Abstract
Dendritic cells (DCs) are key antigen-presenting cells central to the induction of primary immune responses. Despite the prevalence of respiratory disease in sheep and the increasing use of the ovine lung as a model for human disease, ovine respiratory tract DCs (RTDCs) have not yet been characterized. Using single and double immunocytochemical staining, expression of a number of potential DC markers (MHC class II, CD1b, SIRPα, and CD205) by ovine RTDC populations has been determined. MHC class II staining revealed widespread populations of DCs either adjacent to respiratory airway epithelium or within the lung parenchyma. CD1b was expressed by a small subpopulation of both airway and parenchymal RTDCs. Expression of SIRPα was limited to a small subpopulation of airway RTDCs but was absent from the lung parenchyma. CD205 was widely expressed by airway RTDCs but expressed only by a small subpopulation of parenchymal RTDCs. In addition, the majority (87%) of parenchymal CD205+ cells exhibited a non-DC-like morphology and did not express MHC class II, suggesting that these single CD205+ cells were not DCs. Phenotypic differences between airway and parenchymal RTDCs may be related to functional differences between the two populations.
D
Despite the prevalence of ovine respiratory disease and the increasing use of the ovine lung as a model of human lung diseases including asthma (Bischof et al. 2003), bronchitis (Nikula and Green 2000), acute lung injury (Murakami et al. 2004), and neoplasia (Mornex et al. 2003), respiratory tract (RTDC) populations within the ovine lung have not been characterized. This is in contrast to the situation in the rat (Holt and Schon-Hegrad 1987; Holt et al. 1988; Schon-Hegrad et al. 1991; Gong et al. 1992), the mouse (Gonzalez-Juarrero and Orme 2001; Calder et al. 2004; von Garnier et al. 2005), and the human lung (Holt et al. 1989; Schon-Hegrad et al. 1991) in which extensive RTDC populations expressing high levels of MHC class II antigen have been identified in both the airway mucosa and lung parenchyma.
In addition to MHC class II expression, studies of ovine and bovine DC populations have identified a number of other potential RTDC markers. Expression of CD1b, a cell-surface glycoprotein thought to present lipid and glycolipid moieties to T cells (Porcelli et al. 1998), has been demonstrated in 60-90% of afferent lymph DCs (ALDCs) and DCs in the dermis and dermal-epidermal junction of the skin (Dutia and Hopkins 1991; Rhind et al. 1996; Hopkins et al. 2000; Ryan et al. 2000). SIRPα, a member of the signal regulatory protein (SIRP) family thought to mediate binding of CD4+ T cells to DCs (Brooke et al. 1998), is expressed by dermal and epidermal DCs in cattle and between 60 and 90% of bovine ALDCs and 60-75% of ovine ALDCs (McKeever et al. 1991; Howard et al. 1999; Gliddon et al. 2004; Watkins et al. 2005). Finally, expression of the cell surface protein CD205 (DEC-205), a member of the C-type multilectins thought to be an antigen uptake receptor (Gliddon et al. 2004), has been well characterized in cattle and is strongly expressed by all bovine ALDCs and by bovine intestinal, dermal, and epidermal DCs. CD205 characterization in sheep has been limited to analysis of afferent lymph in which ~65-90% of ALDCs express CD205 (Ryan et al. 2000; Watkins et al. 2005). Of these potential DC markers, CD1b and CD205 are not expressed by monocyte/macrophages, whereas SIRPα expression has previously been demonstrated in bovine monocytes (McKeever et al. 1991).
In this study we have used a combination of single and double immunocytochemical (ICC) staining to identify and characterize the expression of CD1b, SIRPα, and CD205 by MHC class II+ RTDC populations within the ovine lung. Antibodies specific for ovine CD14, an ovine monocyte/macrophage marker that is weakly expressed or absent from ovine ALDCs (Gupta et al. 1996; Ryan et al. 2000; Watkins et al. 2005), were used to differentiate DCs from macrophages. From these results, more detailed analyses of the role of RTDCs in the regulation of ovine immune responses and generation of lung pathology will be possible.
Materials and Methods
Reagents
Primary antibodies used in this study and their respective sources and dilutions are listed in Table 1. Envision Plus horseradish peroxidase for mouse immunoglobulin was purchased from DakoCytomation (HRP; Ely, UK). Alexa Fluor-568 tyramide substrate was obtained from Molecular Probes (Eugene, OR). Mouse-absorbed rabbit anti-rat IgG fluorescein-5-isothiocyanate-conjugated antibody was purchased from Vector Laboratories (FITC; Peterborough, UK), and donkey anti-rabbit IgG Alexa Fluor-488 was purchased from Molecular Probes.
