Abstract
Lycopene, the major carotenoid found in tomatoes, is a potent antioxidant associated with the prevention of degenerative diseases such as breast cancer. This effect could be due to the interaction between lycopene and retinoic acid receptors as well as the stimulation of gap junction communication and synthesis of connexin 43. The expression of the RARα, RARβ, and Cx43 proteins was analyzed using immunohistochemistry in two breast cancer cell lines, MCF-7 and MDA-MB-231, and in a fibrocystic dystrophy cell line, MCF-10a, after a 48-hr exposure to 10 μM lycopene. A real-time quantitative PCR analysis was then performed to measure mRNA expression. RARα and Cx43 expression were increased at both mRNA and protein levels in two breast cell lines.
T
Materials and Methods
Cell Lines
MCF-7 and MDA-MB-231 cells (American Type Culture Collection), originating from a pleural effusion of patients with invasive breast carcinoma (Soule et al. 1973; Cailleau et al. 1974), were grown, respectively, in RPMI 1640 medium supplemented with 10% heat-inactivated FBS, 2 mM L-glutamine, 20 μg/ml gentamycin, and 0.04 U/ml insulin; and L-15 Leibovitz medium (Invitrogen SARL; Cergy Pontoise, France) supplemented with 2 mM L-glutamine, 20 μg/ml gentamycin and 15% heat-inactivated FBS. For experiments, cells were grown in T-75 flasks to a density of 106 cells in 15 ml of medium at 37C in a humidified atmosphere, with 5% CO2 for MCF-7 and without CO2 for MDA-MB-231.
The MCF-10a cell line was established from the mammary tissue of a patient with fibrocystic breast disease (Soule et al. 1990). Cells were grown in DMEM/Ham's F12 (Invitrogen SARL) with 20 ng/ml epidermal growth factor, 100 ng/ml cholera toxin, 0.01 mg/ml insulin, 500 ng/ml hydrocortisone, and 10% heat-inactivated horse serum. Cells were grown in a 37C humidified atmosphere with 5% CO2.
Lycopene Treatment
Exposure to lycopene was carried out for 48 hr with 10 μM synthetic lycopene dissolved in 0.25 g/l tetra hydro furanebutylated hydroxyl toluene (Sigma Chimie; St Quentin Fallavier, France). This concentration was previously reported (Chalabi et al. 2004) to induce a G1/S-phase cell cycle arrest with an increase in late-G1 phase cell number. Control cells were plated in medium without lycopene supplementation.
Immunohistochemistry
Four-μm alcohol-formalin-acetic acid-fixed and paraffin-embedded sections were cut using a microtome, mounted on silanized glass slides (Starfrost; Duiven, The Netherlands), and dried overnight at 37C. After deparaffinization and rehydration through graded alcohols (100% and 70%) and distilled water, a heat-induced antigen retrieval method was used, which included a 3-min incubation in citrate buffer, pH 5.95, in a pressure cooker, followed by a 15-min cooldown period in a water bath. Further processing was performed with a NexES automated immunostainer using an AEC kit (Ventana Medical Systems Inc.; Tucson, AZ). Slides were then incubated at 37C for 32 min with anti-RARα (rabbit, sc-5051; Santa Cruz Biotechnology, Santa Cruz, CA), anti-RARβ (rabbit, sc-552; Santa Cruz Biotechnology), or anti-Cx43 (rabbit, sc-909, goat, sc-6560; Santa Cruz Biotechnology) primary antibodies. All antibodies were used at a 1:10 dilution except for rabbit anti-Cx43, which was used at 1:5. Subsequent incubations with a biotinylated secondary antibody, avidin-conjugated peroxidase complex, were carried out in a Ventana NexES immunostainer in accordance with the manufacturer's protocol. Slides were then counterstained with hematoxylin for 3 min, rinsed in distilled water, and coverslipped with an aqueous Faramount mounting media (DAKO; Glostrup, Denmark). The primary polyclonal antibody was omitted and replaced with PBS as a negative control.
RT-QPCR
RNA Extraction. Cells were plated at a density of 3 × 106 cells per T-75-cm2 flasks. At 80% confluence, cells were washed twice with PBS. Total RNA was isolated using 1 ml of RNA-B (Qbiogene; Illkirch, France) according to the manufacturer's protocol. Total RNA samples were dissolved in diethyl-pyrocarbonate—treated water, and A260 measurements were performed to determine their concentrations using a Hitachi spectrophotometer U-2000.
