Abstract
The Vif protein of human immunodeficiency virus-1 (HIV-1) interacts with members of the APOBEC family of cytidine deaminases. In this study, we isolated RNA from renal cortex as well as from isolated glomeruli and tubulointerstitial fractions from two pigtailed macaques that were exsanguinated and perfused with saline. RT-PCR results indicate that APOBEC3G was detected in the tubule fractions but not in the glomerular fractions. Immunoblot analysis using lysates prepared from these same fractions and a monoclonal antibody to APOBEC3G confirmed the RT-PCR findings. To determine which cell types express APOBEC3G, immunohistochemical studies were performed using this monoclonal antibody on renal cortical sections. Our results clearly show that the glomeruli do not express APOBEC3G but that select tubules within the cortex express APOBEC3G at high levels. To further differentiate the distribution of APOBEC3G expression, serial sections were stained with the lectins Dolichos biflorus agglutinin (DBA) and Phaseolus vulgaris erythroagglutinin (PHA-E), which differentially bind to epithelial cells of the tubules and glomeruli. Our results indicate that APOBEC3G expression was restricted to PHA-E-staining tubules and not DBA-staining tubules, suggesting that APOBEC3G expression was restricted to proximal convoluted tubules. These findings suggest that infection of epithelial cells of proximal renal tubules could suppress Vif-defective HIV-1 replication, whereas infection of cells of the glomeruli, a major target of HIV-associated nephropathy, could act as a reservoir for the replication of Vif-defective HIV-1.
A
The interaction of HIV-1 proteins with cellular proteins can influence its replication in a particular cell type. The Vif protein is now known to interact with members of the apolipoprotein-B-editing catalytic polypeptide 3 or APOBEC3 gene family (Sheehy et al. 2002). The human genome contains eleven known members of the APOBEC family, many of which have cytidine deaminase activity (Sawyer et al. 2004). In primates, the APOBEC3 gene cluster consists of eight genes that reside on chromosome 22, which probably arose as a result of gene duplication events (Jarmuz et al. 2002). Recent studies have shown that in the absence of Vif, APOBEC3G is incorporated into the maturing virions and it deaminates deoxycytosine residues to deoxyuridine in newly synthesized minus strand DNA (Yu et al. 2004b). Ultimately, this results in G to A changes in the viral genome, introduction of pretermination codons, and decreased virus infectivity (Harris et al. 2003; Kao et al. 2003; Mangeat et al. 2003; Mariani et al. 2003; Zhang et al. 2003). The interaction of Vif with APOBEC3G results in subsequent degradation by the proteasome, reducing incorporation of APOBEC3G into maturing virions (Dussart et al. 2004; Marin et al. 2003; Sheehy et al. 2003; Stopak et al. 2003; Yu et al. 2003; Liu et al. 2004a; Mehle et al. 2004). Of the APOBEC3 genes, APOBEC3G is most closely related to APOBEC3F (Jarmuz et al. 2002). Studies have now shown that in addition to APOBEC3G, APOBEC3F can also preferentially restrict the replication of Vif (-) HIV-1 (Liddament et al. 2004; Wiegand et al. 2004; Zheng et al. 2004). In addition, APOBEC3B has been shown to have moderate activity against HIV-1, whereas other studies provide evidence of potent anti-HIV-1 activity (Yu et al. 2004a; Doehle et al. 2005). To date, no study has examined the distribution of this important host defense factor in the kidney. In this study, we have examined the distribution of APOBEC3G in the renal cortex using RT-PCR, immunoblot, and immunohistochemical-based methods in the macaque kidney. Our results indicate that APOBEC3G expression is restricted to the proximal convoluted tubules and collecting ducts of the renal cortex and is absent in the glomeruli and distal convoluted tubules of non-human primates.
Materials and Methods
Proteins, Antibodies, and Cells
The recombinant APOBEC3G (catalog number 10,068), APOBEC3F (catalog number 11,097), and APOBEC3B (catalog number 11,099) proteins were obtained from the National Institutes of Health AIDS research and reference reagents branch (Germantown, MD). The mouse monoclonal antibody against APOBEC3G was obtained from Immunodiagnostics (Woburn, MA; catalog number 7105). The HeLa APOBEC3G cell line (catalog number 9907) was also obtained from the National Institutes of Health AIDS research and reference reagents branch. The control antibodies used in this study were a rat monoclonal antibody against the rat F4-80, a 160-kDa surface glycoprotein that is a member of the EGF-TM7 family of proteins (Serotec, Raleigh, NC; MCAP497), and a mouse monoclonal antibody against the HIV-1 Gag p27 protein (AG3.0; National Institutes of Health AIDS research and reference reagents program). Both antibodies gave the same results.