Animals and Tissues
Respiratory tracts of three adult blackface ewes were sourced from Moredun Research Institute (Edinburgh, UK) at postmortem, and samples of trachea, right mainstem bronchus, and right caudodorsal lung lobes were collected. All experimental procedures involving animals were approved by The University of Edinburgh's Biological Services Ethical Review Committee and were performed under license as required by the UK's Animals (Scientific Procedures) Act 1986. Tissue samples other than lung parenchyma were cut to blocks of ~1 cm2 and embedded in optimal cutting temperature (OCT) compound (Tissue-Tek; Sakura-Finitek, Zoeterwoude, The Netherlands) before snap freezing in isopentane/dry ice. For lung parenchyma, the appropriate lung lobe was inflated with a 1:1 mixture of OCT compound and PBS before cutting into 1-cm3 blocks and freezing as previously described. Tissue sections were cut to 6-μm thick, mounted on poly-L-lysine-coated slides (BDH; Poole, UK), and stored at −80C prior to use. Representative tissue sections from each source animal were evaluated by a qualified histopathologist after staining with hematoxylin and eosin and were found to be free of pathological changes.
Single-labeling ICC Staining
Slides were removed from −80C and air dried before fixation in 100% methanol at −20C for 10 min. After washing in PBS, endogenous peroxidase was quenched by incubation in 0.3% hydrogen peroxide in methanol for 20 min at room temperature. Slides were then washed in PBS before loading into a Sequenza immunostaining center (Shandon; Runcorn, UK). Nonspecific tissue antigens were blocked by incubation in 10% normal goat serum in PBS/0.5% Tween 80 (PBS/T80) for 1 hr at room temperature prior to incubation with the primary antibodies overnight at 4C. Controls were provided by replacing the primary antibody with an IgG fraction from normal mouse or rat serum at 2 μg/ml as appropriate. All monoclonal antibodies (MAbs) and controls were diluted in PBS/T80 containing 10% normal goat serum.
Details of monoclonal antibodies (MAbs) used in single and double immunocytochemical (ICC) staining

Kindly provided by Prof. John Hopkins (University of Edinburg, Edinburgh, UK).
Purchased from Serotec Ltd; Oxford, UK.
Kindly provided by Ian Anderson (Moredum Research Institute, Midlothian, UK). CD, cluster designation; DC, dendritic cell; MHC II, major histocompatability complex class II; RT, room temperature.
Slides were washed twice in PBS and secondary antibody (peroxidase-labeled polymer conjugated to goat anti-mouse immunoglobulins, included in the EnVision system kit) was applied to sections for 30 min at room temperature. After a final PBS wash, sections were incubated with the 3,3′-diaminobenzidine (DAB) for 7.5 min at room temperature, washed in distilled water, counterstained with hematoxylin, dehydrated in graded alcohols, cleared, and mounted in DPX mounting medium (Fischer Scientific; Loughborough, UK).
Dual-labeling Immunocytochemical Staining
Tissue sections were fixed, blocked, and incubated with the mouse MAbs CC98, ILA-24, and CC20 as described in the previous sections. After overnight incubation at 4C, sections were washed twice in PBS and secondary antibody (peroxidase-labeled polymer conjugated to goat anti-mouse immunoglobulins; DakoCytomation) was applied to sections for 30 min at room temperature. Slides were washed in PBS and incubated for 10 min with Alexa Fluor-568 tyramide (Molecular Probes).
Sections were then blocked for 30 min in 10% normal mouse serum in PBS/T80 before incubation with the second primary antibody, rat MAb SW 73.2, for 1 hr at room temperature. After PBS washing, slides were incubated for 1 hr at room temperature in 2 μg/ml of mouse-adsorbed rabbit anti-rat IgG FITC-conjugated antibody, washed, and then incubated for 1 hr at room temperature in 2 μg/ml donkey anti-rabbit IgG Alexa Fluor-488. Sections were mounted in Mowiol mounting medium (Calbiochem-Novabiochem; San Diego, CA) and allowed to set for ≥1 hr at room temperature before microscopic analysis. Controls were provided by replacing the primary antibody with an IgG fraction from normal mouse or rat serum at 2 μg/ml as appropriate. All MAbs and controls were diluted in PBS/T80 containing 10% normal goat serum for first primary antibody detection and 10% normal mouse serum for second primary antibody detection.
Image Acquisition and Analysis
Brightfield images were acquired with a Sony DXC-390P 3CCD color video camera (Scion Corporation; Frederick, MD) mounted on an Axiovert 100 inverted microscope (Carl Zeiss; Welwyn Garden City, UK). The RGB video signal from the camera was digitized using Scion Image (Scion Corporation) installed in a G4 Macintosh computer (Apple Computer; Cupertino, CA) fitted with a CG-7 frame grabber (Scion Corporation).
Confocal images were acquired using an MRC-600 confocal laser-scanning microscope (CLSM; Bio-Rad Laboratories, Hemel Hempstead, UK) mounted on an Axiovert 100 inverted microscope equipped with Plan-Apochromat objective lenses (Carl Zeiss). Fluorophores were excited and imaged sequentially using the 488-nm (FITC) and 568-nm (Alexa Fluor-568) lines from a 15-mW Kr/Ar laser (Bio-Rad Laboratories).
Images were prepared using Object-Image (Vischer et al. 1994) and Photoshop (Adobe Systems; Uxbridge, UK). Object-Image is a public domain software package based on NIH Image (Rasband and Bright 1995), developed by Norbert Vischer (The University of Amsterdam, Amsterdam, The Netherlands), and is available via the Internet at http://simon.bio.uva.nl/object-image.html.
