Abstract
We have recently reported that in chronic myocardial ischemia, adult mammalian cardiomyocytes express P-glycoprotein (P-gp). We now investigate if P-gp is also expressed in acute regional ischemia followed by reperfusion. Adult conscious sheep underwent 12-min occlusion of the mid-left anterior descending artery (inflatable cuff). Successful ischemia-reperfusion was confirmed by monitoring percent systolic left ventricular anterior wall thickening (sonomicrometry) during the whole is chemic period and every 10 min over 2 hr following cuff deflation. At 3, 24, and 48 hr after reperfusion, P-gp expression was investigated by immunohistochemistry and Western blot and
Keywords
T
In the present study we investigated if cardiomyocytes of adult conscious sheep undergoing acute, nonlethal regional myocardial ischemia followed by full reperfusion express P-gp.
Materials and Methods
Surgical Preparation
Nine Corriedale sheep weighing 27 ± 2 kg were operated. All procedures were done in accordance with the Guide for the Care and Use of Laboratory Animals, published by the US National Institutes of Health (NIH publication No. 85-23, revised 1996). After premedication with acepromazine maleate (0.3 mg/kg), anesthesia was induced with sodium thiopental (20 mg/kg) and maintained with 3% enflurane carried in oxygen through a Bain tube connected to a volume-driven ventilator. A sterile thoracotomy was performed at the left fifth intercostal space, and the pericardium was opened. A pressure microtransducer (P7 transducer; Konigsberg Instruments, Inc., Pasadena, CA) was inserted in the left ventricle (LV) through a stab wound in the apex together with a fluid-filled catheter for later calibration of the microtransducer. The left anterior descending (LAD) artery was dissected free from surrounding tissue just distal to the emergence of the second diagonal branch, and a pneumatic cuff occluder was positioned around it.
To measure LV wall thickness, a pair of 5-MHz piezoelectric crystals (one sutured to the epicardium and the other lying in the subendocardium) was placed well within the zone to be rendered ischemic. Cables and catheters were tunneled SC to emerge between the scapulae, and the thoracotomy was repaired. The venous and LV catheters were flushed daily with heparinized saline. Cephalomycin (1 g) was given IV immediately after surgery and continued for 3 days (1 g/day IM).
Experimental Protocol
Seven to 10 days after surgery the animals were studied in the conscious, unsedated state. The Konigsberg transducer and the ultrasonic crystals were connected to a System 6 Physiological Monitoring System (Triton Technology; San Diego, CA), and the fluid-filled catheter was connected to a pressure transducer (DT-XX; Viggo-Spectramed, Oxnard, CA) previously calibrated using a transducer calibration system (Xcaliber; Viggo-Spectramed). The zero pressure point was set at the level of the right atrium, and the signal generated by the Konigsberg transducer was adjusted to match that of the external transducer. Ultrasonic crystals were calibrated in millimeters using the sonomicrometer internal calibration.
The experiment consisted of 20 min of stabilization and 12 min of ischemia followed by reperfusion. This duration of ischemia was previously shown to produce a significant degree of myocardial stunning without permanent cardiomyocyte injury (Negroni et al. 2002).
Measurements were acquired just before ischemia (basal condition), at the time of cuff deflation, every 10 min during the first hour, and every 20 min during the second hour of reperfusion. At each acquisition time, the signals of consecutive steady beats were digitized at 4-msec intervals over 15 sec using a personal computer equipped with an A/D converter (National Instruments Lab-PC; Austin, TX) and software developed in our laboratory.
At 3 (
Regional Function Measurements
End diastole was defined to occur at the onset of the rapid upstroke of the digitally obtained time derivative of left ventricular pressure (dP/dt). End systole was defined as the time point where the descending limb of the negative dP/dt reached 10% of its minimal value and end ejection was established to occur at peak negative dP/dt (del Valle et al. 2001). Percent regional wall thickening fraction (%WTh) was calculated as %WTh = 100X(WThe-WThd)/WThd, where WThe is maximum wall thickness between end systole and end ejection, and WThd is end diastolic wall thickness.
Signal processing and %WTh calculation were performed at each time of data acquisition from each recorded beat. The average of processed beats (15 to 30 beats) was the sample value assigned to the corresponding acquisition time. Ischemia and reperfusion recovery were expressed as percent of basal condition taken as 100%.
