Abstract
The aerobic biodegradability of acrylate copolymers for warp sizing was studied using the shaking-bottle incubating method for their COD removal rates after being cultivated for 30 days. Influences of acrylate unit chemical structure, including the alkyl chain length of the ester group, α-methyl group, and molar content of acrylate units, on the biodegradability of the sizes were investigated. The acrylate copolymers were prepared via free radical copolymerization of acrylates with acrylic acid in ethanol. The biodegradability of acrylate copolymers strongly depended on the structure and molar content of the acrylate units. After being cultivated for 30 days, poly(MA-co-AA) was biodegraded substantially and the COD removal rate exceeded 50%. However, the biodegradability of poly(BMA-co-AA) was poor and the removal rate was 10%.
Considering both discharge quantity and effluent composition, wastewater produced in the textile industry seriously damages the environment. 1 , 2 More than 2.4 billion tons of textile industry wastewater, with over 300,000 tons of COD (Chemical Oxygen Demand) producing components, were discharged in China in 2010 (National Bureau of Statistics of China, 2010). Of the COD producing components discharged in textile finishing, more than half are sizing agents. 3 About 300,000 tons of sizes are consumed every year in China. However, the sizing agents in the desizing wastewater were discharged without recycling. Therefore, biological treatment of textile sizes should be carried out to reduce sizes contribution to wastewater pollution. In recent years, new strict regulations, with increased enforcement, have been mandated in many countries to lower the COD value of discharged textile industry wastewater.
Conventional sizing agents used are starch, polyvinyl alcohol (PVA), and acrylate copolymeric sizes. Of these, starch possesses the best biodegradability PVA has been restricted or banned in sizing process in many European countries. 4 Acrylate copolymeric sizes possess good adhesion to synthetic fibers compared with starch and PVA; however, few researchers have studied the environmental impact of acrylate copolymeric sizes.
Acrylate copolymeric sizes are copolymers of various acrylates with acrylic acid, mainly through solution or emulsion polymerization. 5 , 6 In the copolymer, the molar content of acrylate units, which play a major role in end-use performance, exceeds 50%. 7 , 8 Acrylic acid monomer endows the copolymers with better water solubility or dispersity to make sizes mix and desize more easily. The most commonly used acrylate monomers include methyl acrylate, ethyl acrylate, butyl acrylate, methyl methacrylate, and butyl methacrylate. The structural formula of acrylate copolymeric sizes is shown in Scheme 1.
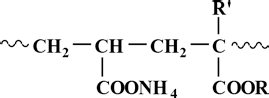
Acrylate ester copolymer structures.
The molecular structure of acrylate copolymers depends on the types of R and R’ groups (Scheme 1). It was reported that the structural parameters of acrylate units, including the alkyl chain length of ester groups, α-methyl group, and molar content of acrylate units, determine the end-use performance of water-soluble acrylate copolymers. 9 , 10 However, little is known about the relationship between the structure of acrylate copolymers and environmental performance. If the dependence of biodegradability on the structure of acrylate copolymeric sizes were known, acrylate copolymeric sizes with good environmental performance could be designed and produced. Unfortunately, no previous investigations have given this problem serious consideration.
Primary aerobic biodegradability of organic contaminants in wastewater can be evaluated by the COD removal rate.11-13 COD was used as a measure of the oxygen equivalent of organic matter content for a sample that was susceptible to oxidation by a strong chemical oxidant. 14 The higher the removal rate, the better the biodegradability. By measuring COD before and after cultivation, aerobic biodegradability of acrylate copolymers in water can be evaluated by COD removal. As a result, a relationship between biodegradability and molecular structure of acrylate copolymers was determined. Therefore, the structures of copolymers having good environmental performance can be determined after copolymers with the optimal type and number of acrylate units were prepared and tested.
Experimental
Materials
Methyl acrylate (MA), ethyl acrylate (EA), butyl acrylate (BA), methyl methacrylate (MMA), butyl methacrylate (BMA), acrylic acid (AA), and benzoyl peroxide (BPO) were all chemically pure grades and used directly as received. Ethanol, potassium dichromate, silver sulfate, ferrous ammonium sulfate, potassium dihydrogen phosphate, calcium chloride, ferric chloride hexahydrate, and other chemicals were analytical grade and used directly as received.