Sequences of Assay-on-Demand, primers, and Taqman probes used for the quantification of RARα, RARβ, Cx43, and 18S transcripts

Microchip Gel Electrophoresis. The Agilent 2100 Bioanalyzer and RNA 6000 Nano LabChip kit (Agilent Biotechnologies; Palo Alto, CA) were used to assess the RNA quality of tissue samples. RNA samples were diluted, and 1 μl [which corresponds to ∼1.5% (v/v) of each probe] was transferred to the Nano LabChip along with 1 μl of RNA 6000 ladder (Ambion; Courtaboeuf, France). Analyses were performed according to the manufacturer's instructions, and results were shown as electropherograms.
cDNA Synthesis. One μg of total RNA was used for the synthesis of first-strand cDNA using the First Strand cDNA Synthesis kit (Amersham Pharmacia Biotech; Uppsala, Sweden) following the manufacturer's instructions.
RT-QPCR. mRNA levels were quantified using an ABI Prism 7700 Sequence Detection System (Applied Biosystems; Courtaboeuf, France) and Assay-on-Demand (Applied Bio-systems), except for the 18S and Cx43 genes. 18S and Cx43 probes and primers were designed so that they overlapped splice junctions, thereby avoiding the potential amplification of genomic DNA. Sequences were chosen using Primer Express (Applied Biosystems) software and are presented in Table 1.
Twenty-five ng of cDNA was used in the RT-QPCR for mRNA expression in a total volume of 25 μl reaction mixture containing 12.5 μl TaqMan Universal PCR Master Mix (2×). The mix was optimized for 5′ nuclease assay using TaqMan probes and contained AmpliTaq Gold DNA Polymerase, AmpErase uracil-DNA glycosylase, and desoxyribonucleotide triphosphates with 2′-desoxyuridine 5′-triphosphate. Two-hundred nM of RARα or RARβ Assay-on-Demand, or 50 nM Cx43 and 18S primers and TaqMan probes were added. Amplification reactions were performed by a three-step thermal cycling method, which consisted of a 2-min step at 50C and 10 min at 95C followed by 40 cycles at 95C for 30 sec and 60C for 1 min. RT-QPCR assays were conducted in quadruplicate for each sample. Relative quantitation of mRNA level was done using the comparative CT method (Fink et al. 1998), which is the quantitative normalization of cDNA in each sample to an internal control (i.e., 18S rRNA) to normalize quantity and quality of cDNA samples. To ensure the reproducibility of mRNA determinations, two independent total RNA extractions were performed. Two independent reverse transcription reactions were carried out for one RNA extraction, whereas only one was performed for the second extraction. Each RT was analyzed in triplicate and expressed as a mean value ± standard deviation (Favy et al. 2000).
RARα, RARβ, and Cx43 immunostaining in MCF-7, MDA-MB-231, and MCF-10a breast cell lines upon a 48-hour exposure to 10 μM lycopene
Cyt, cytoplasm; N, nucleus; Mb, membrane. Scale of relative expression intensity: (−) no expresion, (±) low intensity, (+) moderate intensity, (++) high intensity, (+++) very high intensity.
Results
Immunohistochemistry
Expression patterns of RARα, RARβ, and Cx43 appeared heterogeneous among the breast cell lines studied. Major differences emerged in nuclear and cytoplasmic localizations (Table 2).