Manual cell counting, measurements of epithelial length, and stereology (Cavalieri method) (Gundersen and Jensen 1987; Gundersen et al. 1988) were performed on images of DAB-stained sections as appropriate, using custom software developed for Object-Image.
Single-labeled tissue sections were analyzed as follows: to determine the number of positive cells adjacent to the epithelium of the trachea and bronchi, positive cells were counted in 10 × 40 fields/slide. For cell counts adjacent to bronchioles, the number of positive cells adjacent to five end-on bronchioles/slide was counted. For lung parenchymal cell counts, the number of positive cells in ten random × 40 fields was assessed. Cell counts were expressed as the number of cells/mm epithelium for airways, and the number of positive cells/mm2 lung tissue for lung parenchyma (determined by stereology). Statistical analysis of single-labeled cell counts was performed using a paired t-test. Double-labeled tissue sections were analyzed using an MPV II fluorescence microscope (Leitz; Wetzlar, Germany). Five hundred fluorescently labeled MHC class II+ cells were counted per section and the number of single and double positive cells recorded. From this, the percentage of CD1b+, SIRPα+, and CD205+ cells coexpressing MHC class II, and the percentage of the total number of MHC class II+ cells coexpressing each DC marker were calculated.
Results
Single Immunocytochemical (ICC) Staining of DC Markers in Normal Ovine Lower Respiratory Tract Tissue
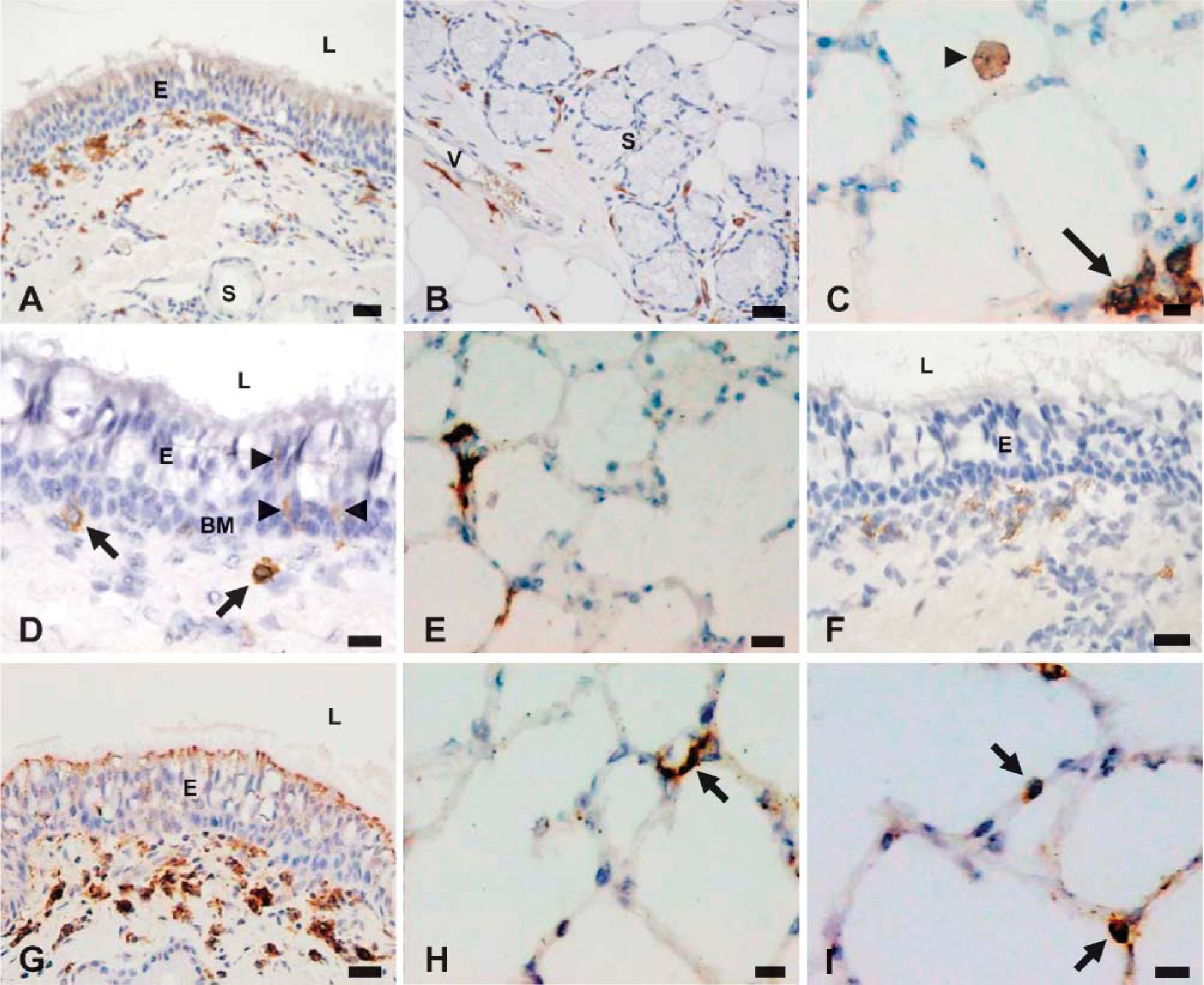
