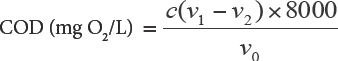Abstract
The effects of chain extender used in the extension of isocyanate-terminated pre-polymer during the preparation of waterborne-polyurethane (WPU) sizes were determined based on properties such as adhesion to fibers, film properties, desizability, and aerobic biodegradability. The extenders used included ethylene glycol (EG), butanediol (BD), hexanediol (HD), and ethylenediamine (EDA). The resulting effects and properties were strongly depended on the extenders. Diols were superior to EDA in increasing the molecular weights of extended WPUs, enhancing the adhesion to polyester fibers, and improving the film properties and aerobic biodegradability. EDA was unsuitable for the extension. For the diols, BD was the most preferred. Comparisons on the properties of the resulting WPUs versus PVA and starch were made.
In past decades, a wide variety of waterborne polyurethanes (WPU) have attracted interest due to their non-polluting properties, chemical resistance, excellent abrasion resistance, and relatively good stability in biological environments.1–2 WPU mechanical properties vary from tough, abrasion-resistant coatings for floors to highly elastic films for plastics and textiles, 3 since polyurethanes can be tailor-made. 4 Because of these versatile and unique properties, WPUs have an almost unlimited spectrum of applications. 5 Polyurethanes find applications in textiles, including breathable waterproof textiles, synthetic leathers, shrink-proof wools, and adhesives. 6 Also, they have been used to modify cotton textiles to improve resistance to creasing and shrinking. 7 A recent paper 8 found potential use for WPU in warp sizing. An industrial trial 9 confirmed that the incorporation of WPU into a sizing formulation led to evident improvement of sized polyester/cotton yarn quality.
Currently, aqueous dispersion of WPUs can be produced by techniques such as acetone, pre-polymer mixing, ketimine– ketazine, and melt dispersion processes. 10 Undoubtedly, the pre-polymer mixing process is preferred since it is extremely simple, no solvent intermediation is needed, and the pre-polymer is directly dispersed in water.11-12 Therefore, this method is commercially predominant for preparing aqueous dispersions of WPUs.12-13
The pre-polymer mixing method consists of two main steps: 1) preparation of the isocyanate (–N=C=O group, abbreviated as NCO)-terminated pre-polymer, which has lower viscosity and does not need solvent; and 2) chain extension of the pre-polymer for enhancing WPU mechanical properties. 14 The molecular structure of a WPU depends on the extenders used because some segments of WPU molecules are formed by extending a terminal diisocyanate with a low molecular weight diol or diamine. 15 Obviously, use of different extenders induce differences in polarity, cohesive energy, and intermolecular forces in the resulting WPU. This means that the extenders influence the end-use ability of WPUs in warp sizing.
Currently, industrial development of WPU sizing agents suffers from a lack of detailed information on the extenders. Accordingly, efforts have been directed toward revealing the effect of chain extenders on the properties of WPU warp sizes. In this study, the chain extenders ethylene glycol (EG), butanediol (BD), hexanediol (HD), and ethylenediamine (EDA) were used to examine their influences upon the characteristics and properties of WPU sizes. The extenders used were determined based on molecular weights, solubility, sizing film properties, adhesion to fibers, desizability, and aerobic biodegradability of the resulting WPU sizes. Additionally, the properties of the WPUs were compared with those of traditional sizing agents, such as starch and polyvinyl alcohol (PVA).
Materials and Methods
Materials and Reagents
Polypropylene glycol (PPG, 1000 MW) was purchased from Haian Petrochemical Plant and dried in a vacuum oven at 100 °C for 2 h before use. Dimethylol propionic acid (DMPA), obtained from Zhongan Chemical Co. Ltd., was desiccated in vacuum oven at 80 °C for 1 h before use. Toluene diisocyanate (TDI) was obtained from Grand Central Plains United Co. Ltd. Ethylene glycol (EG), butanediol (BD), hexanediol (HD), ethylenediamine (EDA), triethylamine (TEA), and dibutyltin dilaurate (DBDL) were purchased from Sinopharm Chemical Reagent Co. Ltd. These chemicals were all laboratory-grade and were dried with 4A molecular sieves before use.
