Abstract
Newborn rats were treated with sodium alendronate to study how enamel is formed and the effect of alendronate during early odontogenesis. Ultrastructural analysis combined with high-resolution immunocytochemistry for amelogenin was carried out. Twelve rats were subjected to daily SC injections of sodium alendronate (2.5 mg/kg/day) for 3 days on their dorsal region, whereas three rats were daily injected with saline solution as a control. Molar tooth germs from 3-day-old rats were fixed under microwave irradiation in 0.1% glutaraldehyde + 4% formaldehyde buffered at pH 7.2 with 0.1 M sodium cacodylate. The specimens were left undecalcified, postfixed with osmium tetroxide, dehydrated, and embedded in LR White resin. Ultrathin sections were incubated with a chicken anti-24-kDa rat amelogenin antibody, a secondary antibody, and finally with a protein A-gold complex. Large patches of amelogenin were present over the unmineralized mantle dentin and at early secretory ameloblasts. At more advanced stages, they were also detected at the enamel matrix, as well as in the mineralized dentin, at the periodontoblastic space of the dentinal tubules, and at the predentin. It is likely that the main effect of alendronate at early stages of odontogenesis is the increase of synthesis/secretion of amelogenin, promoting its deposition within the forming dentin and enamel.
O
Amelogenins are the major components of the developing enamel matrix as they comprise ∼90% of all enamel proteins secreted by ameloblasts (Nanci and Smith 1992). Amelogenins are hydrophobic proteins rich in proline, histidine, and glutamine. They are present in small amounts at the beginning of amelogenesis where they are immunodetected as early as the differentiation of ameloblasts starts, i.e., before the secretion of the other enamel proteins (Nanci et al. 1998). It is more evident when highly sensitive methods of immunodetection combined with transmission electron microscopy are applied (Arana-Chavez and Nanci 2001). Many amelogenin cleavage products from proteinase activity and isoforms translated from alternatively spliced mRNA transcripts have been characterized by several researchers in the last 20 years [reviewed in Veis (2003)] Alternative splicing of amelogenin mRNA results in the formation of proteins ranging from 5 to 28 kDa in size (Simmer and Snead 1995). The amelogenin sequences at both carboxy- and N-terminal regions promote their self-assembly into 20-nm-diameter nanospheres, which are thought to play a key role in guiding the growth of enamel crystallites (Fincham et al. 1999; Moradian-Oldak 2001). The action of matrix metalloproteinases, especially MMP-20, on the amelogenin molecules cleavage is crucial for their self-assembly, association with the mineral phase, and degradation and removal from the extracellular matrix [reviewed in Smith (1998)].
Bisphosphonates are pyrophosphate analogs that, because of their similarity to natural pyrophosphate, avidly bind to bone (Fleisch 1987). This property, in addition to their ability to inhibit osteoclastic bone resorption, has led to the use of bisphosphonates in the treatment of skeletal disorders such as osteoporosis, Paget's disease, osteogenesis imperfecta, and metastatic bone diseases (Fleisch 1998; Vasikaran 2001). The molecular mechanisms of action of bisphosphonates on osteoclasts have recently been elucidated. Depending on the presence of a nitrogen atom in the alkyl chain of the molecule, bisphosphonates act either by causing toxic effects on osteoclasts or by interfering with specific intracellular pathways in those cells. Although several studies have suggested that bisphosphonates do not adversely affect the osteoblast activity (Sato et al. 1991; Nii et al. 1994; Rodan and Fleisch 1996; Evans 2002), cells other than osteoclasts may also be affected by bisphosphonates in other mineralized tissues. It has also been reported that bisphosphonates act as inhibitors of mineralization in bone (Francis 1969; King et al. 1971; Schenk et al. 1973) and tooth (Reynolds et al. 1973; Beertsen et al. 1985; Ogawa et al. 1989; Takano et al. 1998; Sakai et al. 1999; Jayawardena et al. 2002). Indeed, administration of bisphosphonates at high and successive doses has been shown to affect mineralization of both dentin and enamel (Simmelink 1987; Izumi 1989; Ohya et al. 1989; Fejerskov et al. 1990; Weile et al. 1990, 1993; Fouda et al. 1991; Wakamatsu 1991; Takano et al. 1998). Some light microscopy studies have shown that rats injected with 1-hydroxyethylidene-1, 1-bisphosphonate (HEBP) develop enamel hypoplasia due to a disturbance in the enamel protein secretion (Fuangtharnthip et al. 2000; Yamada et al. 2000).
Alendronate is a potent nitrogen-containing bisphosphonate that inhibits enzymes of the mevalonate pathway, thus preventing the prenylation of small GTP-binding proteins that are responsible for the cytoskeletal integrity and for intracellular signaling in osteoclasts (Fisher et al. 1999; van Beek et al. 1999; Bergstrom et al. 2000). Although the mode of action of alendronate is mainly being investigated in bone, little is known about its effects on the formation of dental hard tissues, in particular on enamel.
To test the hypothesis that alendronate may induce some alterations on the early development of enamel, resembling the effects of other bisphosphonates, we have carried out an ultrastructural study on molar tooth germs from alendronate-treated 3-day-old rats combined with the high-resolution immunocytochemical labeling for amelogenin, the major enamel protein secreted at early stages of odontogenesis. Analysis of the effects caused by sodium alendronate on the ameloblast metabolism may elucidate some aspects of the amelogenin function during early odontogenesis. As alendronate is usually administered orally in treatments for osteoporosis and other bone diseases, it would be difficult to determine comparable serum concentrations in the newborn rats. Thus, we have given a daily high dose (2.5 mg/kg/day) of alendronate SC, based on a previous study of the growth plate from long bones in mice (Evans et al. 2003).
Materials and Methods
Animals, Alendronate Treatment, and Tissue Processing
Principles of Laboratory Animal Care (NIH Publication 8523, revised in 1985) and Brazilian national laws on animal use were observed for the present study, which was authorized by the Ethical Committee for Animal Research of the University of São Paulo, Brazil.
Fifteen 3-day-old Wistar rats weighing ∼7 g were studied. Twelve rats were subjected to daily SC injections of 2.5 mg/kg/day (Evans et al. 2003) of sodium alendronate (Matéria Prima-Farmácia de Manipulação; São Paulo, Brazil) for 3 days on their dorsal region, whereas three rats were daily injected with saline solution as a control. Two hr after the third injection, the tooth germs were removed and quickly placed in a fixative containing 0.1% glutaraldehyde + 4% formaldehyde (freshly prepared from paraformaldehyde) and buffered at pH 7.4 with 0.1 M sodium cacodylate (Arana-Chavez and Nanci 2001). Briefly, the specimens were immersed in a beaker containing 40 ml of fixative at room temperature, which was subsequently placed in a 20 × 20-cm glass recipient filled with ice and immediately placed inside a Pelco 3440 laboratory microwave oven (Ted Pella; Redding, CA). The temperature probe of the oven was submersed into the fixative, and the specimens were exposed to microwave irradiation at a 100% setting for three periods of 5 min with the temperature programmed to a maximum of 37C (Massa and Arana-Chavez 2000). After microwave irradiation, specimens were transferred into fresh fixative and left overnight at 4C. They were then washed in 0.1 M sodium cacodylate buffer, pH 7.4, for 1 hr, left undecalcified, and postfixed in 0.1 M cacodylate-buffered 1% osmium tetroxide for 1 hr at room temperature or left unosmicated. They were then dehydrated in graded concentrations of ethanol and embedded in LR White resin. Toluidine blue-stained 1-μm-thick sections were examined under a light microscope, and regions containing the sequence of initial stages of odontogenesis were trimmed for ultrathin sectioning. Eighty-nm-thick sections were cut with a diamond knife on a Leica Ultracut R ultramicrotome (Leica Inc.; Buffalo, NY), collected onto 200-mesh nickel grids coated with 2% parlodion in amyl acetate, stained with uranyl acetate and lead citrate, and examined in a Jeol (Tokyo, Japan) 1010 transmission electron microscope operated at 80 kV.
High-resolution Postembedding Immunocytochemistry
Ultrathin sections of osmicated samples were pretreated with an aqueous solution of 5% sodium metaperiodate for 60 min (Bendayan and Zollinger 1983) and washed with distilled water for 15 min. Unosmicated sections were directly processed for immunolabeling. Grids were incubated with a chicken egg yolk anti-rat 24-kDa amelogenin antibody (Nanci et al. 1996) diluted 1:150 for 5 hr and then incubated with a rabbit anti-chicken IgG secondary antibody (Sigma; St Louis, MO), diluted 1:2000 for 1 hr. The sites of antibody-antigen binding were revealed with a protein A-gold complex, prepared as described in Bendayan (1995). Grids were washed with PBS between incubation steps and blocked with PBS-1% ovalbumin for 15 min. After the protein A-gold, the grids were jet washed with PBS followed by distilled water. All steps were carried out at room temperature. Sections were then stained with uranyl acetate and lead citrate and examined in a Jeol 1010 transmission electron microscope operated at 80 kV. Controls for the specificity of the labeling consisted of incubating the sections with preimmune serum, with the secondary antibody followed by protein A-gold, or with protein A-gold alone.
Results
Ultrastructural Morphology
Examination of ultrathin sections from the alendronate-treated specimens revealed evident alterations in the extracellular matrix at all stages of odontogenesis when compared with the control group. As an example, Figure 1 shows the comparison between a specimen from the alendronate-treated group (Figure 1A) and another from the control group (Figure 1B). At later stages, large patches of electron-opaque granular material were present between the plasma membrane of the ameloblasts at the newly formed enamel matrix. The enamel crystals were placed somewhat disorderedly in the alendronate-treated specimens when compared with the control group (Figures 1A and 1B).