Macaques and Processing of the Kidneys
The kidneys from two pigtailed macaques (Macaca nemestrina) were used to examine for the presence of APOBEC3G and were controls for an unrelated study. At the time of euthanasia, animals were anesthetized by administration of 10 mg/kg ketamine (IM) followed by sodium pentobarbital at 20-30 mg/kg IV. A laparotomy was performed on each animal, and the animals were exsanguinated by aortic canulation. The left ventricles were canulated, the right atria were nicked, and the animals were perfused with 2 liters of cold pyrogen-free Ringer's saline. All aspects of the animal studies were performed according to the institutional guidelines for animal care and use at the University of Kansas Medical Center. The kidneys were removed, the renal cortices were removed, and portions were immediately frozen in liquid nitrogen for RNA isolation. Other pieces of renal cortex were placed in 10% neutral buffered formalin for subsequent paraffin embedding, sectioning, and staining (hematoxylin and eosin) for routine histological analysis. No histological lesions were found in the renal tissues examined, and no significant levels of blood cells within blood vessels of the renal cortex were found.
Fractionation of Glomerular and Tubulointerstitial Fractions
Freshly harvested kidney tissue was sieved for isolation of glomeruli and cortical tubules using the sieving method of Savin and Terreros (1981). Glomerular and tubulointerstitial fractions were isolated for evaluation of protein and RNA as previously described (Gattone et al. 1998; Stephens et al. 1998,2000). We have found that the material retained by a 150 mesh screen was rich in glomeruli, whereas the material passing through the 200 mesh screen contained mainly tubules and mononuclear cells. The glomeruli retained by the 150 mesh sieve were further purified by preparing a series of 2-fold dilutions in DMEM medium supplemented with 10% fetal bovine serum. These dilutions were plated into 24-well plates, and those wells containing only glomeruli and no tubules (determined by microscopy) were pooled and concentrated by centrifugation. The purity of these glomerular preparations was greater than 99%. The glomerular and tubulointerstitial fractions were retained for DNA and RNA isolation.
RNA Extraction and RT-PCR Analysis
RNA was extracted from the renal cortex or from renal cortical fractions using Trizol reagent (Invitrogen; Carlsbad, CA) following the manufacturer's protocol. RNA samples were treated with DNase I to remove any contaminating DNA. The integrity of the RNA samples was confirmed by formaldehyde gel electrophoresis (data not shown) and amplification of the β-actin gene using RT-PCR and oligonucleotide primers previously described (Hill et al. 2006). For amplification of APOBEC3G from RNA samples, we used nested RT-PCR using the Titan One-Tube RT-PCR system (Roche; Indianapolis, IN) and APOBEC3G-specific oligonucleotide primers 5′-GAATACCGTCTGGCTGTGCTACG-3′(sense) and 5′-AGAAGTAGTAGAGGCGGGCAAC-3′ (anti-sense) in the first round. The conditions for the RT-PCR were a 30-min incubation at 42C, 10 cycles of denaturation at 94C for 30 sec, annealing at 55C for 30 sec, and elongation at 68C for 45 sec. This was followed by 25 cycles of denaturation at 94C for 30 sec, annealing at 55C for 30 sec, and elongation at 68C for 2 min. For the nested PCR, 1 μl of the first-round reaction was used in DNA PCR using oligonucleotide primers 5′-CAAAGATCTTTCGAGGCCAGGTG-3′ (sense) and 5′-AAAGATGGTCAGGGTAACCTTCGG-3′ (antisense). The conditions for the nested DNA PCR were 30 cycles of denaturation at 92C for 1 min, annealing at 55C for 1 min, and primer extension at 72C for 3 min. The resulting amplified product was visualized in agarose gels and with a predicted product size of 209 bp. These primers were designed such that they will not amplify the APOBEC3F gene (data not shown).