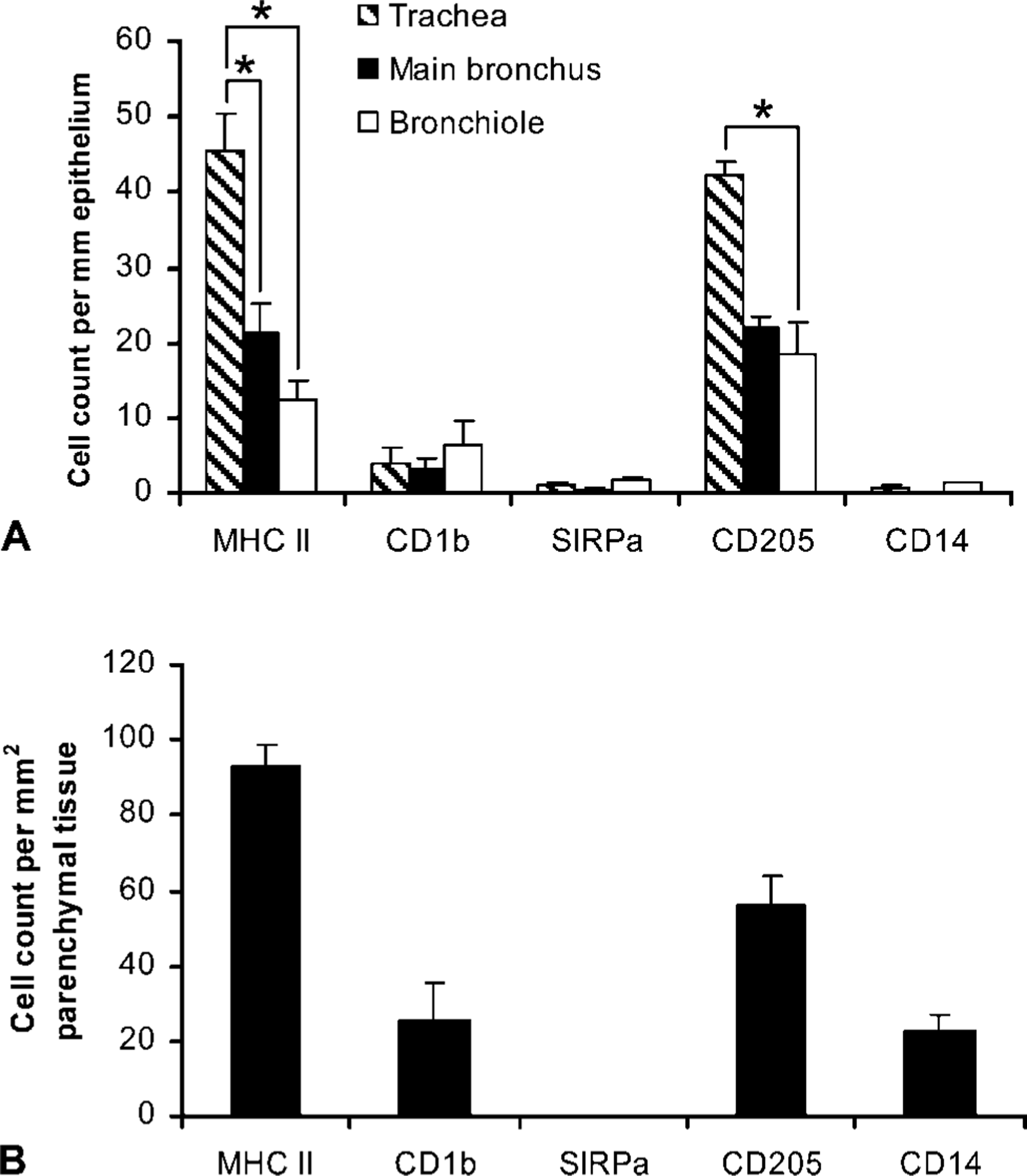
Samples of trachea, right mainstem bronchi, and right caudodorsal lung lobes from three adult sheep were subjected to single ICC staining for the DC markers MHC class II, CD205, SIRPα, and CD1b. Representative images of stained tissue sections are shown in Figure 1. The numbers of positively stained cells adjacent to airways and within the lung parenchyma are shown in Figures 2A and 2B, respectively.
Widespread labeling of MHC class II was observed throughout the respiratory tract. Two separate populations of cells were discernible adjacent to respiratory tract airways. The first was comprised of strongly positive cells spaced at relatively regular intervals along the airway epithelium (Figure 1A). These cells often appeared to be closely associated with the epithelial basement membrane occasionally penetrating the membrane and were generally large with irregular morphology. A second population of highly pleomorphic MHC class II+ cells was observed in the underlying mucosa, generally adjacent to submucosal glands and blood vessels (Figure 1B). Occasional small aggregations of positive cells were present in the submucosa of trachea and bronchi. Larger aggregations were present adjacent to bronchioles. There was a sequential reduction in epithelial MHC class II+ cells from larger to smaller airways, with significantly lower numbers of positive cells in bronchi compared with trachea (p≤0.05) and significantly lower numbers of positive cells adjacent to bronchioles compared with main bronchi (p≤0.05) (Figure 2A).