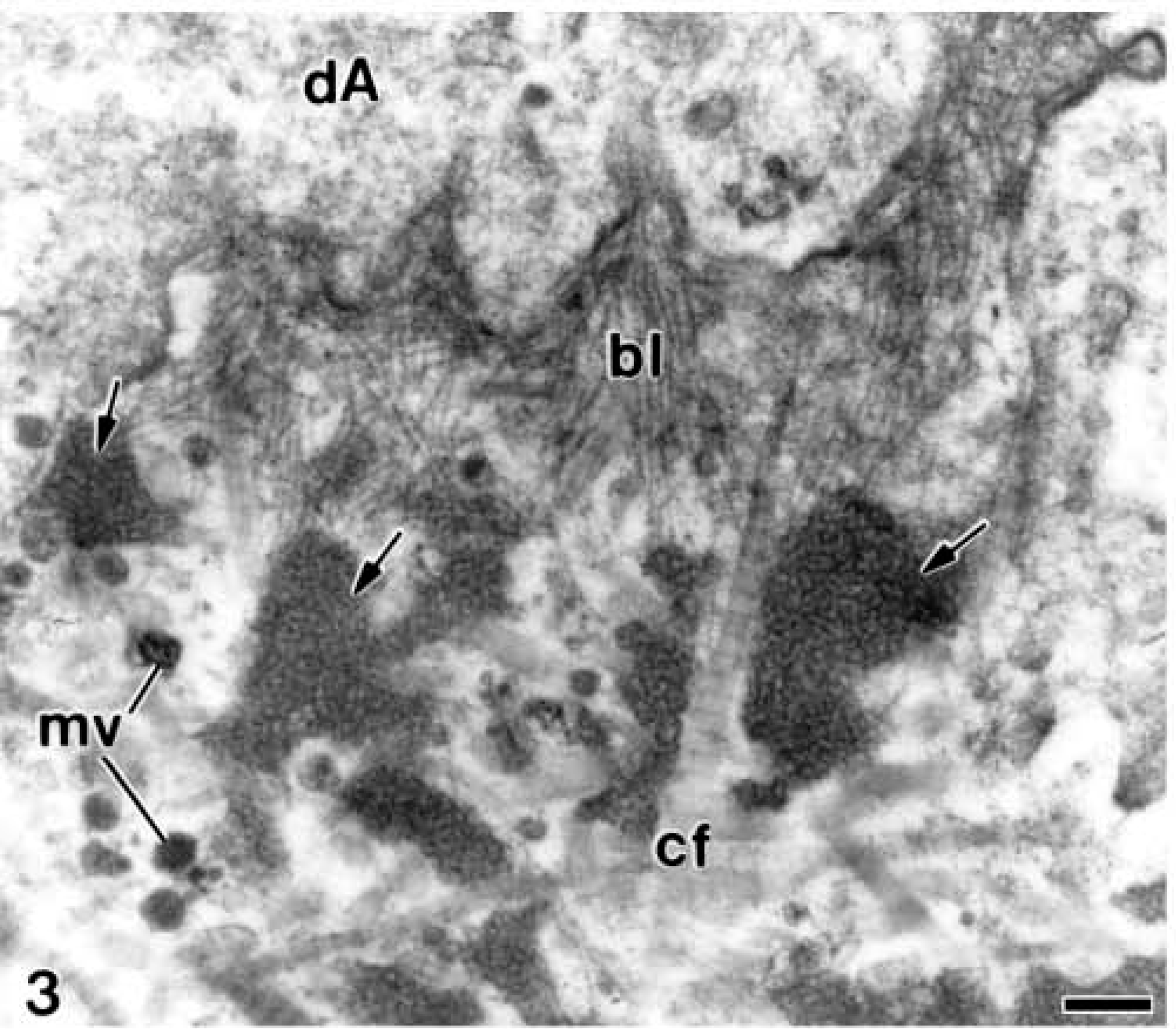
At very early stages before the undifferentiated ameloblasts initiated their elongation and reversed their polarity, patches of a conspicuously granular electronopaque material appeared at their proximal end. They were present in the early unmineralized mantle dentin between the collagen fibrils and matrix vesicles. Electron-opaque granular material was also found along the basolateral surface of ameloblasts among the collagen fibrils and other components of the mantle dentin matrix (Figure 2). Thus, during the first stages of odontogenesis the patches of electron-opaque granular material were identified in close relation to the distal surface of differentiating ameloblasts and among the disrupted basal lamina (Figure 3). At these stages, a few granular deposits were also observed at the apical surface of the differentiating odontoblasts (Figure 2).
In stages in which the unmineralized dentin matrix formed a continuous layer, electron-opaque patches present at the distal end of differentiating ameloblasts were smaller than those observed in proximity to the distal end of differentiating odontoblasts (Figure 4). When the developing mantle dentin started its mineralization in relation to the matrix vesicles and then spread to the adjacent collagen, the patches of granular electron-opaque material continued to accumulate (data not shown).
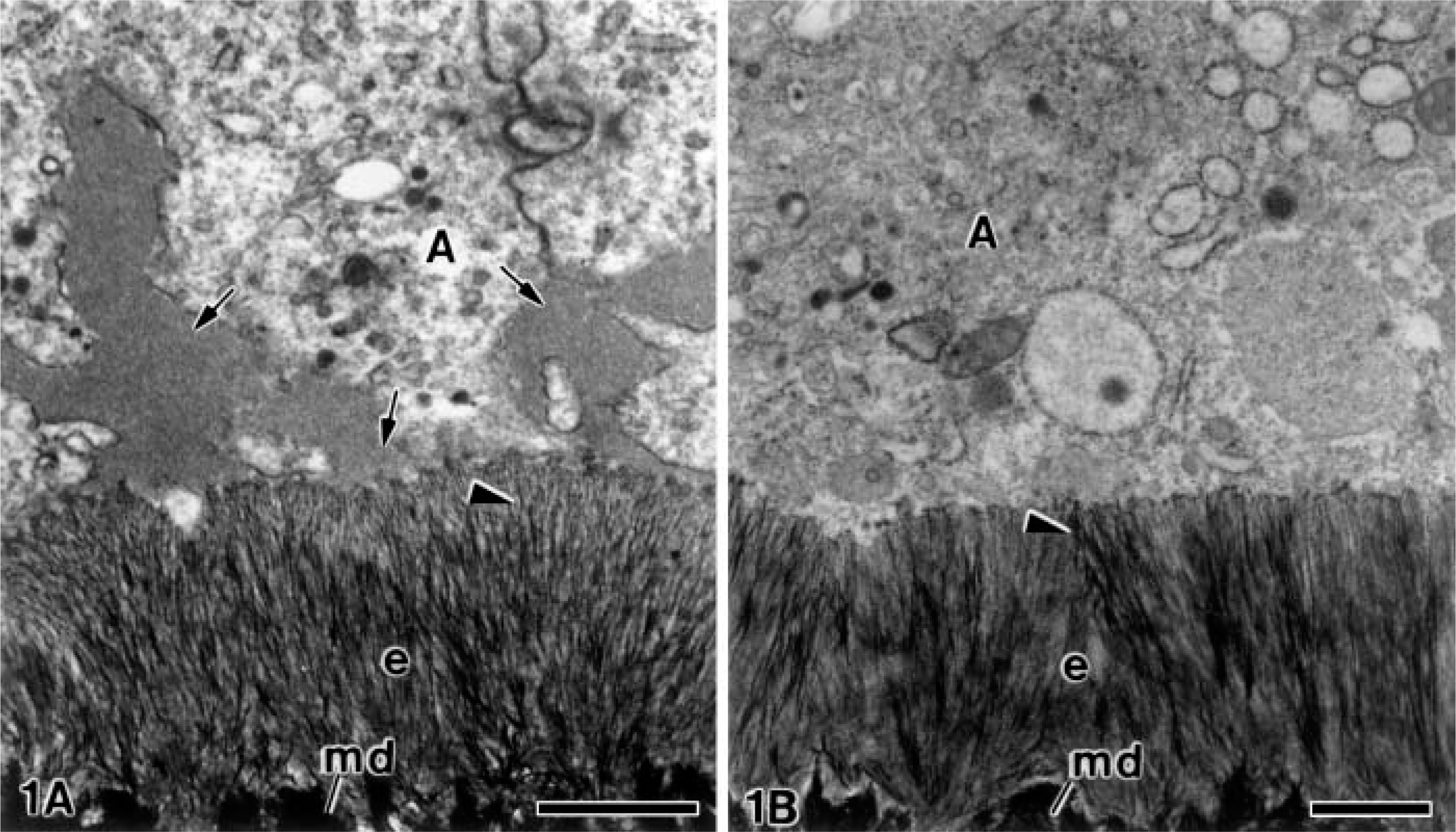
Alendronate-treated and non- treated molar tooth germs. Electron micrographs showing the secretion stage of amelogenesis. (
In more advanced phases, when the mineralized mantle dentin established a continuous band and the secretory stage of amelogenesis began, numerous patches of granular material were present at the distal surface of ameloblasts. They were present at the intercellular spaces (Figure 5), as well as into the ameloblast cytoplasm, inside secretory granules (Figure 6). As this granular material kept on accumulating, the developing enamel matrix started its mineralization. The nascent enamel crystallites were surrounded by the excess of granular matrix, which was interposed between the ameloblast plasma membrane and the crystals (Figure 6). The mineralized dentin presented small patches of granular material in the interior of the dentinal tubules, occupying the periodontoblastic space in close relation to the odontoblast process (Figure 7). In contrast, the patches of granular material were numerous and larger in the predentin, where they appeared to be located below the dentinal tubules and mainly in proximity to the odontoblast processes (Figure 7). The patches of electron-opaque granular material were observed between the odontoblasts but not into the cytoplasm of odontoblasts or at the subodontoblastic region of the dental papilla (see below).
Post-embedding Immunocytochemistry for Amelogenin
As described above, only the early stages of dentinogenesis were somewhat similar between both groups, because from a given time point the presence of large patches were clearly identified in the alendronate-treated group. Thus, at early stages, when the nontreated specimens (control group) were incubated with the anti-amelogenin antibody, the amelogenin 24-kDa molecule was present at the distal cytoplasm of the differentiating ameloblasts and dispersed within the unmineralized dentin matrix in close relation to the basal lamina (Figure 8A). At slightly more advanced stages, no patches immunoreactive for this protein were observed in the developing dentin matrix (Figure 8B).
In the alendronate-treated specimens, the patches of electron-opaque granular material described above were strongly immunoreactive for amelogenin at all studied stages of dentinogenesis and amelogenesis. Amelogenin was detected at early stages of ameloblast differentiation when these cells started the organization of their synthesis and secretion apparatus. The colloidal gold particles were observed inside secretory granules at the distal end and at the early unmineralized dentin matrix where they appeared in close relation to the basal lamina and the newly released matrix vesicles (Figure 9) The immunoreactivity for amelogenin at these early stages was somewhat more intense than that observed in the control specimens (compare with Figure 8A). In a slightly more advanced stage, the gold particles formed patches at the unmineralized dentin, which were more concentrated adjacent to the differentiating ameloblasts (Figure 10). Later in dentin formation, they were in close relation to the distal surface of odontoblasts, as well as to some projections of the odontoblast cell bodies (Figure 11). In the newly mineralized mantle dentin, the patches of amelogenin, which were larger and in a higher amount, were located close to the differentiating odontoblasts and their process (Figure 12). At the odontoblast layer, patches strongly immunoreactive for amelogenin were surrounded by projections at the distolateral regions of odontoblasts (Figure 13). Other patches with smaller size were also detected between the odontoblasts, at their basolateral sides (see below).

Electron microscopy of alendronate-treated molar tooth germs. A region of mantle dentin (d) during early stages of dentinogenesis in which some patches of electron-opaque granular material (arrows) are easily observed within the collagen fibrils (cf) and the matrix vesicles (mv). dA, differentiating ameloblasts; dO, differentiating odontoblasts. Bar = 1 μm.
Electron microscopy of alendronate-treated molar tooth germs. A higher magnification view of a stage of odontogenesis in which the basal lamina (bl) appears partially degraded. Some patches of electron-opaque granular material (arrows) are near the distal surface of differentiating ameloblast (dA). cf, collagen fibrils; mv, mineralizing matrix vesicles. Bar = 0.2 μm.