Immunoblot Analysis for APOBEC3G
Renal cortex or renal cortical fractions were homogenized to a final 10% weight/volume in 1X PBS, pH 7.4. PMSF and aprotinin were added to final concentrations of 5 mM and 1 mM, respectively. Sample-reducing buffer was added to 2 mg tissue equivalents, and samples were boiled for 5 min, cooled, and loaded onto a 10% SDS-PAGE. Cells were lysed in 1X radioimmunoprecipitation assay buffer on ice for 30 min. Lysates were centrifuged to remove the nuclei, and then 4 vols of 100% cold methanol was added and incubated at −80C for 30 min followed by centrifugation at 11,000X g for 30 min. The protein pellets were washed with 100% cold methanol followed by centrifugation at 14,000 rpm for 15 min. The protein pellets were dried, re-suspended in 150 μl of sample-reducing buffer, boiled for 5 min, cooled, and loaded onto a 10% SDS-PAGE. Equal amounts of protein from each fraction were loaded onto the SDS gels. Blots were blocked in 4% BSA (Sigma; St Louis, MO) in TTBS (0.3 M NaCl, 40 mM Tris-HCl, pH 7.5, 0.1% Tween-20) overnight at 4C. APOBEC3G was detected using an anti-APOBEC3G monoclonal antibody (Immunodiagnostics, catalog number 7105; 1:10,000 dilution) for 1 hr at room temperature. Blots were washed four times for 5 min in TTBS, followed by incubation with a biotinylated anti-mouse IgG (Vector Laboratories, Burlingame, CA; 1:1000 dilution) for 1 hr. Antibodies were diluted in 3% BSA in TTBS. Blots were washed four times in TTBS, and chemiluminescent signal was acquired using Vectastain ABC-AmP Western blotting immunodetection kit according to the manufacturer's instructions (Vector Laboratories). Immunoblots were imaged with the BioRad ChemiDoc XRS system in conjunction with Quantity One software (Hercules, CA).
Immunohistochemistry
For immunohistochemistry, the renal cortex was fixed in 10% neutral buffered formalin for 7 days. The tissue was then cryoprotected in 30% sucrose in 0.1 M PBS, pH 7.2, and frozen sectioned at 40 μm using a sliding microtome (Hill et al. 2006). Sections were washed free floating (three times in PBS), quenched for endogenous peroxidase in 0.6% H2O2 for 30 min, and blocked in 10% normal serum matched to the secondary antibody in PBS for 1 hr. Sections were incubated in primary antibody overnight at room temperature. For detection of APOBEC3G, the primary antibody used was a mouse monoclonal anti-human APOBEC3G (1:1000 dilution, Immunodiagnostics catalog #;7105). Sections were washed three times at 10 min per wash, and incubated in biotinylated horse anti-mouse IgG (Vector Laboratories; 1:200) for 1 hr. Sections were then washed three times in PBS, and incubated in avidin-biotinylated enzyme complex (Vector Labs) for 1 hr. The sections were rinsed two times in PBS and incubated with 0.5% diaminobenzidine containing 0.1% H2O2 for 2 min until color development was observed. Negative controls included the use of an irrelevant monoclonal antibody (Serotec catalog number MCAP497) that binds to anti-F4/80, as well as controls in which no primary antibody was added. Slides were viewed using a Nikon TE300 microscope and photographed using a SPOT camera system (Tokyo, Japan).
For identification of proximal and distal convoluted tubules/collecting ducts, serial sections from the renal cortex were reacted for 1 hr with biotinylated Dolichos biflorus agglutinin (DBA) lectin (Vector Labs; 1:1000 dilution) or biotinylated Phaseolus vulgaris erythroagglutinin (PHA-E) lectin (Vector Labs, 1:2000 dilution). Sections were washed three times in PBS, and incubated in avidin-biotinylated enzyme complex (Vector Labs) for 1 hr. The sections were rinsed two times in PBS and incubated with 0.5% diaminobenzidine containing 0.1% H2O2 for 2 min until color development was observed. Sections were then mounted on slides and coverslipped for viewing. The negative control included no lectin controls. Slides were viewed using a Nikon TE300 microscope and photographed using a SPOT camera system, and common regions from serial sections were compared for lectin and APOBEC3G immunoreactivity.
Results
RT-PCR Analysis Reveals that the Tubulointerstitial Fractions but Not Glomeruli Express APOBEC3G
We analyzed unfractionated and fractionated renal cortex for the presence of APOBEC3G mRNA. The results shown in Figure 1A indicate that APOBEC3G mRNA was expressed in the unfractionated renal cortex. As our controls, we used RNA isolated from macaque peripheral blood mononuclear cells (PBMCs; positive control) and RNA from HeLa cells, which do not express APOBEC3G (negative control).
Immunoblot Analysis Confirms the Expression of APOBEC3G
Because the results from the RT-PCR indicated that APOBEC3G mRNA was expressed in the tubulointerstitial but not glomerular fractions of the renal cortex, we performed immunoblot analysis to detect protein expression. We prepared tissue homogenates from renal cortex and the two renal fractions in the presence of protease inhibitors and used these in immunoblot analysis with a monoclonal antibody directed against APOBEC3G. The results shown in Figure 2A indicate that C8166 cells expressed a protein with an Mr of 48,000 (predicted size of APOBEC3G), whereas HeLa cells, which do not express APOBEC3G, were negative for this protein. Western blot analysis demonstrated expression of APOBEC3G protein in tubulointerstitial fractions but not glomerular fractions of the kidney, thus confirming the RT-PCR analysis.
