial efficiency with minimal surface charge. 3-(Trimethoxysilyl)-propyldimethyloctadecyl ammonium chloride (Fig. 2) is a quat organosilicone or a quat silane salt.
8
It demonst
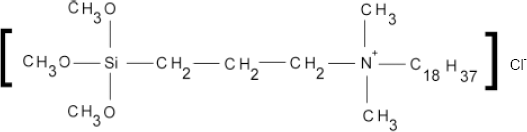
Chemical structure of 3-(trimethoxysilyl)-propyldimethyloctadecyl ammonium chloride. 9
These organofunctional silanes have hydrolyzable groups on the silicon atom, therefore under proper conditions, these can be hydrolyzed to silanols. Hence, silanol groups can react with each other on the fibrous surfaces. The chemical bonding on the surface is formed in two steps. First, the ion exchange process occurs in which cations of the silane quat compound replace protons from water on the surface-water interface. The interface between water and the surface generates a negative electrical charge. Second, the compound, by means of its silicon functionality, polymerizes and covalent bonds can be formed between the surface and the chemical compound. 9 , 10
Positive charges on the nitrogen atoms in the quat compound cause damage to microbial cell membranes, protein denaturation, and disruption of cellular structures. 11 Because bacterial cells generally include functional groups containing sulfur, nitrogen, or oxygen, 12 quats also eradicate microbes by death or inactivation (Fig. 3). In addition, the ionic interaction between the highly-positive charge on the surface and the cell membrane leads to neutralization of membrane components. As a result, the membrane loses its integrity and cell death occurs. 7 In this “nonleaching” method, antimicrobial efficiency is maintained as long as the compound is bound to the material's surface. 11
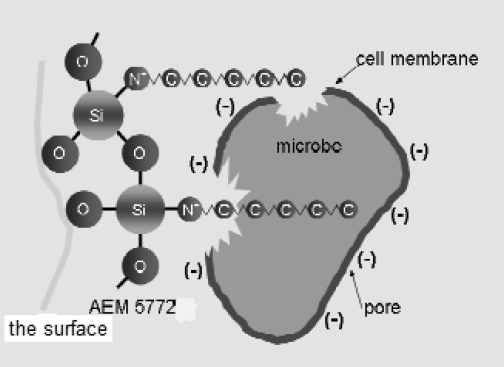
The physical process of antimicrobial activity between a quaternary ammonium compound and the cell membrane of microbes. Reprinted with permission from JepsenHealthCare. 18
Density of the quat groups has a strong relationship with antimicrobial efficiency 7 , 13 , 14 Murata, et al., 13 developed a technique to analyze the density of quat groups on the surface, assuming a ratio of 1:1 for fluorescein (sodium salt) to quat group. In this method, the treated surface was immersed in a solution of disodium fluorescein with distilled water. After removing unreacted molecules, treated samples were placed in 3 mL of 0.1% ocetyltrimethylammonium chloride in distilled water to desorb the dye via an orbital shaker. Absorbance of the resultant solution was measured at a wavelength of 501 nm after adding 10% 100 mM aqueous phosphate buffer at pH 8.0. Finally, the density of the dye bound to the quat groups was calculated assuming a ratio of 1:1 fluorescein:quat to determine the quat group ratio.
Murata, et al., 13 prepared antimicrobial poly(quaternary ammonium) (PQA) polymer by surface-initiated atom transfer radical polymerization of (2-dimethylamino) ethyl methacrylate (DMAEAM). Afterwards, polyDMAEAM was quaternized with alkyl bromides to achieve surface antimicrobial activity. The effective surface charge density for biocidal surfaces was accepted to be within 1 × 1015to 5 × 1015 charge units/cm2. If the surface charge density was greater than 3 × 1015, the cells died within a short period of time. It was found that this effect did not depend on the density and chain length of the PQA compound. Furthermore, it was believed that if the surface contained greater than 5 × 1015 charge units/cm2, it would kill at least a monolayer of Escherichia coli.
Kügler, et al., 7 obtained grafted quaternized poly(vinylpyridine) chains on glass surfaces and varied the charge density within the organic outer layer between ∼1012 to ∼1016 charge units/cm2. Their studies, however, documented the charge density of only the outer part of the polymer layer since the fluorescein was too bulky to fully penetrate the coating. Only the outer layer charge can be bonded by fluorescein complexation. In addition, Kügler, et al., 7 concluded the outer-layer charge density (OLCD) should be ∼1014 charge units/cm2 for E. coli and Staphylococcus epidermidis under low division conditions of the bacteria. Yet, under high division conditions, the OLCD needs to be ∼1013 charge units/cm2 for S. epidermidis and ∼1012 charge units/cm2 for E. coli.