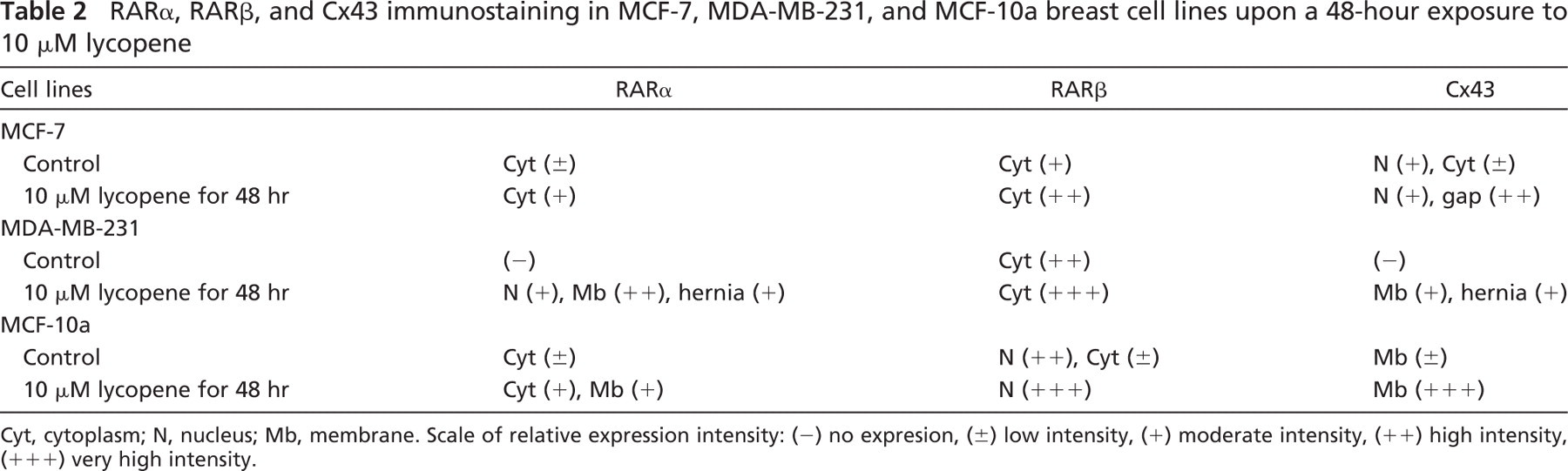
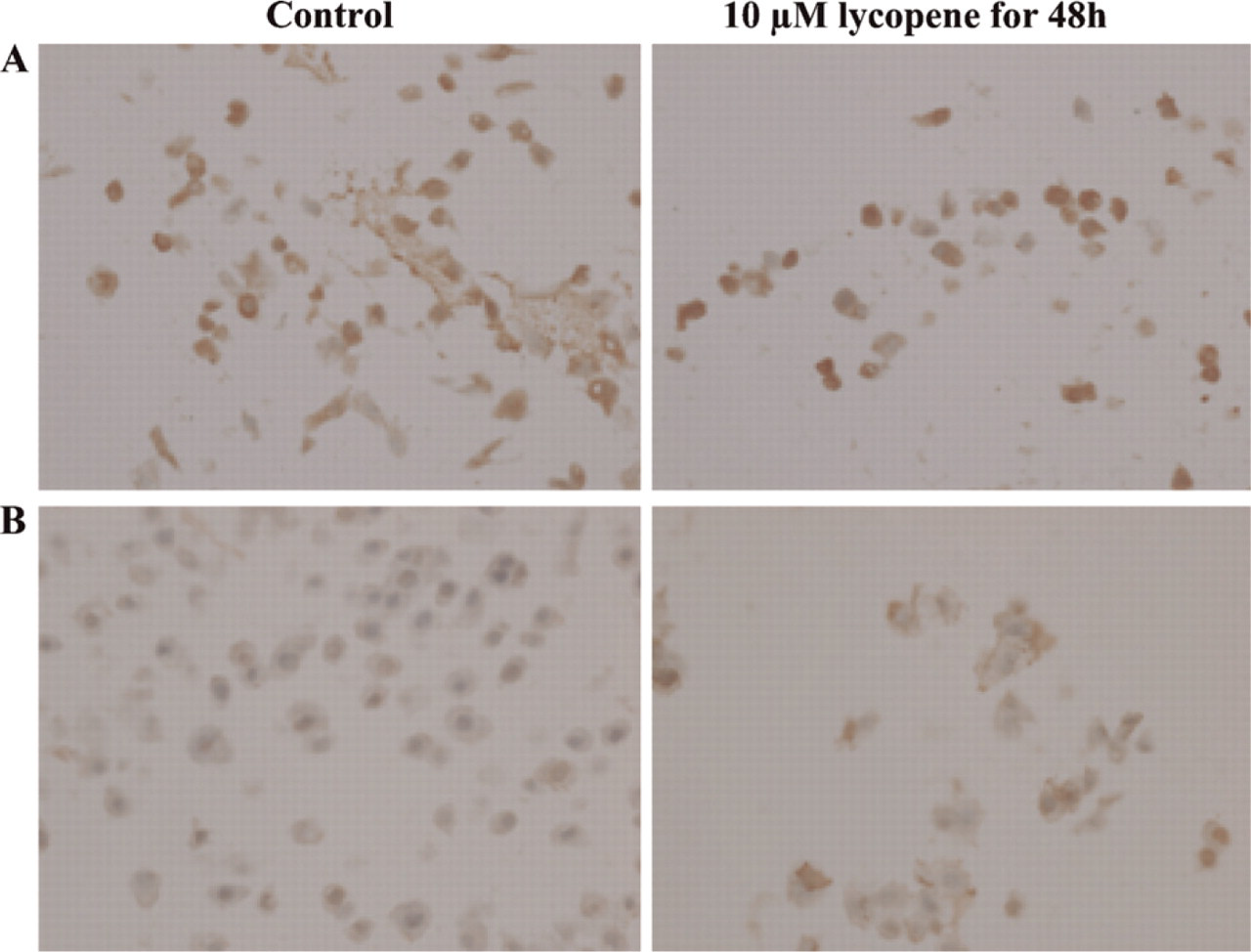
MCF-7 Cell Line. In the estrogen-positive MCF-7 breast cancer cell line, patterns for RARα and RARβ retinoic acid receptors displayed a cytoplasmic staining that was increased in lycopene-treated cells (Figures 1A and 1B). Nuclear expression and moderate Cx43 cytoplasmic expression were observed in control cells. After lycopene exposure, a nuclear expression was observed along with an increase in gap junctions (data not shown).
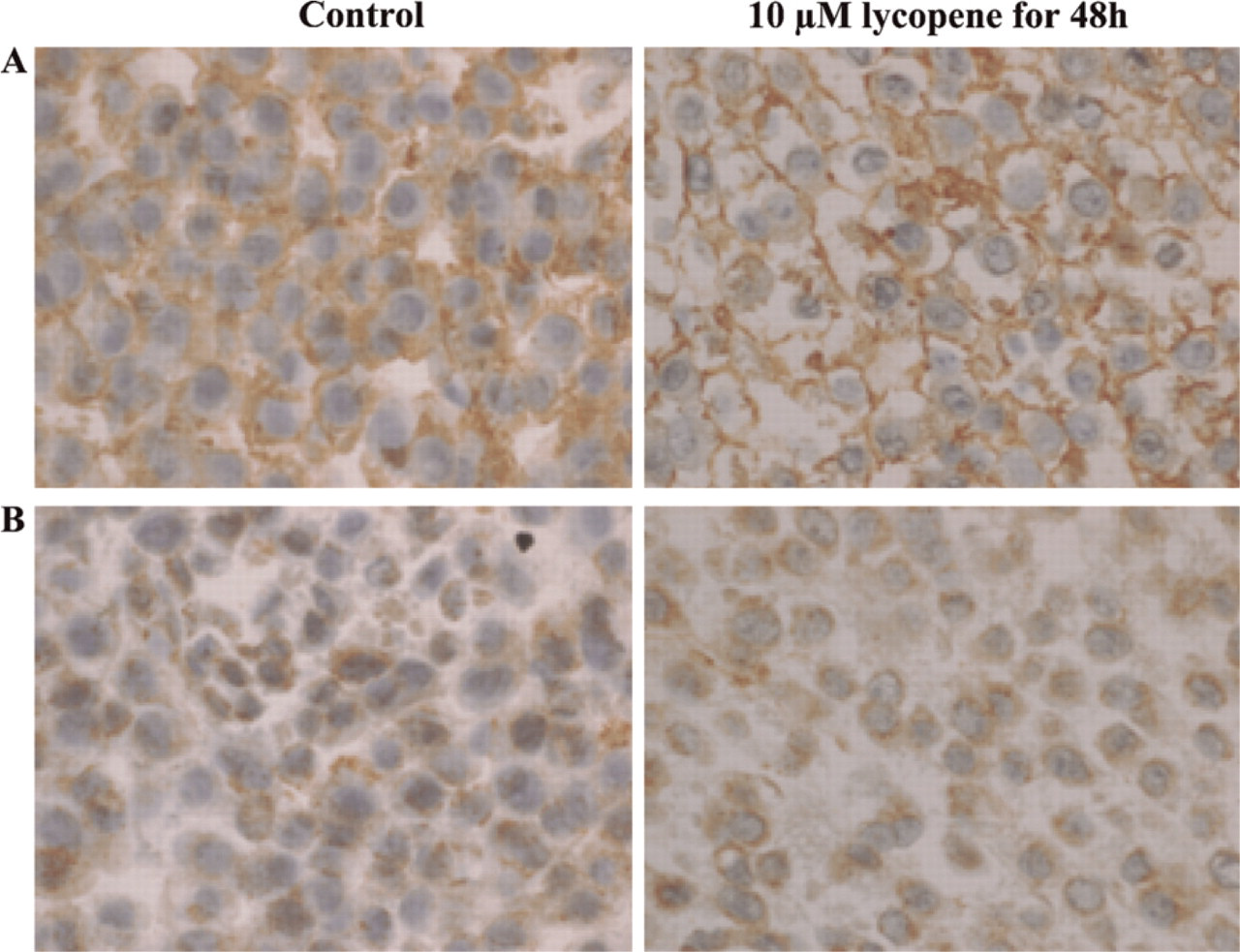
MDA-MB-231 Cell Line. A 48-hr exposure to 10 μM lycopene increased nuclear RARα expression in membranes along with the appearance of herniæ (Figure 2A). In