PVA 1799 (degree of polymerization = 1700, degree of alcoholysis > 99% mole) were obtained from Beijing Oriental Petrochemical Co. Commercial cornstarch, with an apparent viscosity of 46 mPa?s, was supplied by Fenda Starch Co. Ltd. Prior to use, cornstarch was acid-thinned with HCl to an apparent viscosity of 8 mPa?s in accordance with prior work. 16 Pure cotton roving (372 tex, 98 in twist factor) was supplied by Huamao Textile Co. Ltd. Polyester roving (361 tex, 49.8 in twist factor) was obtained from Yizheng Chemical Fiber Co. Ltd. Fiber length and fineness were 27.0 mm and 1.69 dtex for pure cotton roving, and 38.0 mm and 1.73 dtex for all-polyester roving, respectively.
Synthesis of WPU
PPG (0.06 mol, number-average MW = 1000 g/mol) and DMPA (0.06 mol) were added to a dried reaction flask equipped with a reflux condenser, mechanical stirrer, a nitrogen inlet, and a thermometer. The mixture was heated to 70 °C and stirred for 1 h. TDI (0.21 mol) was added slowly to the reaction mixture under a nitrogen atmosphere. Note: All appropriate safety precautions must be taken when working with TDI, including working in a properly ventilated fume hood with appropriate personal protective equipment. For details, consult a TDI Material Safety Data Sheet (MSDS).
After 1.5 h, DBDL (0.02% by weight of total reactants) was added to the flask, and the reaction was allowed to proceed for another 1.5 h. Then, the chain extender (0.08 mol) was added by dropping funnel and the NCO-terminated polyurethane was reacted with the extender under mechanical stirring for 2 h. The reaction mixture was then cooled to 40 °C and TEA was added to neutralize the product to pH 7. Acetone was added to dilute the system if the product was too sticky. Finally, deionized water was added under vigorous stirring and acetone (if added) was removed under vacuum at 60 °C.
Spectroscopy
Fourier transform infrared (FT-IR) spectra were taken using a Nicolet Nexus 470 Spectrometer (Thermo Electron Co.) to examine the extended polyurethanes. To avoid interference by moisture, specimens were dried at 105 °C for 12 h before analysis. Spectra were recorded using the total reflection technique over a wave number range between 400 and 4000 cm−1. A spectral resolution of 4 cm−1 was used and 32 scans were acquired for each spectrum.
Characterization
Apparent viscosity was measured using an NDJ-79 viscometer (Electrical Mechanical Plant of Tongji University) at a 4% (w/w) paste concentration with a shear rate of 2028 s−1 at 25 °C. Each sample was measured in duplicate and average values were reported.
Transmittance of aqueous polyurethane pastes was measured according to Craig's method. 17 Polyurethane was diluted with distilled water to form a 1% (w/w) paste by heating to 95 °C and stirring for 1 h. After the paste was cooled to 25 °C, transmittance of the paste was measured with a UV-2802S spectrophotometer at (Unico Instrument Co. Ltd.) 220 nm against a distilled-water blank.
Average molecular weights were determined by gel permeation chromatography (Waters). A Waters 600 High Performance Liquid Chromatography (HPLC) System was equipped with a Waters 2410 Refractive Index Detector and a Waters M32 chromatography workstation. Chromatographic separation was achieved on Ultrahydrogel Linear 300 mm × 7.8 mm ID × 2 chromatography columns (Waters). A 0.1 M sodium nitrate solution was used as a mobile phase and the flow rate was 0.9 mL/min.
Adhesion to Fibers
Adhesion of WPU to cotton and polyester fibers was measured in accordance with the method described in the prior publication. 18 In each case, thirty samples were measured and average values were reported after abnormal values had been rejected by statistical analysis.
Film Properties
WPU film was prepared by incorporating 40% (w/w) acid-thinned starch. Starch was used to make separation of the cast film from the film-forming substrate easy. Due to its strong adhesive power, pure WPU film could not be peeled of from the substrates.
