Abstract
Nitric oxide synthase (NOS) and interleukin-6 (IL-6) are constitutively expressed in hypothalamic cells. However, phenotypic and functional aspects of these cells remain unknown. We have studied the expression pattern of these two molecules in hypothalamic cells expressing corticotropin-releasing factor (CRF) and arginin-vasopressin (AVP), two major regulatory peptides in the hypothalamus-pituitary system, using immunofluorescence, intracerebroventricular injection of colchicine, and the study in parallel of the labeling pattern of axons in the median eminence. Within AVP cells, we distinguished two different populations: large, intensely stained AVP cells coexpressing IL-6; and large, intensely stained AVP cells coexpressing IL-6 and NOS. Within the CRF cells, we distinguished three different populations: large, intensely stained CRF cells immunonegative for AVP, NOS, and IL-6; large cells weakly stained for CRF and AVP, immunopositive for NOS and immunonegative for IL-6; and small cells intensely stained for CRF and AVP and immunonegative for IL-6 and NOS. In addition, we also found AVP cells containing IL-6 in the suprachiasmatic nucleus. These results suggest that neuronal NOS and IL-6 may be involved in different modulatory processes in hypophysiotropic and non-hypophysiotropic cells.
C
The proinflammatory cytokine interleukin-6 (IL-6) and the short-lived, unstable free radical nitric oxide (NO) have been involved in neuroendocrine functions playing regulatory roles on the HPA axis. IL-6 induces the synthesis and secretion of CRF (Lyson and McCann 1991, 1992; Navarra et al. 1991) and AVP (Mastorakos et al. 1994; Raber et al. 1997) at the hypothalamic level and ACTH and glucocorticoid secretions directly at the pituitary (Naitoh et al. 1988; Lyson and McCann 1992) and adrenal levels, respectively (Salas et al. 1990). In addition, a recent study shows that IL-6 is coexpressed with AVP in SON and PVN cells (Ghorbel et al. 2003). On the other hand, nitric oxide synthase (NOS), the enzyme responsible for the synthesis of NO, has been detected at different levels of the HPA axis, including oxytocin, AVP, and CRF cells in the PVN and SON (Bredt et al. 1990; Arevalo et al. 1992; Calka and Block 1993; Miyagawa et al. 1994; Sanchez et al. 1998; Nylen et al. 2001), axon terminals in the posterior pituitary lobe (Sagar and Ferriero 1987; Ceccatelli et al. 1993), and gonadotrophs and folliculostellate cells in the anterior pituitary lobe (Ceccatelli et al. 1993; Gonzalez-Hernandez and Gonzalez 2000); and also in the SCN (Decker and Reuss 1994; Reuss et al. 1995; Wang and Morris 1996; Chen et al. 1997; Caillol et al. 2000), where NO has been involved in the transduction of retinal inputs (Ding et al. 1994; Watanabe et al. 1994; Weber et al. 1995; Amir and Edelstein 1997; Mitome et al. 2001; Golombek et al. 2004). The administration of NO-related drugs in different experimental models has suggested that it exerts a tonic modulatory action on the basal activity of the HPA axis (Chiodera et al. 1996; Srisawat et al. 2000; Givalois et al. 2002; Stern and Zhang 2005), so that IL-6 and NOS appear as two modulatory molecules constitutively expressed in hypothalamic cells and potentially exerting antagonistic actions. It would be interesting to know whether all hypothalamic AVP cells, including those in the SCN, express IL-6, whether IL-6 is also expressed in hypothalamic cells containing other peptides, and whether IL-6 and NOS are coexpressed in the same hypothalamic cell subpopulation.