contrast, a high RARβ staining that was stronger in the presence of 10 μM lycopene for 48 hr was found in control cytoplasms (Figure 2B). No Cx43 staining was observed in control cells, but lycopene treatment resulted in an intense Cx43 expression in hernia on membrane (Figure 2C).
In MCF-7 cells: (
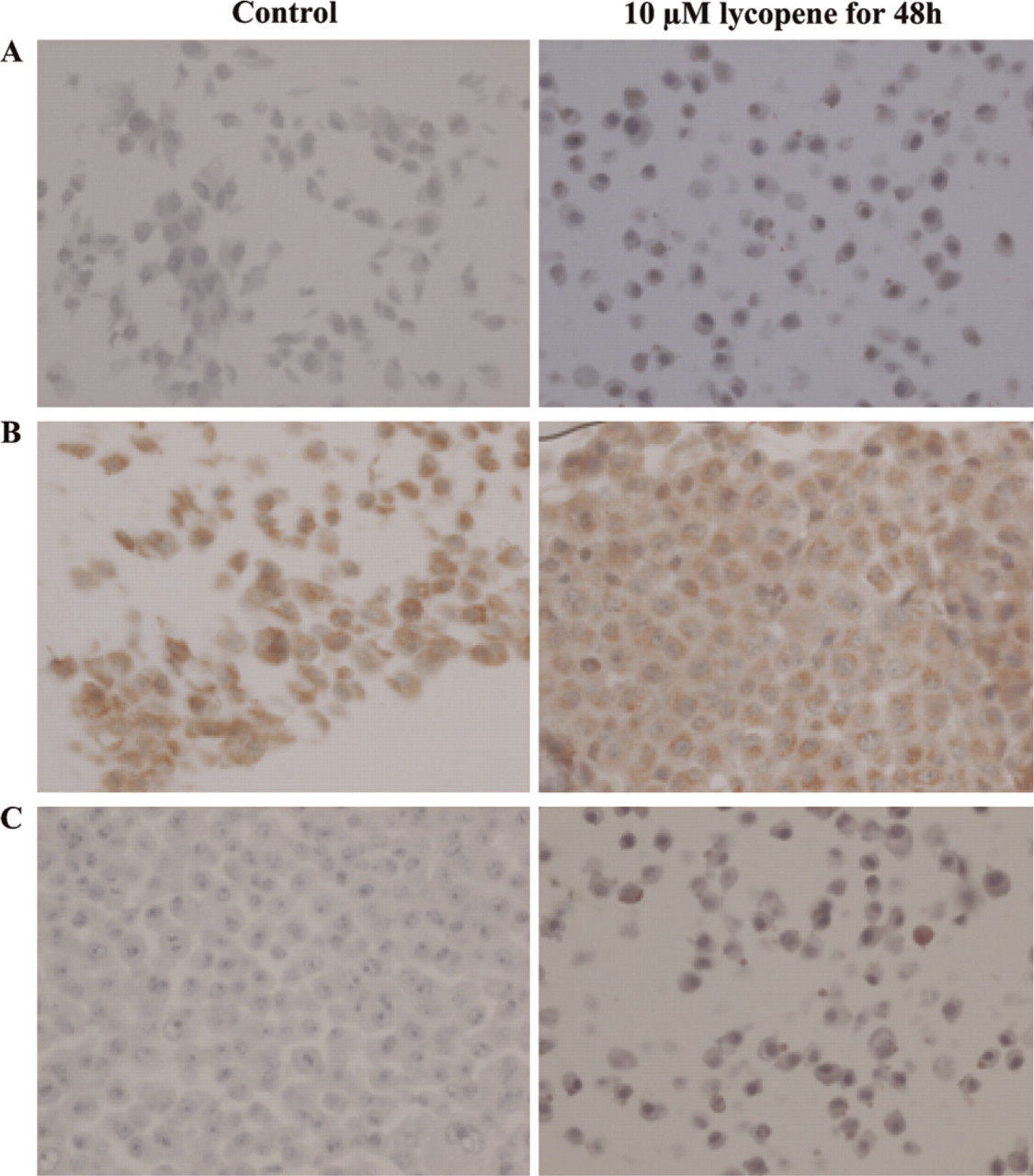
MCF-10a Cell Line. In the MCF-10a fibrocystic dystrophy cell line, a moderate RARα cytoplasmic expression was observed in control cells. Upon lycopene treatment, this intensity increased moderately within the cytoplasm and the membrane (data not shown). A strong RARβ staining that increased within the nucleus of lycopene-treated cells was revealed in the nucleus and cytoplasm