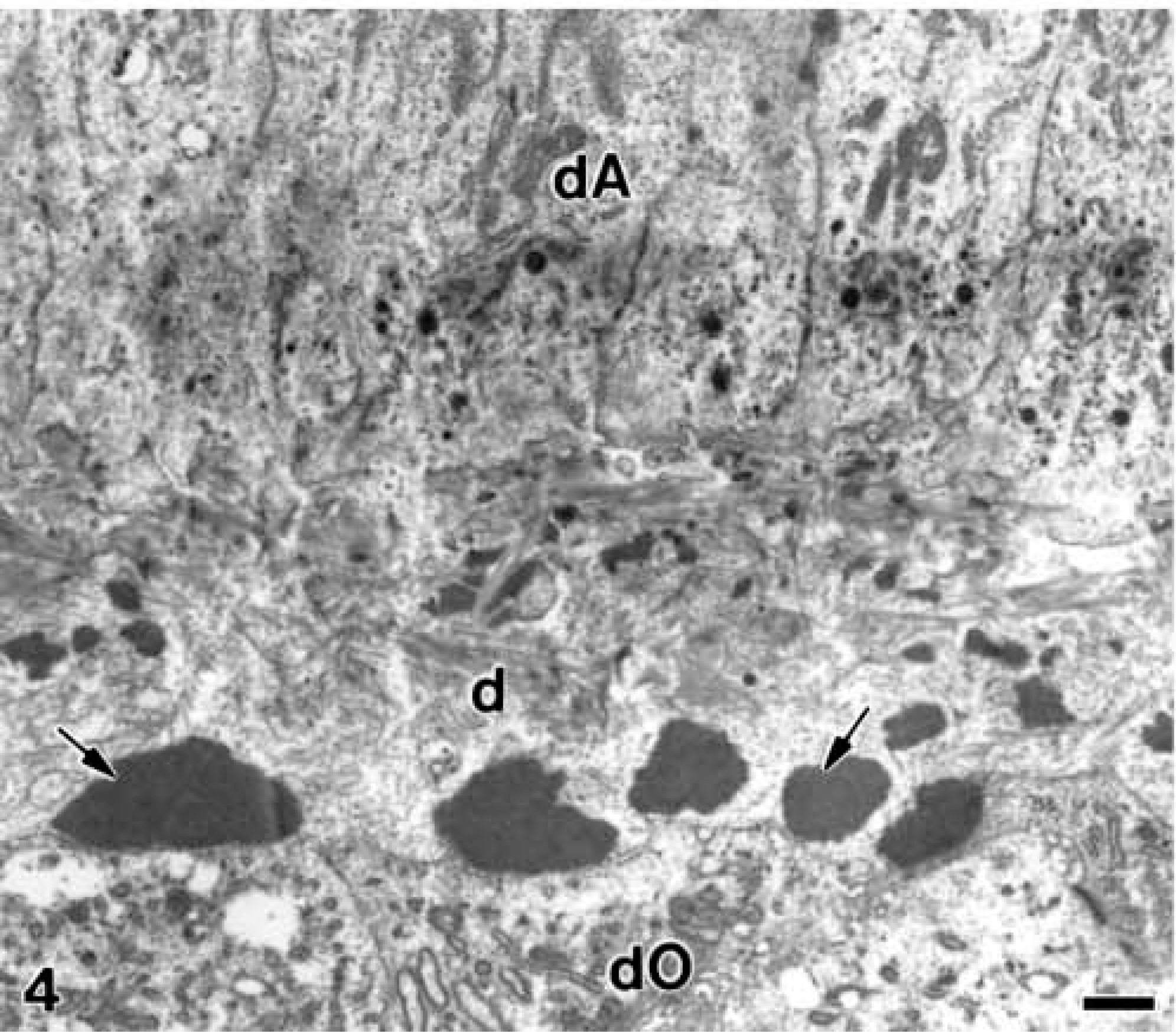
Electron microscopy of alendronate-treated molar tooth germs. A continuous layer of unmineralized mantle dentin matrix (d) in which large patches of electron-opaque granular (arrows) are seen, especially adjacent to the distal end of differentiating odontoblast (dO). dA, differentiating ameloblast. Bar = 1 μm.
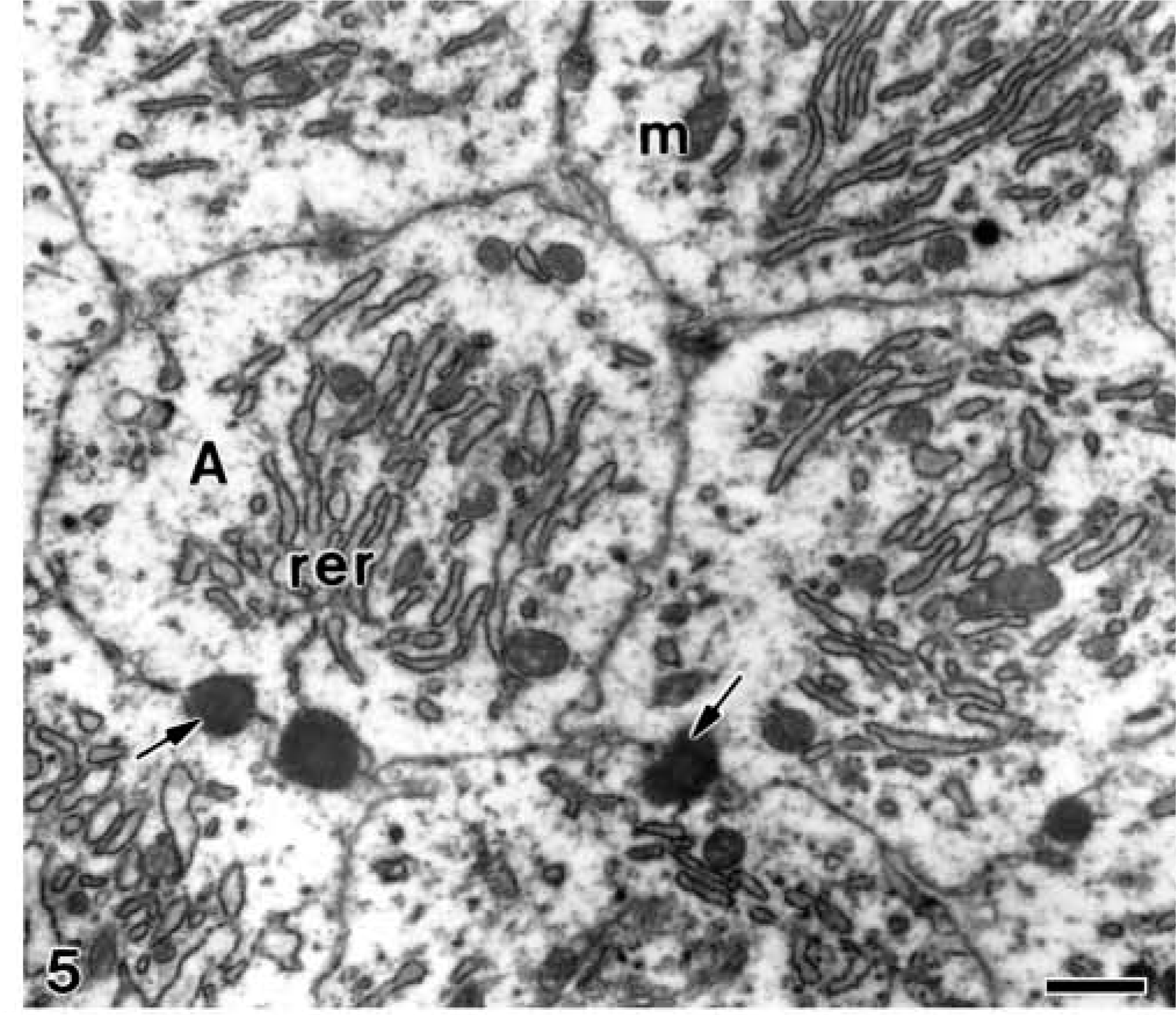
Electron microscopy of alendronate-treated molar tooth germs. Electron-opaque granular patches (arrows) are observed at the intercellular spaces of cross-sectioned fully differentiated ameloblasts (A) during the secretion stage of amelogenesis. Note the well-developed rough endoplasmic reticulum (rer) and some mitochondria (m) in the cytoplasm of ameloblasts. Bar = 1 μm.

Electron microscopy of alendronate-treated molar tooth germs. An oblique section at the distal end of some ameloblasts (A) during early secretory stage of enamel proteins at amelogenesis. The electron-opaque granular patches (arrows) are present at the intercellular spaces, as well as at the ameloblast distal end, whereas the enamel layer (e) is forming. Inset shows the nascent enamel crystallites (arrowhead) at some distance from the plasma membrane of ameloblasts due to the presence of interposed granular matrix (arrows). Bar = 1 μm; inset bar = 0.1 μm.