Synthesis and Determination of Acrylate Copolymers
Acrylates were copolymerized with acrylic acid in ethanol solution in a 500 mL flask. The flask, equipped with a condenser, was immersed in a thermostatically-controlled water bath. Ethanol (120 mL) was added to the flask and heated reflux (78 °C). The initiator, BPO (0.72 g), was dissolved in 120 g of acrylate-AA monomer blends at room temperature, and 40 mL of the initiator-monomer solution was added to the flask. Forty minutes later, the remaining initiator-monomer solution was added continuously over 1 h. When the initiator-monomer solution addition was completed, the copolymerization was started. One hour later, more initiator (0.24 g) was added to the flask. The copolymerization was continued under continuous stirring and reflux for 6 h. About 20 mL of reaction product was then removed for subsequent analysis of residual monomer and copolymer composition. Thereafter, the remaining product was neutralized with 10% ammonia solution when the temperature was reduced to 60 °C. The product was diluted with 260 mL of distilled water and evacuated by distillation under reduced pressure (0.01 mPa) to remove ethanol from the product. Finally, the product was diluted with distilled water to ∼440 mL. 7 , 9 Conversion of monomer was determined by the method used in previous investigations. 7 , 15 The apparent viscosity of the acrylate copolymer product was measured using a rotary viscometer (NDJ-79) using a concentration of 4% (w/w) at room temperature as described in a prior method. 7 , 9 Acrylate copolymer products produced in this study were characterized (Table I).
Characteristics of Acrylate Copolymeric Sizes
Determination of COD Removal
The shaking-flask method for primary aerobic biodegradability determination, ISO 7827-1984(E), was used. 16 A phosphate buffer solution was prepared by dissolving KH2PO4 (8.50 g), K2HPO4•3H2O (28.53 g), Na2HPO4•12H2O (67.02 g), and NH4Cl (1.70 g) in distilled water and diluting to 1 L. The pH value of the buffer solution was ∼7.4.
After 0.1 g of sample was added to a volumetric flask and diluted with distilled water to 1 L, 2 mL of the phosphate buffer solution, 1 mL of 27.50 g/L CaCl2 solution, 1 mL of 11.00 g/L MgSO4 solution, 4 mL of 0.15 g/L FeCl3 solution, 1 mL of 40.00 g/L (NH4)2SO4 solution, and 20 mL inoculated water were added to the flask. Inoculated dilution water came from Tai Lake in China. A blank control was also prepared with distilled water.
In each 250 mL culture flask, 150 mL of above-mentioned blend solution was added. After being sealed, the flasks were placed on a constant temperature shaking table (L-24A-1, Xiamen Rapid Co. Ltd.) and cultivated at 20 °C with a rotational speed of 100 rpm. The cultivation period was 30 days. A flask was taken out every five days and the blend solution in the flask was used for COD determination in accordance with the standard analytical method. 14 All experimental runs were performed in duplicate to diminish errors. A percent removal of COD at time R for the sample was calculated using Eq. 1.
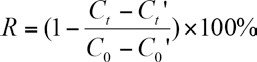
C t and C' t are the COD of sample and blank after culture respectively, and C0 and C'0 are the COD of sample and blank before culture respectively.
Determination of Degradation Kinetic Factors
Generally COD degradation kinetics fits the characteristic first-order reaction kinetics model. 17 , 18 The COD degradation rate was calculated using Eq. 2.
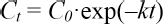
C0 (mg/L) and C (mg/L) are the COD values before culture and after t-day culture, respectively, t (d) is the culture time, and k (d−1) is the degradation rate coefficient.
Eq. 2 expressed as the natural log results in Eq. 3.
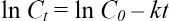
The half-life period (t1/2) of the COD is expressed by Eq. 4.
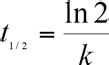
Statistical Analysis
Kinetic factors, including k values and the correlation coefficient r were calculated and analyzed using SPSS 13.0 software.
Results and Discussion
Basic characteristics of the prepared acrylate copolymers are summarized in Table I. The apparent viscosity of the copolymers was observed to decrease with the increase in MA unit content and the alkyl chain length of the ester group in the acrylate units. The acrylate copolymers become more hydrophobic with increased MA unit content and increased ester group alkyl chain length. Increased hydrophobicity decreases the intermolecular forces between the copolymers and water. Therefore, the apparent viscosity of the copolymers decreased. Solid content of the acrylate copolymers was similar to that of most acrylate copolymers used in warp sizing. Moreover, monomer conversions for all the acrylate copolymers were higher than 94%, indicating that most monomers were converted to polymers.