of control cells (Figure 3A). A moderate Cx43 staining that increased sharply after lycopene treatment was found on the membrane of control cells (Figure 3B).
In MDA-MB-231 cells: (
RT-QPCR
To quantify RARα, RARβ, and Cx43 mRNA expression upon exposure to lycopene, an RT-QPCR study using the Taqman method was performed in MCF-7, MDA-MB-231, and MCF-10a breast cell lines (Figure 4). Expression of each mRNA species in lycopene-treated cells was normalized to their expression level in control cells, which was set to the arbitrary value of 1.
Lycopene significantly increased RARα mRNA expression in MCF-7 (p<0.05) and MCF-10a (p<0.01) cells. However, no variation in RARα mRNA expression was observed in MDA-MB-231 cells. Although an increase in RARβ mRNA levels in MDA-MB-231 cells (p<0.01) was measured, a decrease was shown in MCF-7 and MCF-10a cells. Lycopene treatment resulted in a significant increase in Cx43 mRNA levels in MDA-MB-231 and MCF-10a (p<0.01), but no variation was obtained in the MCF-7 cell line.
Discussion
In this work, we performed an immunohistochemical study of RARα, RARβ, and Cx43, which are three markers potentially involved in breast cancer in three breast cell lines (MCF-7, MDA-MB-231, and MCF-10a), upon exposure to 10 μM lycopene for 48 hr. This concentration was previously reported to induce a G1/S cell cycle arrest, during which BRCA1 and BRCA2 were highly expressed (Chalabi et al. 2004).