It should be noted that the fluorescein complexation method does not always detect all charges present in the organic layer because of difficult dye molecule penetration into the layer. It becomes more difficult to determine the charge density for thicker organic layers.
The primary goal of this work was to develop a new method to estimate the nitrogen cation charge density of 3-(trimethoxysilyl)-propyldimethyloctadecyl ammonium chloride based coating the polyester fabric surface by dyeing it with commercial C.I. Direct Red 47 dye. The focus was to design a simple and more effective method than previous techniques. In addition, the nitrogen cation density of the samples (charge units/cm2) were compared, calculated (from the applied amount of quat-silane), and measured (from dyeing experiments). The effects of charge density on K/S values, antibacterial efficiency water absorbency speed, and surface potentials were also studied.
Experimental
Materials and Methodology
Knitted 100% polyester (PET) fabric samples were treated with different loads of 3-(trimethoxysilyl)-propyldimethyloctadecyl ammonium chloride (quat-silane) up to 0.30%. Treated and control (untreated) samples were used in this study (Table I). Treated fabrics were provided by Aegis Environments.
Characteristics of PET Fabrics Tested
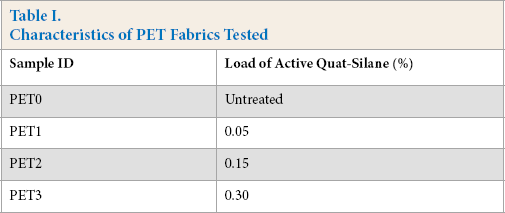
All fabrics were obtained as finish free, clean, and without any impurities. Solutions of quat-silane were diluted with distilled water to deliver different concentrations of the active antimicrobial ingredient to the fabrics. The fabrics were then padded and wet pick-up was adjusted to ∼100%.
After treatment, the substrates were dried and polymerization was conducted at 100 °C for 5 min. Samples were then washed and rinsed with de-ionized water repeatedly to remove any unbound quat-silane.
High purity Direct Red 47 (C.I. 14985) was used to dye treated and untreated fabrics. The chemical structure of this dye is shown in Fig. 4. The charge density was calculated and the color strength (K/S) value was observed for each dyed sample. Following the dyeing procedure, the remaining dye content in the solutions was kept and sealed for evaluation with a Cary 3E UV-Visible spectrophotometer. Based on the spectrophotometer results, the amounts of dye absorbed by the fabrics were determined. Details are given in subsequent sections.
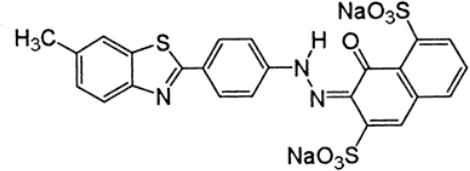
Chemical structure of Direct Red 47 dye. (Society of Dyers and Colourists and AATCC, 2009) 19
PET Fabric Dyeing and Charge Density Determination
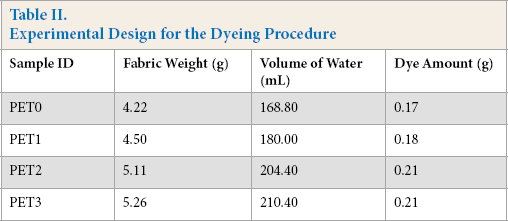
Fabric coloring was performed using a Werner Mathis AG type CH-8155 laboratory dyeing machine with a liquor ratio (LR) of 40:1. Table II outlines the experimental design for the dyeing procedure. After dyeing, the fabrics were removed immediately from the baths, with 10 mL of the dyebath solutions kept in sealed glass containers for spectroscopic analysis of the dye concentrations. To spectroscopically determine the amount of dye in the solutions, the remaining dye solutions were diluted 50 times with de-ionized water. Light absorbance of the Red 47 dye solutions was measured at λmax = 523 nm. According to the Beer-Lambert law, the standard (linear direct) calibration curve was obtained with a correlation coefficient of greater than 0.999.
Experimental Design for the Dyeing Procedure
The 50-fold dilution was performed after the experiment was carried out to find the actual amount of dye in each solution. A detailed explanation of the charge density calculation is provided in Results and Discussion section.