Immunohistochemistry Indicates that APOBEC3G Expression Is Confined to Tubular Regions but Not Glomeruli
Recently, we showed that a monoclonal antibody directed against the APOBEC3G could be used in fixed, cryoprotected tissues to detect APOBEC3G-expressing cells (Hill et al. 2006). As a positive control for our immunohistochemical experiments, we immunostained sections of macaque spleen tissue, which is rich in lymphocytes and macrophages and known to express APOBEC3G. As shown in Figures 3C and 3D, APOBEC3G expression was detected in the spleen tissue, particularly in cells of the red pulp, which contains numerous macrophages and T and B cells. Immunohistochemistry on serial sections of the spleen revealed that APOBEC3G expression was detected in the same region as cells expressing CD4 (Figures 3A and 3B). As a negative control, we showed that the F4-80 antibody did not stain the spleen tissues (Figures 3E and 3F). These results indicate that this monoclonal antibody could be used in immunohistochemistry to detect APOBEC3G-expressing cells in our formalin-fixed cryopreserved tissues.
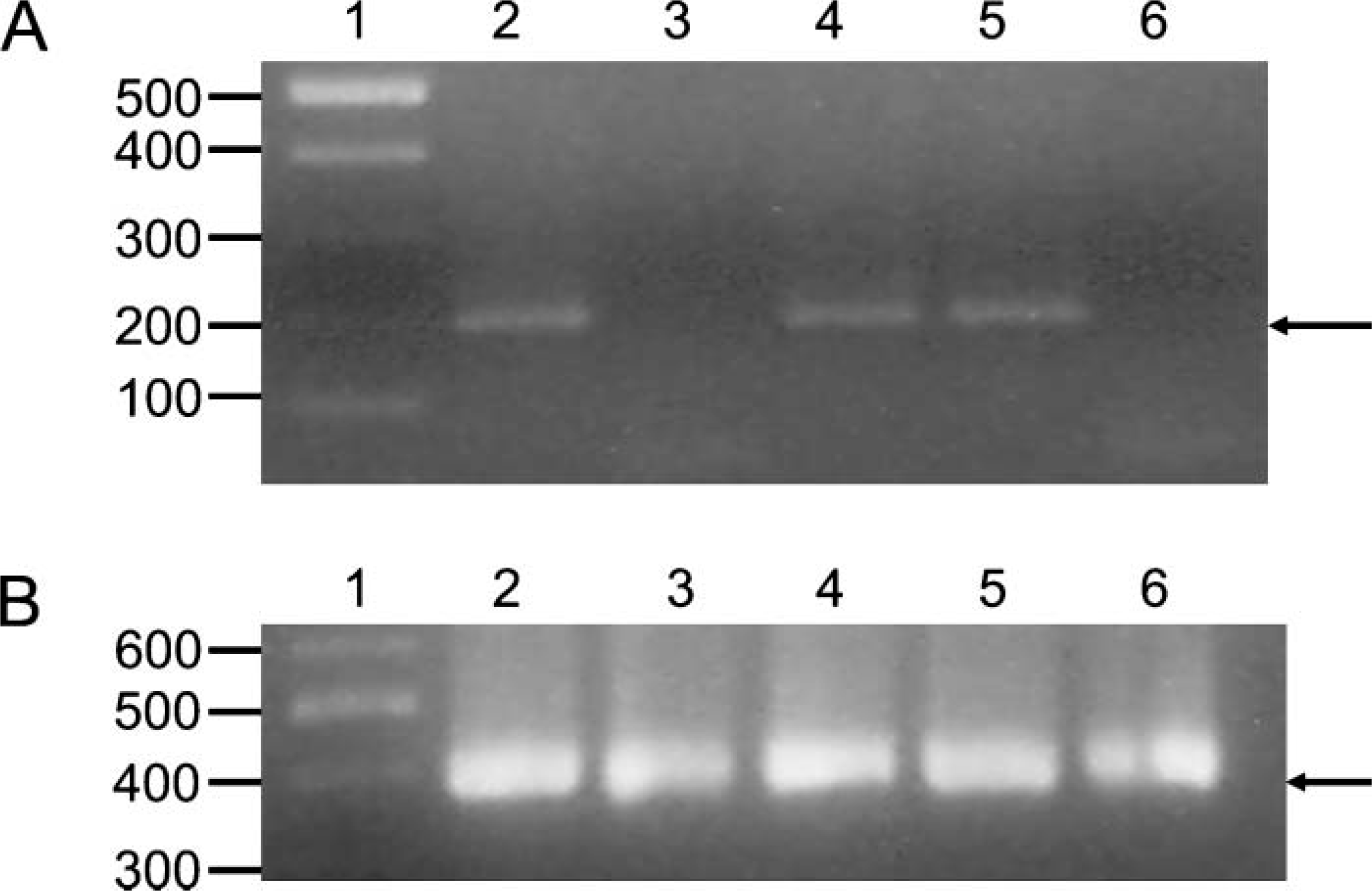
RT-PCR indicates that expression of APOBEC3G is confined to the tubulointerstitial fraction of the renal cortex. A pigtailed macaque was exsanguinated and perfused with saline to remove as much contaminating blood from the kidneys as possible. The kidneys and the renal cortex were removed, and the RNA was isolated using Trizol reagent according to the manufacturer's instructions. The RNA preparations were Dnase treated, heat inactivated, and then used in RT-PCR using oligonucleotide primers to amplify either APOBEC3G (
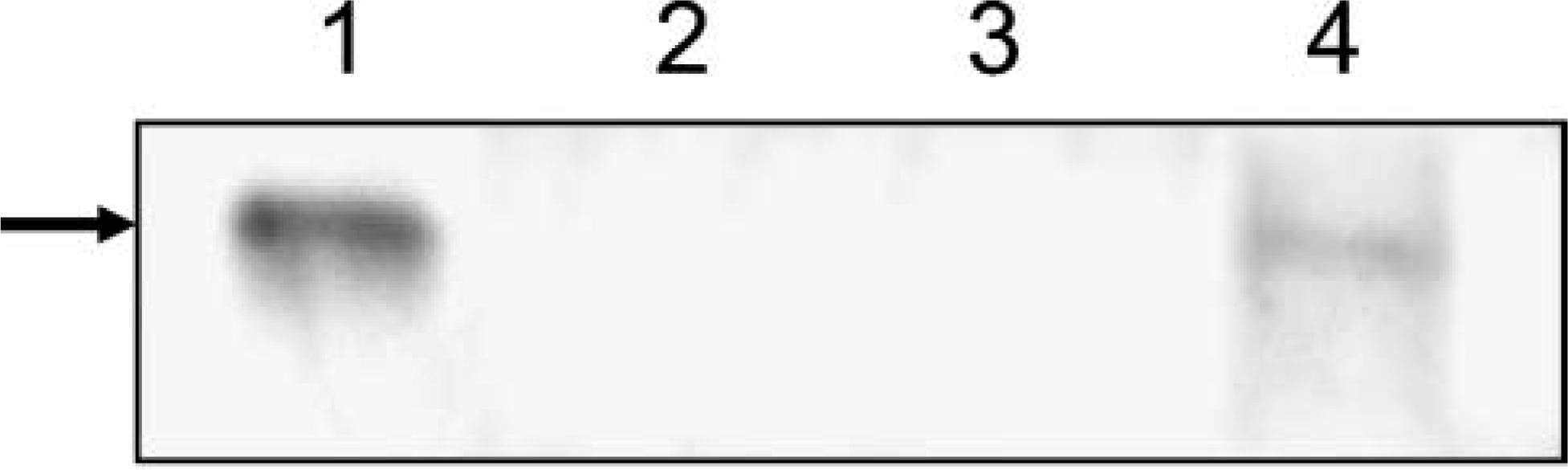