The WPU and starch were dispersed with distilled water to form a 1% aqueous dispersion and the dispersion was pasted by heating it to 95 °C under mechanical stirring. After the paste was maintained at 95 °C for 1 h, it was poured completely onto 650 × 400 mm cellulose film spread on a same size glass plate and dried at 65% relative humidity (RH) and 25 °C. Ten, the dried film was peeled of, tailored into strips, and measured in accordance with our previous work. 19
Desizing
Desizing behavior of the WPUs was determined by the time required to break the film strips in excess water at 80 °C. Before being tested, the film was cut into strips (100 × 20 mm) and the strips were kept at 65% RH and 20 °C for 24 h. By gripping one end of the strip over a water bath, the other end of the strip was suspended in water at 80 °C under a tension of 490 dynes by fixing a 0.5 g metal load on the end. The period of time was recorded beginning from the time when one end of the strip was put into water and ending at the time when the strip was broken. Ten strips were determined to obtain the mean value reported.
Aerobic Biodegradability
Chemical oxygen demand (COD) and biological oxygen demand within 5 days (BOD5) were determined using the Standard Methods for the Examination of Water and Waste-water described in the prior article. 20 COD was measured as follows: 10 mL of 0.250 M potassium dichromate was added to 20 mL of sample (0.1 g/L). Sulfuric acid, silver sulfate, and ferroin were added in sequence (used as reaction medium, catalyst, and indicator, respectively). After refluxing for 2 h, non-reduced potassium dichromate was titrated with ammonium ferrous sulfate (0.10 M). The amount of ammonium ferrous sulfate reacted was converted to COD using Eq. 1.
c is the ammonium ferrous sulfate standard solution concentration (M), v and v2 are the ammonium ferrous sulfate standard solution volumes (mL) in blank and sample, respectively, v0 is the sample solution volume (mL); and 8000 was the conversion value of the O2 molar mass (mg·O2/L).
BOD5 was examined by using clarified water obtained by settling sludge from a local lake. The procedure consisted of filling with 30 mL of 1 g/L cooked starch paste and 1 mL of the water into an airtight bottle and incubating the bottle at 20 ± 1 °C for 5 days. Dissolved oxygen was measured before and after incubation, and the BOD5 (mg·O2/L) was calculated from the difference between initial and final dissolved oxygen. The data reported were the mean values of duplicate individual tests for each case.
Results and Discussion
Effect of Chain Extension
FT-IR spectra of the WPUs extended by EG and EDA are showed in Figs. 1a and 1b, respectively. The isocyanate (NCO) absorption peak at 2270 cm−1 disappeared in the spectra of both 1a and 1b, indicating that the isocyanate groups of the pre-polymers reacted completely after the extensions. The absorption peaks that appeared at 3300, 1720, 1535, and 1220 cm−1 were attributed to -NH stretching, carbonyl adsorption, N-H bending, and C-N stretching of carbamate 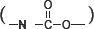 and carbamide
and carbamide 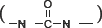 units The peak at 1070 cm−1 was assigned to C-O-C stretching. These peaks indicated that carbamate or carbamide units existed in the polyurethanes and the monomer units were linked together through these units.
units The peak at 1070 cm−1 was assigned to C-O-C stretching. These peaks indicated that carbamate or carbamide units existed in the polyurethanes and the monomer units were linked together through these units.
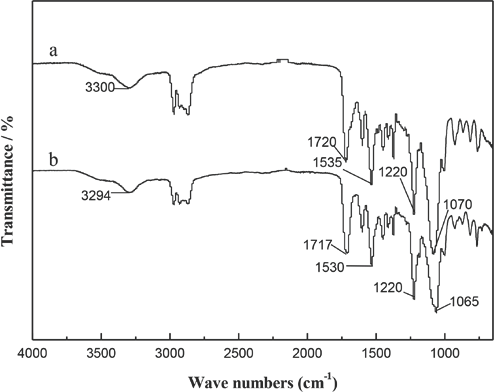
FTIR spectra of polyurethanes extended by a) ethylene glycol and b) ethylenediamine.
Despite being very hazardous itself, TDI is a reaction monomer that is consumed during the condensation of TDI's isocyanate groups with diols or diamine to form part of the WPU chain. TDI's hazardous isocyanate groups could not exist in a waterborne system because the groups react with water quickly. Furthermore, isocyanate groups are not formed during WPU degradation in nature. Therefore, the resulting polyurethanes themselves are non-toxic, as confirmed in the literature. 1
The effects of chain extenders on the characteristics of WPUs are demonstrated in Table I. Molecular weights of the WPUs prepared varied from 1.93 × 104 to 5.26 × 104, depending on the extenders. BD and EG were the preferred extenders because they greatly increased the molecular weights of terminal products. Solid contents of the copolymers were in the range of 22%–30%, similar to the commercial sizing agents of acrylate copolymers. 20 The extenders also had a significant impact on the solubility and physical form of the WPU dispersion. The extension with diamine led to a milky white and opaque liquid. The extension with diols resulted in good solubility and their dispersions showed good stability in water. This meant that the diols were superior to the diamine in the extension of the pre-polymer for preparing WPU sizing agents.
