Abstract
The aim of this study was to evaluate the adverse effects of cyclosporine A (CsA) toward renal morphogenesis and to test the renoprotective natural antioxidants such as provinol (PV). Pregnant rats were divided into four groups. Group I was injected SC with olive oil. Group II was treated with oral administration of PV and was used as control. Group III animals were injected SC daily with CsA, and group IV animals were injected daily with CsA and PV for 21 days of pregnancy. Five pups per litter were killed and the kidneys removed and treated by morphological and immunohistochemical (IHC) methods. IHC analysis considered two proteins responsible for nephrotoxicity in adult rats: inducible nitric oxide (iNOS) and matrix metalloproteinase-2 (MMP2). Pregnancy outcomes among CsA-treated rats demonstrated a reduced number of pups. Pups that were exposed antenatally to CsA presented several pathologic findings in all immature parenchyma and an increase in iNOS and MMP2 expression. These side effects were not observed in kidney of litters born from CsA + PV-treated mothers. Our study indicates that CsA induces morphological alterations in renal parenchyma of neonates and that PV plays a protective role against these side effects.
F
However, experience regarding CsA use in human pregnancy is limited, and no final conclusion may be drawn with regard to the harmlessness or other effects of the drug. Experimental studies have instead resulted in further concern and suggested that (a) in rat, CsA induces fetal growth retardation in organ morphogenesis (Rezzani et al. 1997) and produced alterations in T-cell maturation with suppression of lymphoproliferative responsiveness to mitogen activation (Padgett and Seelig 2002); and (b) in rabbit, CsA determines an impaired nephron differentiation (Papaccio and Esposito 1990), a permanent nephron deficit inducing systemic hypertension, and progressive renal insufficiency (Tendron et al. 2003; Armenti 2004; Tendron-Franzin et al. 2004). In particular, Rezzani et al. (1997) demonstrated that rats born from treated mothers showed temporary defects in the development of some hemopoietic organs (thymus, spleen, liver, and bone marrow). Tendron-Franzin et al. (2004) showed that CsA exposure in rabbit induced a vacuolization of proximal tubular cells and a number of defects in nephrogenesis.
The main aims of the current study were to better address morphological alterations in renal morphogenesis of the litters from CsA-treated mothers and also the possible protective role of antioxidants such as polyphenols against these negative effects. To help to address these questions we studied the activity and expression of two proteins responsible for CsA nephrotoxicity in adult rats: inducible nitric oxide synthase (iNOS, NOS2) (Rezzani et al. 2003; Buffoli et al. 2005) and matrix metalloproteinase-2 (MMP2) (Eberhardt et al. 2000; Khanna et al. 2002).
In particular, polyphenols are common constituents of the human diet present in most foods and beverages of plant origin (Manach et al. 2005), and the beneficial effects of fruits, vegetables, or red wine may, in part, be explained by the presence of these substances having a multitude of biological activities including antioxidant and free-radical-scavenging properties and inhibition property of vascular smooth muscle cell proliferation (Middleton et al. 2000). Recently, we showed that administration of provinol (PV), a polyphenol obtained from red wine, protects against nephrotoxicity during CsA treatment in adult rats (Buffoli et al. 2005).
To our knowledge, there are no data regarding the protective role of PV against adverse renal effects on litters from CsA-treated mothers. In our study, we sought also to demonstrate that PV is a useful antioxidant in the hope that this could provide clues regarding the development of therapeutic strategies during pregnancy and immunosuppressive treatment.
Materials and Methods
Animals and Experimental Treatments
Protocols were approved by the Italian Ministry of Health and complied with Guiding Principles in the Use of Animals in Toxicology.
Ninety-day-old virgin female and male Wistar rats weighing 250-300 g were used. Animals were housed in cages under controlled temperature with a 12-hr light/12-hr dark schedule and access to food and water ad libitum. Female animals were checked every morning, and when pregnancy was assumed with a vaginal plug (Rezzani et al. 1997) in 32 rats, they were removed, kept in separate cages, and divided into four groups of eight rats each. These groups were treated during pregnancy from day 0 to day 21. In particular, group I was injected SC with olive oil, the CsA vehicle; group II was treated with oral administration of PV (40 mg/kg/day; Société Francaise de Distillerie, Vallont Pont d'Arc, France) according to Buffoli et al. (2005). These animals were used as control groups. Group III animals were injected SC daily with CsA (15 mg/kg/day; Sandoz, Basel, Switzerland), and group IV animals were injected daily with CsA and PV for 21 days of pregnancy.
The end of treatments corresponded to spontaneous delivery for all groups; pups were weighed within 2 or 3 hr after birth. Five pups per litter were randomly chosen and killed using terminal ether anesthesia. Kidneys were removed and washed briefly in phosphate buffer (0.1 M, pH 7.4). A median slice was quickly cut from each organ (parallel to the short axis of the kidney). One slice was frozen at −20C, another was quickly frozen and powdered in liquid nitrogen, and the remaining slice was immediately fixed in 4% paraformaldehyde prior to embedding in paraffin wax according to standard procedures.
Morphological and Immunohistochemical Analysis
Paraffin-embedded slices were cut at 5 μm on a standard microtome. Cross-sections included the full thickness of the cortex and medulla and were serially used for histopathological [hematoxylin-eosin (H&E)] and immunohistochemical (IHC) evaluation.
Before IHC analysis, sections were deparaffinized, rehydrated, and then immersed in 3% hydrogen peroxide (H2O2) in methanol for 30 min to block endogenous peroxidase activity.