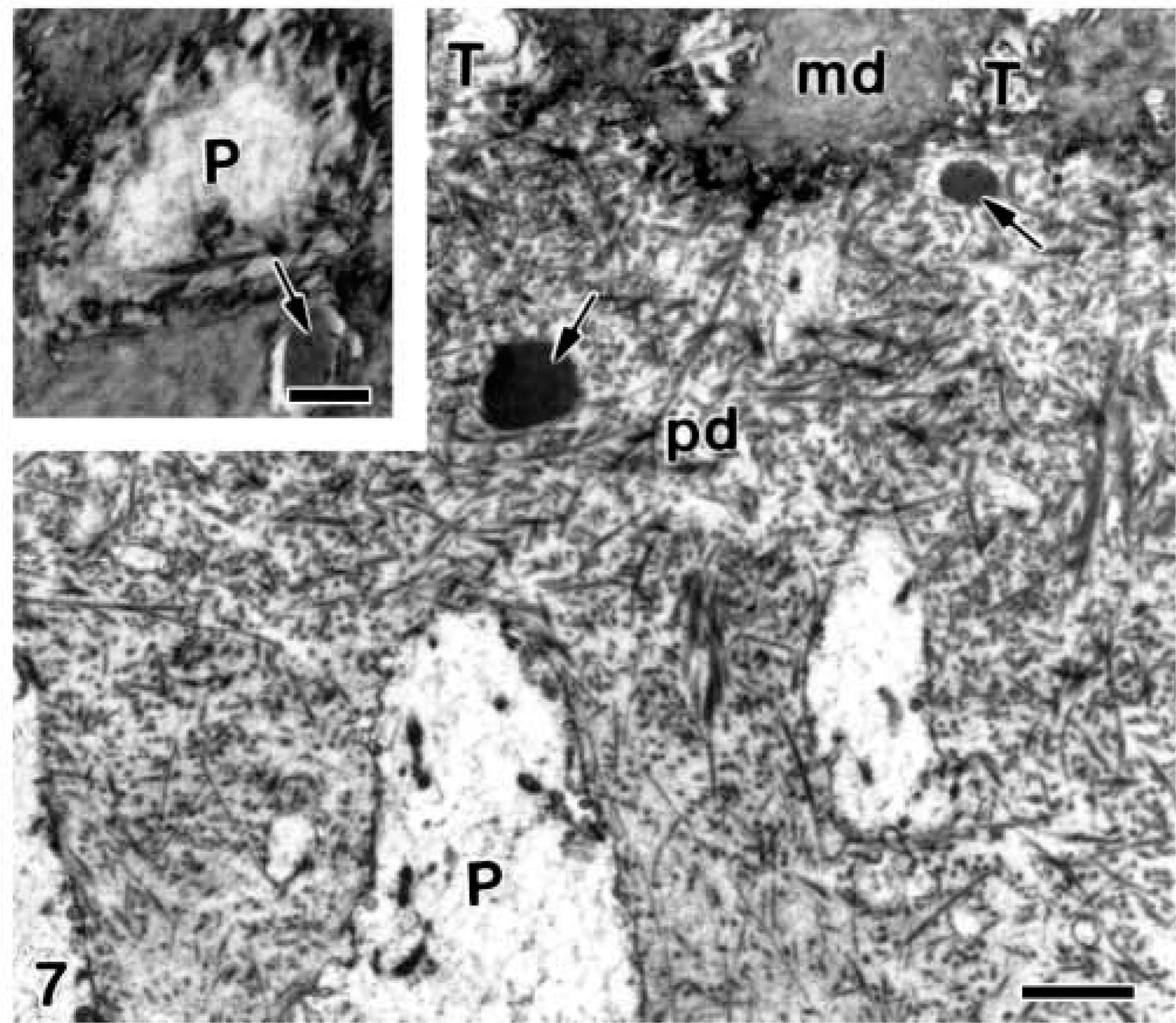
Electron microscopy of alendronate-treated molar tooth germs. A longitudinal section at the interface between the predentin (pd) and the mineralized dentin (md). Electron-opaque granular patches (arrows) are present below two dentinal tubules (T). Inset illustrates a transversal section of the mineralized dentin in which an odontoblast process (
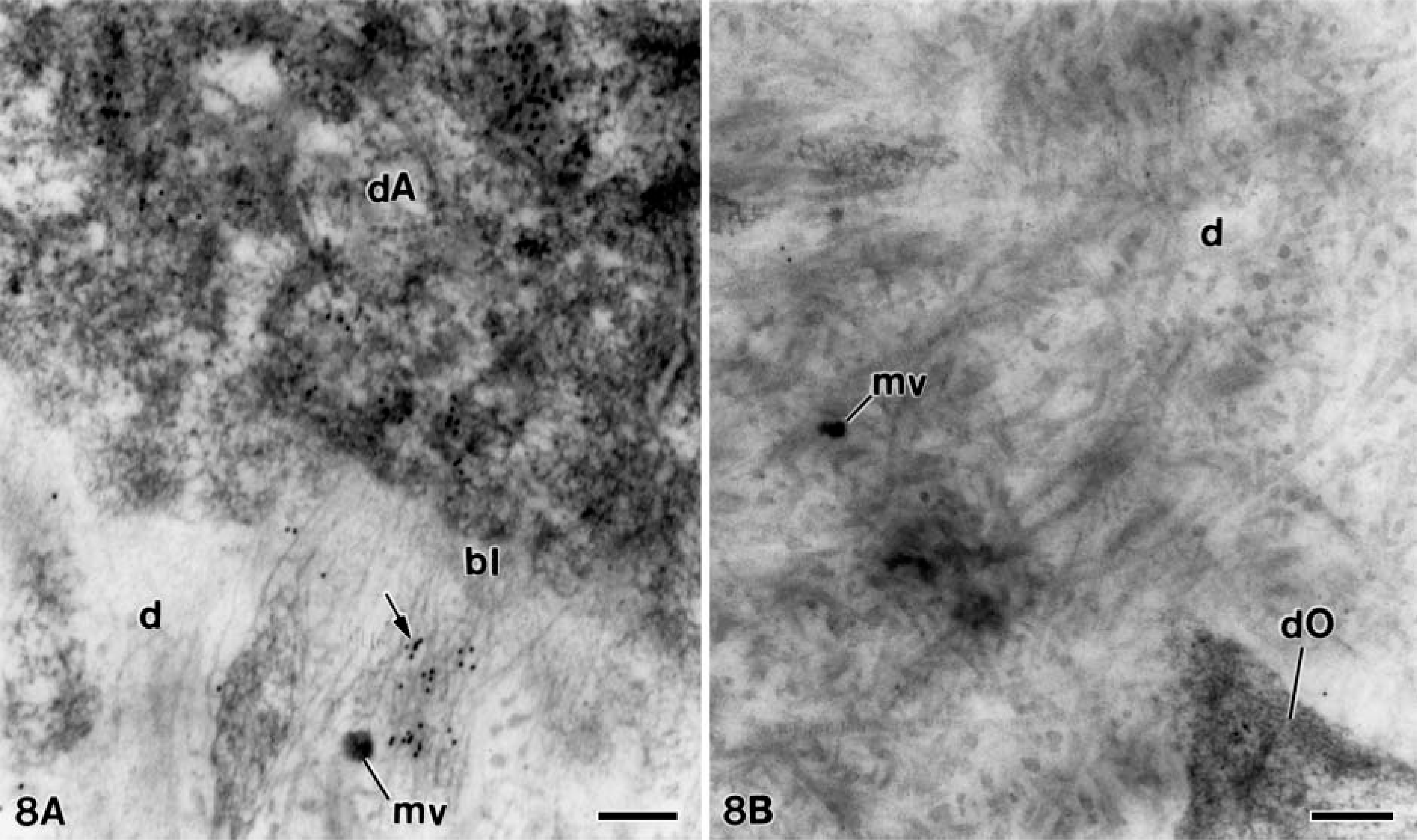
Electron microscopy of non-treated molar tooth germs incubated with the anti-amelogenin antibody. (
At later stages, when the mantle dentin was fully mineralized, the ameloblasts were fully differentiated thus starting the secretory stage of amelogenesis. Antibody-colloidal gold complexes immunoreactive for amelogenin were located at perinuclear multivesicular bodies (Figure 14) and inside secretory granules at the Golgi apparatus of ameloblasts (Figure 15). Amelogenin was also immunodetected forming patches at the intercellular spaces between the distolateral surfaces of ameloblasts, as well as at the developing enamel matrix, where appeared in close relation to the enamel crystals (Figure 16). At these stages, small patches of particles were evident inside the dentinal tubules at the periodontoblastic space, whereas the mineralized dentin was not labeled (Figure 17). The patches observed at the predentin as well as at the intercellular spaces between odontoblasts were also strongly immunoreactive for amelogenin (Figure 18A), whereas the odontoblast cytoplasm was never labeled for amelogenin (Figures 18A and 18B).
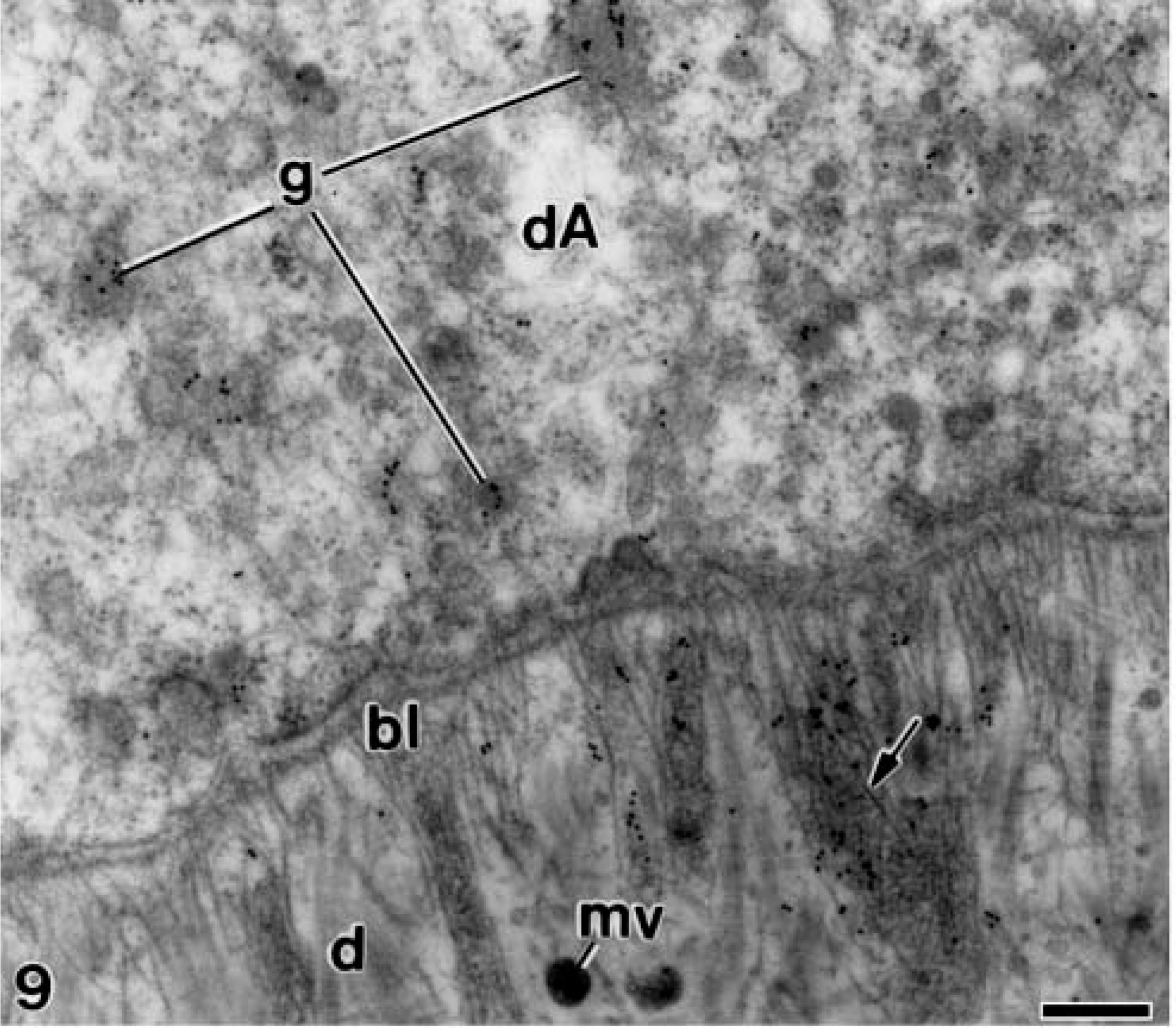
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. The basal lamina (bl) appears continuous at early stage of dentinogenesis. Patches of electron-opaque granular material (arrow) that is immunoreactive for amelogenin appears between the sparse collagen fibrils and the matrix vesicles (mv). The distal portion of an early differentiating ameloblast (dA) contains some secretory granules (g) with gold particles in their interior. d, mantle dentin. Bar = 0.2 μm.
