Abstract
In early postnatal mouse skin, the NG2 proteoglycan is expressed in the subcutis, the dermis, the outer root sheath of hair follicles, and the basal keratinocyte layer of the epidermis. With further development, NG2 is most prominently expressed by stem cells in the hair follicle bulge region, as also observed in adult human skin. During telogen and anagen phases of the adult hair cycle, NG2 is also found in stem cell populations that reside in dermal papillae and the outer root sheaths of hair follicles. Ablation of NG2 produces alterations in both the epidermis and subcutis layers of neonatal skin. Compared with wild type, the NG2 null epidermis does not achieve its full thickness due to reduced proliferation of basal keratinocytes that serve as the stem cell population in this layer. Thickening of the subcutis is also delayed in NG2 null skin due to deficiencies in the adipocyte population.
N
The developmental expression pattern of the NG2 chondroitin sulfate proteoglycan, also known as the melanoma chondroitin sulfate proteoglycan, makes it useful as a marker of progenitor cells in a variety of tissues. NG2 is expressed by immature progenitors in several types of normal tissues and in most cases is downregulated with progenitor differentiation. Immature cells that express NG2 include oligodendrocyte progenitors in the central nervous system (Nishiyama et al. 1996; Stallcup 2002; Lin et al. 2006), chondroblasts and osteoblasts in skeletal tissue (Nishiyama et al. 1991; Fukushi et al. 2003), myofibroblasts and smooth muscle cells in the intestine and macrovasculature (Grako and Stallcup 1995; Ozerdem et al 2001; Terada et al. 2006), and nascent pericytes in microvasculature (Ozerdem et al. 2001, 2002).
Adding further support to the NG2/progenitor cell connection, two recent reports document the expression of the proteoglycan by stem cells associated with human interfollicular epidermis and hair follicles (Legg et al. 2003; Ghali et al. 2004). As with other types of progenitor cells, a possible role for NG2 in epidermal stem cell migration is noted in these studies. On the basis of these reports, we were spurred to investigate the pattern of NG2 expression in wild-type (WT) mouse skin and to use the NG2 null mouse to determine the consequences of NG2 ablation during skin development. Our results show that NG2 expression in mouse skin mimics in many respects the pattern seen in human skin. Moreover, NG2 ablation has effects on specific layers of the developing skin, namely, epidermis and subcutis.
Materials and Methods
Mice
C57Bl/6 WT and NG2 null mice (Grako et al. 1999) were maintained as separate colonies in the Association for Assessment and Accreditation of Laboratory Animal Care-accredited Burnham vivarium. All experimental work was carried out according to Office of Laboratory Animal Welfare guidelines, subsequent to approval by the Burnham Institutional Animal Care and Use Committee.
Antibodies and Reagents
Rabbit and guinea pig antibodies against NG2 have been described previously (Ozerdem et al. 2001, 2002). Antibodies against CK-5, CK-10, CK-15, and Ki67 were obtained from Abcam, Inc. (Cambridge, MA). Anti-FABP-4 was from R&D Systems (Minneapolis, MN), and anti-BrdU was from Serotec (Oxford, UK). Secondary antibodies were obtained from Molecular Probes (Eugene, OR). BrdU was purchased from Sigma-Aldrich (St Louis, MO).
Histology and Immunocytochemistry
Dorsal skin samples were dissected from the caudal area of mice ranging in age from late embryogenesis to adulthood. These specimens were fixed overnight at 4C in 4% paraformaldehyde. Tissue was embedded in paraffin and cut into 5-μm sections for immunohistochemistry or embedded in optimal cutting temperature compound, frozen, and cut into 10-μm sections for immunofluorescence. Immunofluorescence labeling was performed as previously described (Fukushi et al. 2003). Immunoperoxidase labeling was performed using a Dako Envision kit (Dako; Carpinteria, CA) according to the manufacturer's instructions.
For BrdU labeling, pregnant females at day 17 of gestation were injected IP with 80 μg BrdU per gram of body weight. Pups from these females were taken at various ages postnatally for determination of BrdU incorporation via immunolabeling with BrdU antibody (Ozerdem and Stallcup 2004).
For Oil Red O staining, cryosections were washed with PBS, incubated for 30 min at room temperature in a 0.5% solution of Oil Red O in 60% isopropanol, and counterstained with hematoxylin.
In Situ Hybridization
A 1-kb fragment corresponding to mouse NG2 base pairs 5458–6358 (AN2) (Schneider et al. 2001) was amplified from mouse RNA by RT-PCR and inserted into the pBluescript II KS(+) vector (Stratagene; La Jolla, CA). Digoxigenin-labeled sense and anti-sense cRNA probes were generated by T7- or T3-primed RNA polymerase reactions, respectively (Roche Molecular Biochemicals; Mannhein, Germany). RNA in situ hybridization was performed on 4% paraformaldehydefixed paraffin sections using a biotinylated tyramine protocol (Kerstens et al. 1995).