Effect of Acrylate Structure
Figs. 1 and 2 show the influence of the alkyl chain length of the ester group in acrylate units on the COD over time and on COD removal rates (i.e., R in Eq. 1), respectively, for the copolymers. The COD over time and COD removal rates were greatly dependent on the ester group alkyl chain length. The increase in the ester group alkyl chain length increased COD and reduced COD removal rates. Thus, poly(MA-co-AA) was more biodegradable than copolymers with longer ester group alkyl chains.
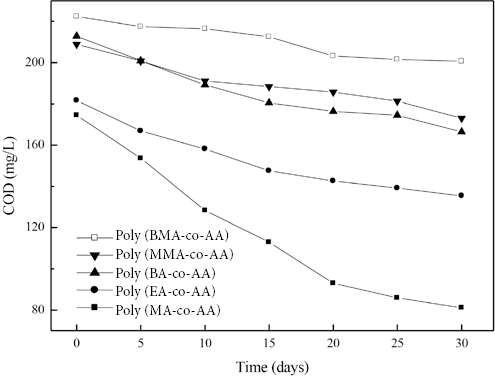
Effect of chemical structure of acrylate units on COD of acrylate copolymers over time.
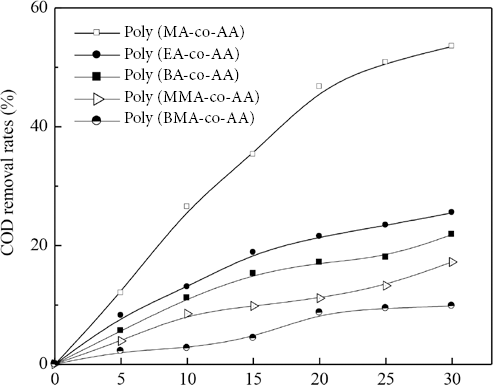
Effect of chemical structure of acrylate units on biodegradability of acrylate
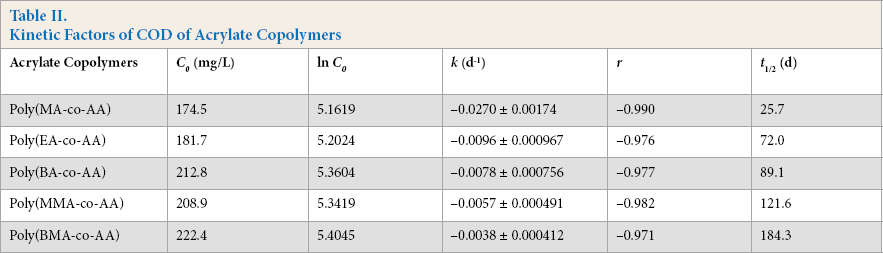
Table II presents the effects of alkyl chain length of the ester group in acrylate units on the kinetic factors of the copolymers, such as degradation rate coefficient (k) and half-life period (t1/2) of COD. With the increase in the ester group alkyl chain length, the absolute value of k decreased sharply and t1/2 substantially increased. For example, the k of poly(MA-co-AA) was about three times greater than that of poly(BA-co-AA). The absolute values of the r values of the first-order kinetic equations were all above 0.97. Based on variance analysis of the data, confidence levels of the COD kinetic equations were all above 95%. The kinetic factors obtained from the equations were in agreement with the experimental COD removal rate results.
Kinetic Factors of COD of Acrylate Copolymers
It is reported that poly(alkyl acrylate) is generally resistant to biodegradation due to their hydrophobicity19–21 The random copolymer poly(acrylate-co-acrylic acid) is easily degraded by microorganisms due to the incorporation of hydrophilic acrylic acid into the copolymeric macromolecular chain. Microorganisms generally degrade polymers either starting from their terminal groups or by degrading along their molecular chains at random. 22 Biodegradation of acrylate copolymers proceeds in both ways. Additionally, a small amount of side ester groups are hydrolyzed during the degradation. 21
As the oxidation site is known to be located on microorganism cells, the polymer has to contact microorganisms during biodegradation. 22 With the increase in the alkyl chain length of the esters, side chains of the copolymers extend and the hydrophobicity increases. Longer hydrophobic side chains have poorer water-solubility and exhibit a greater tendency to aggregate. As a result, the contact probability between microorganisms and copolymeric chains decreases, inhibiting degradation. Polyacrylates with longer aliphatic side chains also have poorer water solubility. In other words, they have better lipid solubility. The cell membrane of the microorganisms is mainly composed of phospholipids.23,24Transfer of polyacrylate molecules with longer aliphatic side chains through the cell membrane for intracellular oxidation should occur more smoothly. However, intracellular oxidation is based on good contact between the polyacrylate molecules and the microorganisms—less likely for polyacrylates with longer aliphatic side chains. 25 As a result, transfer of molecules with longer side chains through the cell membrane is more difficult because of the lower probability of the contact of the molecules with the microorganisms. Intracellular oxidation loses one of the most important prerequisites. Consequently polyacrylates with longer side chains are less biodegradable than the ones with shorter side chains.