Histology and Immunohistochemistry
The LV was cut transversally at a plane lying ∼0.5 cm above the epicardial crystal. Correct positioning of the endocardial crystal was assessed, and a 1-cm-thick slice of the whole LV wall close to (but not containing) the crystal pair was cut and fixed in 10% buffered formaldehyde for 48 hr. The slice was then sectioned into four pieces corresponding to the interventricular septum and the anterior, lateral, and posterior LV walls. Each piece was further divided into two halves. All eight fragments were embedded in paraffin. Four-μm-thick tissue sections were stained with hematoxylin-eosin, periodic acid-Schiff, and Masson's trichrome. For immunohistochemistry, antigen retrieval was done by incubating the hydrated sections in 10 mM sodium citrate buffer (pH 6) in a microwave oven during 5 min. To detect P-gp in a double-check fashion, we used two different specific monoclonal antibodies: clone C494 (1:100, 200 μg/ml; Signet Laboratories, Dedham, MA) or clone MDR88 (1:100,10 mg/ml; Biogenex, San Ramon, CA). Clone C494 antibody detects an epitope present only in the
Western Blot
In the six hearts submitted to Western blot analysis, an extra 1-cm slice immediately distal to the slice used for histological analysis was cut, divided into eight fragments as previously described, and deep frozen in liquid nitrogen. Protein extracts were obtained by the method of Maniatis (Sambrook et al. 1989) from ischemic-reperfused and control myocardial fragments. Samples containing 100 μg of protein (10 mM KCl, 1.5 mM MgCl2, 10 mM Tris Cl, pH 7.4, 0.5% SDS, 2 μg/ ml leupeptin, 2 μg/ml aprotinin, and 1 μg/ml E64) were subjected to SDS-PAGE in 7% gels and then electroblotted to Hybond P membrane (Amersham Life Science; Arlington Heights, IL). The primary antibody used was the monoclonal C494 (Signet Laboratories) or anti-β actin (Santa Cruz Biotechnology; Santa Cruz, CA) as internal protein loading control. The secondary antibody was a peroxidase-conjugated goat anti-mouse IgG (Dako; Carpinteria, CA) coupled with the detection system ECL reagent (Amersham) according to the manufacturer's instructions. Densitrometric analysis was performed using Gel-Pro Analyzer software (version 3.1; Media Cybernetics, Silver Spring, MD). Tissues with high constitutive expression of P-gp (liver, colon, and kidney from rat and sheep) were used as positive controls. Results obtained in control and ischemic-reperfused tissue samples were expressed in optical density (OD) units.
mRNA Isolation and RT-PCR Analysis
Total RNA was isolated (Trizol reagent; Gibco BRL, Grand Island, NY), treated with DNase I (Promega; Madison, WI), quantitated, and reverse transcripted (random hexamers; Perkin-Elmer, Boston, MA). Primers 5′-AGCCCATTCTGTTTGACTGC-3′ and 5′-TCAAGTCTGCGTTCTGGATG-3′ were designed to amplify the 3371- to 3742-bp region of the sheep
Statistical Analysis
Percent WTh was compared at each time point using Student's one-sample
Results
Effect of Ischemia-Reperfusion on Regional Myocardial Function
In all animals, ischemia-reperfusion lead to myocardial stunning. Basal, preocclusion WTh was 39.6 ± 4%. Figure 1 shows the time course of wall thickening fraction expressed as a percent of basal wall thickening, from the moment of cuff deflation up to 2 hr of reperfusion. At the onset of reperfusion, systolic %WTh showed a negative value (-14.2 ± 5.9%), indicating regional dyskinesia. Following cuff deflation, %WTh recovered progressively, reaching 70.5 ± 5.1% of the basal value after 2 hr of reperfusion. We did not measure %WTh at the time of sacrifice. However, because no permanent ischemic damage occurs in this established model of myocardial stunning, complete recovery of regional myocardial function is to be expected, especially at the 24- and 48-hr time points, as previously reported (del Valle et al. 2001; Negroni et al. 2002).