Results and Discussion
NG2 Expression During Mouse Skin Development
In early postnatal mouse skin, there is NG2 expression associated with blood vessels (data not shown), consistent with our previous reports of the expression of the proteoglycan by microvascular pericytes (Ozerdem et al. 2001, 2002). In addition, the skin also exhibits extensive non-vascular expression of NG2, which we investigated in more depth as a function of developmental age. The proteoglycan was not detected in prenatal skin but was first evident at a low level on the day of birth. At postnatal day 1 (Figure 1A), NG2 is expressed throughout the dermis (arrow) and even more strongly in the outer root sheath of hair follicles (arrowhead). Significantly, superficial to the strongly labeled dermis, NG2 is also found in lesser quantities in the basal layer of keratinocytes (Figure 1B, arrow), the progenitor population of the epidermis (Jones et al. 1995; Jensen et al. 1999). At day 3 (Figure 1C), NG2 expression is still strong in the outer layer of hair follicles (arrowhead) but is becoming weaker in the dermis (arrow). At 7 days of age (Figure 1D), NG2 has almost vanished from the dermis (arrow) and is strongly expressed mainly in the bulge region of the hair follicle (arrowhead). This situation is maintained at postnatal day 10 and beyond (Figure 1E), with NG2 expression detectable primarily in the hair follicle bulge region (arrowhead).
In the absence of NG2 expression in the dermis, the low proteoglycan level present in basal keratinocytes is more easily seen at day 10 (Figure 1F, arrow) and into adulthood. Expression of NG2 in the interfollicular epidermis is a hallmark of adult human skin (Legg et al. 2003; Ghali et al. 2004). The more prominent labeling of clusters of NG2-positive basal keratinocytes seen in adult human epidermis may be due to the presence of more extensive areas of interfollicular epidermis in the human. In the mouse, hair follicles are more densely packed than in the human, leaving less space for clusters of NG2-positive basal keratinocytes. NG2 expression in the dermis was not noted in the two studies of human skin. This may be due to the fact that these studies did not examine neonatal skin (Legg et al. 2003; Ghali et al. 2004), the stage at which we have detected strong NG2 expression in the mouse dermis.