Microbiological Analysis
Antimicrobial tests were carried out using ASTM E2149-10 test method. This is a dynamic shake flask test and a quantitative method to determine the antimicrobial activity in test tubes using 1.0 g sample, 50 mL of 0.3 mM KH2PO4, 1 × 105 E. coli/mL, and 0.01% non-ionic wetting agent.
K/S Data Analysis
Dyed fabrics were left to stand for 24 h at room temperature, and then analyzed using an X-Rite Model SP64 spectrometer equipped with Color iControl software to obtain K/S data. K/S values were determined using the Kubelka-Munk equation. 15
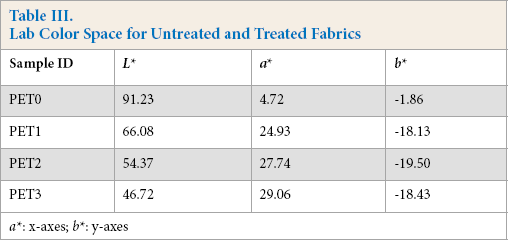
Lab color space data are given in Table III. L represents whiteness (0 is black, and 100 is white), a* shows the red-green axis. If the values are greater than zero, it is in red, and if the values are less than zero it is in green. In addition, b* demonstrates the yellow-blue axis. Positive values are in yellow, but negative values are in blue. “Zero” for a* and b* values indicates neutral tone.
Lab Color Space for Untreated and Treated Fabrics
: x-axes;
: y-axes
Water Absorbency Determination
Water absorbency speed for each fabric sample was determined using AATCC Test Method (TM) 79-2000. In this technique, the time length can be determined for fabrics to absorb a drop of water. In a typical experiment, a drop of water from a dropper is placed onto the fabric from 1 in. above, and then the average time to absorb into the sample is recorded. The test included washing with AATCC TMs 61-2A and 135 using 5, 10, 20, 30, and 40 wash cycles.
AATCC TM 61-2A is an accelerated laundering procedure, which is performed at 49 ± 3 °C with 50 steel balls and 0.15% AATCC standard detergent. AATCC TM 135 is carried out in a regular home washing machine for a 9-min cycle by adding 46 g of AATCC liquid reference detergent at cool temperature and then dried 40 min by home dryer at low heat.
Surface Potential Assessment
Monroe Electronics Fieldmeter Model 177 and the probe-type model 1036E were used to observe the surface potential of the samples. 16 Electric field is defined as the electric force (F) per unit charge (q). The relationship between the electric force and the charge is determined from Eq. 1 via the vibrating reach approach. It can be displayed with SI units of Newton per coulomb (N/C) or, equivalently, volts per meter (V/m).
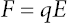
E is the electric field strength and is determined by the magnitude and locations of the other charges.
Results & Discussion
Charge Density Determination
Negatively-charged sulfonate groups of a typical direct dye (anionic) molecule such as CI. Direct Red 47 dye can easily bind to the positively-charged quat group in the dynamic dyeing experiment. 9 In the current work, the light absorbance of the remaining Red 47 dye solution after the dyeing process for the treated fabric showed a strong relationship between direct dye uptake and active quat-silane percent for the fabric coating. It gave an indication that the coating structure was sufficiently accessible to the dye molecules. The concentration of the dye in the remaining solutions after the dyeing process decreased as the quat-silane content increased (Table IV). Therefore, Red 47 dye uptake as the number of molecules for each fabric surface could be determined. Table V represents the percent dye absorbed by each fabric and the number of moles, which was determined from the molecular structure of Red 47.17,19
Concentration of Direct Red 47 Dye in Remaining Solutions after the Dyeing Process
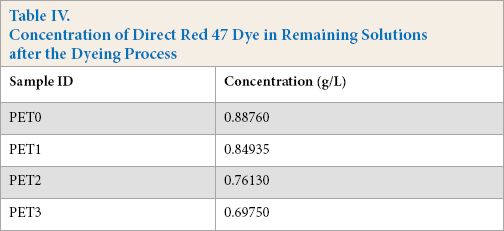
It is believed that the quat-silane coating and dye molecules were uniformly distributed for each fabric and the interaction between the positively-charged quat nitrogen atoms were accessible to Red 47. There are two possibilities for this interaction: (1) two sulfonate groups from one molecule of Red 47 were attached to two different nitrogen cations (Case I), or (2) only one sulfonate group on the Red 47 molecule was attached to one nitrogen ion in the quat molecule (Case II) during dyeing processes.
