Abstract
A novel route of partial carboxymethylation, termed pad-dry-pad-cure, was investigated using conventional fabric processing equipment. The effect of process parameters such as concentration of sodium hydroxide, curing time, and concentration of ammonium monochloroacetate on the carboxymethylation content was probed. The treatment was characterized for uniformity by laser scanning confocal microscopy and for changes in fiber cross-sectional geometry by scanning electron microscopy (SEM). Confocal micrographs revealed the occurrence of treatment uniformly across the fiber cross sections, whereas SEM showed the resultant oval cross sections of the treated fibers.
Introduction
Carboxymethyl cellulose (CMC) is an important polymer with extensive uses in industry. Diverse applications of CMC include use as a crosslinker in bone repair, 1 a film forming material in edible coatings,2,3 a thickening agent in the food industry, 4 an emulsifier and viscosity modifier, 5 in superabsorbent polymers (SAPs),6,7 and as size in the textile industry. 8
Numerous production routes are available that involve multiple steps. Key reagents such as sodium hydroxide and monochloroacetic acid, or its salts, are used.9–13 CMC is produced industrially in two stages. In the first step, the cellulose is treated with sodium hydroxide (NaOH) to activate it. In the second step, monochloroacetic acid or its sodium salt is added. 14 The first step includes shredding of naturally-occurring cotton fibers, followed by dissolution and conversion to alkali cellulose in presence of solvents including isopropanol and water, 15 N,N-dimethyl acetamide (DMA)/LiCl, 16 N-methylmorpholine-N-oxide, 17 aqueous cellulose solvent Ni(tren)(OH)2 [tren-tris(2-aminoethyl) amine], 18 and triethylamine (TEA). 19 Recently, partial carboxymethylation of cellulosic fabric has been carried out based on methods involving no dissolution and cellulose shredding using conventional textile wet processing equipment and methods. The resulting anionic cellulose can be applied to interesting technologies such as producing wrinkle-resistant fabrics.20–22
Recently, two important routes were used to achieve anionic cellulose: 1) treatment with NaOH, followed by impregnation with sodium monochloroacetate and batching, and 2) impregnation with a mixture of NaOH and monochloro-acetate and curing. In the first route, treatment times were extremely long, ranging up to 18 h, whereas in the second method, the treatment suffered from early loss of active chloroacetate reagent due to reaction between the mixture ingredients.23,24 In a novel route described in this paper, NaOH and ammonium monochloroacetate were used as key reagents in a unique process sequence that eliminated the above problems and produced significantly improved results over the above two routes.
Experimental
Materials
A desized and scoured plain woven cotton fabric (Testfabrics Inc.) was used as cellulose substrate for the treatment after bleaching with a peroxide recipe. This fabric weighed 120 g/ m2. Sodium hydroxide (NaOH), acetic acid, hydrochloric acid, monochloroacetic acid, and ammonium carbonate were laboratory grade.
Methods
Carboxymethylation of Cotton Fabric
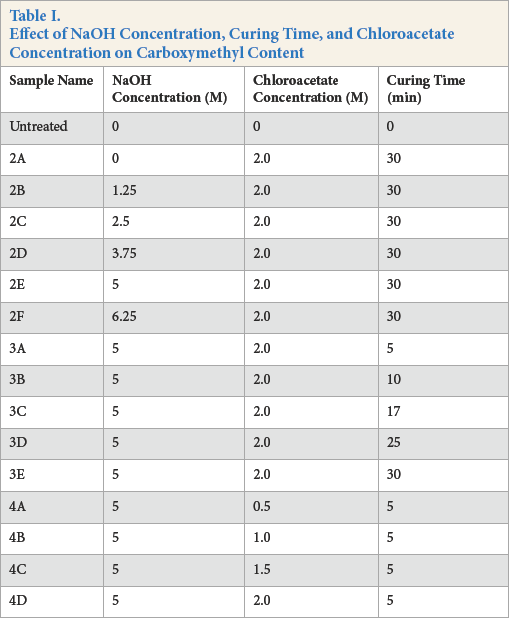
The treatment method termed as “pad-dry-pad-cure” is a route of cellulose carboxymethylation that was not explored before. 11 Treatment involved the two-step Williamson ether synthesis (Fig. 1). 5 In Step 1, samples were soaked in a NaOH solution of known concentration for 5 min, squeezed to contain the solution weight equal to the fabric weight (100% wet pickup, wpu), and dried at 45 °C for 12 min. For example, 100 g of cellulose absorbed 100 g of 1M NaOH solution, effectively providing 4 g of NaOH for reactions in the subsequent treatment step. In Step 2, varying concentrations of monochloroacetic acid were neutralized by ammonium carbonate to form chloroacetate aqueous solutions. The NaOH-treated cellulosic fabrics were impregnated with chloroacetate using a laboratory roller squeezer without soaking and squeezed to 100% wpu. Since temperatures above 70 °C were known to be optimal for carboxymethylation reactions, 25 samples were subjected to 85 °C for a given time in an air-circulated oven, followed by washing with water, acidifying with 2 g/L acetic acid, again washing with water, and then air drying. Carboxymethyl contents were analyzed using the procedure described by Hashem. 20 An experimental sequence was designed as shown in Table I to study the effect of important process parameters viz. NaOH concentration, curing time, and chloroacetate concentration on carboxymethyl contents.