Figs. 1 and 2 also reveal the marked influence of α-methyl groups in acrylates on the biodegradability of acrylate copolymers. This observation demonstrates that use of methacrylates in monomer formulation impairs the biodegradability and induces difficulties in treating desizing wastewater.
Table II also presents the influences of α-methyl groups in acrylates on the kinetics of copolymer degradation. Presence of α-methyl groups in acrylates led to an abrupt decrease in the absolute value of k and a substantial increase in t1/2 values. For instance, the t1/2 value of poly(MA-co-AA) is only about one-fifth that of poly(MMA-co-AA). The kinetic factors obtained from the equations conform to the experimental results of COD removal rates.
There are three major reasons for the decrease in COD removal rates induced by the presence of α-methyl groups in acrylates. The presence of α-methyl groups increases the hydrophobicity of acrylate copolymers. In addition, the α-methyl group occupies a larger space than the hydrogen atom, thereby blocking microorganism contact with acrylate copolymeric chains. Furthermore, α-methyl groups inhibit the degradation of acrylate copolymers from terminal groups. 26 As a result, the biodegradability of methacrylate copolymers is worse than that of acrylate copolymers. Kawai 22 and Iwahashi 26 also found that α-methyl groups in methacrylic acid units decreased the biodegradability of acrylic acid homopolymers.
Effect of Acrylate Content
The molar ratio of MA/AA in poly(MA-co-AA) was determined by 1 H-NMR spectral analysis (Fig. 3a–c). Chemical shift peaks at 3.5 ppm and 12.3 ppm corresponded with methyl protons (-COOCH3) in MA and carboxylic acid protons in AA (-COOH), respectively. The molar ratio of MA/AA in the copolymer as determined by 1H-NMR was in good agreement with the MA/AA feed ratio during copolymerization (Table III).