Effects of Chain Extenders on the Characteristics of WPU
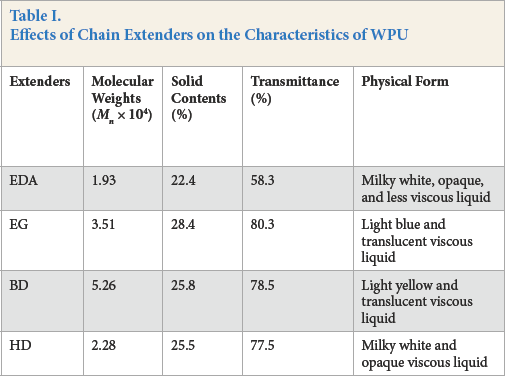
Chain extension occurred between the –NH2/–OH groups of the extenders and the isocyanate group of the pre-polymer. The –NH2 group was basic and could react with two isocyanate groups 21 to form biuret units, thereby leading to crosslinking during extension. Crosslinking reduced solubility and dispersibility of the WPU in water. Variations in solubility caused by diol extensions were mainly attributed to the number of carbon atoms or chain length of the diols between their two hydroxyl groups. The increase in the number of carbon atoms reduced the hydrophilicity of WPU and thus decreased water solubility.
Effect on Adhesion to Fibers
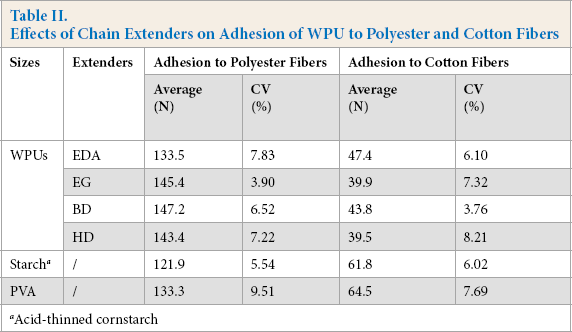
Table II shows the influence of extenders on the adhesion of WPUs to fibers. The extenders exhibited a marked effect on adhesion. Diol extension presented stronger adhesion to polyester fibers, whereas the diamine extension showed better adhesion to cotton fibers. To achieve strong adhesion, the polarities of sizing agents and fibers should be equal. 22 Strong bonding can be achieved only when sizes and fibers were either both polar or nonpolar, with polar/ nonpolar pairs never forming strong adhesion. The uramino units formed by the extension with diamine was polar—the same as cotton in polarity, but different than polyester. Therefore, strong adhesion would occur between cotton and the WPU extended with EDA, with weaker adhesion to polyester. WPUs extended with the diols possessed weaker polarity than WPUs extended with diamines, thereby providing stronger adhesion to polyester fibers and weaker adhesion to cotton fibers. Therefore, the WPUs extended with diols should be applied for sizing polyester yarns.
Effects of Chain Extenders on Adhesion of WPU to Polyester and Cotton Fibers
Acid-thinned cornstarch
Table II also indicates the effect of carbon chain length of diols on the adhesion to both kinds of fibers. Of the WPUs extended with diols, BD extension presented the strongest adhesion to polyester fibers. Considering the solubility, film performances, and adhesion, BD should be adopted as chain extender for extending the isocyanate-terminated pre-polymer in the preparation of WPUs.
Comparisons of WPUs’ adhesion to fibers versus traditional sizing agents are shown in Table II. For polyester fibers, adhesions of the WPUs extended with diols were stronger than those for both starch and PVA. WPUs extended with either diol or diamine could not match the ability of starch and PVA to achieve strong adhesion to cotton. Therefore, WPUs could be used for promoting size blend adhesion to polyester-containing warps, but not for sizing pure cotton warps.