In MCF-10a cells: (
The present results showed heterogeneous expression patterns among these cell lines. Immunohistochemical expression of RARα indeed demonstrated a cytoplasmic staining in the MCF-7 and MCF-10a cell lines, which increased upon exposure to lycopene. In contrast, no expression was found in MDA-MB-231 control cells, whereas a nuclear staining was observed upon exposure to lycopene, with a high expression within herniæ on membranes. These results were consistent with those obtained using RT-QPCR. While the RARβ cytoplasmic staining in MCF-7 and MDA-MB-231 increased upon a 48-hr exposure to 10 μM lycopene, a nuclear RARβ staining that increased after exposure to lycopene was observed in the MCF-10a dystrophic cell line. In contrast, RT-QPCR experiments showed a decrease in RARβ mRNA expression in MCF-7 and MCF-10a cell lines. An inverse relationship was previously found between mRNA and protein expressions for BRCA1 and BRCA2 genes (Chalabi et al. 2004). These results were due to posttranscriptional and posttranslational modifications. Although no variation was observed for BRCA1 and BRCA2 proteins upon exposure to lycopene, increased levels of phosphorylated BRCA1 and BRCA2 proteins were observed (Chalabi et al. 2005). It would therefore be of interest to assess the phosphorylation status of RARβ protein, along with its acetylation or ubiquitination levels. RARα plays a major role in retinoidmediated growth inhibition of human breast cancer cells in vitro. Moreover, RARα protein was reported to be expressed at significantly higher levels in tumors with greater proliferative activity, suggesting that RARα expression may be altered with tumor progression (van der Leede et al. 1996). It has been established that RARα expression is greater in estrogen receptor (ER)-positive cell lines, whereas RARβ is more commonly expressed in ER-negative cell lines (Roman et al. 1992). Furthermore, in the ER-positive MCF-7 cell line, RARα levels were about twice as high as in ER-negative MDA-MB-231 cells (Han et al. 1997), which could account for the lack of expression of RARα observed in the untreated ERα-negative MDA-MB-231 cell line. These data were consistent with results reported by Lu et al. (2005), in which ERα and RARα protein levels were compared in five breast cancer cell lines, including MCF-7 and MDA-MB-231; ERα and RARα were expressed in MCF-7 cells but not in MDA-MB-231 cells (Lu et al. 2005). Moreover, ERα has been shown to be a direct regulator of RARα transcription, and an estrogen response element was identified in the RARα promoter (Roman et al. 1993; Rishi et al. 1995; van der Leede et al. 1995; Han et al. 1997; Lu et al. 2005).
Previous studies reported that lycopene enhances GJIC (Bertram et al. 1991; Zhang et al. 1991; Sies and Stahl 1998; Aust et al. 2003). Loss of GJIC is one of the hallmarks of carcinogenesis (Yamasaki et al. 1995, 1996) and its restoration or upregulation is associated with decreased proliferation (Heber and Lu 2002). In this work, we studied the expression of Cx43 in breast cell lines upon a 48-hr exposure to 10 μM lycopene by immunohistochemistry. Cx43 was indeed thought to be a relevant marker of early oncogenesis in breast tissue (Laird et al. 1999). Moreover it has been demonstrated that carotenoids, including lycopene, upregulate Cx43 at both protein and mRNA levels (Zhang et al. 1992). Our results showed a nuclear and cytoplasmic pattern of Cx43 expression in the MCF-7 cell line and an increase in gap junctions upon lycopene supplementation. No Cx43 expression was observed in untreated MDA-MB-231 cells, whereas lycopene-treated MDA-MB-231 cells showed a positive pattern in nuclear membranes and hernia. A slight positive pattern that was significantly increased upon exposure to lycopene was observed on the membrane of MCF-10a control cells. RT-QPCR experiments confirmed immunohistochemistry results. Several studies have shown expression of connexins in tumor cells, but these have been abnormally located and accumulated within the cytoplasm. Such observations have been made both in vitro and in vivo and did not depend on the origin of the tumor (Mesnil et al. 2005). It has been demonstrated that relocation of Cx43 to the membrane is associated with the induction of GJIC and decreased cell growth in vitro (Roger et al. 2004). The relationship between the Cx43 location on the membrane and growth regulation could be associated with the phosphorylation of Cx43, which would result in the cytoplasmic location to the endosomes through the activation of the extracelular signal-regulated kinase/mitogen-activated protein kinase pathway (Mograbi et al. 2003).