Detection of APOBEC3G protein in the renal cortex. Renal cortex was isolated and fractionated into glomerular and tubulointerstitial fractions as described in the Materials and Methods section. Tissue homogenates (10% w/v) were prepared on ice in PBS, pH 7.2, containing a cocktail of protease inhibitors. Equal aliquots (based on protein content) of the fractionated homogenates were mixed in 2X sample-reducing buffer and boiled. The lysates were run on Western blots and probed using a mouse monoclonal antibody directed against APOBEC3G (1:10,000). Chemiluminescent signal was acquired using VectaStain ABC-Amp and a BioRad gel imager. Lane 1, C8166 cells (positive control); Lane 2, HeLa cells (negative control); Lane 3, isolated glomerular fraction; Lane 4, tubulointerstitial fraction. Arrow is at predicted size of APOBEC3G.
We next used this antibody in immunohistochemical analysis of the renal cortex, and the results are shown in Figures 4A and 4B. Figure 4A is a low-power image showing that glomeruli were devoid of APOBEC3G immunostaining whereas the tubulointerstial fractions were positive for APOBEC3G expression. Higher magnification images show that the site of intracellular expression was cytoplasmic (Figure 4B). Immunohistochemistry with an irrelevant monoclonal antibody (Figure 4C) or with no primary antibody (data not shown) revealed no staining of the renal cortex. Because the micrographs shown in Figures 4A and 4B indicated a differential immunostaining pattern suggestive of proximal convoluted tubules, we next used differential binding of lectins to differentiate between proximal and distal convoluted tubules/collecting ducts. Renal cortical sections stained with DBA revealed a pattern of reactivity that was consistent with distal tubules and collecting ducts (Figures 4D-4F), whereas sections stained with PHA-E revealed a reactivity pattern that was consistent with proximal tubules and glomeruli (Figures 4G-4I). Of the two patterns of reactivity, the APOBEC3G immunostaining pattern closely resembled that of the reactivity of proximal convoluted tubules stained with PHA-E.