Sections were incubated for 1 hr at room temperature in normal goat serum for iNOS and in normal rabbit serum for MMP2 (diluted 1:5; Dakopatts, Milan, Italy) and serially treated with rabbit polyclonal antibody anti-iNOS and goat polyclonal antibody anti-MMP2 (diluted 1:100; Santa Cruz Biotechnology, Santa Cruz, CA) at 4C overnight. Successively, sections were washed in TBS 0.1 M, pH 7.4, and then incubated with a biotinylated secondary antibody (goat anti-rabbit for iNOS and rabbit anti-goat for MMP2, diluted 1:50; Dakopatts) for 60 min and then with the avidin-biotin horseradish peroxidase complex (ABC kit; Dakopatts) for an additional 1 hr. Sections were then immersed in a solution of 0.05% 3,3-diamino-benzidine tetrahydrochloride (DAB) and 0.03% H2O2. Finally, sections were counterstained with hematoxylin, dehydrated, and mounted. Control reactions were performed in the absence of primary antibody and in the presence of isotype-matched IgGs.
Stereological Analysis for Glomerular Volume and Size
Renal sections cut at 5 μm on a standard microtome and stained with H&E were analyzed according to Pereira et al. (2002). Briefly, an estimate of volume-weighted mean glomerular volume (VWGV) was made using the point-sampled intercepts method. Five microscopic fields were analyzed per section, three sections per kidney, and five animals per group (75 fields per group). A test system consisting of parallel lines associated with test points was superposed on each field. The direction of the lines on the sample was determined by lottery. For each point inside the unbiased counting frame, which hits a glomerulus intercept through the point, measurement of the intercept length was performed using a 32-mm-long logarithmic rule composed of a series of 15 classes, where width of any class is ~17% larger than that of the preceding class. Each individual intercept was cubed, and the mean of all values was multiplied by p/3 in every case to obtain VWGV. Differences among groups were tested by statistical analysis.
For glomerular size (GS), ten randomly selected fields with a known area (0.15 mm2/field) were analyzed for each section (five sections and five animals per group) by a blinded observer. Size of the glomeruli were calculated in the selected fields according to Lucas et al. (1997). Briefly, the average GS was evaluated by means of a graduated eyepiece, each value being the arithmetic mean of two measurements obtained in two perpendicular axes. These data have been studied by statistical analysis.
Quantitative Analysis of Swollen Tubules
The sections cut at 5 μm on a standard microtome, including the cortical and medullary regions, were stained with H&E. They were evaluated for swelling of epithelial cells in proximal and distal tubules, which were identified according to Rezzani et al. (2005). Briefly, proximal tubules were differentiated from distal tubules as follows: proximal tubules had larger diameters than distal tubules. The nuclei of proximal tubule epithelial cells were usually arrayed at the base of the cells. Tubular swelling is identified as detachment or loss of epithelial cells according to histopathological analysis (Bush et al. 1999). Under a light microscope (X1000), ten randomly selected fields (0.04 mm2/field) were analyzed for each section (five sections, five animals per group), and the results were statistically evaluated.
Quantitative iNOS and MMP2 Analysis
For quantitative analysis, the 5-μm-thick sections including cortical and medullary region were evaluated at X100 magnification using an optical microscope (BX50; Olympus, Hamburg, Germany) equipped with an image analyzer (Image Pro Plus; Milan, Italy). The integrated optical density was calculated for arbitrary areas, measuring 10 fields with the same area for each sample. Data were pooled to represent a mean value, and a statistical analysis was applied to compare the results obtained from the different experimental groups.
Immunoblotting Analysis for iNOS and MMP2 Proteins
Samples of kidneys (80 mg of wet tissue) were homogenized in 25 mmol/liter Tris-HCl, pH 7.4, containing 5 mmol/liter EDTA, 50 mmol/liter NaCl, 1 μmol/liter leupeptin, 0.3 μmol/liter aprotinin, 0.1 mmol/liter PMSF, 1 mmol/liter pepstatin, and 1% SDS. After centrifugation (15,000 × g, 20 min, twice) supernatants all containing the same amount of protein (100 μg) were analyzed in SDS-PAGE 10% gels and electrotransferred to a nitrocellulose membrane (pore size 0.45 μm; BioRad, Hemel Hempstead, UK) by wet blotting (100 V for 1 hr). The membrane was blocked with 5% non-fat dry milk in Tris-buffered saline Tween-20 (TTBS) at 4C. After washing with TTBS, proteins were exposed overnight at 4C to a polyclonal rabbit anti-iNOS antibody and a mouse monoclonal anti-MMP2 (both from Santa Cruz).
These were detected using secondary biotinylated anti-rabbit secondary antibody (Dakopatts) for iNOS and a biotinylated goat anti-mouse secondary antibody for MMP2 (Dakopatts) as appropriate and an avidin-peroxidase complex according to the manufacturer's instructions (ABC kit; Dakopatts) with a solution of 0.05% DAB (3,3-diaminobenzedine tetrahydrochloride) and 0.03% hydrogen peroxide. Bands were evaluated with an image analyzer (Image Pro Plus) and statistically analyzed.