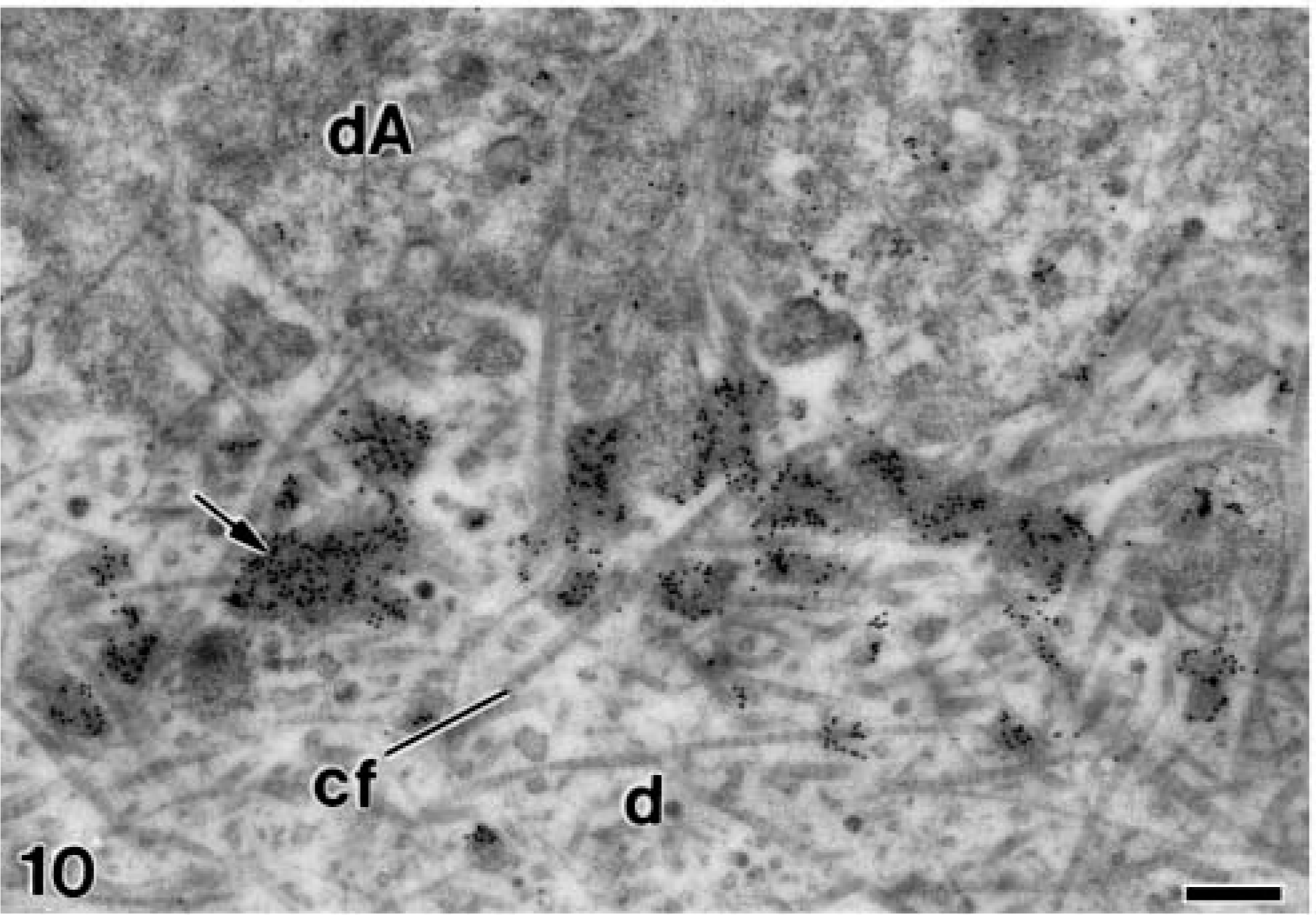
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. A stage of development in which the basal lamina is no longer present and the differentiated ameloblasts (dA) exhibit an undulated distal end. Numerous patches of electron-dense granular material (arrows) contain densely packed colloidal gold particles. cf, collagen fibrils; d, mantle dentin. Bar = 0.25 μm.
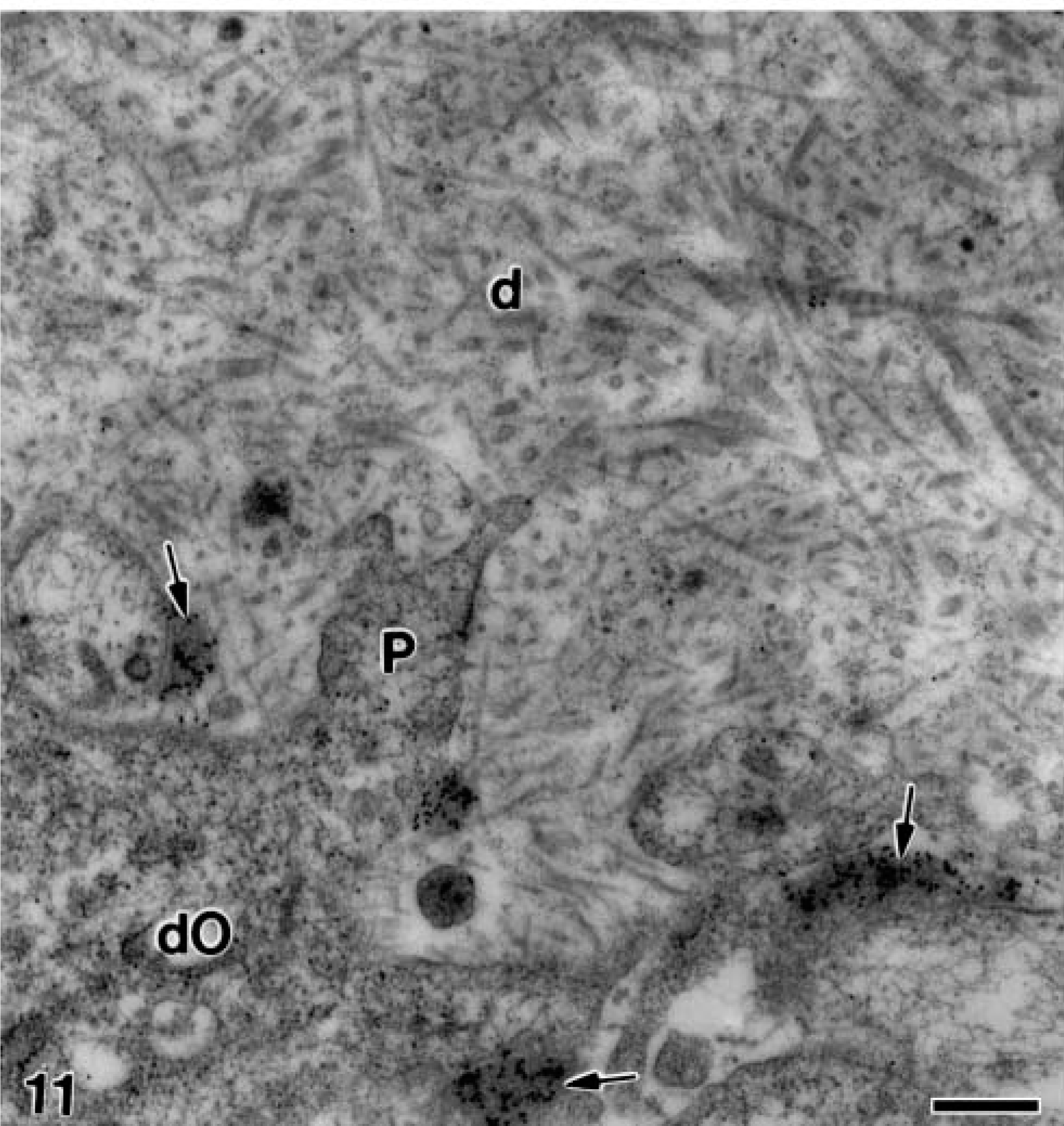
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. The matrix of mantle dentin (d) from a similar stage than that of Figure 10 adjacent to the distal pole of the differentiating odontoblasts (dO) in which numerous gold particles are present at the patches of electron-opaque granular material (arrows), in close relation to the odontoblast plasma membrane. Note some odontoblast projections (P) surrounding the immunoreactive patches. Bar = 0.5 μm.
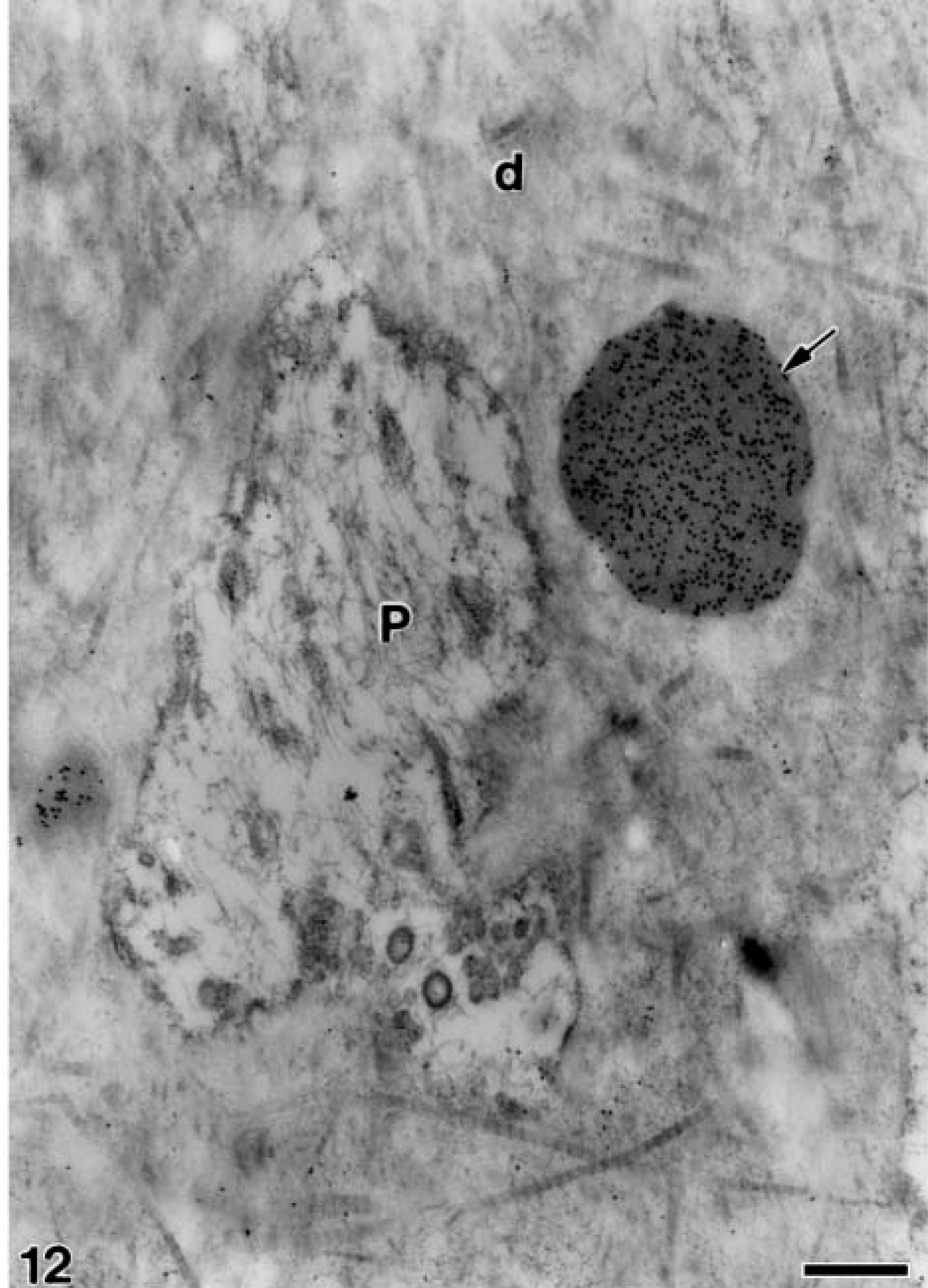
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. A more advanced stage than that of Figure 11 in which a large patch of electron-opaque granular material (arrow) near the odontoblast process (
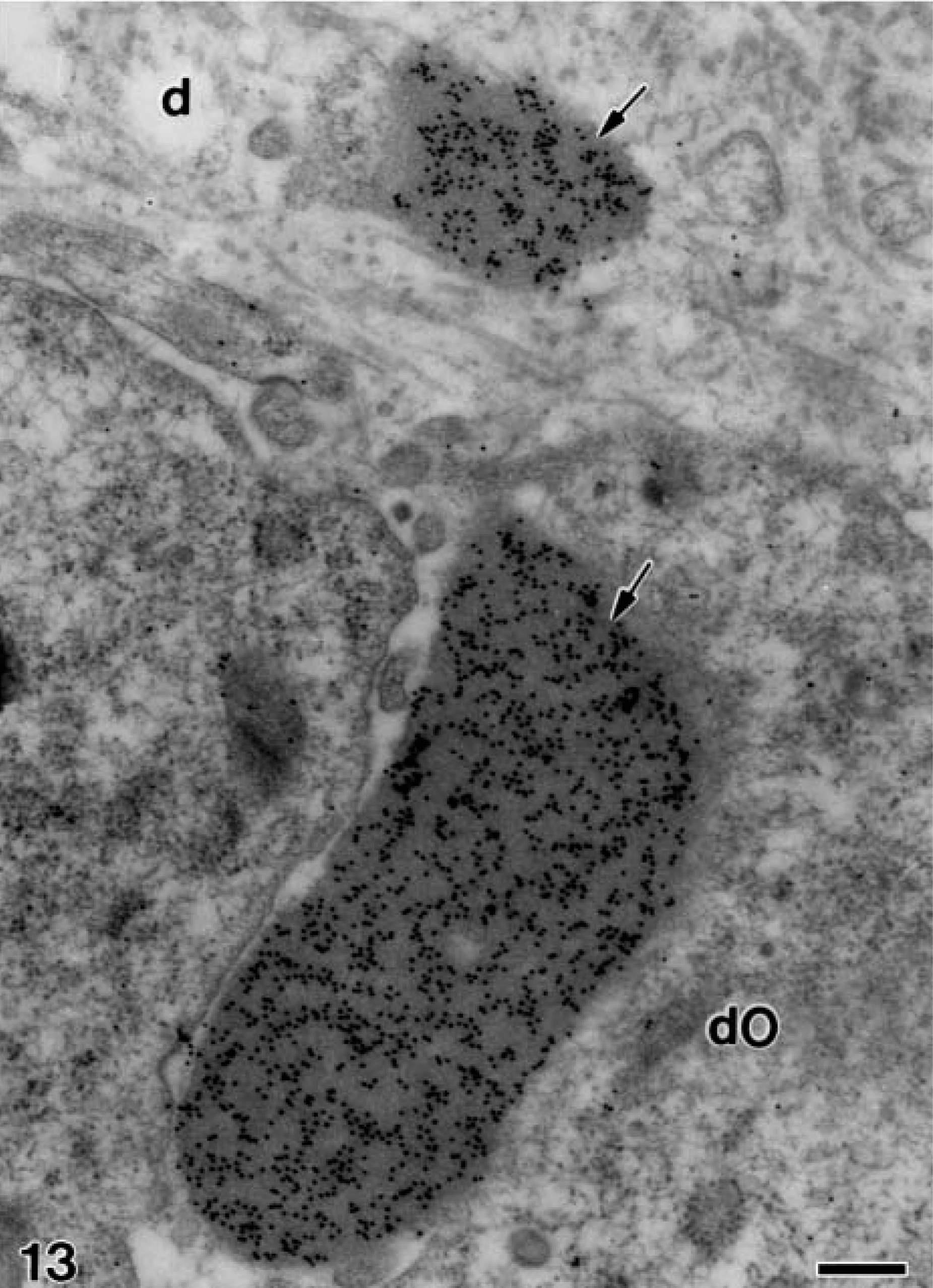
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. The same stage of development as Figure 12 in which two patches of electron-opaque material (arrows) strongly immunoreactive for amelogenin are present at the distal region of differentiating odontoblasts (dO). Note that the large one is located at the intercellular space. d, mantle dentin. Bar = 0.25 μm.
Discussion
The present high-resolution immunocytochemical study showed that sodium alendronate promotes evident disturbances on the early developmental stages of rat molar tooth germs. Numerous amelogenin-containing patches of enamel matrix appeared on the early unmineralized mantle dentin before the onset of the secretory stage of amelogenesis, different from the normal pattern of amelogenin secretion that took place in the control group. Then, amelogenin diffused through the dentin matrix toward the layer of odontoblasts and accumulated in the predentin, assuming an ectopical deposition clearly visible in the alendronate-treated group.
Although it is well established that amelogenin is the major enamel protein and most of the knowledge about its functions has been accumulated in recent years [reviewed in Nanci et al. (1998); Moradian-Oldak (2001)], many questions remain unsolved. The administration of sodium alendronate, which clearly increased the amelogenin secretion by ameloblasts, may help us to elucidate some aspects of the amelogenin function during amelogenesis. As the electron-opaque patches containing granular material strongly immunoreacted with the anti-24-kDa amelogenin antibody, it is reasonable to presume that it consisted of