Isolation of Glomerular and Tubulointerstitial Fractions Confirms the Lack of APOBEC3G Expression in the Glomeruli
To confirm the immunohistochemical analysis, which indicated that cells of the glomeruli did not express APOBEC3G, glomerular and tubulointerstitial fractions were isolated from the macaque renal cortex. These fractions were used either in RT-PCR analyses or in immunoblot analyses to assess the presence or absence of APOBEC3G. As shown in Figures 1B and 2B, we were unable to amplify APOBEC3G from RNA extracted from the glomerular fraction by RT-PCR or detect APOBEC3G in immunoblot analyses. In contrast, we detected APOBEC3G mRNA and protein in the tubulointerstitial fractions. These data confirm our immunohistochemical results.
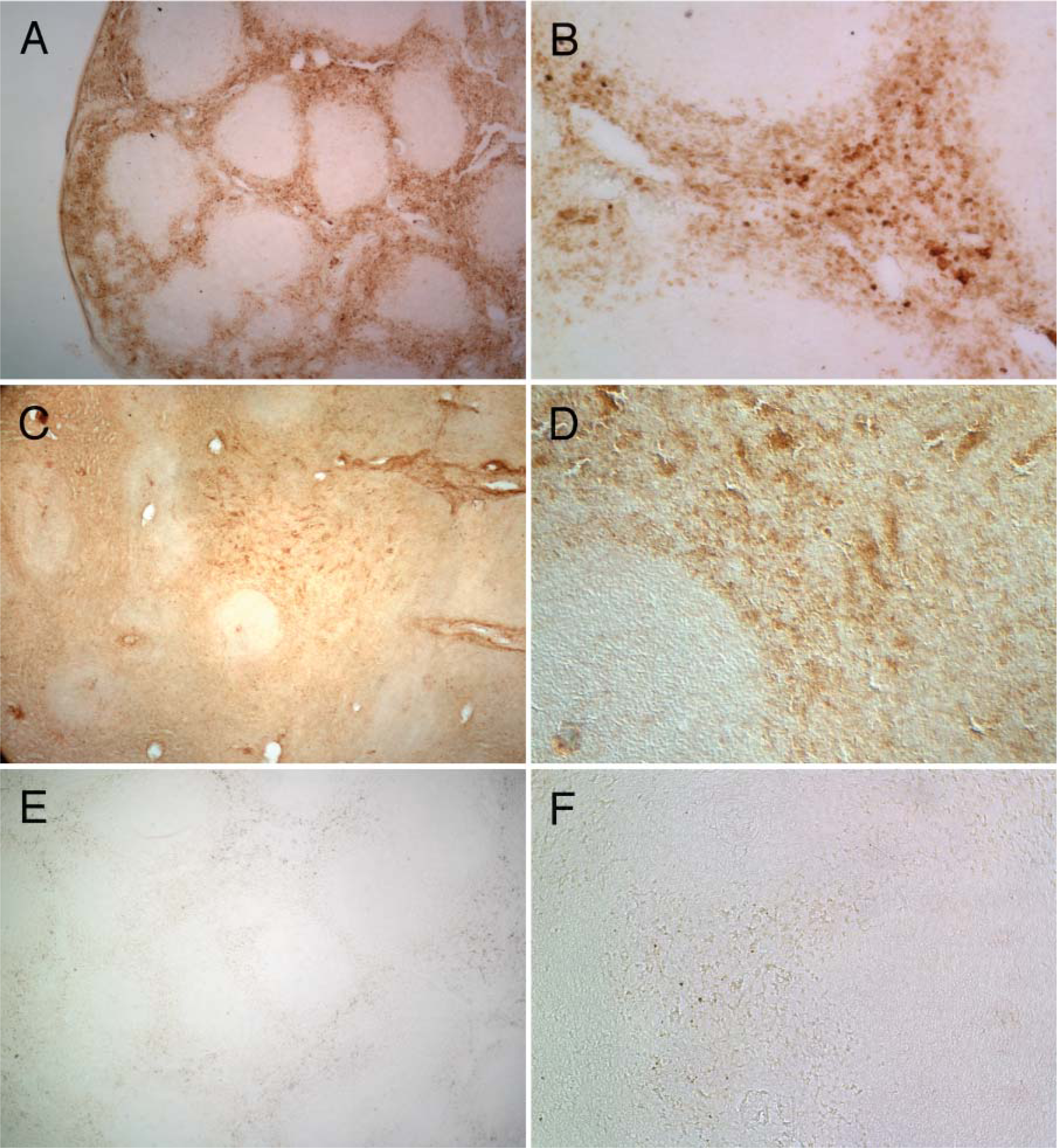
Expression of CD4 and APOBEC3G in the same region of the spleen. Frozen sections of spleen (20 μm) were stained for CD4 (
Discussion
Macaques are known to develop a nephropathy following inoculation with simian immunodeficiency virus (SIV) or simian-human immunodeficiency virus (SHIV) and have served as models for studying various aspects of HIV-1 pathogenesis (Alpers et al. 1997; Stephens et al. 1998; Liu et al. 1999). In the present study, we have examined the mRNA and protein expression of the cytidine deaminase APOBEC3G in renal tissues. Our results clearly indicate that APOBEC3G is expressed in tubules of the renal cortex but not in the glomeruli. We confirmed the lack of expression of APOBEC3G in the glomeruli by the isolation of glomerular and tubulointerstitial fractions followed by analysis by RT-PCR. By using plant lectins that differentially bind to different carbohydrate structures on the tubules of the renal cortex (i.e., proximal convoluted tubules versus distal convoluted tubules and collecting ducts), we showed that APOBEC3G expression is confined to the proximal convoluted tubules.