MMP2 Zymographic Analysis
MMP2 activity was detected by gelatinase zymography (Rawdanowicz et al. 1994) on slices that were powdered in liquid nitrogen. The powders were resuspended in ice-cold lysis buffer, incubated on ice for 15 min, and then centrifuged (12,000 × g, 15 min, 4C). Supernatant fractions all containing the same amount of protein (100 μg) were retained, and protein concentrations in samples were equalized using a Bio-Rad protein assay. Samples were mixed in SDS-PAGE 10% gel (Minigel apparatus; BioRad) containing gelatin (0.2%) with non-reducing conditions and separated by electrophoresis. Subsequently, gels were washed twice in 2.5% Triton X-100, incubated in zymography digestion buffer (50 mmol Tris, 5 mmol CaCl2, pH 7.6; Sigma Chemical Co., St Louis, MO) overnight at 37C, and then immersed for 3 hr in staining solution (0.25% Coomassie Blue R250, 10% glacial acetic acid in water). Gelatinolytic activity was visualized as clear areas of lysis in the gel (Rizzoni et al. 2004).
Statistical Analysis
Samples were analyzed and scored blindly. Results are presented as means ± SEM. Statistical significance of differences between the experimental groups was estimated using the ANOVA and Bonferroni test; p<0.05 was considered significant.
Results
Pregnancy Outcomes and Pup Mass among Control, CsA-, and CsA + PV-treated Rats
Data on pregnancy outcomes and on body weight of pups are reported in Table 1. Gestation lasts 21 days in rats, and CsA treatment had no adverse effects on the duration of gestation. Most CsA-treated mothers showed a significant decrease in maternal weight in comparison with the control in relation to the number of pups per litter that was statistically reduced. No difference in body weight among pups in the control and CsA groups was observed. Moreover, there was no difference in body weight between litters from CsA + PV-treated mothers and those from control group.
Effect of maternal cyclosporine A (CsA) exposure on pregnancy outcomes
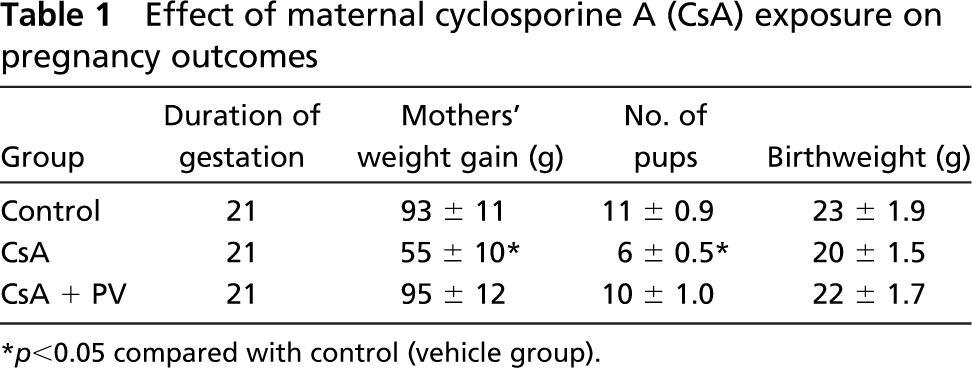