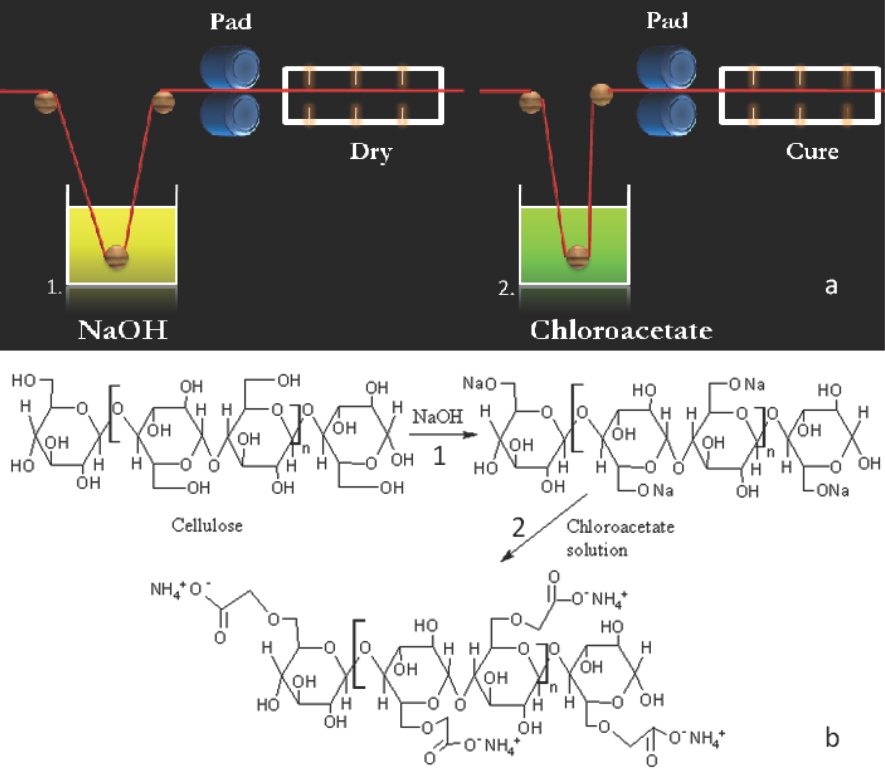
a) Experimental setup and b) schematic of carboxymethylation reaction.
Characterization by Laser Scanning Confocal Microscopy
Confocal microscopy is a non-destructive technique for examination of fiber cross sections and surfaces. It uses a fluorescent cationic dye that selectively stains anionic carboxymethyl cellulose. The dye absorbs energy at one wavelength and emits it at a longer wavelength-producing fluorescence in the visible region. 26 In this study, confocal microscopy was used to qualitatively determine whether the carboxymethylation reaction occurs all across the fiber interior or only on its surface. Since the results for treated Sample 4D (cf. Table I) showed potential for applications in technologies such as producing wrinkle-resistant cotton fabrics,23,24 it was characterized (along with an untreated sample for comparison) using a Leica TCS NT laser scanning confocal electron microscope system. At least three replicates per sample were observed under the microscope after staining with the dye.
The dye staining procedure for the samples was as follows. A buffer was prepared by adjusting 0.2 M NH4Cl to pH 9.3 using ammonia. A solution (0.1%) of a fluorescent basic dye, Sevron Brill Red 4G 200%, was made using this solution. Samples in beakers were stained with this dye using a 40:1 liquor-to-goods ratio at 50 °C for 16 h with continuous agitation in a mechanical shaker at 150 strokes/min. The samples were then given an initial rinse in the buffer solution to remove the excess surface dye layer, given two 20 min rinses in buffer solution with a 150:1 liquor-to-goods ratio at room temperature with vigorous shaking (150 strokes/min), and then dried in air. 27 The dried samples were then scanned by confocal microscopy for radial cross-sections in FITC mode using a 40× objective, and a zoom factor of 4.0.