To improve our knowledge of the role of IL-6 and the neuronal isoform of NOS in the hypothalamus, and particularly in the AVP and CRF systems, we have investigated the expression of IL-6 and NOS in CRF-and AVPergic neurons in different rat hypophysiotropic regions (PVN, SON, and ME) and the SCN. We have used immunohistochemistry, double immunofluorescence, and intracerebroventricular injection of colchicine, a chemical that disrupts the assembly of microtubulin and blocks the axonal transport, and that is frequently used to increase the immunostaining signal of peptides in neuron somata (Norstrom 1975; Banks 1976; Lantos et al. 1995)
Materials and Methods
Animals and Tissue Preparation
A total of 22 adult male Sprague-Dawley rats weighing 270-290 g were housed under conditions of controlled temperature (24 ± 1C) and light (8:00
Tissue Processing
Sections were collected in four to six parallel series and processed for single immunostaining or double immunofluorescent labeling. Floating sections for single immunostaining were immersed for 20 min in 3% H2O2 to inactivate endogenous peroxidase, and incubated for 60 min at room temperature (RT) in the preincubation solution (PIS): 4% normal goat serum (NGS) (Jackson ImmunoResearch; West Grove, PA) or 4% normal donkey serum (NDS) (Jackson Immuno-Research) in 0.1 M PBS, pH 7.4, containing 0.05% Triton X-100 (Sigma), and incubated overnight in PIS containing one of the following primary antibodies: a rabbit anti-AVP polyoclonal antibody (AVPr), 1:4000 (INC; Aurora, Ohio); a rabbit polyclonal antibody against amino acids 25-41 with N-terminally added lysine of the C terminus fragment of human CRF (CRFr), 1:2000 (Sigma); a goat anti-CRF polyoclonal antibody against the C terminus fragment of human CRF (CRFg), 1:300 (Santa Cruz Biotechnology; Santa Cruz, CA); a goat anti-IL-6 polyclonal antibody (IL-6 g), 1:600 (Santa Cruz Biotechnology); a mouse anti-neuronal NOS monoclonal antibody (NOSm), 1:2000 (Sigma); or a rabbit anti-neuronal NOS polyclonal antibody (NOSr), 1:2000 (Zymed; San Francisco, CA). After several rinses, sections were incubated for 2 hr in either biotinylated goat anti-mouse antiserum (1:1200; Jackson ImmunoResearch), biotinylated goat anti-rabbit antiserum (1:1200; Jackson ImmunoResearch), or biotinylated donkey anti-goat antiserum (1:1000; Jackson ImmunoResearch) and 1:200 NGS or 1:200 NDS in PBS. Immunoreactions were visible after incubation for 1 hr at RT in ExtrAvidin (1:5000; Sigma) in PBS, and after 10 min in 0.005% DAB and 0.001% H2O2 in cacodylate buffer, pH 4.5. After several rinses in PBS, slides were dehydrated, cleared in xylene, and coverslipped with DPX (BDH Chemicals; Poole, England).
For double labeling, sections were immersed for 60 min in PIS, and overnight in PIS containing a mixture of two of the primary antibodies used in the single immunostaining made in different species and at double concentration. They were combined in six different pairs according to the following schedule: AVPr/CRFg, AVPr/IL-6 g, AVPr/NOSm, CRFr/IL-6 g, CRFr/NOSm, and NOSr/IL-6 g. After several rinses, immunofluorescent labeling was visible after incubation for 3 hr in a mix containing either 1:150 lissamine rhodamineconjugated goat anti-rabbit IgG (Jackson ImmunoResearch) and 1:150 fluorescein isothiocyanate-conjugated goat antimouse IgG in PBS containing 1:200 NGS, or 1:150 Cy2-conjugated donkey anti-goat (Jackson ImmunoResearch) in PBS containing 1:200 NDS followed by 1:150 lissamine rhodamine-conjugated goat anti-rabbit IgG (Jackson ImmunoResearch) in PBS containing 1:200 NGS. After several rinses, sections were mounted on gelatinized slides, air dried, coverslipped with Vectashield (Vector Laboratories; Burlingame, CA), and examined under epifluorescence microscopy using appropriate filters. For each type of immunohistochemistry and immunofluorescence, control experiments were performed by removing the primary antibodies, resulting in negative staining. The contrast in fluorescent images was improved by using the Adobe Photoshop program.
Analysis
The topographic distribution of stained neurons was studied in immunohistochemical and immunofluorescent material following the hypothalamic division of Nylen et al. (2001) and Xiao et al. (2005) for the hypophysiotropic nuclei and Moore et al. (2002) for the SCN. To get a semiquantitative estimate of the topographical distribution and the neurochemical profile of the different cell populations, single- and double-labeled neuronal profiles were counted in seven rats. In each rat, eight randomly selected sections 120-160 μm apart in the rostroacudal axis were examined. Only cell profiles including nucleus were analyzed, and profile counts were corrected using Abercrombie's formula (Abercrombie 1946; Guillery 2002). Quantitative data are expressed as mean ± SEM.