p<0.05 compared with control (vehicle group).
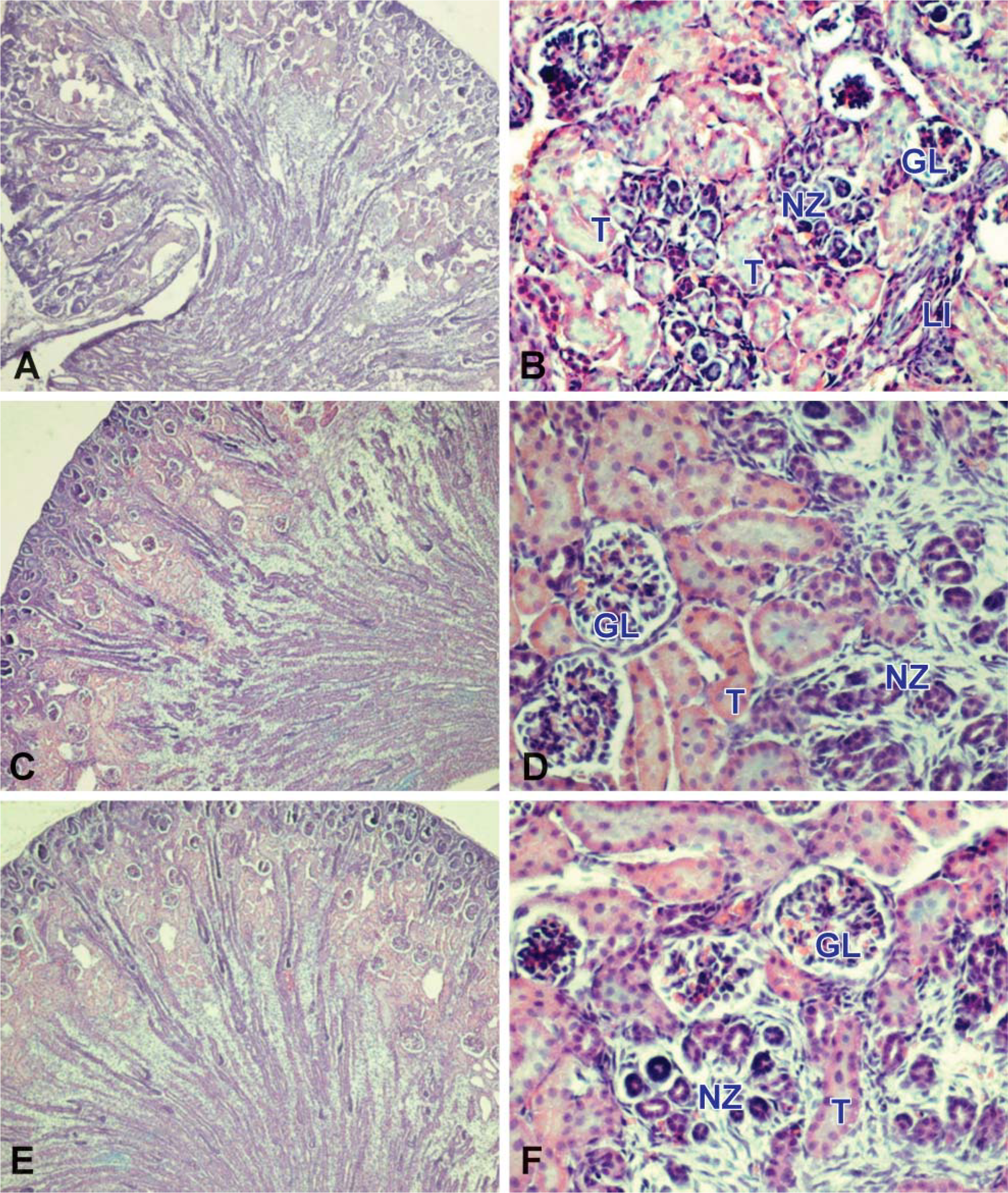
Renal histology of pups born from CsA- (
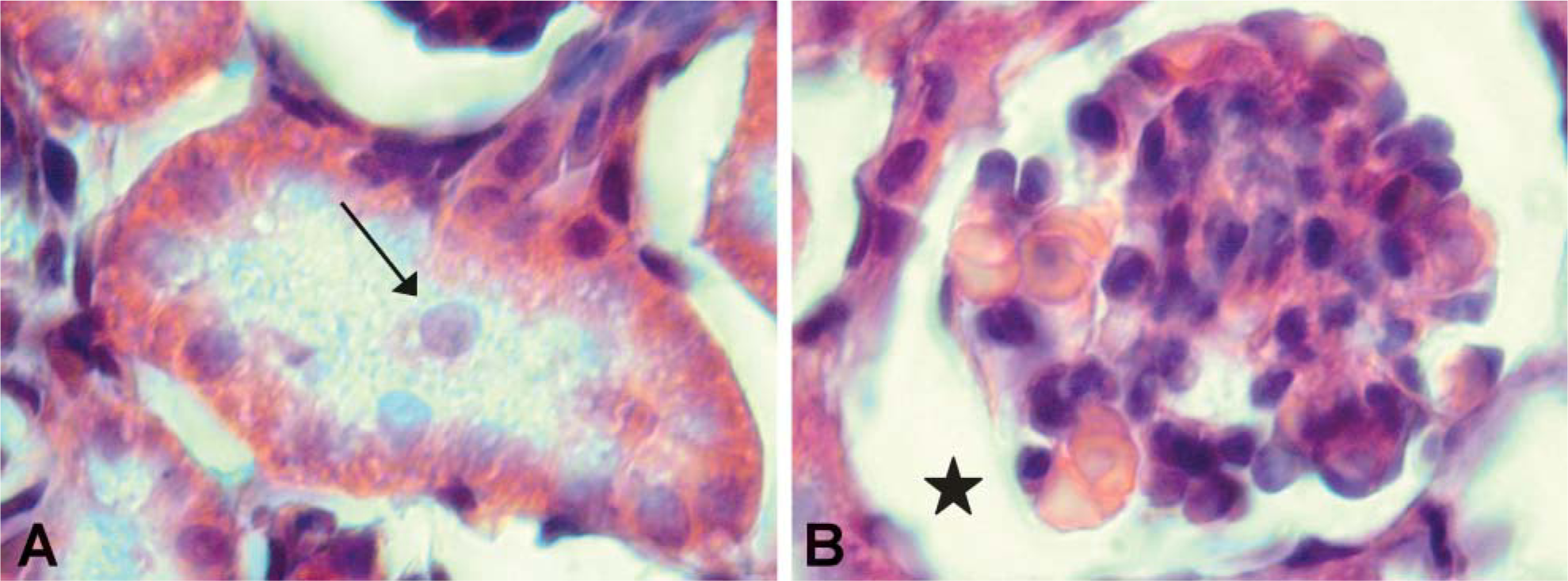
Renal histology of pups born from CsA-treated rats stained with H&E (X1000). Arrow indicates tubular swelling (
Renal Morphological Features of Pups Born to CsA-treated Mothers
Neonatal kidneys from CsA-treated mothers displayed several pathologic findings in all immature parenchyma. As shown in Figure 1A, the cortex showed a disorganization in the nephrogenic zones; signs of tubular and glomerular alterations were evident. At high magnification, distal tubules were not clearly evident in the nephrogenic zones, whereas there was a number of lymphocytic infiltrates (Figure 1B). Moreover, size of the apparently unaltered glomeruli was considerably reduced.
Tubulopathy was observed in most proximal tubules; cellular lesions such as tubular swelling were also evident (Figure 2A). In particular, statistical analysis of tubular swelling is reported in Table 2.
Several glomeruli were damaged, and glomerular sclerosis such as tuft adhesion to Bowman's capsule and prominent urinary chamber were present (Figure 2B). Signs of lesions were also observed in the medulla because the lumen of Henle loops was enlarged.
Renal Morphological Features of Pups Born to Control and CsA + PV-treated Rats
In rats, nephrogenesis begins on embryonic day 12 and is completed between 10 and 15 days postnatally (Balbi et al. 2004). As illustrated in Figures 1C and 1E, several generations of nephrons have already been induced. At the periphery, the nephrogenic zone contained the mesenchyme with connective tissue and a number of tubules already formed. In the cortex, we found a high number of mature proximal tubules, whereas distal tubules were not well developed and were included in the nephrogenic areas. Many glomeruli displayed normal cytoarchitecture, and a minor number showed no Bowman's capsule completely defined (Figures 1D and 1F).