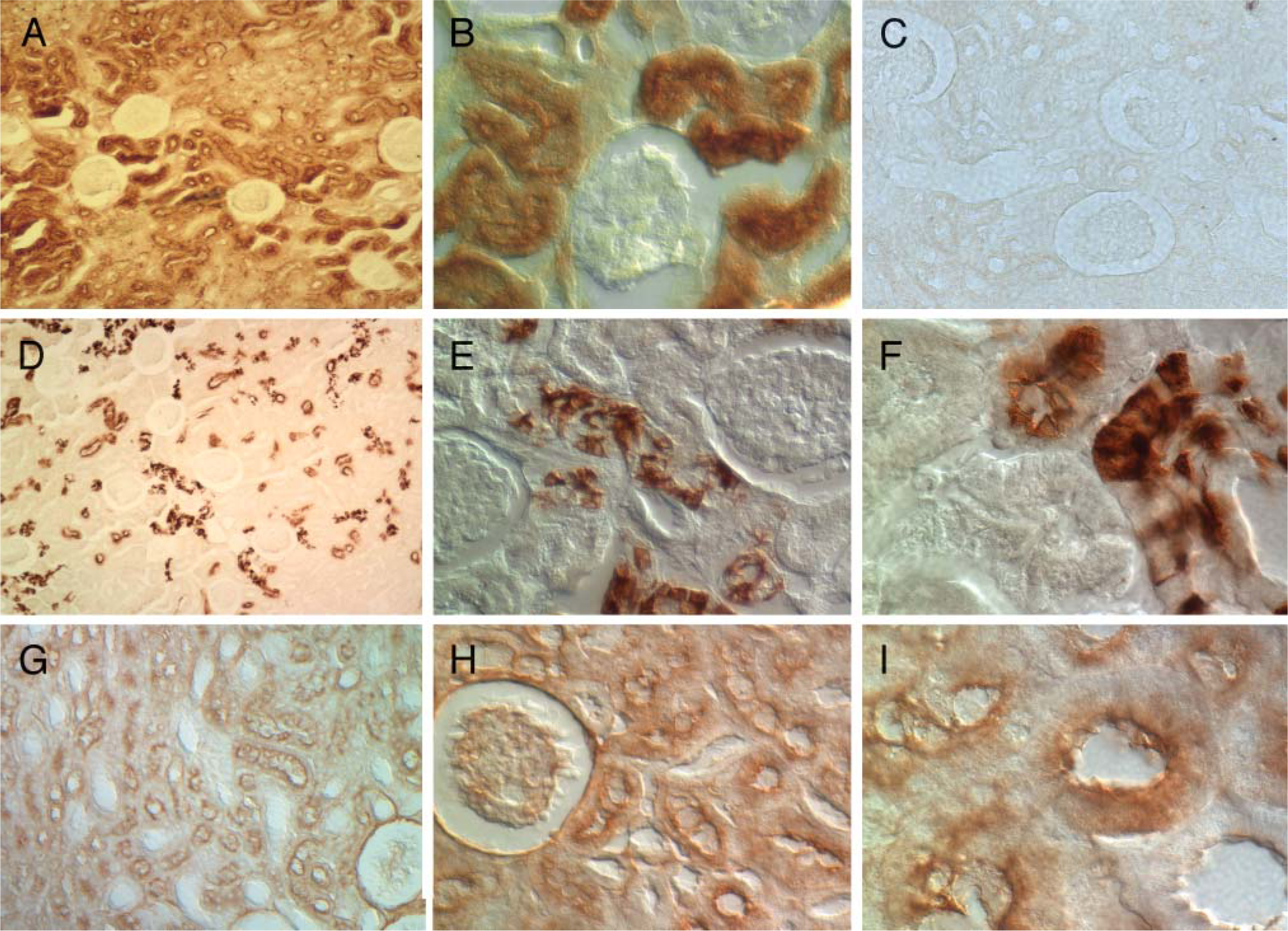
Immunohistochemical analysis of the renal cortex of a macaque showing APOBEC3G-positive cell types, Dolichos biflorus agglutinin (DBA)-reactive distal convoluted tubules, and Phaseolus vulgaris erythroagglutinin (PHA-E)-reactive proximal convoluted tubules and glomeruli. (
HIV-1 is capable of infecting different cell types of the kidney. Studies have shown that the renal tubular epithelial cells can be infected with HIV-1 via a CD4-independent pathway and that infected mononuclear cells can transmit virus to these renal epithelial cells (Cohen et al. 1989; Ray et al. 1998). Other studies have shown that highly active anti-retroviral therapy (HAART) for HIV-1 patients reduces the incidence of HIVAN, but that ESRD may occur as a side effect of HAART (Lucas et al. 2004; Roling et al. 2006). Expression of HIV-1 gene products in podocytes also appears to be important in the pathogenesis of HIVAN. In a transgenic mouse model of HIV-1 nephropathy, expression of parts of the HIV-1 genome can result in a nephropathy similar to that seen in HIV-1 patients (Bruggeman et al. 1997). In a recent study using transgenic mice, it was shown that expression of vif, vpr, nef, tat, and rev under the control of a podocyte-specific promoter resulted in the full spectrum of pathologic changes observed in patients with HIVAN (Zhong et al. 2005). Expression of nef alone in podocytes leads to dedifferentiation of podocytes, which is also observed in patients with HIVAN (Schwartz et al. 2001; Husain et al. 2002,2005; Sunamoto et al. 2003). Finally, in a recent study using in situ PCR, investigators detected viral sequences in podocytes, parietal cells, renal tubular epithelial cells, and infiltrating leukocytes from patients with HIVAN (Tanji et al. 2006). Thus, these studies indicate that HIV-1 can infect the glomerular and tubulointerstitial compartments of the renal cortex.