Characterization by SEM
The untreated sample and sample 4D were used for micrograph studies in vacuum mode (charge reduction mode) using a Hitachi S-3200 electron microscope operated at 20 kV. Micrographs discerned the effect of treatment on fiber cross sections confirming the results seen by confocal microscopy. At least three replicates per sample were observed under the microscope.
Results and Discussion
NaOH Concentration
Samples 2A-F (Table I) were prepared to discern the effect of NaOH concentration used in Step 1 on the carboxymethyl content. This step is similar to mercerization wherein cellulose is converted to its caustic form using concentrated NaOH solution. Samples that were dried after treatment with NaOH were padded with ammonium chloroacetate without soaking to avoid reaction of ammonium chloroacetate with the excess NaOH in the fabric required for the subsequent reaction. An ammonia odor and reduced cellulose carboxymethylation occurred if samples were soaked in chloroacetate, whereas the ammonia odor was not observed when the fabric was padded without soaking. 23
Cellulose carboxymethylation is a bimolecular nucleophilic substitution reaction, 25 the occurrence of which necessitates the presence of cellulosate ions. Fig. 2 shows that, at lower concentrations of NaOH, the number of cellulosate ions is inadequate, leading to lower carboxymethylation. As NaOH concentration increased from 3.75 M to 5 M, anionic cellulose content increased. This was attributed to the sufficient number of reactive cellulosate anions available to complete the carboxymethylation reaction. At NaOH concentrations higher than 5 M, the decrease in carboxymethyl content could be ascribed to the side reaction between NaOH and monochloroacetate both present in the fabric, thus depleting the reactive chloroacetate concentration (Fig. 2). Monochloroacetate hence acts as a limiting reactant being consumed in both reactions viz. 1) reaction between cellulosate ions and monochloroacetate and 2) reaction between NaOH and monochloroacetate. Optimal carboxymethyl content was obtained using 5 M NaOH.
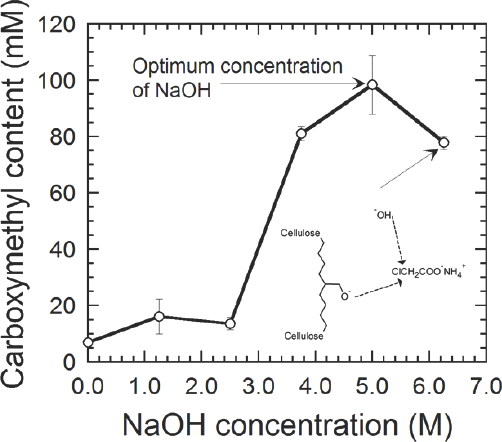
Effect of NaOH concentrations on carboxymethylation reaction (values based per 100 g of sample). Solid line connecting the points serves as a visual guide. Result corresponding to optimal NaOH concentration is marked. Error bars denote the standard error. The schematic represents the reaction of NaOH with ammonium monochloroacetate during Step 2.
Curing Time
Carboxymethylation is a temperature dependent endothermic reaction governed by the Arrhenius equation. The reaction was described to have an activation energy in the range of 85 to 93 kJ/mol underlining the curing step at 85 °C in the treatment. 5 The inset of Fig. 3 (Table I, Samples 3A–E) depicts the effect of curing time on carboxymethyl contents. After a 5 min duration time, values of carboxymethyl content were ∼90 mM/100 g cellulose. These values are desired for commercial application 23 and indicated that the reaction kinetics are fast at these conditions when compared to previous studies.20,21
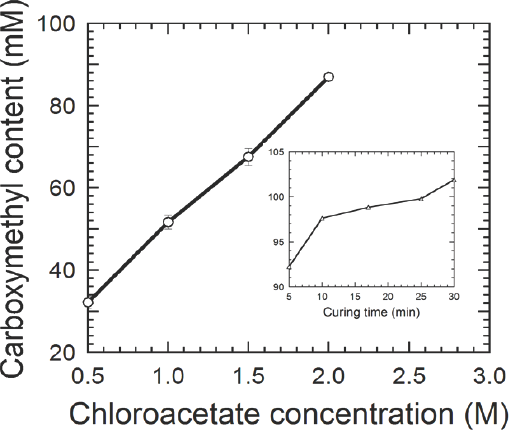
Effect of ammonium monochloroacetate concentration on carboxymethylation reaction. Inset shows how the curing time affects the reaction. Solid lines serve as a visual guide. (values based per 100 g of sample)
Effect of NaOH Concentration, Curing Time, and Chloroacetate Concentration on Carboxymethyl Content
Detailed reaction kinetics studies have not yet been performed. However, it is hypothesized that the temperature profile reaches equilibrium within the fibers in a short time span before significant transport of reaction solution occurs from the fiber interior to the fiber surface. Subsequent fast kinetics produced uniform reaction across fiber cross sections. This was confirmed by confocal microscopy as described later in this paper.