Statistical analysis of Tubular Swelling (TS)

p<0.05 compared with control (vehicle group). Units of measurement were expressed as percentage of renal damaged tubules.
Estimation of VWGV and GS among Pups from Control, CsA-, and CsA + PV-treated Mothers
VWGV was significantly higher in pups of control and CsA + PV-treated mothers (8.7 ± 1.1 and 9.3 ± 1.7, respectively) than in those born from CsA-treated rats.
GS was significantly decreased in pups born from CsA-treated mothers (100 ± 5) with respect to control and CsA + PV-treated rats (178 ± 2 and 173 ± 3, respectively).
Both VWGV and GS analysis are reported in Table 3.
Expression and Activity of iNOS and MMP2 in Kidneys of Litters from CsA-treated Mothers
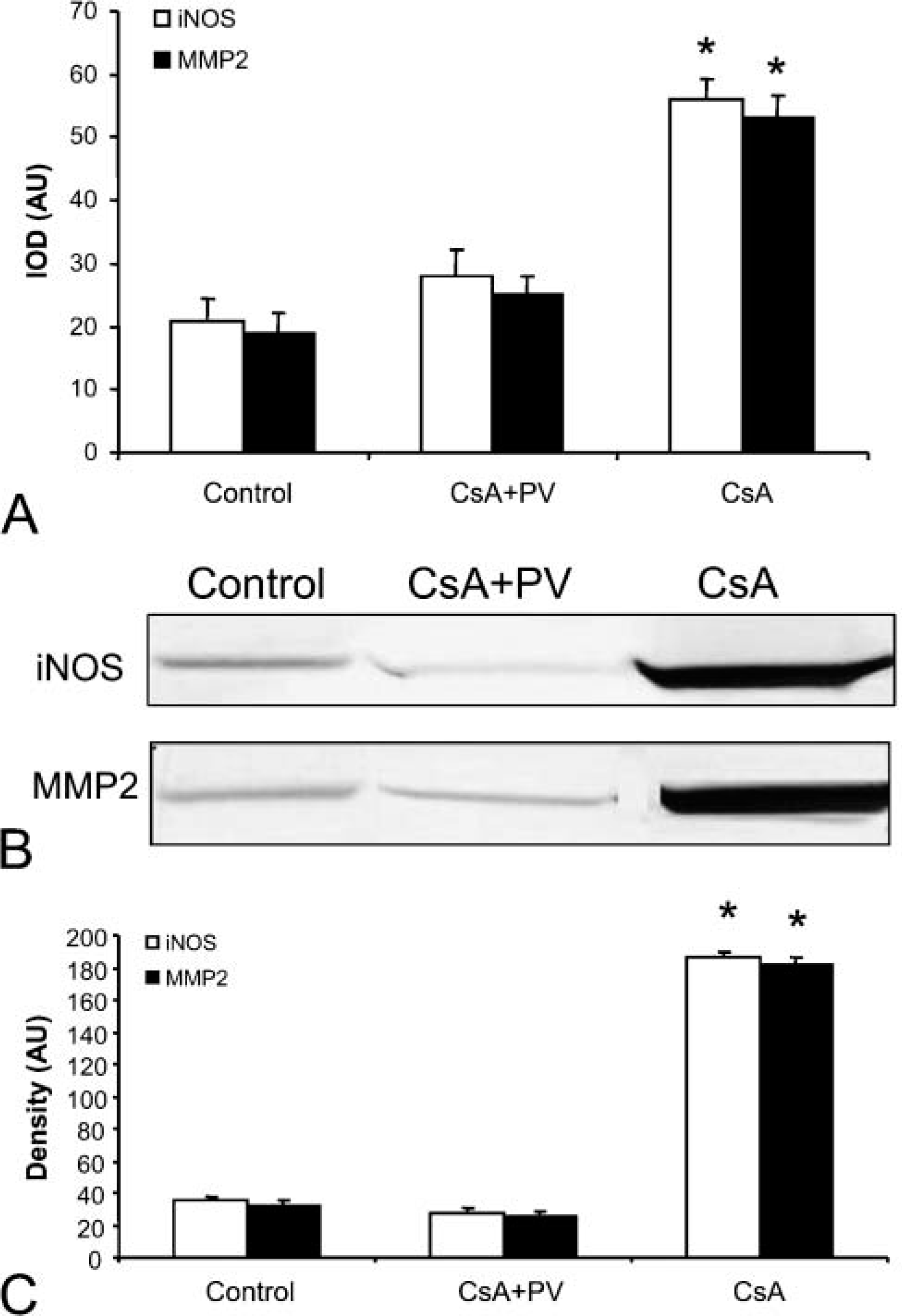
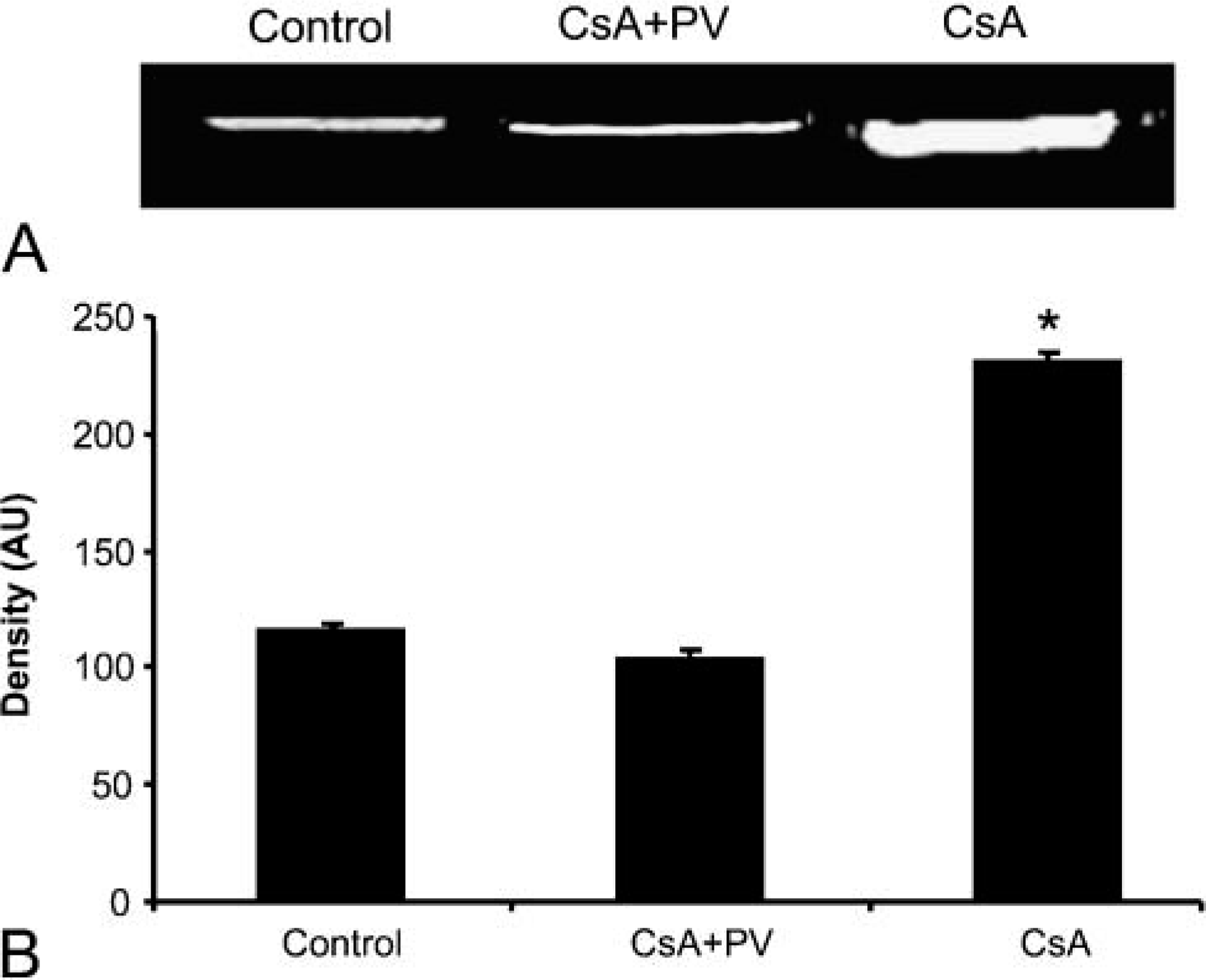
iNOS and MMP2 immunostaining showed a similar positivity in renal cortex and medulla. As shown in Figure 3A, iNOS immunostaining displayed moderate and diffuse positivity in epithelial cells of proximal tubules and a strong positivity in glomeruli and in nephrogenic areas. In these zones it was difficult to distinguish distal tubules. The Henle loops also showed an increase in iNOS and MMP2 positivity that was localized mainly in epithelial cells near the lumen (Figures 4A and 4B). Figure 5 shows quantitative IHC and immunoblotting analysis for iNOS and MMP2. Moreover, Figure 6 shows zymographic analysis for MMP2.
Estimation of volume-weighted glomerular volume (VWGV) and glomerular size (GS)

p<0.05 compared with control (vehicle group). Units of measurement were expressed for VWGV as μm3 × 105 and for GS as μm.