Although not readily detected in our immunofluorescence analysis, developing adipocytes in the subcutis also express NG2, as shown at postnatal days 1 and 7 by the use of in situ hybridization (Figures 1G and 1H). Identification of these cells as adipocytes was confirmed by their location in the subcutis, their morphology, and their expression of fatty acid-binding protein (FABP)-4, also known as aP2 (Hunt et al. 1986; Taylor-Jones et al. 2002; Damon et al. 2006) (data not shown). The finding of NG2 transcripts in adipocytes adds these cells to the list of cell types that express the proteoglycan. It is of interest that adipocytes exhibit a high degree of developmental plasticity and share lineage relationships with chondroblasts, osteoblasts, and pericytes (Schor and Canfield 1998; Sims 2000), all of which are cell types previously shown to express NG2.
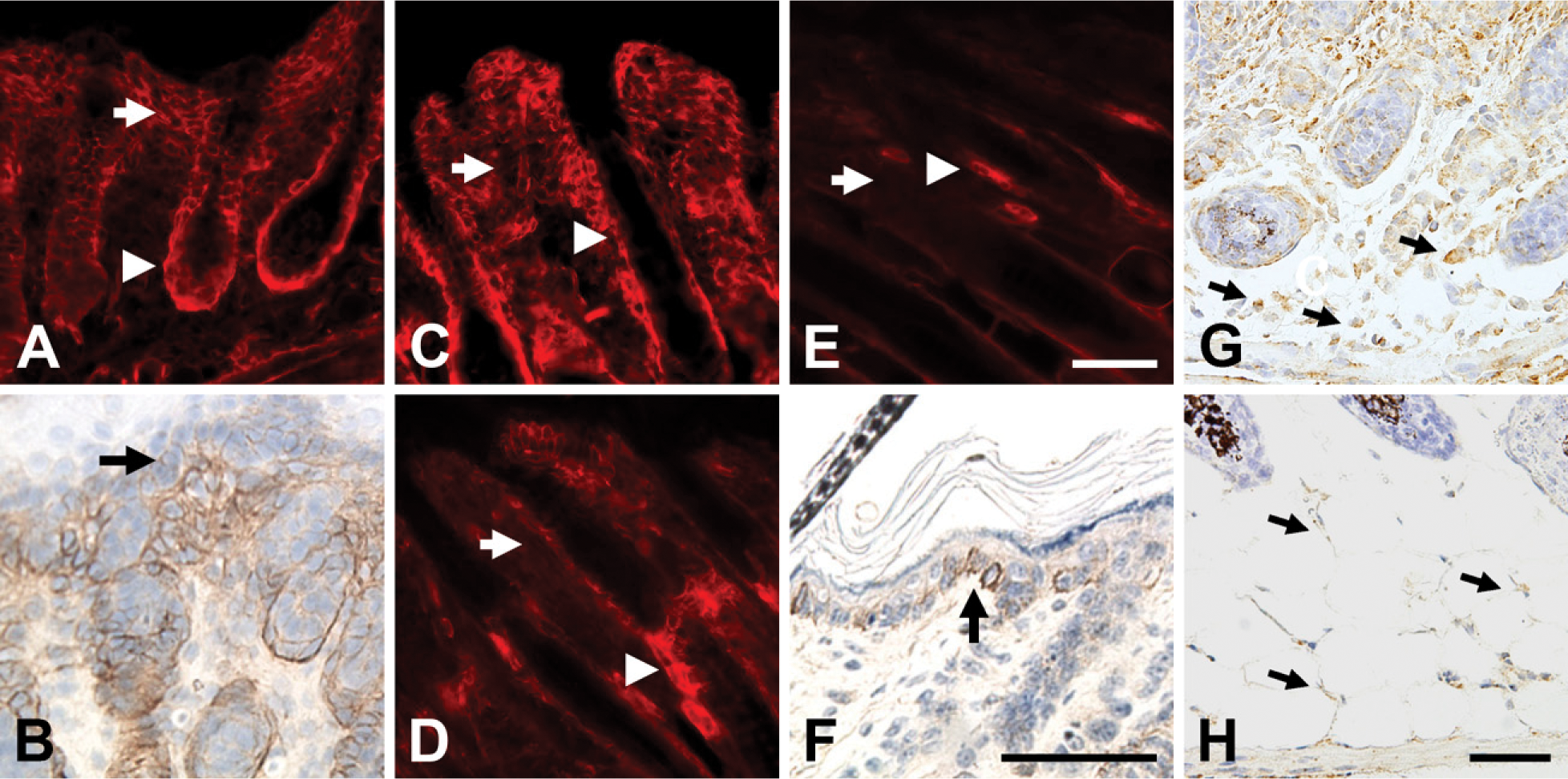
NG2 expression during early postnatal skin development. On postnatal day 1 (
Along with the basal layer of keratinocytes, the bulge region of hair follicles is of particular interest as a stem cell niche. The bulge region not only contributes to hair development but also serves as a reservoir of progenitor cells for skin development and regeneration (Cotsarelis et al. 1990; Reynolds and Jahoda 1993; Taylor et al. 2000; Oshima et al. 2001). Prominent expression of NG2 in the hair follicle bulge region has been reported in studies on human skin (Legg et al. 2003; Ghali et al. 2004). In the adult mouse, we confirmed NG2 expression in this niche by comparing NG2 labeling with that of cytokeratin (CK)-15, which is expressed by stem cells in the bulge region (Lyle et al. 1998). We also used CK-15 expression as a tool for studying changes in NG2 expression as a function of the mouse hair cycle. Phases of the adult hair cycle were determined according to Muller-Rover et al. (2001). During the telogen (quiescence) and anagen (active growth) phases (Figures 2A–2D), NG2 expression is strong not only in the CK-15-positive bulge region (asterisks in all panels) but also in dermal papillae (marked with arrow in telogen phase, Figures 2A and 2B) and in the outer root sheath of hair follicles (arrowheads). In contrast, during the regressive catagen phase of the cycle (Figures 2E and 2F), NG2 expression is restricted to CK-15-positive stem cells in the bulge region. Thus, NG2 expression by bulge region stem cells is a constant throughout the hair cycle. The NG2 expression pattern in stable (telogen) or growing (anagen) hair follicles is also suggestive. The outer root sheath of the hair follicle is contiguous with the interfollicular epidermis, sharing some of the same properties as a stem cell niche (Sonnenberg et al. 1991; Commo et al. 2000; Panteleyev et al. 2001). Dermal papillae, in addition to providing inductive signals that orchestrate follicle growth (Jahoda et al. 1984; Reynolds and Jahoda 1991), also serve as a source of mesenchymal stem cells for renewal of adipocytes, chondrocytes, osteoblasts, hematopoietic cells, and even neural cells (Fernandes et al. 2004; Shi et al. 2004; Hoogduijn et al. 2006).