Recently, we examined APOBEC3G expression in the central nervous system (CNS) and found that expression was restricted to neurons (Hill et al. 2006). Because HIV-1 in humans and SIV or SHIV in macaques replicate predominantly in microglial cells and to a lesser extent in astrocytes but not neurons, this has implications for the viruses that reside in the CNS (Tornatore et al. 1994; Bagasra et al. 1996; Ludwig et al. 1999). That is, if APOBEC3G is not expressed within the microglial cells, there is no selective pressure to express a functional Vif protein to counteract the actions of APOBEC3G cytidine deaminase. Thus, it is possible that viruses expressing non-functional Vif proteins may reside in the CNS. Our findings here also show that cells of the glomeruli (podocytes, endothelial cells, mesangial cells) do not express APOBEC3G. Thus, it is possible that podocytes of the glomeruli and some renal tubular epithelial cells (such as the distal convoluted tubules) may serve as reservoirs for the replication of Vif-defective HIV-1. In contrast, infection of the proximal convoluted tubule epithelial cells, which have been shown to express APOBEC3G, should select against the replication of the Vif-defective HIV-1. A potential caveat is that although APOBEC3G does not appear to be expressed in cells of the glomeruli, perhaps other members of the APOBEC3 family (APOBEC3F, APOBEC3B, APOBEC3C) with activity against HIV-1 (or SIV) could be expressed. However, APOBEC3F, which is most closely related to APOBEC3G at the amino acid level, appears to have the same tissue distribution as APOBEC3G. APOBEC3B is poorly expressed in human tissues (and mostly likely macaques). Also, a previous study on HIVAN suggested compartmentalization of HIV-1 sequences in the kidney (Marras et al. 2002). In this study, renal tubular epithelial cells were microdissected from patients with HIVAN and the V3 sequences of the HIV-1 envelope gene were compared with those of PBMCs. Phylogenetic analysis of the viral sequences from these two compartments revealed evidence of tissue-specific evolution. It will be of interest to determine whether HIV-1 viruses expressing nonfunctional Vif proteins can be detected in the podocytes or renal tubular epithelial cells from patients with HIVAN or macaques that develop SIVAN or SHIVAN.
Footnotes
Acknowledgements
This study was supported by National Institutes of Health Grants AI-064019 and AI-051981 to E.B.S.