Single immunocytochemical (ICC) staining of ovine respiratory tract for MHC class II (
Two distinct populations of MHC class II+ cells were identified within the lung parenchyma (Figure 1C). The first consisted of highly pleomorphic, generally strongly staining MHC class II+ cells within the alveolar septal walls. A large number of these cells exhibited a flattened and irregular morphology and were often seen spanning across two adjacent alveoli. The second population consisted of large vacuolated weakly positive cells found attached to the air side of the alveolar wall or within the alveolar airspace. In addition, occasional aggregations of MHC class II+ cells were found within the parenchyma, often adjacent to small blood vessels. Parenchymal MHC class II+ cells were present at a density of ~90 cells/mm2 lung tissue (Figure 2B).
Distribution of DC markers in the ovine airways (
CD1b staining identified low numbers of moderately positive cells throughout the respiratory airways, either in a subepithelial location occasionally penetrating the epithelial basement membrane (Figure 1D) or adjacent to submucosal glands and small blood vessels. These cells were generally large with irregular cell membranes. Occasional aggregations of CD1b+ cells were present in the submucosa of trachea and bronchi or adjacent to bronchioles and small vessels. No significant differences in the numbers of CD1b-positive cells were seen among trachea, bronchi, and lower airways (Figure 2A).
In lung parenchyma, CD1b staining identified a population of large strongly staining cells with a flattened and irregular morphology within the alveolar septal walls, often spanning across two adjacent alveoli (Figure 1E) and present at a density of ~25 cells/mm2 lung tissue (Figure 2B).
SIRPα+ cells were found throughout the respiratory tract airways in low numbers and consisted of large, generally weakly staining cells with irregular morphology, primarily in a subepithelial location (Figure 1F). Occasional small aggregations of SIRPα+ cells were present in the submucosa of trachea and bronchi. Larger aggregations were present adjacent to bronchioles and often exhibited typical organized lymphoid tissue structure with distinct cortical and follicular areas. SIRPα staining was restricted to cortical areas and absent from follicular areas. No significant differences in SIRPα+ cell counts were observed among trachea, main bronchus, and lower airway (Figure 2A). No SIRPα+ cells were identified within the lung parenchyma (Figure 2B).
CD205 staining was widespread throughout the whole respiratory tract and identified two distinct populations of cells in respiratory airways in a similar distribution to MHC class II staining. The first comprised strongly positive cells spaced at relatively regular intervals along the respiratory epithelium with large and irregular morphology (Figure 1G). A second population of highly pleomorphic CD205+ cells was also observed in the underlying mucosa, generally adjacent to submucosal glands and blood vessels. Occasional small aggregations of CD205+ cells were present in the submucosa of trachea and bronchi. Larger aggregations were located adjacent to bronchioles and often exhibited a degree of organized lymphoid tissue structure with cortical and follicular areas. CD205 staining appeared to be concentrated in follicular areas, although scattered CD205+ cells were also seen in cortical areas. This follicular staining pattern was confirmed by analysis of ovine mediastinal lymph node sections for CD205. Positive cells were concentrated within lymphoid follicles, with more scattered CD205+ cells within the cortex (data not shown). There were significantly lower numbers of positive cells adjacent to bronchioles compared with trachea (p>0.05). However, no significant differences in CD205+ cell numbers were observed between trachea and main bronchi (Figure 2A).
Within the lung parenchyma, two distinct populations of CD205+ cells were identified. The first consisted of large strongly stained cells with a flattened and irregular morphology present within the alveolar septal walls, often spanning across two adjacent alveoli, and was the least abundant (Figure 1H). The second and more numerous population consisted of strongly stained cells located within the alveolar septal walls with a smaller, rounded morphology (Figure 1I). Lung parenchymal CD205+ cells were present at a density of ~60 cells/mm2 lung tissue (Figure 2B).
CD14+ cells were identified infrequently throughout the respiratory tract airways, either in a subepithelial location or adjacent to submucosal glands and blood vessels and exhibited a large and rounded morphology (data not shown). No significant differences in the numbers of CD14+ cells were seen among trachea, bronchi, and lower airways (Figure 2A). Within the lung parenchyma two separate populations of CD14+ cells were present. The first population consisted of large, rounded, and vacuolated cells found attached to the air side of the alveolar wall or within the alveolar airspace. The second population consisted of cells with irregular morphology within the alveolar septal wall (data not shown). Lung parenchymal CD14+ cells were present at a density of ~20 cells/mm2 lung tissue (Figure 2B).
Double ICC Staining of DC Markers in Normal Ovine Lower Respiratory Tract Tissue
Single ICC staining identified populations of MHC class II+ cells with DC-like morphology either associated with the respiratory epithelium or within the alveolar septal walls. In addition, staining of a number of DC markers (CD1b, SIRPα, and CD205) was observed in a similar location to MHC class II+ cell populations. To further characterize ovine RTDCs, a double-staining protocol was employed in which potential DC markers were costained with MHC class II. The percentage colocalization for each potential DC marker and the percentages of the total number of MHC class II+ cells coexpressing each DC marker were calculated and are shown in Table 2. Representative images of double ICC staining are shown in Figure 3.