It should be also considered that dye can be adsorbed indirectly by fabric surfaces even if there are no positively-charged quat nitrogen atoms on the surface. This would be considered as background activity. Therefore, it was plausible to subtract the “number of nitrogen ions” found on the untreated sample (without quat-silane treatment) from all other samples to account for this background data. Table VI shows the total number of nitrogen ions (charge) by using the number of moles of Red 47 from Table V for Case I and Case II for only the top fabric layer. This data came from the experiment called “measured” number of nitrogen ions.
Percent Absorbed Direct Red 47 Dye for Each Fabric
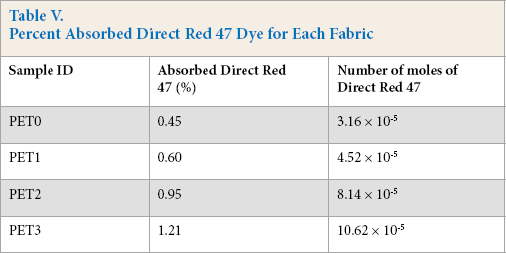
In Table VI, the total number of nitrogen ions for the treated fabrics with the quat-silane were determined after subtracting the number of nitrogen ions detected on the untreated fabric (without quat-silane treatment). The total number of nitrogen ions for PET1 was lower than that for PET0 (Table VI). Consequently, density (units/cm2) was calculated by taking into account the fabric surface area.
Total Number of Nitrogen Ions for Case I and Case II (Experimental Results) for Top Surface of Each Fabric
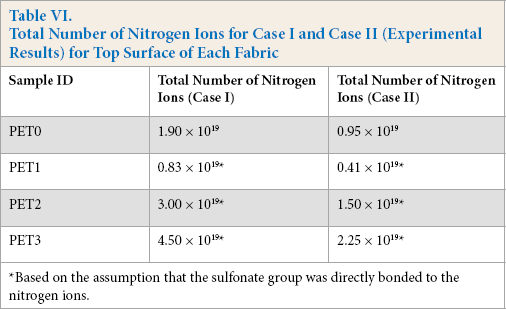
Based on the assumption that the sulfonate group was directly bonded to the nitrogen ions.
The formula for calculating the total number of nitrogen ions (TNC, units/cm2) for the treated fabric is given in Eq. 2.
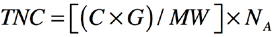
C is the weight percent of the active quat-silane content (Table I), G is the fabric weight (the fabric weight of PET used in this experiment was 0.0195 g/cm2), MW is the molecular weight of active quat-silane (the molecular weight of the quat-silane used in this experiment was 435.189 g/mol), and NA is the value of Avogadro's number (6.022 × 1023 mol−1).
An example of calculation of the nitrogen ions per cm2 on PET1 surfaces is shown below:
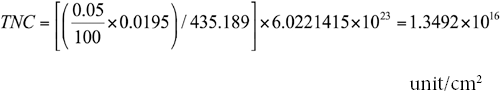
unit/cm2
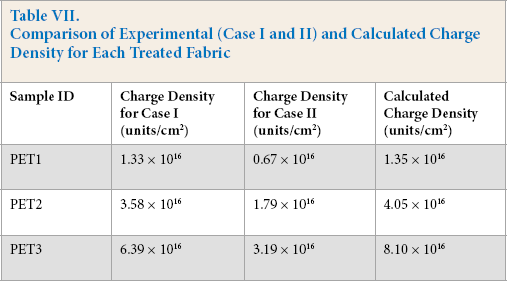
The charge densities for all treated fabrics were calculated in the same way and are given in Table VII under “Calculated Charge Density.”
The results (Table VII) indicate that the two Red 47 sulfonate groups bonded with two different nitrogen ions on the fabric because the charge density for Case I was closer to the calculated charge density (Eq. 2). Interestingly, after increasing the active amount of quat-silane on the fabric surface, the two Red 47 sulfonate groups lose the efficiency to bond with two different nitrogen ions. Hence, it is likely that only one sulfonate group on the dye molecule was attached to one nitrogen ion for each quat-silane molecule in most cases.