Effect on Film Properties
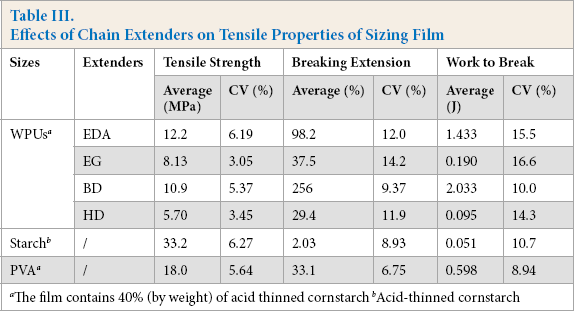
The influences of chain extenders on tensile properties, moisture regain, and wear loss of sizing film are shown in Tables III and IV. WPU extended with EDA gave the strongest film strength, but the worst abrasion resistance. WPU extended with BD showed the greatest film extension, work to break, and abrasion resistance. Tensile properties were often related to the polarity of constituent units in the backbones of macromolecular chains. The polarity of carbamide units produced by diamine extension was stronger than that of carbamate units formed by diol extension. Therefore, EDA extension enhanced the intermolecular interaction and produced the stronger film strength than diol extension did.
Effects of Chain Extenders on Tensile Properties of Sizing Film
The film contains 40% (by weight) of acid thinned cornstarch
Acid-thinned cornstarch
Effects of Chain Extenders on Moisture Regain, Time Required to Break, and Wear Loss of Sizing Film
The film contains 40% (by weight) of acid thinned cornstarch
Acid-thinned cornstarch
However, EDA could not be used as chain extender due to its poor solubility (see Table I) and abrasion resistance. These shortcomings might cause difficulty during desizing and result in reduced protection to warp yarns. Diol extension produced carbamate units with weaker polarity and lower cohesion. When BD was adopted to extend the isocyanate-terminated pre-polymer, the film exhibited the longest breaking extension, greatest work to break, and best abrasion resistance. Therefore, BD could be used as chain extender for the preparation of WPU sizing agents.
Compared with starch and PVA, the WPUs were weak in film strengths. However, all of the WPUs were greatly superior to acid-thinned starch in film extension and work to break. The WPU extended with BD was evidently better than PVA in film extension and work, and was similar to PVA in film abrasion resistance. Insufficient extension of starch film often causes the size film on sized yarn surfaces to rupture, resulting in size shedding and lint and thereby reducing warp yarn protection. Film flexibility is particularly important because sized warps must be able to withstand repeated and extensive bending in weaving operation without damage to size film. 23 Therefore, long extensions were favored to reduce the brittleness of starch film.
Effect on Desizing
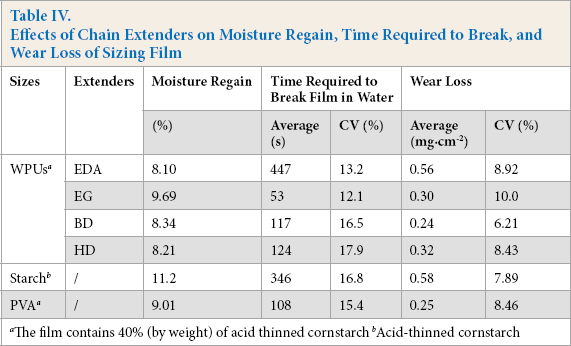
The time required to break sizing film in water was used to estimate the desizing behavior of a sizing agent. Data in Table IV indicate that the extenders showed an evident effect on the breaking time required. The film breaking time of WPU extended with EDA was much longer than that for WPU extended with diols because of the previously mentioned crosslinking. For diol extensions, the film breaking time was increasingly prolonged as the length of carbon chains in the extenders increased due to the enhanced hydrophobicity imparted to WPU by longer hydrophobic carbon chain. For this reason, reduced extension carbon chain length favors the removal of WPU from sized warp yarns during desizing.
Times required to break films of WPUs extended with diols were shorter than that of starch and were similar to that of PVA, indicating no difficulty in desizing. Comparison of moisture regain showed that the ability of WPUs to pick up water was similar to that of PVA. This indicates that application of WPUs as a binder component of the size in sizing operations would not result in tacky films at weave room humidity.