CD1b was expressed on a small subpopulation (1.68.4%) of MHC class II+ cells throughout the respiratory tract, with the percentage of MHC class II+ cells expressing CD1b increasing from trachea to lower lung; 88-95% of CD1b+ cells in the lower airways and lung parenchyma expressed MHC class II (Figure 3A). However, in the tracheal mucosa, ~40% of CD1b+ cells did not express MHC class II. Expression of SIRPα was limited to a small subpopulation (2.3-4.4%) of MHC class II+ cells in airways and decreased from trachea to lower airways. All SIRPα+ cells also expressed MHC class II (Figure 3D). CD205 was widely expressed by MHC class II+ cells in all airways, with ~60% of MHC class II+ cells coexpressing CD205 in these locations. Of CD205+ cells in this location, 97-100% were MHC class II+ (Figure 3G). However, in lung parenchyma, CD205 was expressed by <10% of MHC class II+ cells, and the majority (87%) of parenchymal CD205+ cells did not express MHC class II.
Discussion
MHC class II staining of ovine respiratory tract revealed widespread populations of airway and parenchymal DC consistent with those seen in other species (Holt and Schon-Hegrad 1987; Holt et al. 1989; Gonzalez-Juarrero and Orme 2001). The number of epithelial DCs decreased from upper to lower airways. This is in agreement with previous studies of rat airway DCs and may reflect increased exposure of upper airways to inhaled antigens (Schon-Hegrad et al. 1991). Interestingly, type II pneumocytes, which have been reported to express MHC class II antigen in humans (Peters et al. 1990; Cunningham et al. 1994) and rats (Steiniger and Sickel 1992), did not stain for MHC class II antigen in this study. MHC class II+ cells within the alveolar septal walls were primarily flattened in morphology and entirely located within the alveolar septal wall, whereas type II pneumocytes are rounded in shape and generally protrude into the alveolar airspace.
Evaluation of a number of potential DC markers revealed that the majority of airway epithelial DCs express CD205, whereas expression of CD1b and SIRPα was limited to smaller subsets of epithelial DC. CD205 expression was almost exclusively restricted to MHC class II+ cells, consistent with the finding that the number of CD205+ cells decreased from upper to lower airways, mirroring MHC class II expression. SIRPα was also restricted to MHC class II+ cells with DC morphology and appeared to be a specific DC marker, although only weakly expressed. Identification of macrophage populations using CD14 staining revealed a distinct population of cells in low abundance and with non-DC-like morphology adjacent to airways, indicating that epithelial DCs identified in this study were not macrophages.
CD1b expression was restricted to MHC class II+ cells in lower airways, but in the trachea 40% of CD1b+ cells did not express MHC class II. These single CD1b+ cells were smaller and more rounded than CD1b+/MHC class II+ cells. CD1b expression in sheep has previously been thought to be a specific DC marker in tissues outside the thymus (Dutia and Hopkins 1991; Rhind et al. 2000), although previous studies have employed only single-staining techniques. As MHC class II expression is a hallmark of DC, CD1b+/MHC class II- cells do not appear to be DCs. Cortical thymocytes are known expressors of CD1b in a number of species including sheep (Dutia and Hopkins 1991; Hopkins et al. 2000), cattle (Howard and Naessens 1993a), and humans (Martin et al. 1987). Given the lack of MHC class II expression and the small rounded morphology, CD1b+/MHC class II- cells may represent a subpopulation of lymphocytes. Indeed, single CD1b+ cells were found within airway epithelium, a known location for lymphocytes within the ovine respiratory tract (Chen et al. 1989). Costaining of lymphocyte markers with CD1b would be required to confirm this hypothesis.
Double ICC staining of potential DC markers and MHC class II a

Double ICC staining of potential DC markers with MHC class II antigen was performed in the trachea, lower airway, and lung parenchyma of three normal sheep.
Values expressed are the mean percentages of MHC class II+ cells expressing CD1b, SIRPα, and CD205 ± SE of the mean.
Values expressed are the mean percentages of CD1b+, SIRPα+, and CD205+ cells expressing MHC class II ± SE of the mean. NA, not applicable.