Comparison of Experimental (Case I and II) and Calculated Charge Density for Each Treated Fabric
Antibacterial Efficacy
Quat-silanes are frequently used as antibacterial agents for a broad range of materials used in our daily lives and/or medical applications. The quat-silane drastically increases the death rate of infectious bacteria by disturbing the cell membrane of microbes even if a very small amount is used.
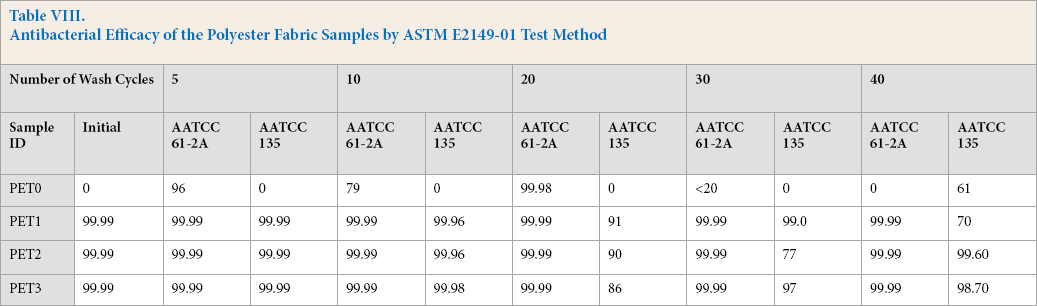
Antibacterial activity of untreated and quat-silane treated fabrics are given in Table VIII. As expected, the untreated PET fabric did not show any antibacterial activity before washing and after 30 wash cycles of AATCC TM 135. However, some antibacterial activity (61% bacteria reduction) was seen after 40 cycles. A high number of wash cycles probably led to accumulation of some antimicrobial agent on fiber surfaces, which might come from the AATCC standard detergent that was used during the washings, and/or from changes of the surface morphology that was caused by washing.
Antimicrobial activity of the untreated fabrics fluctuated after multiple washings with AATCC TM 61-2A (Table VIII). Before washing, the fabric did not demonstrate any antibacterial activity. However, after five washings, the untreated fabric showed 96% bacteria reduction and the percent bacteria reduction decreased to 79% after 10 wash cycles. Use of steel balls and detergent during washing most probably lead to the antibacterial activity of the untreated fabric. Interestingly, maximal antibacterial activity was observed after 20 wash cycles with the value of 99.98% for the untreated sample. At this point, mechanical abrasion and deterioration of the untreated fabric and detergent led to releasing some active compounds to the surface which may have caused the greatest antibacterial activity. A greater number of wash cycles (30 and 40) probably removed the most of the chemicals that accumulated on the surface, leading to untreated fabrics without antimicrobial activity.
On the other hand, all treated fabrics gave excellent antimicrobial efficacy with a bacteria reduction rate of 99.99 % before washing, even when a very low loading of 0.05% quat-silane was used (Table VIII). All treated fabrics had superior antibacterial performance (99.99% bacteria reduction rate) determined using AATCC TM 61-2A with steel balls and after 40 wash cycles. Therefore, the quat nitrogen sites in the quat-silane coating on the sample were very durable and resistant against washing under these conditions.
PET treated with various amounts of quat-silane showed a significant degree of bacterial growth inhibition. Interestingly, the antimicrobial performance of treated samples decreased to 91% after 20 wash cycles using AATCC TM 135 (Table VIII), even though the samples killed 99.99% of the bacteria after 5 and 10 wash cycles. Use of different amounts of detergent at the cool temperature without steel balls for AATCC TM 135 were the main differences compared to use of AATCC TM 61-2A. Therefore, it was concluded that the effect of detergent quantity and wash temperature were more dominant than the presence of steel balls. After 30 wash cycles with AATCC TM 135, the percent bacteria reduction fluctuated for various loads of quat-silane- treated PET from 0.05% to 0.30% (Table VIII). Percent bacteria reduction differences for the various loads of the quat-silane-treated PET became meaningful after 40 wash cycles. PET2 and PET3, with a quat-silane amount of 0.15 and 0.30 respectively, gave a percent bacteria reduction of 99.99% after 40 washes. PET1 (0.05% quat-silane) gave only 70% percent bacteria reduction. The performance of PET1 after 40 washes was very close to PET0 (untreated with the quat-silane) after the same number of wash cycles. In this case, a very low load of quat-silane was used, probably leading to partial removal of quat-silane.
The comparison of unwashed and washed fabrics after even 40 wash cycles with the different washing test methods demonstrates that the quat-silane has a high-potential as a stable antibacterial agent.