Effect on Aerobic Biodegradability
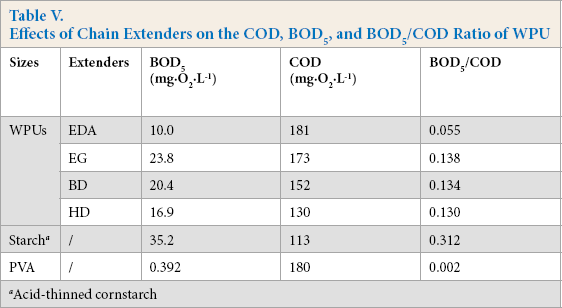
Aerobic biodegradability of organic contaminants in wastewater can be assessed by the BOD5/COD ratio. The ratio gives a gross index of the proportion of the organic materials present that are aerobically degradable within five days. 24 Therefore, the biodegradability of WPUs was assessed by measuring the BOD5/COD ratio. The influence of chain extenders on the COD, BOD5, and BOD5/COD ratios of the resulting WPUs are shown in Table V.
Effects of Chain Extenders on the COD, BOD5, and BOD5/COD Ratio of WPU
Acid-thinned cornstarch
The BOD5/COD ratio was found to be dependent on the extenders. Diols were superior to EDA in increasing the ratios for the WPUs. Therefore, WPUs extended with diols possess better aerobic biodegradability than WPUs extended with EDA. It was also observed that the ratios for WPUs were much greater than that for PVA, but lower than that for acid-thinned cornstarch. This observation suggests that the WPUs were more easily biodegradable than PVA, but less so than starch.
During biodegradation, WPU chains contact the oxidation site located on microorganism cells. 25 EDA extension led to shrinkage of WPU random coils in aqueous paste because of chemical crosslink formation. This shrinkage reduced the probability of contact between the microorganisms and WPUs, inhibiting microbial degradation, and therefore reducing the biodegradability. For the WPUs extended with diols, the chain length between two hydroxyls determined the dispersibility of WPU macromolecules in water. With a decrease in the chain length, the backbones of the resulting WPUs were less hydrophobic, thereby increasing their water dispersibility. For this reason, shorter carbon chains between the two hydroxyls favored improved WPU biodegradability.
Based on the commercial prices of raw materials used in the preparation of WPUs, the WPU was much more expensive than starch but not much more costly than PVA. Starches and PVAs have been extensively used as warp sizing agents for many years. However, starches are brittle and their use in size formulation often results in size shedding and lint. 19 PVA biodegrades slowly in nature and causes problems in wastewater management. 16
It is well known that an unblended size base material seldom meets all of the demands for a specific sizing application. It is common to blend base materials together during sizing. WPU extended with BD might be applied to blends with base materials for imparting desirable properties such as strong adhesion to polyesters and flexible size film to size blends. Of course, the blending ratio of the WPU to base materials in a size formulation would depend on fiber type, warp fineness, fabric texture, and specification.
Conclusions
Based on the water solubility, adhesion to fibers, film properties, desizability, and aerobic biodegradability of WPUs extended with diols and diamine from isocyanate-terminated pre-polymer, it was concluded that BD was an effective pre-polymer chain extender for improving the properties of WPU sizing agents. The chain extender exhibited marked effects on the physical form and solubility of the resulting WPU dispersion, and showed significant impact on the adhesion to fibers, film properties, and aerobic biodegrad-ability of the WPUs. Water solubility of the WPUs extended with EG and BD was better than that with HD and EDA. The WPUs extended with the diols showed stronger adhesion to polyester fibers but weaker adhesion to cotton fibers, whereas with the diamine extender, higher adhesion to cottons but weaker adhesion to polyesters resulted. Due to poor solubility and abrasion resistance, EDA could not be used for chain extension. Aerobic biodegradability of the WPUs extended with diols was better than that with diamine. Of the extenders, BD was preferred because it gave the longest breaking extension, the greatest work to break, and the best abrasion resistance of sizing films tested, although it was also weak in film strength compared to starch and PVA.
Comparisons of WPUs versus PVA and starch properties demonstrated that the WPU extended with BD was superior to both PVA and starch in adhesion to polyester fibers. Therefore, this WPU could be used to improve the adhesion of size blends to polyester-containing warps. It was also found that the aerobic biodegradability of this WPU was situated between PVA and starch and application of the WPU in sizing operations would not cause difficulty during desizing.
Footnotes
Acknowledgements
This work was financially supported by the Open Project Program of Key Laboratory of Eco-Textiles (Ministry of Education), China (No. KLET0617).