amelogenin. It is possible that when amelogenin is present in high amounts, as in the alendronate-treated specimens, large patches randomly distributed in the unmineralized dentin matrix may be formed due to the hydrophobic N-terminal of the amelogenin molecules similar to the natural aggregation into nanospheres (Fincham et al. 1999). In addition, the amelogenin hydrophobic proprieties, the specific and strong immunolabeling, and the well-preserved ultrastructure of the specimens obtained with the methods applied (Arana-Chavez and Nanci 2001) dismiss the possibility that the patches of granular material found in the present study could be the stippled material caused by poor fixation.
Extracellular granular substance was first described as ‘precursor substance secreted into the extracellular environment where fibrillogenesis and mineralization is taking place’ (Fearnhead 1958), later named ‘stippled material’ (Watson 1960). In the following two decades, studies were carried out investigating the nature of that stippled material during enamel secretion found in different frequency and quantity. However, most of these early ultrastructural studies employed immersion fixation and slow penetrating fixatives such as osmium tetroxide. With the advent of aldehydes and perfusion fixation, stippled material was absent or present in lower amounts, meaning that it depends upon the fixative and the fixation method, i.e., according to Nanci and Warshawsky (1984), stippled material is a fixation artifact.
Immunolabeling of amelogenin over the granular material in the alendronate-treated specimens is consistent with the idea that some chemical agents are capable of provoking secretory alterations on ameloblasts, causing the presence of stippled material. In these cases, the source of the stippled material is not due to poor fixation, but high amounts of amelogenin are actually at the extracellular matrix. There are previous reports of the presence of stippled material when rats were injected with fluoride, strontium, or cobalt ions (Neiman and Eisenmann 1975; Chen and Eisenmann 1984). Similar accumulates of amelogenin appearing as large as patches of granular material have also been detected at the cemento-enamel junction during the earliest stages of root formation in porcine teeth (Bosshardt and Nanci 2004).
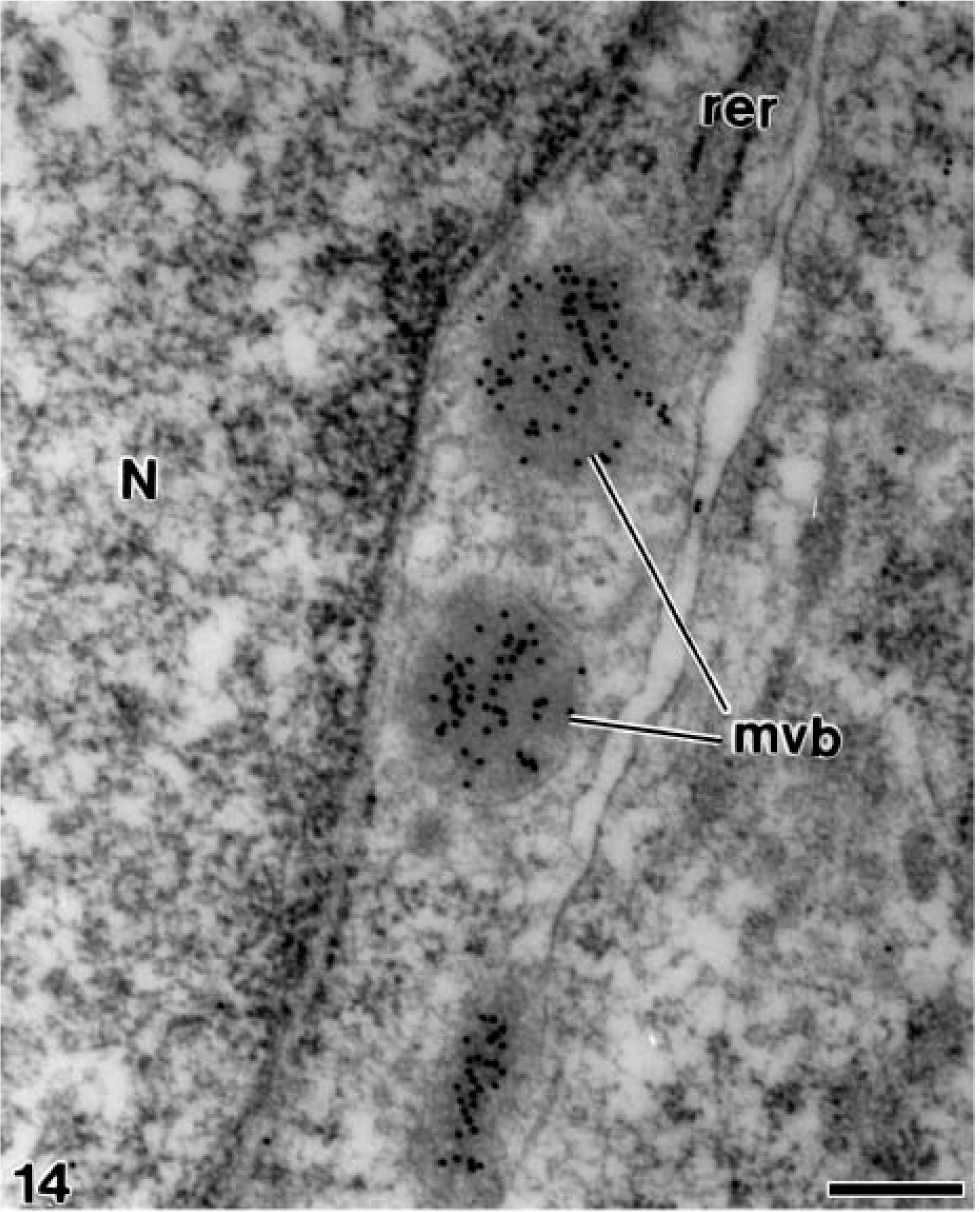
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. Three multivesicular bodies (mvb) containing numerous gold particles are present laterally to the nucleus (
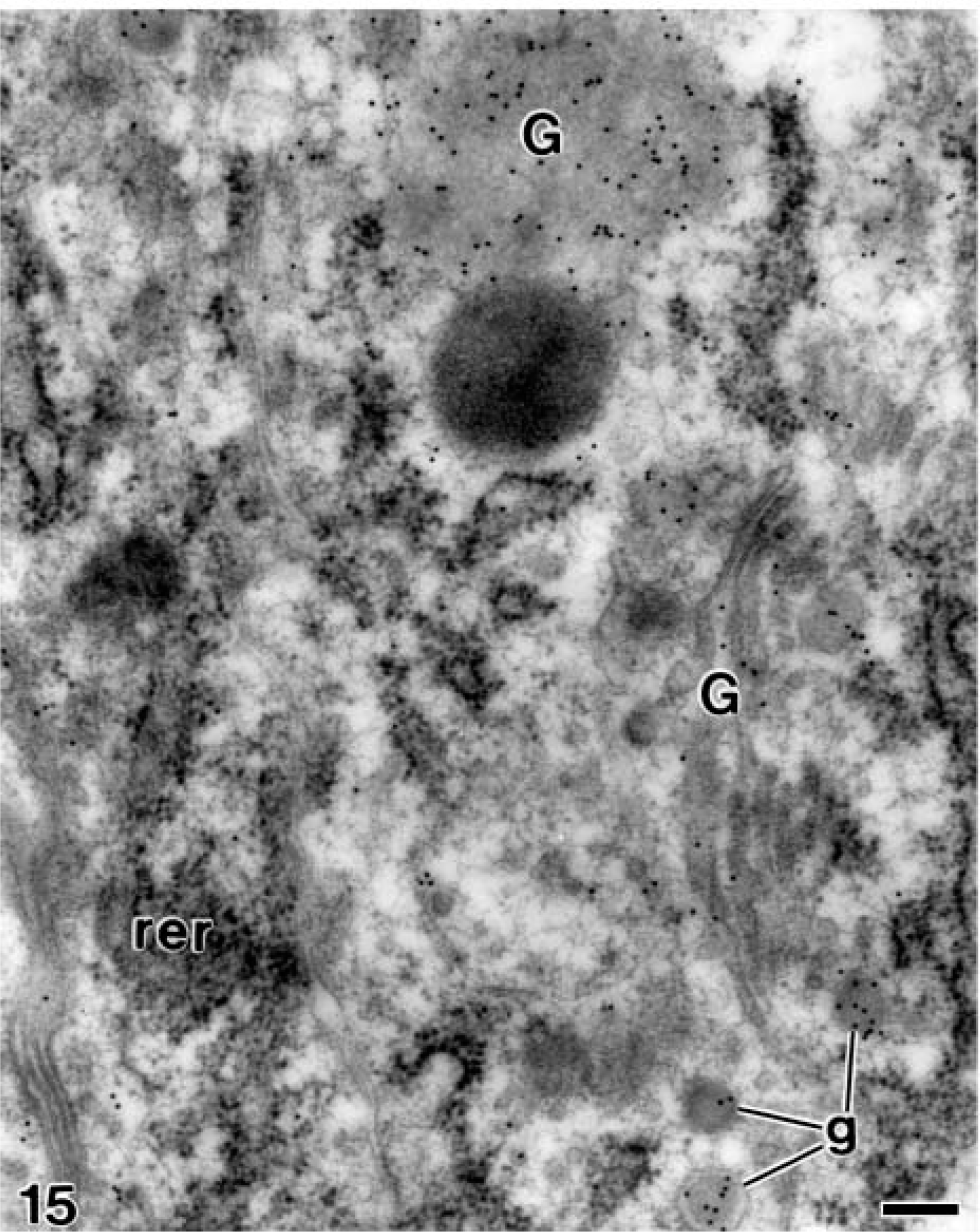
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. Numerous gold particles are observed at the Golgi complex (G) of a fully differentiated ameloblast. rer, rough endoplasmic reticulum; g, secretory granules Bar = 0.2 μm.