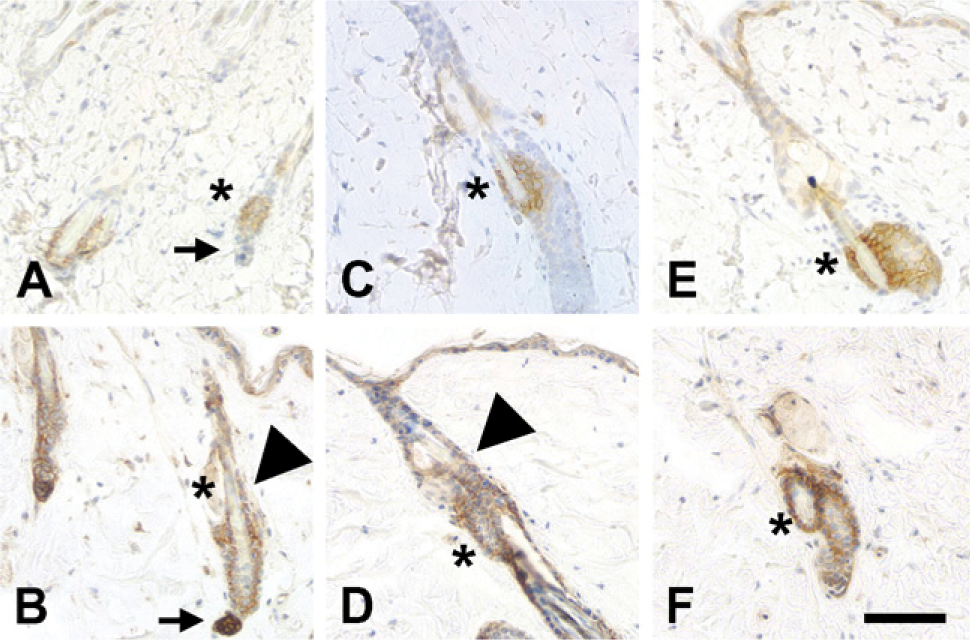
NG2 expression by stem cells as a function of the hair cycle. Cytokeratin (CK)-15 immunoreactivity (
Skin Development in the NG2 Null Mouse
As an initial means of determining a functional role for NG2 in skin development, we compared the morphological features of WT and NG2 null skin (Figure 3A) and quantified the thickness of individual skin layers. Although overall skin thickness increases during the first week postnatally in both WT and NG2 null mice (Figure 3B), thickening of NG2 null skin lags behind that of WT skin during postnatal days 1–5. This analysis was extended to specific layers of the developing skin: the epidermis (Figure 3C), the dermis (Figure 3D), and the subcutis (Figure 3E). Whereas no consistent differences were detected in the dermis or in hair follicle development, changes in both the epidermis and subcutis were evident in the NG2 null mouse. In the WT mouse, the epidermis reaches its maximum thickness at birth and then steadily decreases in width over the course of the first week postnatally. In contrast, the NG2 null epidermis does not attain the full thickness seen in the WT. Instead, the NG2 null epidermis maintains a constant, reduced thickness during postnatal week 1, undergoing a decrease only at 10 days postnatally. The converse situation is seen in the subcutis. The increasing thickness of this layer during week 1 accounts for much of the overall increase in skin thickness seen in the WT mouse. The thickness of the NG2 null subcutis lags behind that of the WT during the first 5 postnatal days.
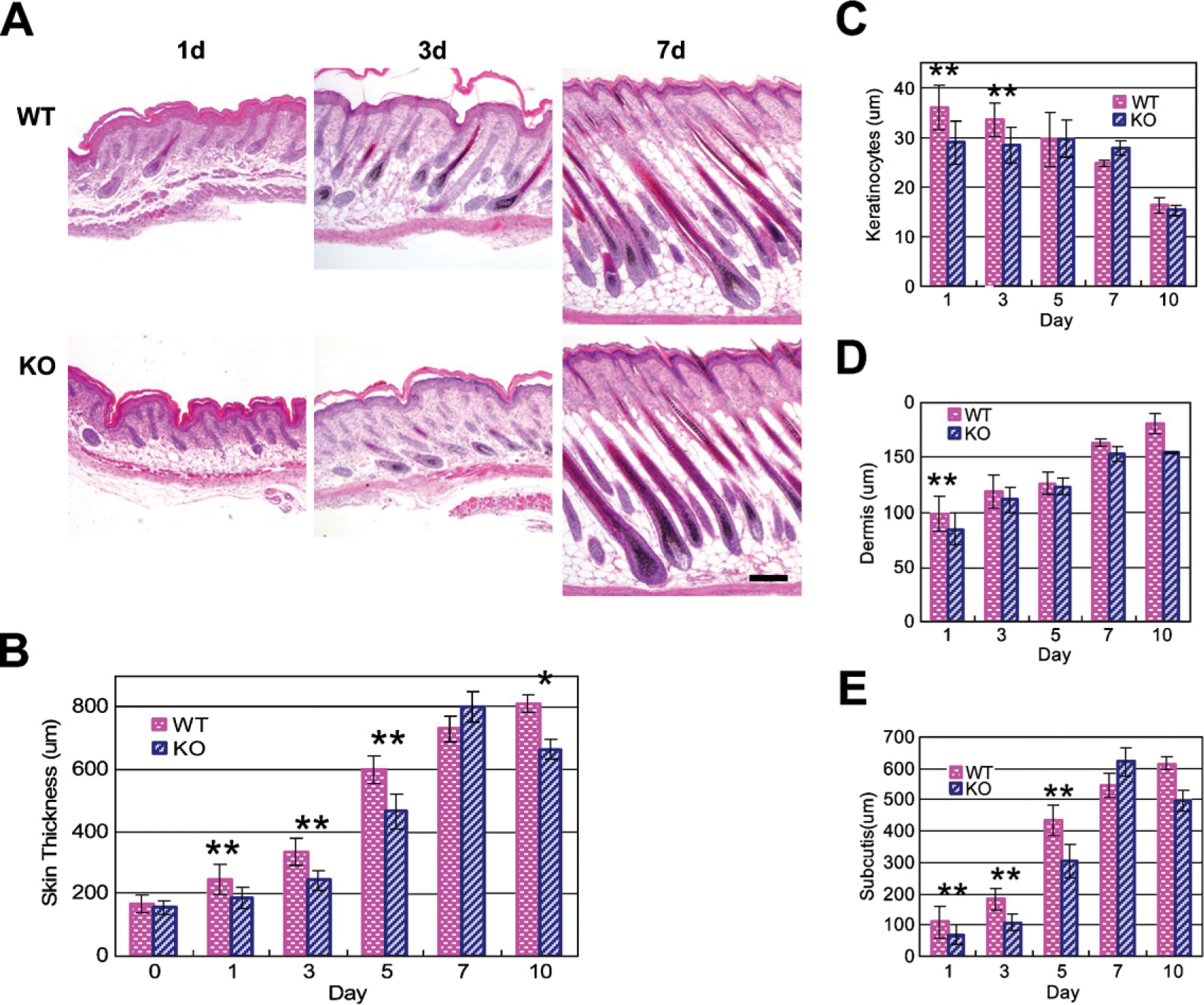
Skin development in the NG2 null mouse. Images of hematoxylin- and eosin-stained skin sections (transverse) were prepared from wild-type (WT) and NG2 null mice during the first 10 days postnatally (
Decreased keratinocyte production in the NG2 null mouse. (
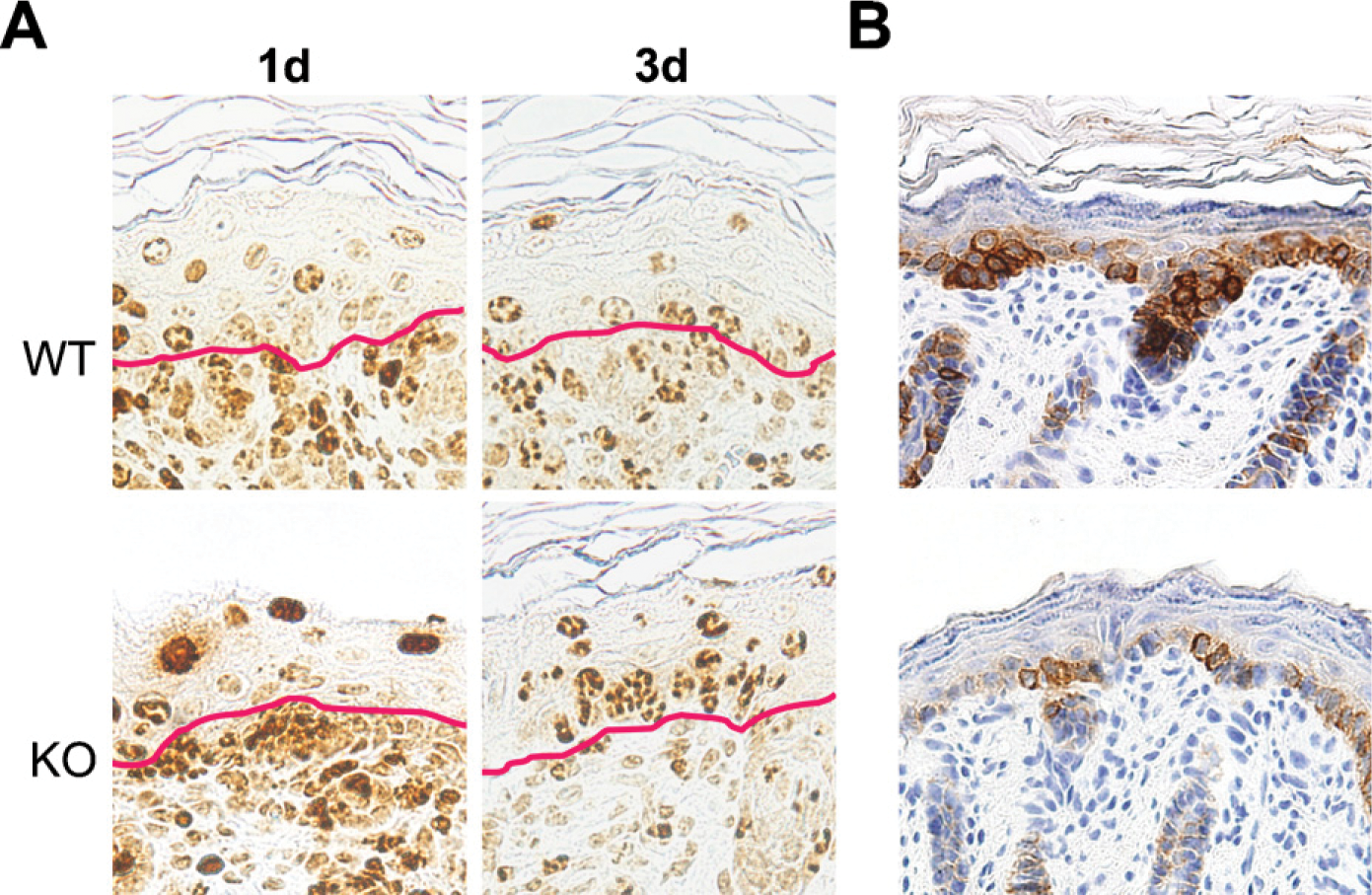