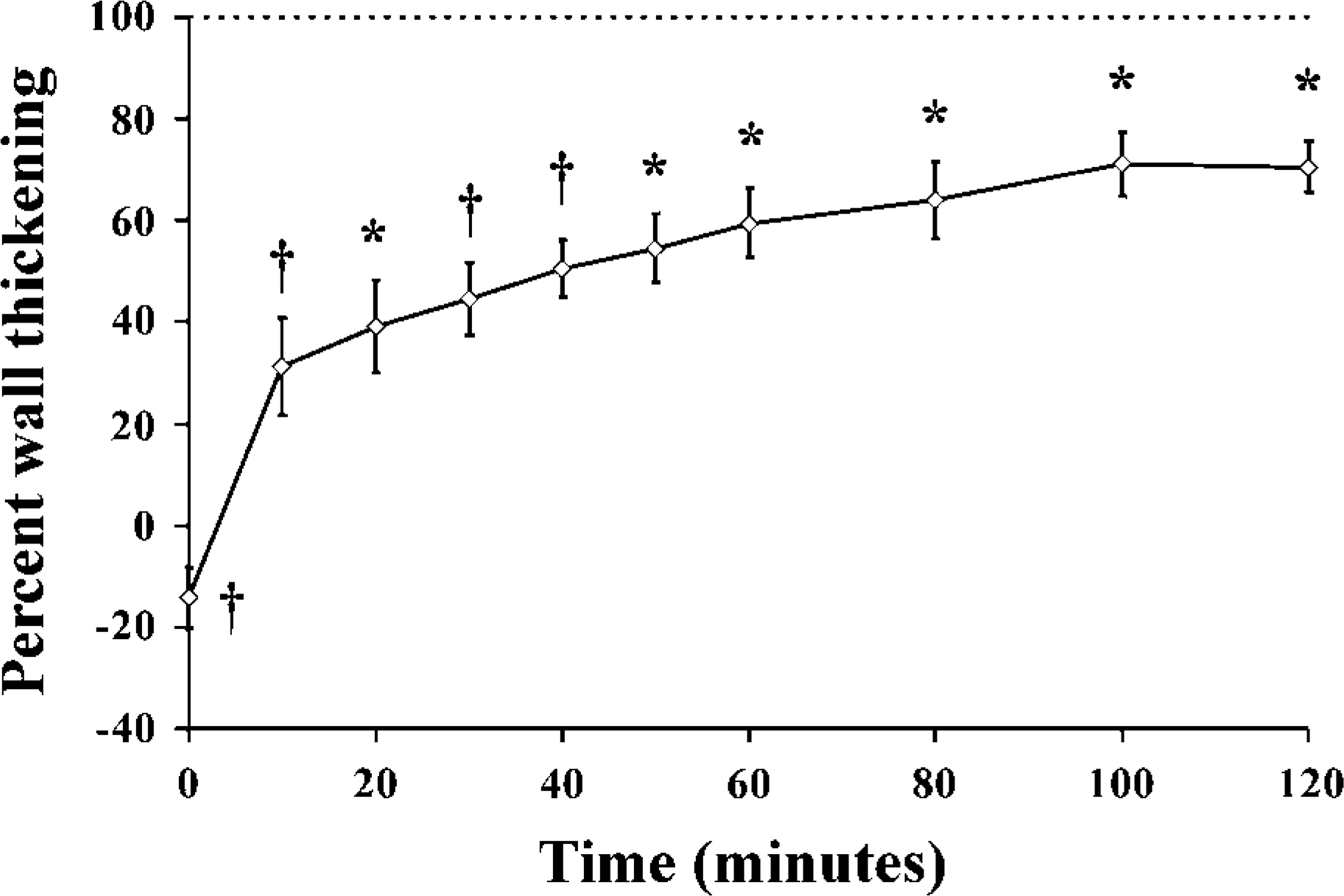
Time course of percent wall thickening in the left anterior descending artery territory. At the onset of reperfusion, the left ventricular anterior wall displays dyskinesia. Over the following 2 hr, percent wall thickening remains significantly lower than baseline, confirming the presence of stunning in the myocardium subjected to ischemia-reperfusion. The 100% value represents the baseline value prior to ischemia. Results are expressed as percent of baseline %WTh. ∗
Immunohistochemistry
In the five sheep killed at 3 hr after the onset of reperfusion, all cardiomyocytes in the tissue blocks corresponding to the LAD territory (anterior septum and anterior ventricular wall) displayed a positive reaction at their sarcolemma and T tubules (Figure 2A). In contrast, cardiomyocytes from the lateral and posterior ventricular wall were consistently negative. In the tissue sections corresponding to the periphery of the ischemic area, transition was abrupt, and intensely P-gp-positive cardiomyocytes were observed alongside negative ones (Figure 2B). In this border zone, the capillaries (Figure 2B) as well as larger arterioles (Figure 2C) displayed a positive reaction.