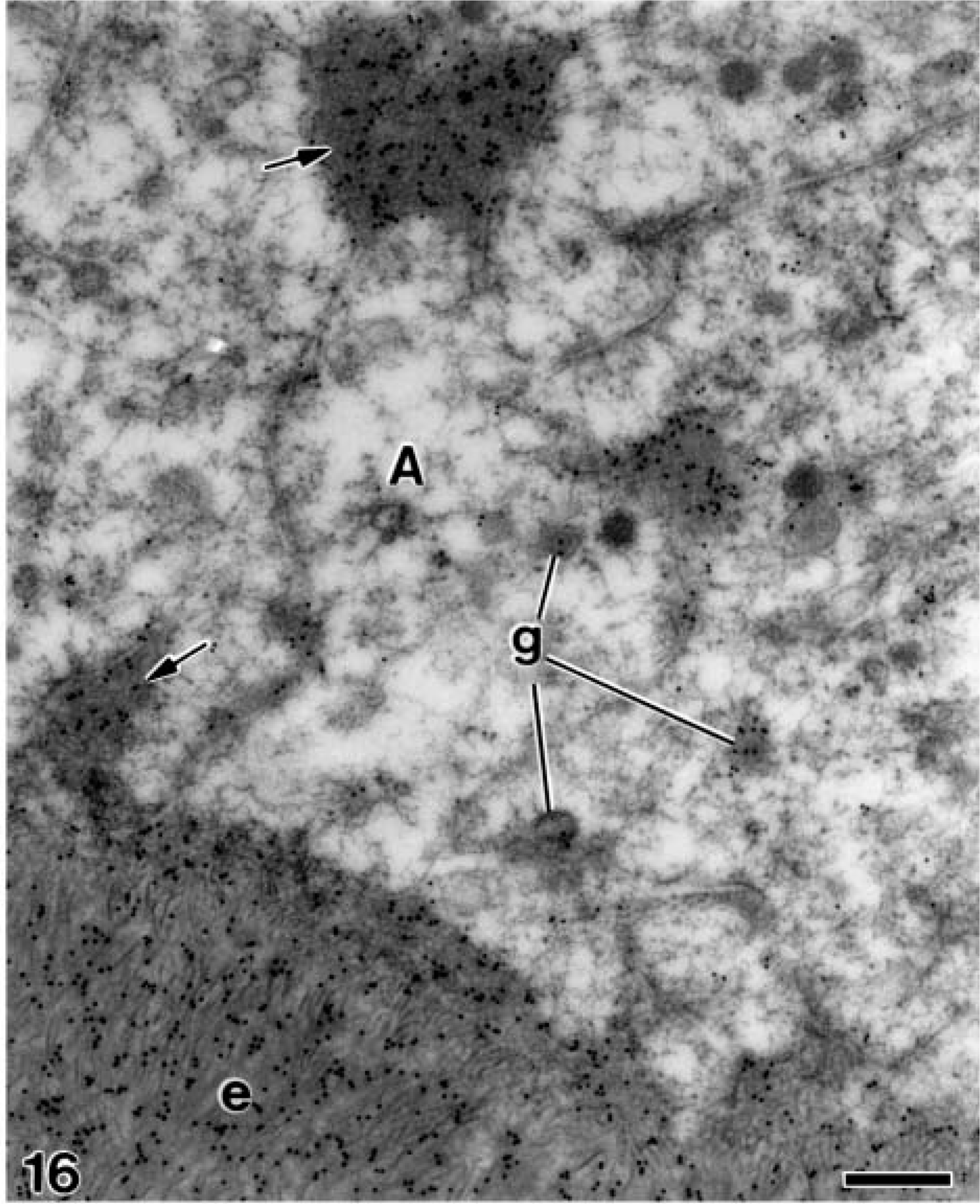
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. The distal region of secretory ameloblasts (A) contains secretory granules (g) immunoreactive for amelogenin. Numerous gold particles are also seen on the patches of electron-opaque granular material (arrows) present at the intercellular regions as well as at the secreting enamel matrix (e). Bar = 0.25 μm.
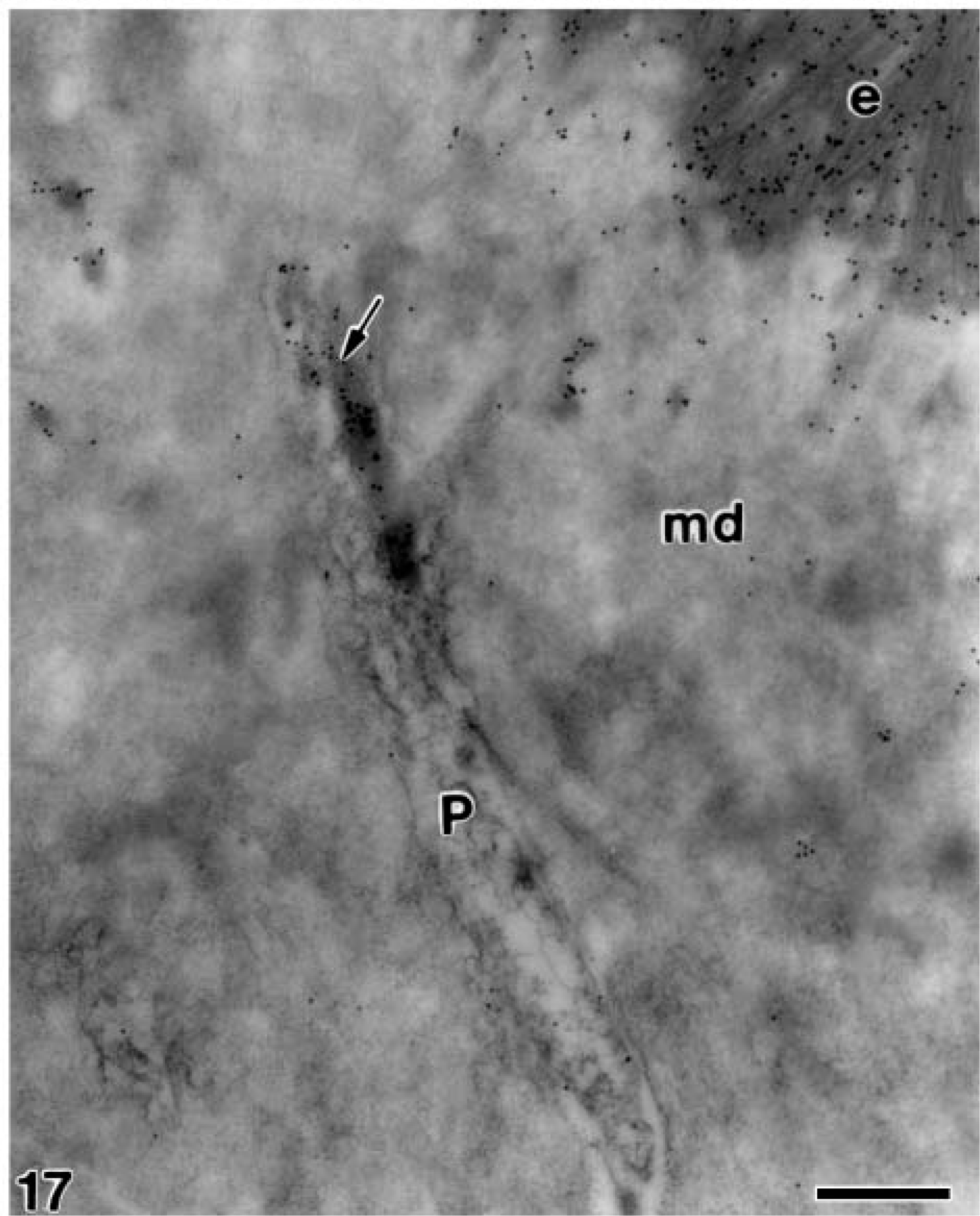
Electron microscopy of alendronate-treated molar tooth germs incubated with the anti-amelogenin antibody. Patches of gold particles (arrow) are present at the interior of a dentinal tubule, adjacent to the odontoblast process (P) at the interface between the mineralized dentin (md) and enamel (e) Bar = 0.5 μm.