Because we have previously shown that NG2 ablation results in a significant decrease in the proliferation of microvascular pericytes (Ozerdem and Stallcup 2004), it seemed likely that decreased cell proliferation may also be a factor in altering skin development in the NG2 null mouse. Evidence for an effect of NG2 ablation on cell proliferation in the keratinocyte layer was obtained via combined use of BrdU and Ki67 labeling and immunolabeling for CK-5, which is expressed by basal layer keratinocytes (Fuchs and Green 1980; Moll et al. 1982). Dividing cells in mouse embryos were tagged by injection of BrdU into pregnant mice on the 17th day of gestation. At this stage, BrdU-labeled cells are abundant in the dermis but are seen much less frequently in the overlying epidermis. BrdU labeling in the epidermis is initially detected in the basal layer of keratinocytes, the progenitor population that expresses NG2. With increasing developmental age, these basal cells progressively differentiate, move upward, and are detected at suprabasal levels on postnatal days 1 and 3 (Watt and Green 1982; Fuchs and Byrne 1994; Lavker and Sun 2000) (Figure 4A).
Keratinocyte proliferation—dilution of BrdU label in suprabasal keratinocytes
Following BrdU injection into pregnant females at 17 days of gestation, the intensity of BrdU labeling was determined at postnatal days 1 and 3 (P1 and P3) for keratinocytes in the suprabasal layer of the wild-type (WT) and NG2 knockout (KO) epidermis. Cells with strong uniform nuclear labeling were scored as ++. Cells with diminished, patchy nuclear labeling were scored as + or +/−, depending on the label intensity (see Figure 4A for examples). Percentages of cells were determined in each of the three labeling categories. At P1, >90% of WT keratinocytes exhibit dilution of the BrdU label compared to only 40% of KO keratinocytes. By P3, these values have become more similar for WT and KO samples.
At day 1, the BrdU label has become diluted in suprabasal WT keratinocytes due to continued proliferation of these cells. In contrast, the BrdU label remains strong in many suprabasal NG2 null keratinocytes at day 1, suggesting a failure of these cells to proliferate to the same extent as WT cells. Table 1 semiquantitatively compares the dilution of the BrdU label in WT and NG2 null keratinocytes, revealing