Ammonium Monochloroacetate Concentration
The effect of ammonium monochloroacetate concentration on the carboxymethyl content of the treated cotton fabric is shown in Fig. 3 (Table I, Samples 4A–D). In the concentration range studied, increased ammonium monochloroacetate concentration was accompanied by an increase in the fabric carboxymethyl contents without leveling of. This indicated that the hydroxyl sites on cellulose chains were not saturated by reaction with chloroacetate.
Confocal Microscopy and SEM Analysis
Since the curing step was performed at 85 °C, reactant migration to the fiber surface was suspected. This would limit carboxymethylation to the fiber surface, leaving the interior untreated.
Confocal microscopy analysis is a powerful tool for analyzing reaction homogeneity across the entire fiber matrix. Confocal micrographs would show ring dyeing if the modification occurred only at the fiber surface. Fig. 4a shows the fiber cross section of the untreated sample, depicting a typical kidney bean-shaped geometry of a cotton fiber. The fluorescence across the cross section in the cotton fiber was non-uniform as there were not enough anionic sites available for staining with the dye. Fig. 4b shows the fiber cross section of Sample 4D having a carboxymethyl content equal to 90 mM/100 g. This cross section was more evenly stained than the untreated sample. No ring dyeing was seen, implying the reaction occurred across the entire cross section of the fibers, a critical requirement in applications such as wrinkle-resistant cellulosic fabrics. The fiber cross-sectional geometries as seen by confocal microscopy were further confirmed by SEM to ascertain that the staining process did not introduce any changes or artifacts in the geometry. SEM proved to be an effective tool in observing the changes in the geometry of fiber cross sections. The SEMs in Figs. 5a and 5b showed cross sectional images of untreated and carboxymethylated (Sample 4D) fibers, respectively. Untreated sample showed the kidney bean shape characteristic of cotton cellulose fibers. Sample 4D showed an oval cross-section, which was ascribed largely to the high concentrations of NaOH used in Step 1 of the treatment, similar to mercerization. The similarity seen in the cross section of the treated fiber, both by confocal microscopy and SEM, indicated that the dye staining process did not affect the fiber cross section morphology. The long-time electron beam exposure of fibers in SEM at high voltages developed holes in the fibers from beam damage.
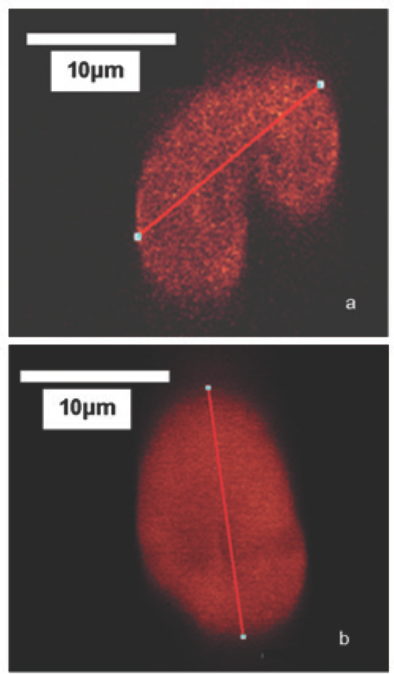
Radial cross-sectional images of a) untreated sample and b) Sample 4D. Micrographs reveal the presence of carboxymethyl groups in the bulk of the treated sample as much as on the surface.

Cross-sectional images of a) untreated sample and b) Sample 4D. The figures confirm the oval cross section of treated fibers as seen by confocal microscopy.
Conclusions
A novel treatment route for carboxymethylation of cotton cellulose suitable for textile finishing applications, such as those creating wrinkle-resistant fabrics, by means of ionic crosslinking has been described. The method involved treatment with NaOH, drying, treatment with ammonium chloroacetate, and curing. The effect of process parameters on carboxymethyl content was considered and optimized based on the chemistry of the reaction, treatment conditions (e.g., NaOH concentration), curing time, and chloroacetate concentration. Confocal microscopy proved conclusive in confirming the occurrence of carboxymethylation treatment uniformly across the fiber cross section and the images showed changes in fiber morphology from kidney shaped in the untreated sample to oval in the treated one. The SEM images confirmed the fiber morphologies seen by confocal microscopy.
Footnotes
Acknowledgment
The authors would like to acknowledge the National Textile Center for funding this research.