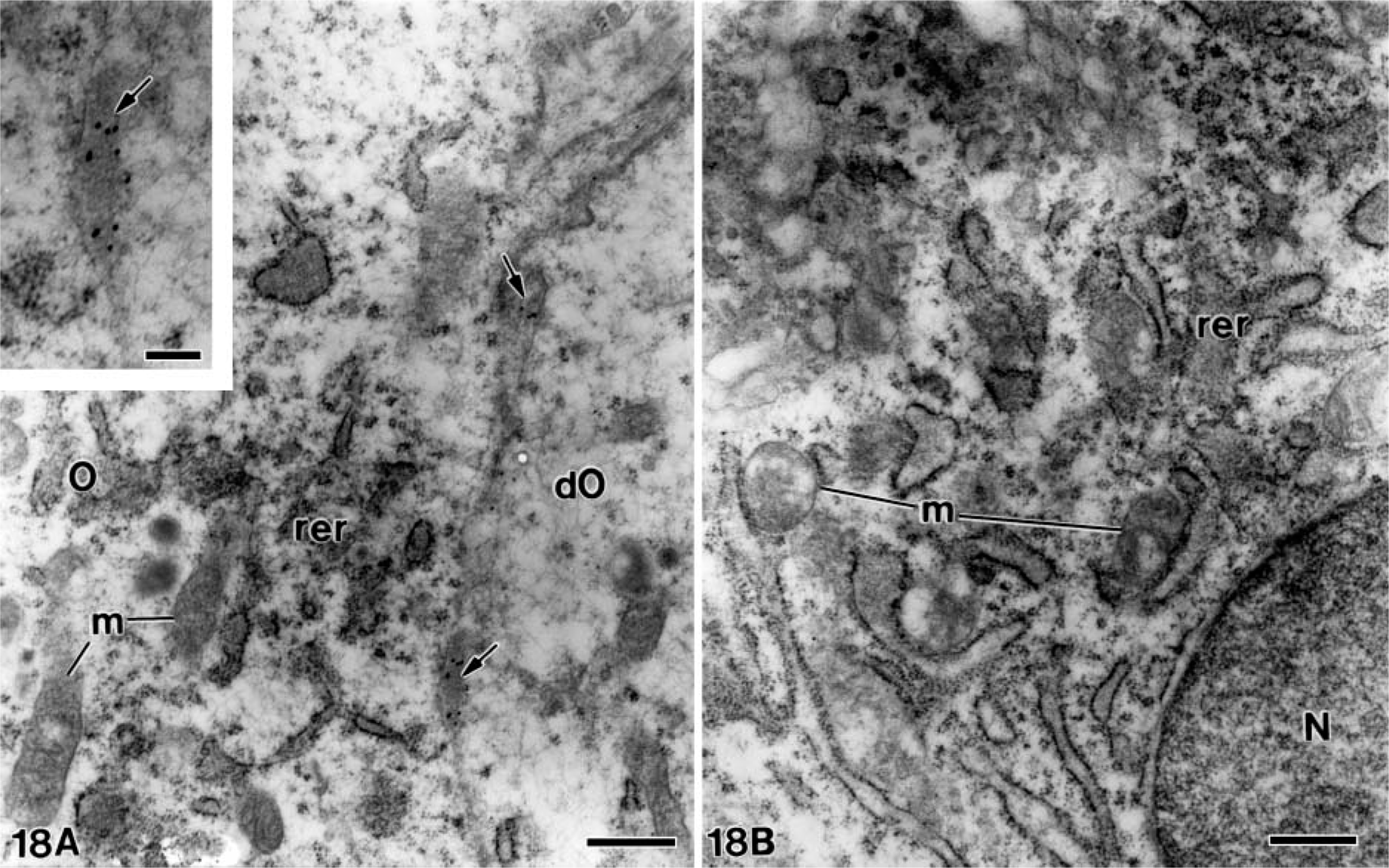
Electron microscopy of alendronate-treated molartooth germs incubated with the anti-amelogenin antibody. (
Immunoreactivity for amelogenin in both the alendronate-treated and the control specimens was clear at the early stages of ameloblast differentiation when these cells had not yet reversed their polarity and the underlying basal lamina was continuous. A short time later, as mantle dentin started its development, the intensity of immunolabeling for amelogenin in the alendronate-treated molar tooth germs became evidently stronger than in the control group. The exact role for the releasing of amelogenin before the secretory phase of amelogenesis is not clear. It has been reported that the small splice products of the amelogenin gene [A+4] and [A-4] may be secreted in small amounts to act differently as signaling molecules affecting ameloblasts and odontoblasts. [A-4] induces odontoblast differentiation but also hinders the differentiation of ameloblasts (Veis 2003; Tompkins et al. 2005a). [A-4] biotin staining was observed in both differentiating ameloblasts and odontoblasts but vanished at the point of dentin mineralization. The lysosome-associated membrane protein 1 (LAMP-1) is present at the plasma membrane as well as the endosomal/lysosomal membrane of cells (Lippincott-Schwartz and Fambrough 1987), and it is believed to be responsible for the internalization of [A-4] into C2C12 cells (Tompkins et al. 2005b). Thus, amelogenin peptides may induce, together with some growth factors such as TGF-1, BMP 2 and 4, and IGF, the postmitotic outer cells of the dental papilla to differentiate into odontoblasts (Slavkin et al. 1988; Thesleff et al. 1995). The increased amount of 24-kDa amelogenin detected during the presecretory phase, before or at the initial stages of dentinogenesis, has not affected the supposing signaling proprieties, because no abnormality was found at the odontoblast phenotype. The diffusion of small amounts of 24-kDa amelogenin toward dentin has been reported in normal odontogenesis (Nanci et al. 1992; Nakamura et al. 1994; Ruch et al. 1995), but it is generally considered an occasional event (Arana-Chavez and Nanci 2001).
Because there was no immunogold labeling for 24-kDa amelogenin within the cytosol or over secretory organelles of odontoblasts, it is presumed that the high amount of amelogenin labeled within the unmineralized dentin matrix and between the odontoblasts in the alendronate-treated specimens is not a result of the synthesis and secretion by these odontoblasts. Even though it has been reported that odontoblasts express amelogenin mRNA (Veis et al. 2000; Oida et al. 2002) and that amelogenin is expressed in relatively small amounts in a temporal-dependent pattern by rodent odontoblasts (Papagerakis et al. 2003), the odontoblasts were never labeled in either the control or treated specimens, thus confirming previous studies with the same antibody (Nanci et al. 1996; Bosshardt and Nanci 1997; Arana-Chavez and Nanci 2001). At least 14 isoforms are translated by an alternative splicing that takes place during amelogenin processing (Tompkins et al. 2005b). Thus, previous studies may have detected other amelogenin molecules in odontoblasts different from that recognized by the antibody used here. In addition, expression of mRNA, where a signal is detected by in situ hybridization, does not necessarily imply that it will be translated into its final protein, or that some nascent molecules may be intracellularly degraded immediately after synthesis by enamelysin (MMP-20) or other matrix metalloproteinases (MMPs) [reviewed in Nanci and Smith (1992); Bourd-Boittin et al. (2004)]. Thus, it is reasonable to assume that the total amount of amelogenin labeled at the forming dentin and between odontoblasts in alendronate-treated specimens was synthesized and secreted by the developing ameloblasts, which yields the amelogenin diffusion toward dentin. Indeed, differentiating and fully differentiated ameloblasts contained many secretory granules labeled with the chicken egg yolk anti-24-kDa amelogenin antibody, whereas the gold particles at the odontoblast layer were observed only at the intercellular spaces.
As odontogenesis progressed, the patches of amelogenin observed in the treated specimens continued to be significantly larger than those observed in the control group. The patches were detected at the forming enamel layer, at the dentinal tubules of the mineralized dentin, and at the predentin near the distal end of the odontoblast. Because the examined ultrathin sections represent individual moments of odontogenesis, which is a dynamic and continuous process, it is likely that the patches represented the ‘footprints’ left by amelogenin during its diffusion pathway toward the dentin.
Although bisphosphonates possess a well-known antiosteoclastic effect (Vasikaran 2001; van Beek et al. 2002; Reszka and Rodan 2004), it has also been recognized that they have high dose-dependent physicochemical proprieties (Baumann et al. 1978) that interfere with the transformation of amorphous calcium phosphate into hydroxyapatite (Fleisch 1998). On the other hand, bisphosphonates have effects on matrix formation (Larson 1974; Van den Boss and Beertsen 1987; Ogawa et al. 1989; Takano et al. 1998; Fuangtharnthip et al. 2000) including disrupting effects on mineralization. In fact, several data have been reported about dentin and enamel hypomineralization in rat incisors induced by HEBP administration: the inhibition of dentin mineralization subsequently promotes the failure of enamel protein secretion and deposition resulting in clear enamel-free zones that interrupt the continuity of the enamel layer (Ogawa et al. 1989; Fuangtharnthip et al. 2000). However, as hydroxyapatite crystals have been clearly discerned in dentin and enamel, it is likely that the nucleation of minerals is not affected by the sodium alendronate treatment. Thus, it may have a lower disrupting effect on mineralization than other bisphosphonates such as HEBP, which produces several enamel hypoplasias when administered SC at similar doses. Whereas those studies evaluated both dentinogenesis and amelogenesis in decalcified rat incisors under light microscopy (Ogawa et al. 1989; Fuangtharnthip et al. 2000), our specimens were intentionally left undecalcified to discern the appearance and distribution of mineral crystals in both dentin and enamel matrices.
Taking the morphological and immunocytochemical results obtained here, it is likely that the main effect of alendronate is a break into the equilibrium of secretion/degradation of amelogenin in enamel matrix that is controlled by ameloblasts throughout the secretory stage of amelogenesis. Indeed, although our results suggest that molecules of amelogenin are oversecreted by ameloblasts, it is possible that they could be insufficiently degraded during the secretory stage of amelogenesis. Because bisphosphonates have been reported to inhibit MMPs (Heikkila et al. 2002), they may act during odontogenesis by affecting the function of MMP-20 (enamelysin) or other enzymes that act during the secretory stage of amelogenesis.
In addition to the above considerations, it is possible that similar morphological alterations would occur in human teeth after administration of high doses of alendronate during odontogenesis. The available experimental studies investigating the effects of bisphosphonates on tooth formation have been made by examining rat incisors (Fuangtharnthip et al. 2000; Yamada et al. 2000), which display continuous growth and eruption. The analysis of rat molar tooth germs carried out here offers the advantage of having the several stages of odontogenesis at a same slope of a given developing cusp. In addition, this model permits a better correlation to the sequential events of odontogenesis that take place in human teeth. It may be important because sodium alendronate is a drug broadly used in therapies for many bone pathologies, including its administration to pregnant women or children (McCarthy et al. 2002; Maasalu et al. 2003). In fact, the stages of molar tooth germs examined in the newborn rats take place intrauterine during the odontogenesis of human primary (deciduous) teeth and during early childhood for development of permanent teeth.
Footnotes
Acknowledgements
This work was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo (Fapesp) (01/13782-0 and 04/05831-9) and Conselho Nacional de Pesquisas (CNPq) (Brazil).
The authors thank Dr. Antonio Nanci (University of Montreal, Montreal, Canada) for supplying the anti-24-kDa rat amelogenin antibody and Miss Sylvia Zalzal for preparing the protein A-gold complex. We also thank Mr. Gaspar F. de Lima for ultrathin sectioning and Mr. Edson Oliveira for printing the electron micrographs.