K/S Data Evaluation
After dyeing the treated fabrics with Red 47, a very strong linear relationship between the color strength (K/S) and the amount of quat-silane on samples (%) was observed with R 2 of 0.9954 as shown in Fig. 5. This proves that increasing charge density (Table VII) by using a greater amount of active quat-silane for the treated fabrics led to a higher number of sulfonic groups from Red 47 dye binding to the quaternary nitrogen sites.
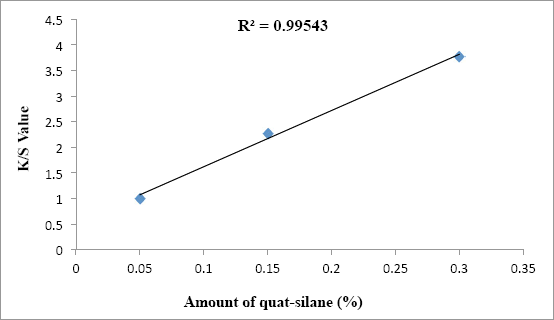
The effect of quat-silane amount on color strength (K/S) values after the dyeing process for treated PET fabrics.
In the Kubelka-Munk equation (Eq. 2), diffuse reflectance spectra can be correlated with dye concentration in the fiber sample. If the absorption coefficient increases, the scattering coefficient will most probably decrease, leading to a higher K/S value.
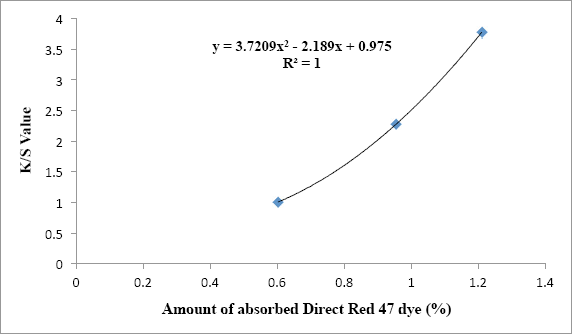
Correlation between K/S values and amount of absorbed Direct Red 47 dye after the dyeing process for treated fabrics.
Increasing the amount of Red 47 dye content for the treated fabrics (the percent of absorbed dye) improved the color strength (K/S) values (Fig. 6) with the perfect correlation for the polynomial trend (Eq. 3) with R 2 of 1. Figs. 5 and 6 show two important experimental findings demonstrating that PET fabrics having a quat-silane coating can be dyed with direct dyes. A lower dye uptake can be seen when a small amount of the quat-silane was used for the treatment of PET fabric.
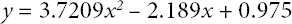
y is the K/S value for treated fabrics and x is the percent absorbed Red 47 dye.
A linear correlation between K/S values and concentration of absorbed Red 47 dye was seen with an R 2 of 0.9807 for the treated fabrics. The R 2 value for the polynomial trend was higher than that of the linear trend. In general, a linear relationship between dye concentration in fibrous substrates and K/S is observed. In this experiment, sometimes one Red 47 dye molecule occupied two different positively charged quaternary nitrogen atoms (Case I), but other times, one Red 47 dye molecule bound to only one positively charged quaternary nitrogen atom (Case II). This became more important as the charge density on the fabrics increased (Table VII). This phenomenon was a reason to observe the polynomial trend instead of the linear trend in the present experiment.
In addition, the correlation between K/S values and charge density (charge units/cm2) had the nearly perfect correlation coefficient of 0.99973 (Fig. 7). Similar analysis between K/S values and calculated charge density (assumed by amount of quat applied) with a linear correlation gave an R 2 of 0.9954. This method was more useful to fully detect almost all the charges present on the fabric surfaces. A 0.05% load of active quat-silane was enough to show 99.99% bacterial reduction (Table VIII). Therefore, a minimum K/S value of 1.007 was required for excellent antimicrobial activity.
Antibacterial Efficacy of the Polyester Fabric Samples by ASTM E2149-01 Test Method
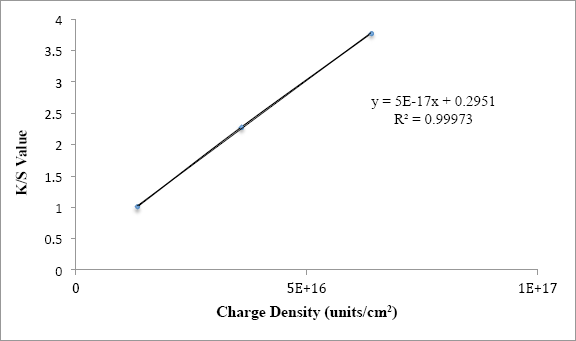
Correlation between K/S values and measured charge density after the dyeing process for treated fabrics.