the retention of label by the NG2 null population at day 1. By day 3, dilution of the label is also apparent in NG2 null keratinocytes, showing that these cells also proliferate, albeit with a slower time course. Impaired proliferation of NG2 null keratinocytes is also reflected by the reduced number of Ki67-labeled basal keratinocytes at day 1 (Table 2). As seen in the BrdU study, proliferation of NG2 null keratinocytes recovers to the level of WT keratinocytes by day 3. These findings are consistent with the observation that the epidermis of the NG2 null mouse is reduced in thickness at postnatal days 1 and 3 (Figure 3C).
The slowly cycling, label-retaining characteristics of NG2 null keratinocytes resemble those of epidermal stem cells. An intriguing idea is that acquisition of NG2 in the WT mouse may mark a developmental transition from slowly cycling epidermal stem cells to more actively proliferating, partially committed keratinocyte progenitors. This would be consistent with our previous observations on NG2 expression in other developing tissues. For example, NG2 is not expressed by multipotent stem cells in germinal zones of the central nervous system or limb bud but is expressed by oligodendrocyte progenitors and chondroblasts, respectively, that arise from stem cells in these zones (Stallcup et al. 1983; Nishiyama et al. 1991, 1996). Our BrdU/Ki67 data for keratinocyte proliferation suggest that stem cells in the NG2 null mouse may have greater difficulty than WT cells in making the transition to more rapidly cycling progenitors.
Keratinocyte proliferation—Ki67 labeling of basal keratinocytes

Using Ki67-immunostained sections counterstained with hematoxylin, we counted the total number of basal keratinocytes and the number of Ki67-labeled basal keratinocytes. These numbers were used to calculate the percent of Ki67-positive basal keratinocytes at P1 and P3 in samples of WT and NG2 null skin. In P1 NG2 KO mice, proliferative basal keratinocytes were clearly reduced in number compared to their WT counterparts. By P3, proliferation in KO specimens recovers to nearly the WT level.