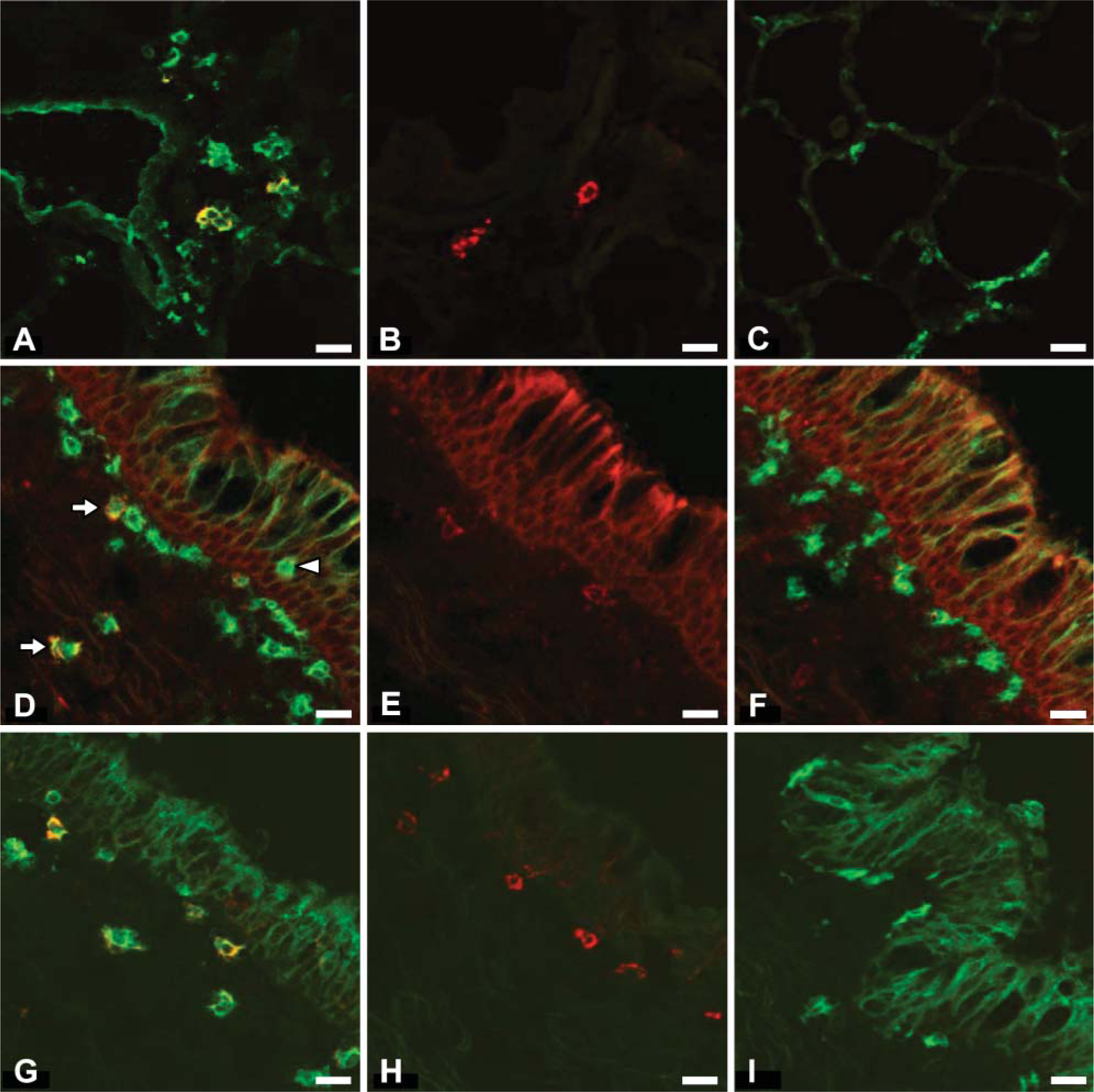
Double ICC staining of ovine respiratory tract (RT) DCs. (
The proportion of airway RTDCs expressing CD205, CD1b, and SIRPα is consistent with previous analyses of ALDCs in cattle and sheep: CD205 is expressed by the majority of ALDCs, whereas CD1b and SIRPα expression is limited to smaller subsets of ALDCs (McKeever et al. 1991; Howard et al. 1997; Brooke et al. 1998; Ryan et al. 2000). However, the proportion of RTDCs expressing CD1b and SIRPα was found to be much lower than reported in ovine and bovine ALDCs. There are a number of possible explanations for this. First, it is likely that CD1b+ and SIRPα+ cells within tissue are an immature phenotype because it is widely thought that DCs in peripheral tissues represent immature cells, with maturation occurring during their migration from the peripheral tissues to the draining lymph nodes (Banchereau et al. 2000). Expression of both proteins may increase with DC maturation and migration from respiratory tract tissue into draining lymphatics. Second, the source of ALDCs in previous studies was afferent lymph draining from the skin into prefemoral or prescapular lymph nodes and, therefore, represent migrating populations of skin DCs. Therefore, the low expression of CD1b and SIRPα by RTDC populations found in this study may reflect innate differences in expression of CD1b and SIRPα between skin and airway DCs. Indeed, comparison of ovine ALDC populations originating from either head mucosae or skin identified higher proportions of SIRPα- within ALDCs migrating from head mucosal surfaces, indicating site-specific expression of SIRPα (Epardaud et al. 2004). Finally, ALDCs are studied primarily using flow cytometry, a technique with potential differences in sensitivity to the standard ICC methods used in this study. These potential differences in sensitivity may contribute to the observed differences in the expression of CD1b and SIRPα between RTDC and ALDC populations.
It is unclear whether CD205+, CD1b+, and SIRPα+ DC represent separate DC populations or are coexpressed. However, ALDC analyses in sheep and cattle would suggest that SIRPα+ and CD1b+ cell coexpress CD205. In one study of sheep afferent lymph, 60-70% and 65-80% of ALDCs were found to express CD1b and CD205, respectively (Ryan et al. 2000), indicating that a proportion of these cells coexpressed both CD205 and CD1b. In addition, up to 75% of CD205+ ovine ALDCs have been shown to coexpress SIRPα (Watkins et al. 2005). Analysis of cattle ALDCs demonstrates widespread CD205 expression, with a subset of these cells expressing SIRPα (Brooke et al. 1998; Howard et al. 1999) and CD1b (Howard et al. 1997). Double staining for CD205, CD1b, and SIRPα would be required to clarify this situation.