As seen in Table III, increasing the charge density also caused an increase in the amount of dye molecules bonded to the treated surface. Hence, the whiteness value decreased from 91.23 to 46.72 (49%) when the active-quat silane increased from 0% to 0.30%. In addition, the red tone increased from 4.72 to 29.06 (516%) when untreated fabric and 0.30% active quat-silane treated fabric were compared. Interestingly, the blue tone did not decrease with increased charge density. Untreated PET fabric had a b* value of -1.86, whereas fabrics treated with 0.05% to 0.30% quat-silane slightly varied from -19.50 to -18.13.
Assessment of Water Absorbency Speed
PET is hydrophobic in nature; so it has many advantages such as quick drying, being easy to wash, able to hold its final form well, etc. After polymerization of quat-silane at 100 °C on the PET surface, a hydrophobic coating film with a crosslinked polymer was formed on the sample surface. AATCC Test Method (TM) 27-2000 is commonly used for measuring effects of fabric wetting. Based on TM 27-2000, the wetting time for the polymerization of quat-silane on the PET surface was determined (Table IX). Water absorbency time for untreated fabric was more than 10 min when measured before washing, however, this time changed significantly depending on the method used and number of wash cycles. For instance, after washing via a regular home washing machine using AATCC TM 135 with wash cycles from 5 to 40 times, the fabric absorbency speed was almost instantaneous. However, if the untreated sample was washed with steel balls using AATCC TM 61-2A, the different number of wash cycles used changed the water absorbency speed. For example, after 5, 30, and 40 wash cycles, the fabric absorbency speed was greater than 10 min. However, after 10 and 20 wash cycles, the water absorbency speed was 3 min and instantaneous, respectively. According to the TM 61-2A results, the water absorbency speed gradually decreased from more than 10 min to an instant after 20 wash cycles respectively. Similar unexpected behavior was also observed during antimicrobial testing after a different number of wash cycles using the same washing procedure (Table VIII).
Water Absorbency Speed (min) for Untreated and Treated Fabrics Before and After Washing
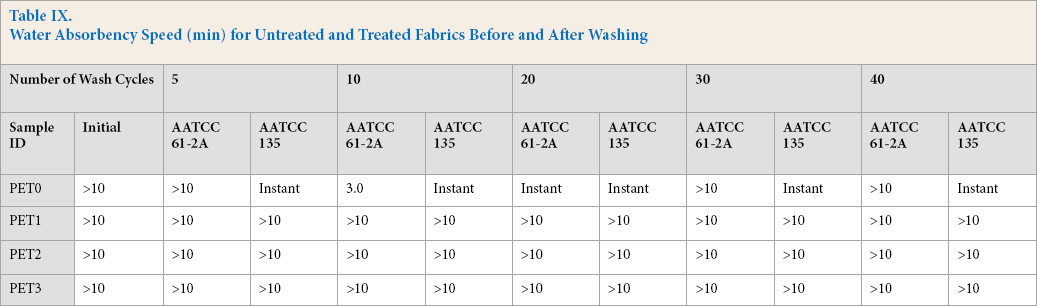
Washing with steel balls and detergent at a high temperature can definitely change the surface morphology. The effect of the steel balls was probably dominant for surface abrasion from 5 to 20 wash cycles, therefore, the water absorbency speed decreased from more than 10 min to instantaneous, respectively. However, increasing the number of wash cycles to greater than 30 changed the total effect of steel balls and detergent at a high temperature on the fabric because the level of surface abrasion and deterioration were not the same after 10 and/or 30 wash cycles.
All treated fabrics showed water absorbency speeds greater than 10 min regardless of the TM used (TM 135 or TM 61-2A) within the 5 to 40 wash cycle range. This indicates that the hydrophobic quat-silane coating on PET fabric surfaces was very durable against long wash cycles up to 40 with different types of washing methods even if they were treated with a low amount of 0.05% quat-silane.
Determination of Surface Potentials
PET fibers do not contain any basic or acidic groups. Therefore, in general, some surfactants can be used to favor dye uptake during the dyeing process.