In sheep killed at 3 and 24 hr after reperfusion, distribution of P-gp in the sarcolemma and T tubules was mostly uniform. In the animals killed at 48 hr after occlusion, most of the P-gp was located in the intercalated discs (Figures 2D and 2E) and lateral gap junctions (Figure 2F).
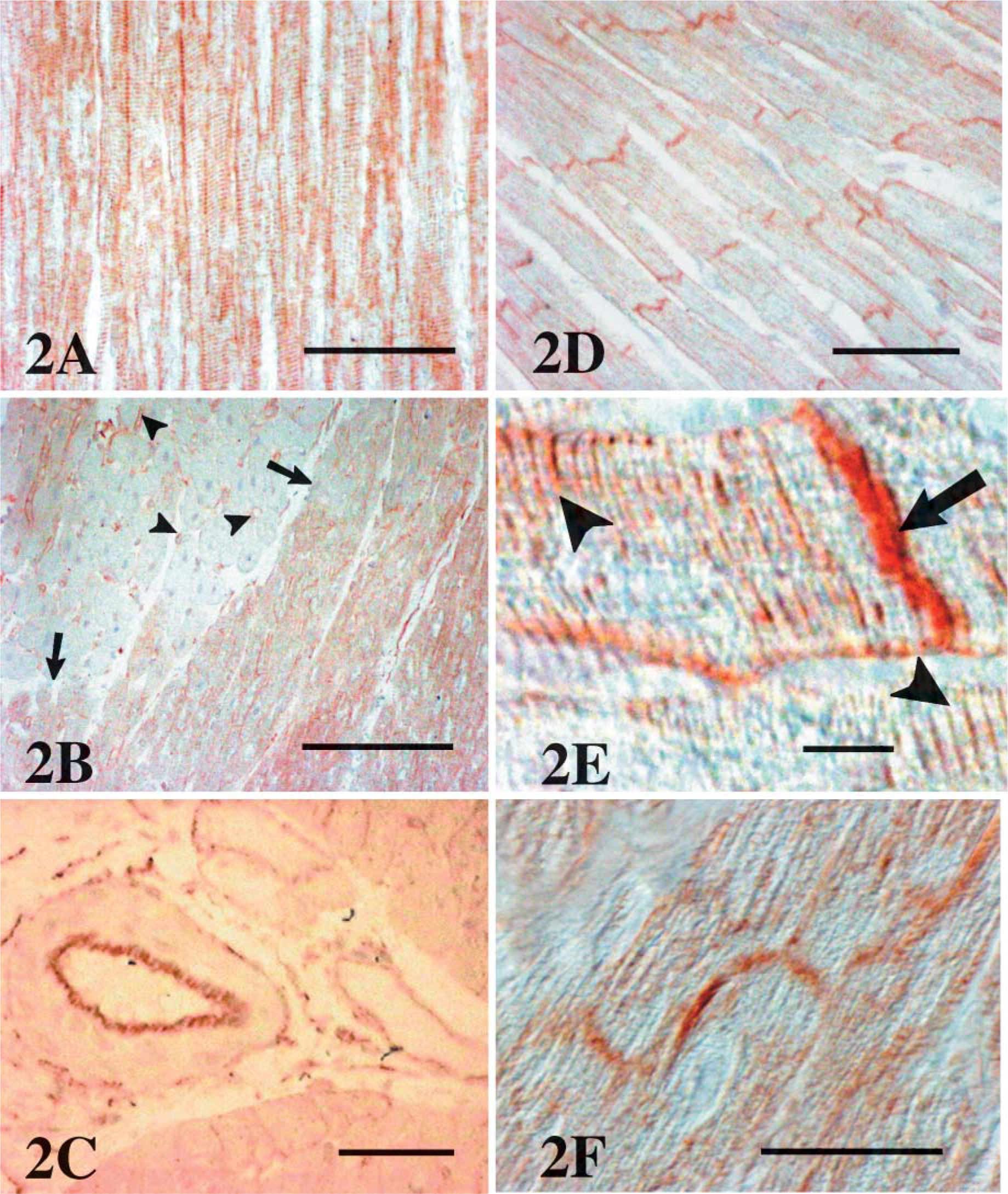
Immunohistochemistry for P-glycoprotein (P-gp). (