CD1b expression by parenchymal DCs was similar to that seen with lower airway DCs, with expression of CD1b being highly restricted to MHC class II+ cells and present in a small subpopulation of DCs. However, expression of SIRPα and CD205 by parenchymal DCs was different from that observed by airway DCs: parenchymal DCs did not express SIRPα, and CD205 was expressed only by a small subpopulation of DCs. It must be noted that MHC class II+ cell populations were more pleomorphic in the lung parenchyma compared with airways, most likely due to staining of other MHC class II-expressing cell types, e.g., B cells, activated T cells, and macrophages (Puri et al. 1987). Therefore, percentages of DCs expressing various DC markers in parenchymal tissue are likely to be underestimated. Unlike the situation in airways where CD205 expression is highly restricted to MHC class II+ cells, CD205 was expressed by a large population of MHC class II- cells in the parenchyma with a small, rounded non-DC-like morphology. CD205 expression has been previously described in γ/δ T cells and B cells (Howard and Naessens 1993a), and it is possible these CD205+/MHC class II- cells represent parenchymal T- or B-cell populations, although B cells would be expected to coexpress MHC class II. In addition, CD205 staining was concentrated in follicular-like structures within cellular aggregations adjacent to bronchioles. These positive cells are most probably follicular B cells, as CD205 has been shown to be expressed by these cells in cattle (Howard and Naessens 1993a; Gliddon et al. 2004), and specific staining of follicular areas within ovine mediastinal lymph node was observed in this study. Therefore, it appears that CD205 is not a highly specific marker for DC populations in the lower lung.
Staining for CD14 identified two populations of macrophages within the lung parenchyma. The first population consisted of cells within the alveolar airspace with characteristic alveolar macrophage morphology, whereas the second population was located within the alveolar septal wall and exhibited a more flattened morphology. This second population of interstitial macrophages exhibited a similar morphology to parenchymal CD1b+ cells and a subset of parenchymal CD205+ cells identified in this study. Therefore, it is possible that CD1b+ and a subpopulation of CD205+ cells within the lung parenchyma represent a population of interstitial macrophages and are not parenchymal DCs. However, given that a number of previous studies have failed to identify CD1b and CD205 expression in ruminant monocyte/macrophages (Dutia and Hopkins 1991; Howard and Naessens 1993a; Rhind et al. 2000; Gliddon et al. 2004), this would appear unlikely. Double staining with CD14 and CD1b/CD205 would be required to confirm this.
Differences in expression of DC markers between airway and parenchymal DC populations may represent two distinct populations of DCs or may represent DCs at different stages of maturation. In either case, it may be that differences in expression may relate to functional differences between the two DC populations. It has been shown that purified parenchymal and airway DCs differ in their antigen presentation capabilities and turnover rates in the rat (Gong et al. 1992; Holt and Stumbles 2000) and the mouse (von Garnier et al. 2005), with antigen presentation and turnover more rapid in airway DC populations. Interestingly, a number of studies have demonstrated functional differences between SIRPα+ and SIRPα- populations originating from the same organ. SIRPα+ ALDCs draining from the skin in cattle has been shown to be more efficient at stimulating T-cell proliferation and antigen presentation compared with SIRPα- ALDCs (Howard et al. 1997), and these two subpopulations of bovine ALDCs have also been shown to differ in their cytokine expression repertoires (Stephens et al. 2003). This is consistent with studies of migrating rat intestinal DCs (iDCs), which found that SIRPα+ iDCs were more immunostimulatory than SIRPα- iDCs (Yrlid and Macpherson 2003). Observations that SIRPα- populations draining from the rat intestine and the ovine head mucosae contain apoptotic material have also lead to the suggestion that SIRPα- subpopulations are involved in self-antigen presentation and tolerance induction (Epardaud et al. 2004; Turnbull et al. 2005). Therefore, the possibility arises that ovine airway DC populations that express limited amounts of SIRPα are more immunostimulatory, and parenchymal cells that are SIRPα- are more immunosuppressive. Studies of purified ovine DCs from airway and parenchymal locations would be required to better characterize these two populations.
In conclusion, the ovine respiratory tract contains a widespread population of DCs, within either a parenchymal or airway location. Expression of CD205, CD1b, and SIRPα differed between airway and parenchymal DC populations: CD205 was expressed by the majority of airway DCs but only by a small subpopulation of parenchymal DCs, CD1b was expressed by small subpopulations of both airway and parenchymal DCs, and SIRPα was expressed by a small subpopulation of airway DCs but not by parenchymal DCs.
Footnotes
Acknowledgements
T.N.M. was supported by a PhD studentship from the Royal (Dick) College of Veterinary Studies, University of Edinburgh, Edinburgh, UK and funding from the European Union (contract no. QLK2-CT-2002-00167).
We thank Jim Williams (Moredun Research Institute) for supplying sheep lung tissue, Prof. John Hopkins (University of Edinburgh) for invaluable advice and support throughout this study, and Dr. Mike Wilkinson (GlaxoSmithKline) for the donation of the Bio-Rad MRC600 confocal microscope.