In parallel with the BrdU/Ki67 studies, we detected a large deficiency in the number of CK-5-positive basal layer keratinocytes in the postnatal day 1 NG2 null mouse (Figure 4B), consistent with a reduction in the ability of these cells to proliferate in the absence of NG2. Significantly, although there were fewer CK-5-positive keratinocytes in the NG2 null skin at postnatal day 1, at later stages of development these cells expressed the differentiation marker CK-10 (Rugg and Leigh 2004) on the same developmental schedule as in WT mice (data not shown). Together with the BrdU/ Ki67 data, these suggest that NG2 ablation affects keratinocyte proliferation more significantly than keratinocyte differentiation.
In future studies it will be of great interest to determine if NG2 ablation is accompanied by changes in the quantity or distribution of β1 integrins and/or extracellular matrix components such as types V and VI collagen, each of which has been shown to be important NG2 ligands. Skin-associated stem cells are normally characterized by high levels of β1 integrin expression (Jones and Watt 1993; Legg et al. 2003). Because NG2 binds to β1 integrins and activates their signaling to promote both cell motility and proliferation (Iida et al. 1995; Eisenmann et al. 1999; Fukushi et al. 2004; Makagiansar et al. 2004, 2007), loss of NG2/integrin interactions may have profound effects on the dynamics of the epidermal stem cell niche. Similarly, NG2 represents an important cell surface receptor for collagens V and VI (Burg et al. 1997; Tillet et al. 1997, 2002; Petrini et al. 2005). Loss of NG2 may therefore perturb cell/matrix interactions within the niche.
Expression of NG2 by adipocytes (Figure 1) provides a possible explanation for the deficiency in subcutis development observed in the NG2 null mouse. We used expression of aP2/FABP-4 as a marker for adipocyte development. At postnatal day 1, aP2/FABP-4 levels are low in the subcutis of both WT and NG2 null mice (not shown). However, adipocyte expression of aP2/FABP-4 increases dramatically in postnatal day 2 WT mice and remains high through day 5 (Figure 5A). Significant aP2/FABP-4 expression in NG2 null mice is not seen until postnatal day 5. This deficit in adipocyte function is also seen in the deposition of lipid, as detected by staining with Oil Red O (Figure 5B). Whereas significant lipid deposition can be detected at postnatal day 3 in the WT mouse, deposition in the knockout (KO) mouse lags behind until postnatal day 7. Even at this time point, lipid distribution in the NG2 null mouse appears extremely disorganized compared with that seen in the WT animal. We cannot determine from the aP2/FABP-4 and Oil Red O data whether NG2 null skin contains fewer adipocyte progenitors than WT skinor whether adipocyte maturation is retarded by NG2 ablation. This distinction will require the use of additional markers for preadipocytes and mature adipocytes.
It has been reported that the aP2/FABP-4 null mouse does not exhibit any developmental deficiencies under normal laboratory conditions, possibly due to the redundant function of FABP-5 (Hotamisligel et al. 1996). It therefore follows that the ablation of NG2 does not produce developmental deficiencies in adipocytes by affecting aP2/FABP-4 function. Instead, the delayed expression of aP2/FABP-4 in NG2 null mice must reflect the effect of NG2 ablation on a more basic aspect of the adipocyte differentiation program. Our evidence points to reduced cell proliferation as one factor that affects both adipocytes and basal keratinocytes in the NG2 null mouse. In this respect, the ability of NG2 to enhance FGF- and PDGF-mediated signaling may provide at least a partial explanation for the deficiencies observed in the NG2 null mouse. Aortic smooth muscle cells derived from the NG2 null mouse fail to proliferate normally in response to PDGF-AA and FGF2 (Goretzki et al. 1999; Grako et al. 1999; unpublished data). Experimental corneal angiogenesis in response to FGF2 is greatly reduced in the NG2 null mouse due to reduced proliferation and recruitment of pericytes to developing vasculature (Ozerdem and Stallcup 2004). Various members of the large FGF family, along with their cognate receptors, play important roles in the proliferation and differentiation of adipocytes (Yamasaki et al. 1999; Hutley et al. 2004; Newell et al. 2006; Zaragosi et al. 2006). Similarly, specific FGF isoforms are known to promote keratinocyte proliferation during both normal skin development and wound healing (Petiot et al. 2003; Beer et al. 2005; Komi-Kuramochi et al. 2005; Braun et al. 2006; Nagy et al. 2006). Along with certain FGF isoforms, PDGF family members can also be potent adipocyte mitogens (Schmidt et al. 1990; Hua et al. 2004). In addition, PDGF promotes re-epithelialization during wound healing (Cheng et al. 2007). Additional work will be required to identify a precise role for NG2 in promoting growth factor signaling in the context of the keratinocytes and adipocytes.