Western Blot
P-gp content (pooled data for all three time points) was 23.9 ± 3.9 OD in control myocardium and 78.4 ± 4 OD in ischemic-reperfused myocardium (
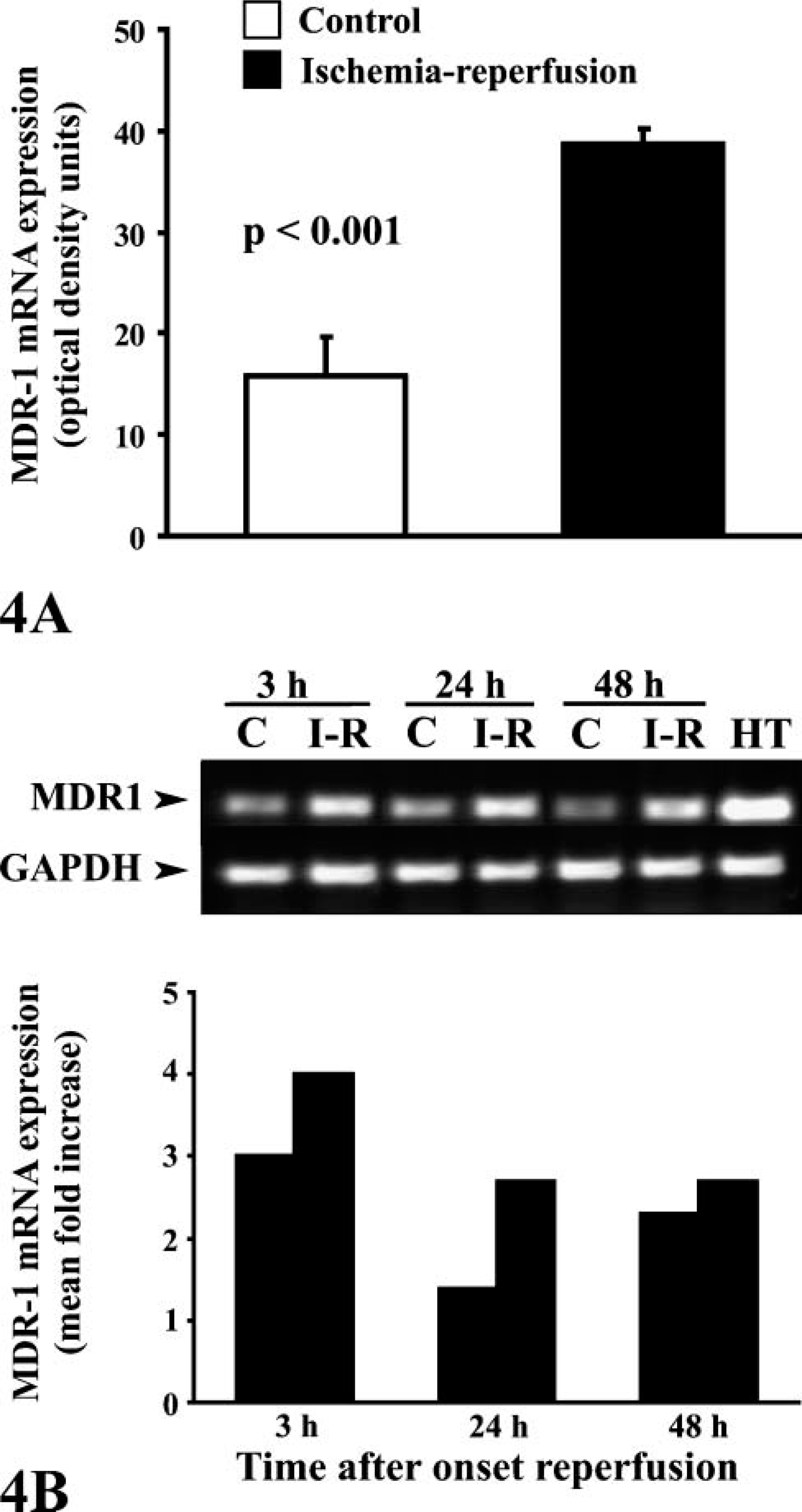
RT-PCR
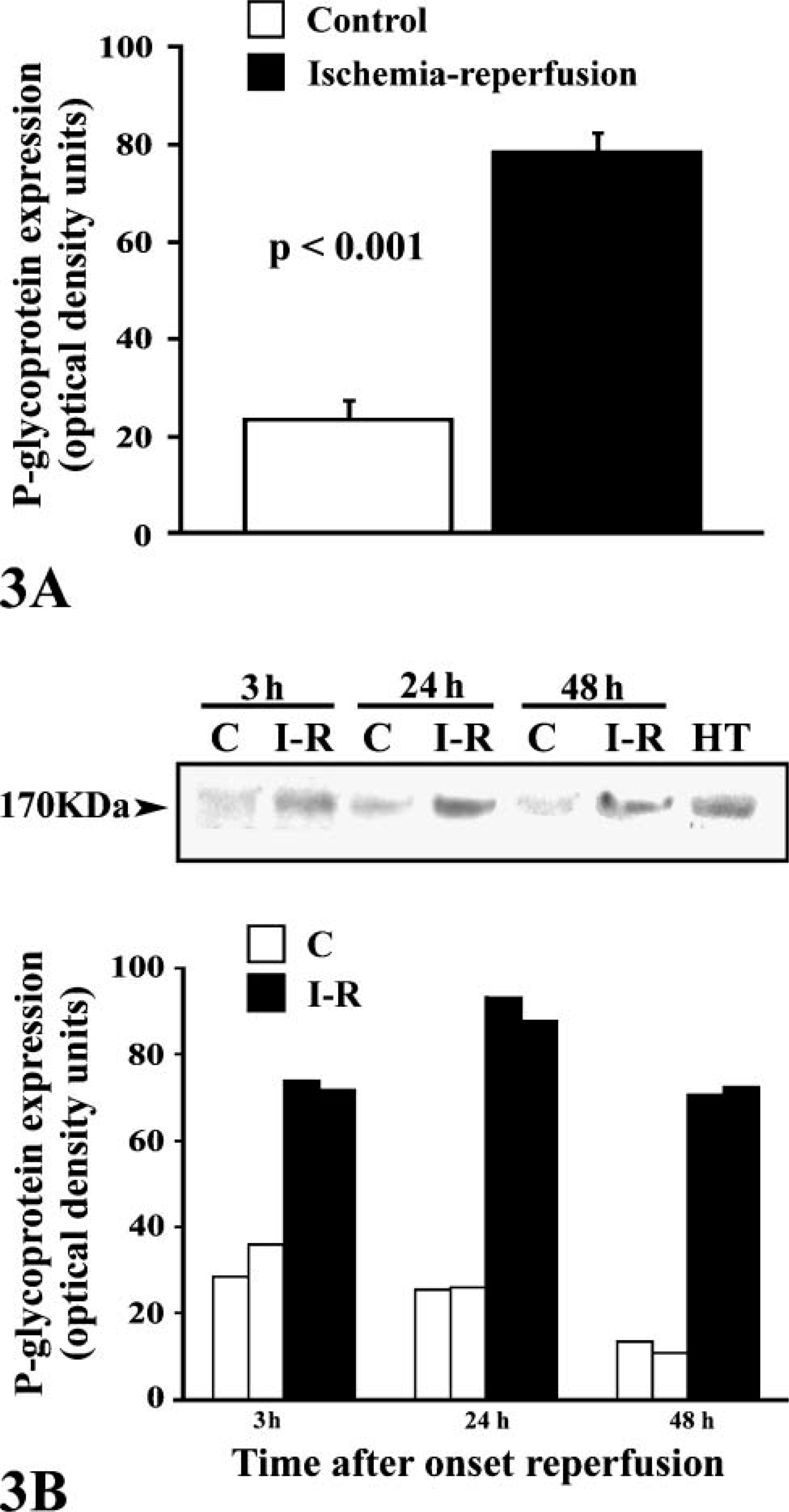
P-gp expression by Western blot analysis. (
Discussion
We have recently reported that P-gp, which is not found in myocytes of mammalian normoperfused myocardium, is expressed when cardiomyocytes are subjected to chronic ischemia (Lazarowski et al. 2005). In the present study we extend our previous observation by demonstrating for the first time that in the setting of acute, non-lethal myocardial ischemia, P-gp expression also occurs as soon as 3 hr after reperfusion and persists for at least 48 hr.