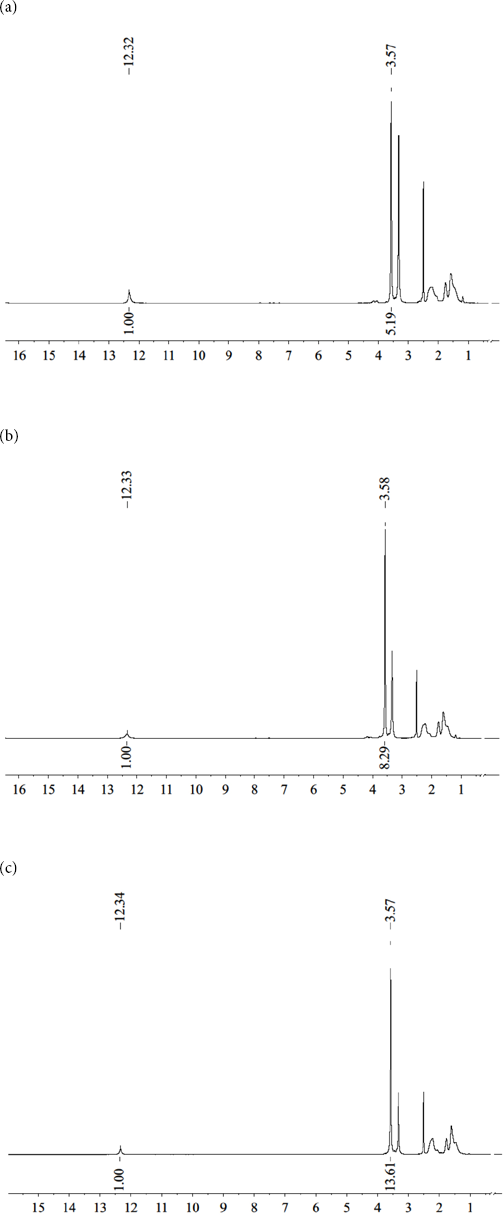
1 H-NMR of acrylate copolymers: the molar content of MA (a) 60%; (b) 70%; (c) 80%.
Molar Content of MA Units in Acrylate Copolymers
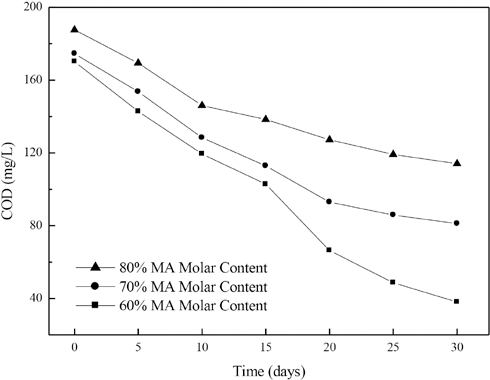
The effect of the MA unit molar content in poly(MA-co-AA) on the COD over time and COD removal rates of the copolymers are shown in Figs. 4 and 5, respectively. With an increase in content from 60% to 80% MA, the COD over time increased and the COD removal rate decreased continuously.
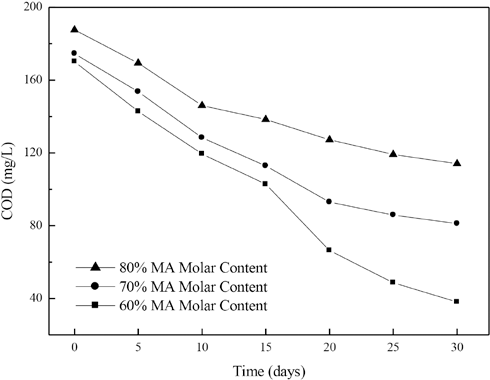
Effect of molar content of MA units in acrylate copolymers on COD over time.
Effect of molar content of MA units in acrylate copolymers on copolymer biodegradability
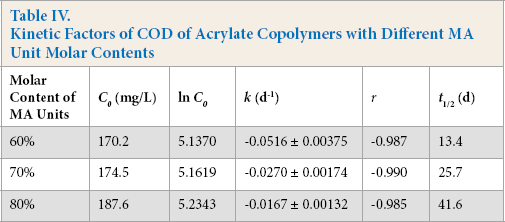
Table IV shows the influences of MA unit molar content in poly(MA-co-AA) on the kinetic factors of copolymer degradation. With the increase in the MA unit molar content, the absolute value of k decreased and the t1/2 value increased remarkably. For example, the k value of 60% MA molar copolymer was about three times greater than that of 80% MA molar content copolymer. The absolute values of r values for the first-order kinetic equations are all above 0.98. The kinetic factors were in good agreement with the experimental results of COD removal rates.
Kinetic Factors of COD of Acrylate Copolymers with Different MA Unit Molar Contents
The COD removal rate results and the factors calculated from the kinetic equation infer that the increase in MA content limited aerobic biodegradability of the copolymers. The increase in MA content decreased the hydrophilicity of acrylate copolymers due to the hydrophobicity of MA units. Therefore, it was more difficult for the polymeric chains to dissolve in a water-based paste. This resulted in poorer direct contact between polymeric chains and microorganisms. Good contact of microorganisms with polymeric chains is a prerequisite for its ability to attack the copolymeric chains. For this reason, the molar content of hydrophobic acrylate monomers should be in an appropriate range so as not to decrease copolymer dispersibility in water.
Conclusions
The biodegradability of acrylate copolymeric sizes was substantially affected by the chemical structure and molar content of acrylate units. The biodegradability decreased with the increase in the alkyl chain length of ester groups of acrylate units, regardless of whether or not the units contained an α-methyl group. The presence of an α-methyl group led to a decrease in the biodegradability of the acrylate copolymers. As the molar ratio of MA to AA increased, the biodegradability of the resultant acrylate copolymer decreased.
Poly(MA-co-AA) gave better biodegradability than any other acrylate copolymer studied in this paper. The COD removal rates of acrylate copolymeric sizes show distinct differences in the degradation period. The COD removal rate of poly(MA-co-AA) showed a substantial increase, exceeding 50% in 30 days, while the rate of poly(BMA-co-AA) showed a relatively slight increase, reaching only 10% in 30 days. Methyl acrylate and acrylic acid in an appropriate molar ratio should be used as monomers for preparing acrylate copolymeric sizes based on the biodegradability of the resulting products.
Future work will include testing acrylate copolymeric size biodegradation using inocula from other water sources.
Footnotes
Acknowledgment
This work was financed by Scientific Research Project of Shaoxing University (No. 2012LG 1014) and Scientific Research Start-up Fund of Shaoxing University (No. 20125028). The work was supported by the Changzhou Key Laboratory of New Textile Material (CM2008304). The authors would like to thank He Yangdong, director of the laboratory for the assistance he supplied.