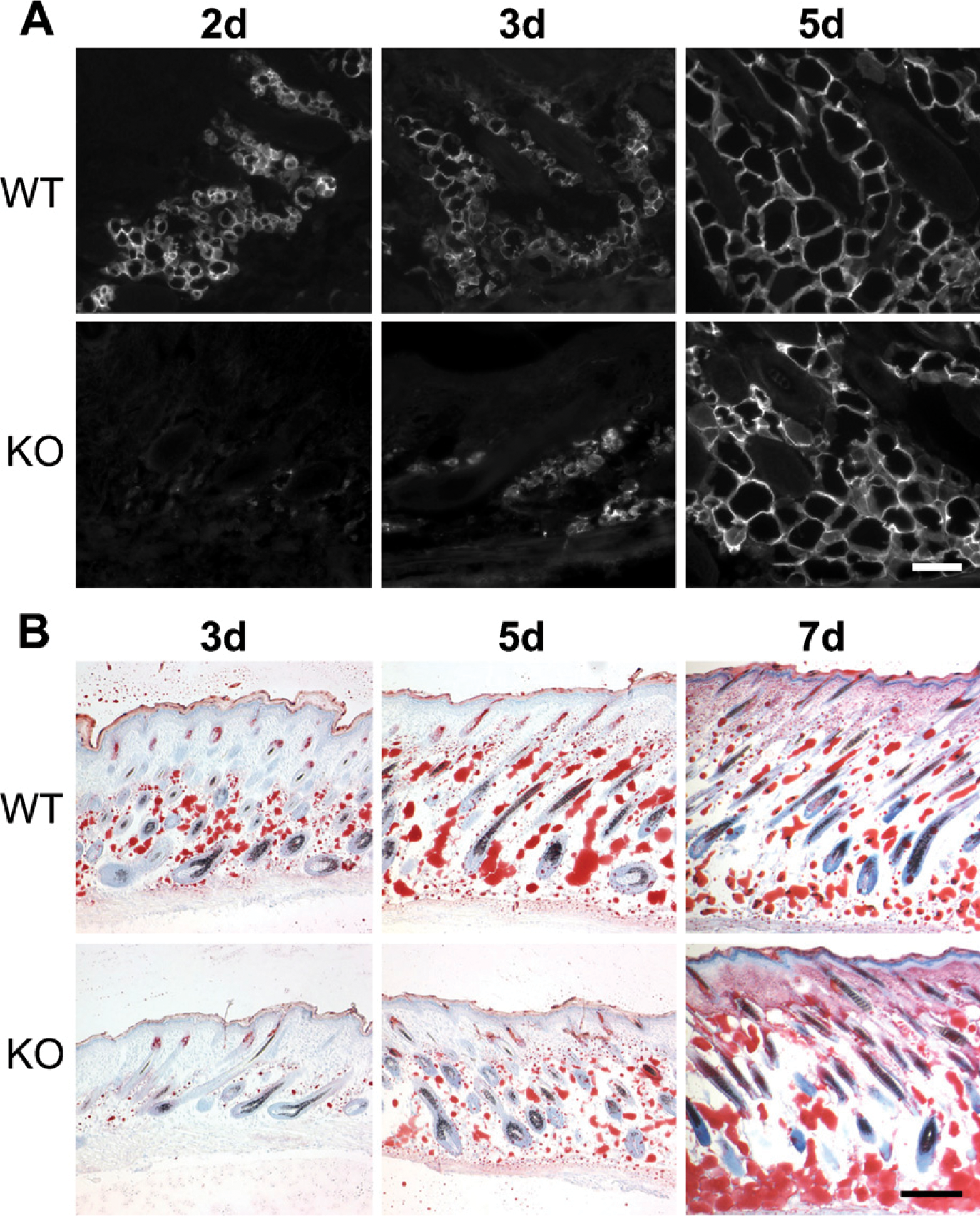
Altered adipocyte development in the NG2 null mouse. (
Surprisingly, development of the NG2 null dermis appears unaffected by NG2 ablation, even though the dermis represents a major site of NG2 expression in newborn WT skin. Similarly, in spite of strong NG2 expression in the WT hair follicle sheath and bulge region, we did not find defects in hair development in NG2 null mice. It is possible that the existence of compensatory mechanisms partially masks the loss of NG2 in early postnatal skin and hair follicles. It is noteworthy that, although we have not detected significant differences in developmental vascularization in WT and NG2 null mice, vascularization in response to a pathological challenge is significantly reduced in the adult KO animal (Ozerdem and Stallcup 2004). It is our hope that future studies on wound healing and hair regrowth will reveal deficiencies in these processes in adult NG2 null mice. Such studies will enhance our knowledge of NG2 as a functional player in the biology of progenitor cells that reside in adult tissues.
Footnotes
Acknowledgements
This work was supported by NIH Grants R01 CA-095287 and P01 HD-025938 (to WBS) and NIH Grants R01 NS-032717 and R01 NS041332 (to YY).
We are indebted to Dr. Minoru Fukuda (Burnham Institute for Medical Research; La Jolla, CA) for use of his brightfield microscope, Jennifer Meerloo (Burnham Institute for Medical Research) for assistance with image analysis, and Regina Kapono (Burnham Institute for Medical Research) for help with the manuscript.