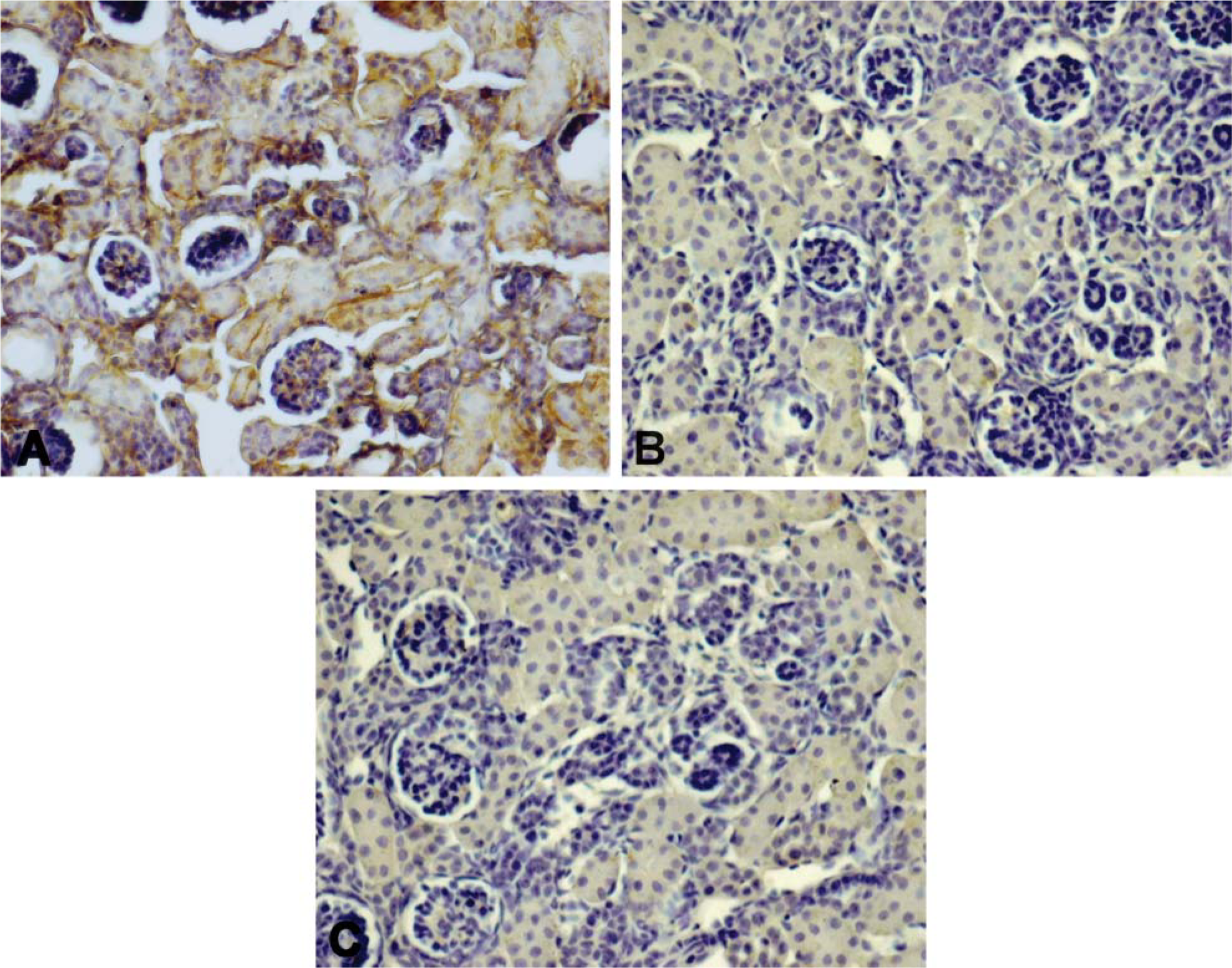
Kidney of pups born from CsA- (
Expression and Activity of iNOS and MMP2 in Kidneys of Pups Born to Control and CsA + PV-treated Rats
In renal cortex, proximal tubules showed very weak positivity, whereas distal tubules and glomeruli were negative (Figures 3B and 3C); in the medulla, the Henle loops and vasa recta were weakly positive (Figures 4C and 4D) as in CsA + PV-treated rats (Figures 4E and 4F). In these structures, staining appeared diffuse in the cytoplasm of epithelial cells. iNOS and MMP2 quantitative IHC and immunoblotting results are shown in Figure 5. MMP2 zymographic analysis was reported in Figure 6.
Discussion
Our results provide evidence that CsA has deleterious effects on renal organogenesis of litters from treated mothers and that CsA + PV treatment prevents these adverse effects.
Negative effects such as prominent glomerular urinary chambers, tubular swelling, interstitial fibrosis, and inflammation are the same characteristics observed in rat kidney exposed to drug treatment (Rezzani 2004). Therefore, these findings showed the ability of CsA to cross the placenta and the importance of controlling the drug level and its metabolites as previously reported by several authors (Nyberg et al. 1998; Venkataramanan et al. 1988).
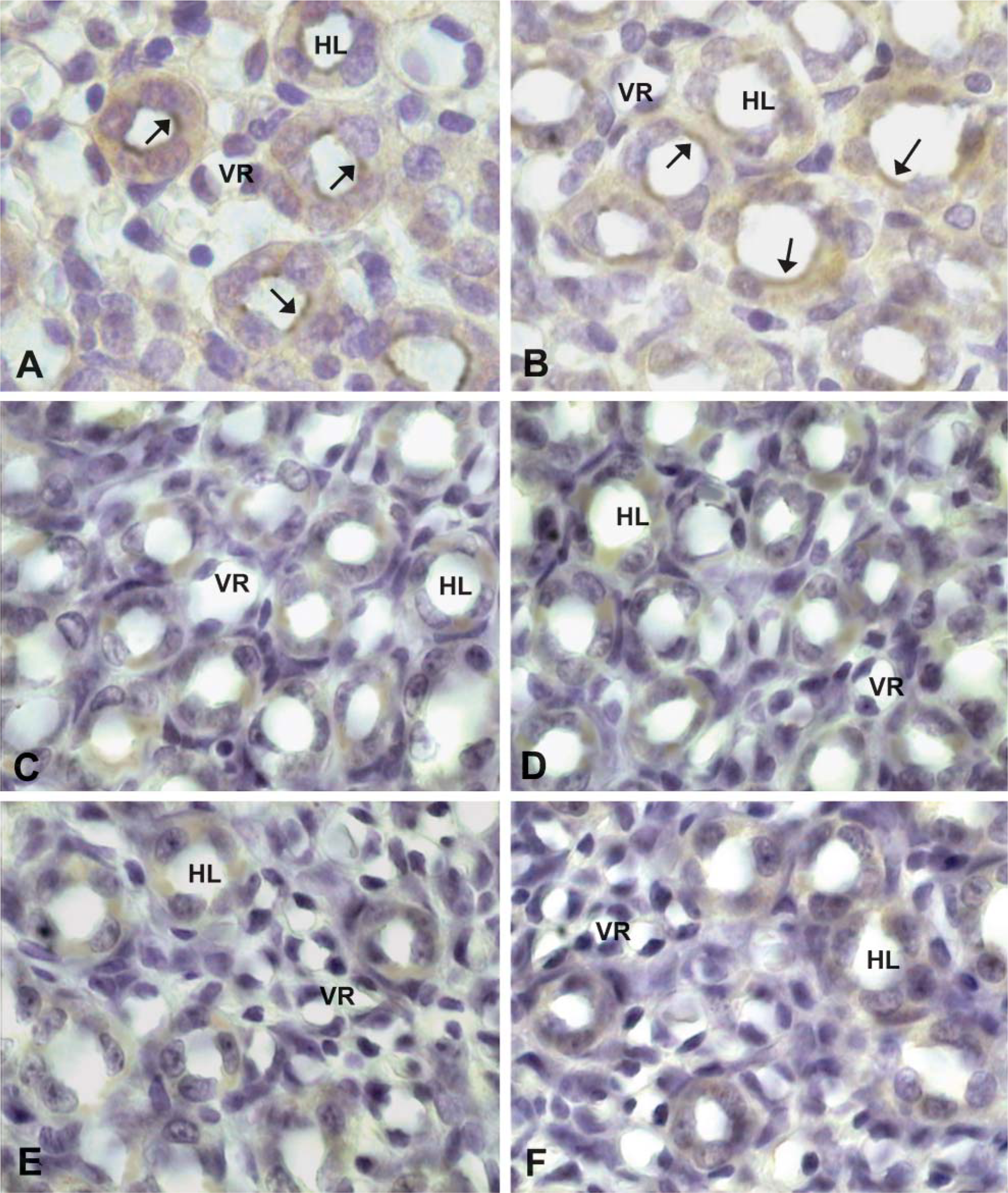
Henle loops in kidney of pups born from CsA- (
(
These data agree with our report and other previous reports indicating that this drug and its metabolites interfere with fetal development (Bermas and Hill 1995; Sgro et al. 2002; Tendron-Franzin et al. 2004). Tendron-Franzin et al. (2004) also showed that the rabbits exposed in utero to CsA initially had, at 1 month of age, a normal arterial blood pressure, but at 11 weeks they developed a marked systemic hypertension worsening with age. These findings can be related to the observed data displaying glomerular and tubular lesions in neonatal rats. We could suggest that this damage participates in the initiation and progression of hypertension as reported in clinical studies linked to renal pathophysiology (Sanchez-Lozada et al. 2003).
(
Moreover, we considered the activity and expression of two proteins (iNOS and MMP2) as playing an important role in nephrotoxicity (Eberhardt et al. 2000; Khanna et al. 2002; Rezzani et al. 2003; Buffoli et al. 2005) and found an induction of these proteins after CsA treatment. Of note, it seems that these data underlie an increase of these proteins in apical membrane of Henle loops. On the basis of these data, we could suggest that CsA initially interferes with the physiological metabolism of Henle loops. In particular, its interference regards the apical membrane, which has a different metabolism from basal membrane as reported by Knepper et