The positive results of the investigation of Red 47 dye adsorption on PET fabrics treated with positively-charged quat-silane led to surface potential studies. Such surfaces might exhibit ionic strength and an increased surface potential.
The surface potential was observed to increase from 1.14 kV to 2.81 kV (146%) when the greatest amount of quat-silane (0.30%) was used on the PET fabric (Table X). It is important to mention that the greatest charge density on the fabric surface was observed when the quat-silane amount of 0.30% was used (see Table VII). The density of positively-charged quaternary nitrogen atoms of between 6.39 × 1016 units/cm2 and 8.10 × 1016 units/cm2 probably changed the surface morphology and increased the ionic strength, which may lead to a higher surface potential.
Surface Potential for Fabrics Before and After Washing
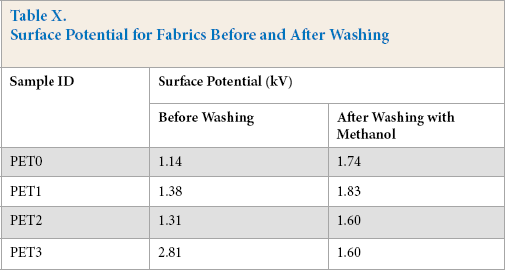
In addition, after washing with methanol, similar surface potentials were observed for all treated and untreated control samples in the range of 1.60 to 1.83 kV (Table X). This effect can likely be attributed to disruption of the ionic charges on fabric surfaces. As a result, after washing with methanol, all treated and untreated samples demonstrated similar behavior.
Conclusion
A new method was developed to determine the charge density of PET fabric treated with a quat-silane antimicrobial agent using Direct Red 47 dye. The experimental charge density value of 1.33 × 1016 charge units/cm2 was very close to the calculated value of 1.35 × 1016 charge units/cm2for 0.05% load quat-silane treated fabric. Hence, it can be concluded that almost all the charges present on the fiber surface were detected by sulfonate groups on the dye molecule at lower concentrations.
This method is significant when analyzing thicker organic layers after quat-silane treatment. The previous method 7 considered only OLCD (outer-layer charge density) because fluorescein dye molecules were unable to bind to all charges present in the organic layer. This occurred because the hydrophobicity of the PET polymer chain backbone hindered dye molecule penetration. In this study, the PET fabric dyeing process with Red 47 was performed under dynamic and active conditions with a relatively long time of more than 75 min. The fluorescein complexation method was performed for 5-10 min under immobile conditions. This new method is more useful for fully detecting the charges present in the layer on fabric surfaces because of the dynamic environment and much longer dyeing time. Furthermore, this technique appears to be easier and simpler than the fluorescein complexation technique.
It should be pointed out that an increased surface charge density caused a higher surface potential after treating the PET fabric with 0.30% quat-silane. The red tone and K/S values increased, but the whiteness value decreased when the charge density increased on the surface based on Red 47 dyeing.
A greater charge density or concentration of the quat groups on the PET surface led to higher color strength values after dyeing with Red 47.
No changes in the water absorbency time were observed after washing with two different methods, AATCC TM 61-2A and AATCC TM 135, at up to 40 wash cycles for all fabrics treated with 0.05% to 0.30% quat-silane. Moreover, after using these washing methods, the coated fabrics maintained a superior antibacterial efficacy as demonstrated by a 99.99% bacterial reduction rate even after 40 wash cycles. This indicates that very strong surface bonds were formed by the quat-silane to the PET fabric surfaces.
For future research in this field, some other types of isolated, pure direct dyes should be used with a different number of sulfonate groups and size. Such an approach would allow a more precise determination of fabric surface charge density. Further studies improving the determination of experimental charge densities at lower and higher surface charge concentrations could be of interest. Such an investigation might reveal a better direct dye type to elicit the minimum surface requirements.
Establishing a correlation between K/S values and surface charge densities is recommended to determine the minimum surface requirements for the desired antimicrobial activity.
Footnotes
Acknowledgments
We thank Professor Stephen Michielsen, Birgit Andersen, and Jeffrey Krauss of the College of Textiles at North Carolina State University for letting us use their analytical facilities. We would also like to thank the Republic of Turkey Ministry of National Education for financial support. We appreciate the valuable comments and suggestions from a reviewer. Matthew Doyle of ORCO Organic Dyestuff Corporation is also acknowledged for a useful recommendation.