Among the variants of ischemic heart disease, ischemia-reperfusion injury is a condition increasingly observed in clinical practice, given the widespread use of primary and rescue coronary angioplasty. When reperfusion occurs early enough to prevent cell death, the resulting feature is myocardial stunning (Braunwald and Kloner 1982), a phenomenon characterized by delayed recovery of ventricular contractile function despite successful reperfusion (Kloner and Jennings 2001; Barnes and Khan 2003). The main mechanisms involved in this condition are oxidative stress with generation of free radicals, principally the hydroxyl radical (Bolli et al. 1987; Farber et al 1988) and perturbation of calcium homeostasis with intracellular free calcium overload (Bolli 1990).
Although it was not the aim of our study to determine the functional role of P-gp in this context, given that the main action of P-gp and other ATP binding cassette (ABC) transporters is to extrude a wide spectrum of cytotoxic compounds to the extracellular space (Litman et al. 2001), it is reasonable to speculate that P-gp may contribute to cardiomyocyte protection against the harmful products derived from ischemia-reperfusion.
In support of this hypothesis, it should be noted that many compounds and signal transduction pathways involved in myocardial stunning have been shown to induce expression of the
The precocity of P-gp expression seen in our model has been previously reported for human lymphocytes after PKC stimulation with TPA (Chaudhary and Roninson 1992), where the
Interestingly, P-gp was located at different regions of the plasma membrane over time. At 3 and 24 hr after reperfusion, P-gp appeared preferentially located in the T tubules, but at 48 hr it was also (and predominantly) present in the intercalated discs and gap junctions. The reasons for these translocations are difficult to explain. However, one can speculate that the preferential presence of P-gp in the T invaginations may serve to extrude dangerous substances from the close vicinity of the contractile apparatus and therefore allow for a more rapid recuperation from stunning. Following the same reasoning, in the intercalated discs P-gp may protect the gap junctions by transporting products of anaerobic metabolism from neighboring myocytes to the narrow (1 to 2 nm) intercellular space between them. Another possibility is that these translocations may be the result of lateral diffusion, a phenomenon that describes trafficking of proteins in the membrane along the lateral surface. A known example for myocyte integral membrane proteins exhibiting significant lateral diffusion is the cardiac ryanodine receptor RyR2 (Peng et al. 2004). In other cell types, glycoproteins including CFTR, an ABC transporter with molecular structure similar to that of P-gp, have been shown to display lateral diffusion (Bates et al. 2006). Therefore, it is reasonable to expect similar behavior for P-gp in the cardiac sarcolemma. Given that in isolated cardiac myocytes hypoxia has been shown to accelerate lateral diffusion (Finch et al. 1985), the translocation pattern observed in our animals may have resulted from a similar influence of the hypoxic conditions on P-gp.
The reason for the persistency of P-gp expression for a relatively long time cannot be explained with the present data, especially because of the low number of animals studied at 24 and 48 hr. However, the possibility that it may be related to the delayed phase of ischemic preconditioning could be considered. The ischemic preconditioning mechanism was described as a phenomenon by which transient myocardial ischemia protects the heart from extensive necrosis (Murry et al. 1986). The protection vanishes in a couple of hours but reappears 24 to 96 hr later, helping to protect the cardiomyocyte from a new ischemic event (Yellon and Baxter 1995). This delayed phase of ischemic preconditioning (also termed second window of protection) may be at least in part afforded by P-gp, extruding from the myocyte the toxic compounds generated by the new ischemic episode.
Finally, our finding of low levels of P-gp expression by Western blot and
In conclusion, in adult sheep undergoing acute, non-lethal myocardial ischemia followed by reperfusion, P-gp is expressed in the sarcolemma of the cardiomyocytes from 3 hr up to at least 48 hr after restoration of myocardial blood flow. Given the known role of P-gp as an extruder of cytotoxic compounds from the intracellular space, our results suggest that P-gp may afford cardioprotection in the setting of ischemia-reperfusion injury.
Footnotes
Acknowledgements
This study was supported by grants from the René G. Favaloro University Foundation.
We thank veterinarians María Inés Besansón, Pedro Iguain, and Marta Tealdo for assisting in anesthesia, and animal house personnel Juan Ocampo, Osvaldo Sosa, and Juan Carlos Mansilla for dedicated care of the animals. We also thank Julio Martínez, Juan Gauna, and Marcela Álvarez for technical help. R.P.L. and A.J.C. are established investigators of Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina.