al. (1999). These authors showed that NaCl transport into the cells occurs via apical membrane of Henle loops and that the potassium ions that enter the cells on the cotransporter are largely recycled into the lumen via apical potassium channels. Sodium ions are actively transported from the cell to the interstitium by the basolateral Na+-K+ATPase. Chloride ions that enter the cells via the Na+-K+-2Cl- cotransporter exit across the basolateral plasma membrane via a chloride channel and/or by K-Cl cotransport.
We previously showed that CsA induces an increase in the expression of different factors such as nuclear factor kappa B (NF-κB) that are able to positively regulate the iNOS genes. These genes can produce an increase in iNOS and a high amount of nitric oxide that can combine with superoxide anions and induce lipid peroxidation and oxidative stress (Buffoli et al. 2005). Regarding the increase of MMP2 expression, Siwik and Colucci (2004) identified that many promoters of MMP have the same binding sites of iNOS and produce an increase in protein expression responsible for the pathological conditions leading to fibrosis. Therefore, the relationship among MMP2, iNOS, and nephrotoxicity could be explained by suggesting that their upregulation is linked to renal oxidative stress and fibrosis that impair renal cytoarchitecture.
The second subject of this study addresses the possible protective role of PV against nephrotoxicity. We showed that CsA + PV treatment is protective against toxic CsA effects in kidney of litters from treated mothers both on cytoarchitecture and on iNOS and MMP2 expression. Recent studies showed that PV is able to counteract CsA-induced negative effects involving the above-reported proteins (Buffoli et al. 2005). Together these data suggest that the protective role played by PV involves the same biochemical pathway observed in CsA-treated adult rats, and we could therefore propose a similar mechanism. In fact, as proposed by Buffoli et al. (2005), red wine polyphenols protect renal damage due to oxidative stress and prevent NF-κB expression during immunosuppressive treatment. This downregulation produces a decrease of iNOS expression. Therefore, reduction of both oxidative stress and iNOS expression via NF-κB pathway may be responsible for the protective effect of PV on CsA-induced structural and functional alterations of the kidney.
In conclusion, our study shows the presence of morphological alterations in renal parenchyma of litters from CsA-treated mothers and the protective role played by PV. These data could be of particular relevance in relation to development of therapeutic strategies during pregnancy and immunosuppressive treatment.
Footnotes
Acknowledgements
The authors thank Miss Stefania Castrezzati for technical assistance.