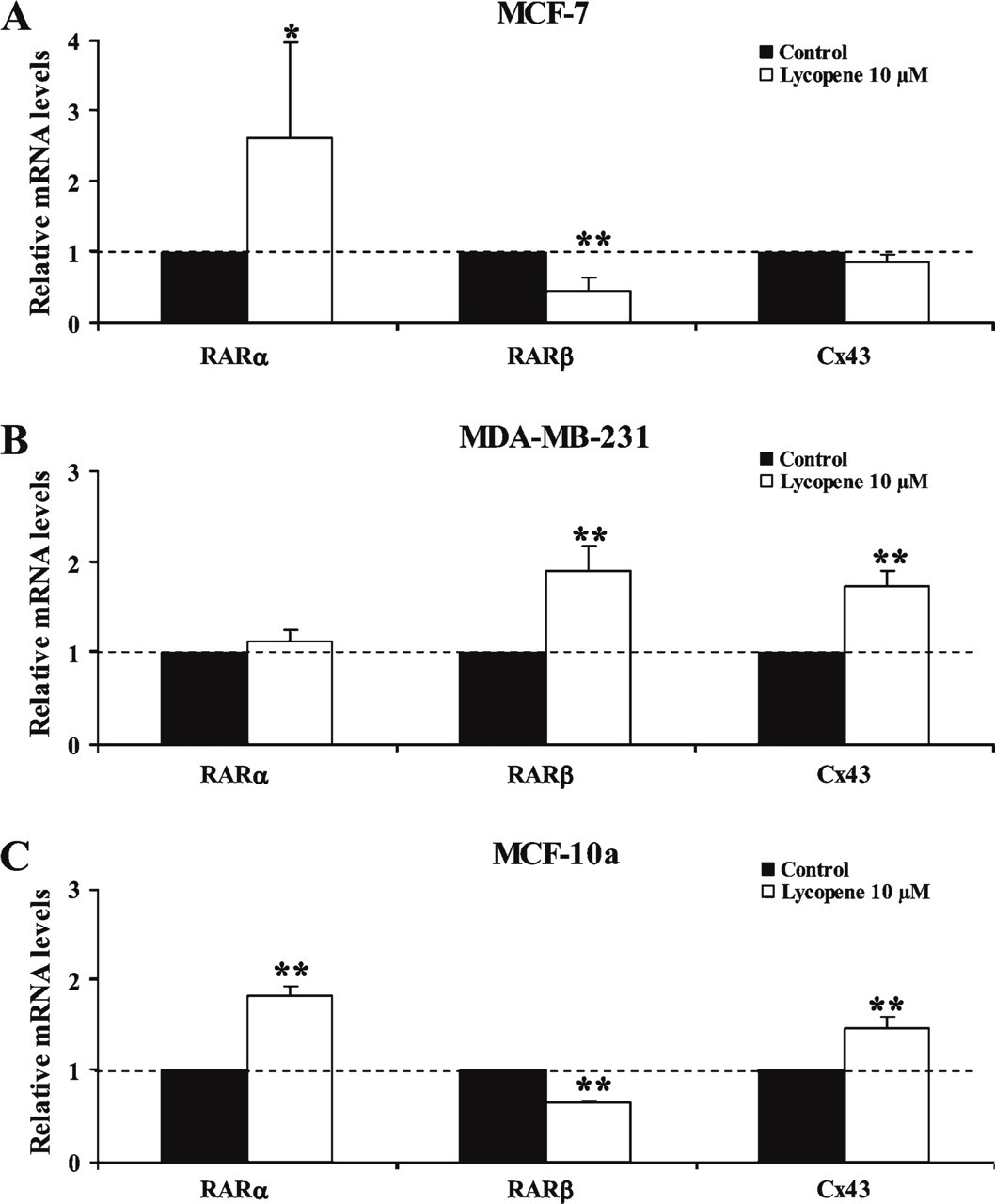
Quantitative RT-PCR of RARα, RARβ, and Cx43 mRNA in the MCF-7 (
In conclusion, this study suggests a putative role for lycopene in RAR and Cx43 expression in breast cell lines. Tomato consumption has been demonstrated in vitro and in clinical studies to have beneficial and protective effects with regard to degenerative diseases such as cancer. Its mechanism of action, however, is not totally clear. Our results would suggest a possible action of lycopene in breast cancer cell lines via retinoic acid receptors. Further investigations are nevertheless necessary to identify other molecular pathways to evaluate their possible biological relevance.
Footnotes
Acknowledgements
This work was supported by grants from La Ligue Nationale Française de Lutte Contre le Cancer and Le Comité du Puy-de-Dôme. N.C. is the recipient of a grant from the Fondation pour la Recherche Médicale; L.D. is the recipient of a grant from the Association pour la Recherche sur le Cancer; S.S. is the recipient of a grant from the Département d'Oncogénétique of Centre Jean Perrin.
We are grateful to Christelle Picard and Jacqueline Avinain for their technical assistance, and to Stéphanie Arnould for correcting the English.