Results
Because our results involve hypothalamic nuclei with different functional meanings, to better explain our findings, we will first describe those concerning hypophysiotropic nuclei, including mostly PVN, SON, and ME, and those found in the SCN.
Hypophysiotropic Nuclei
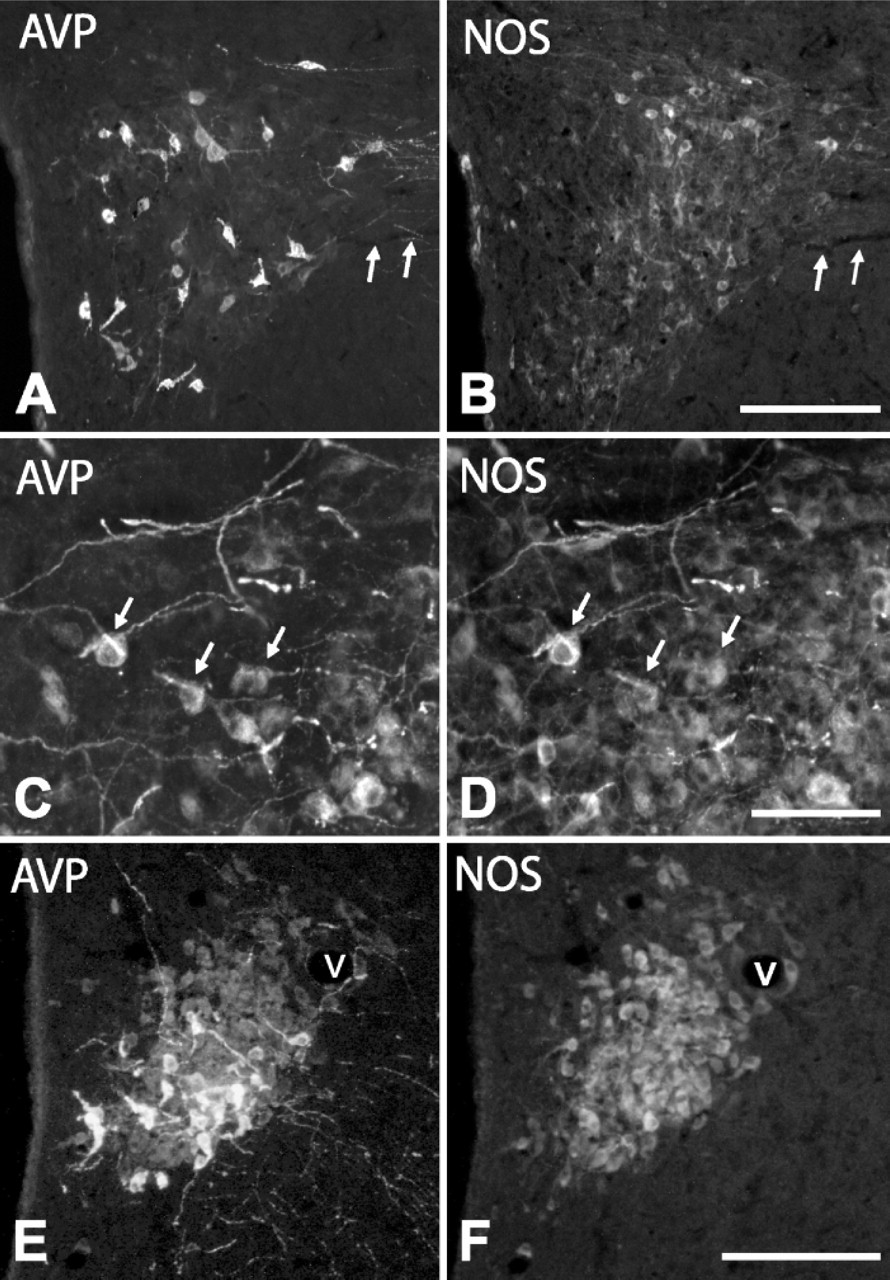
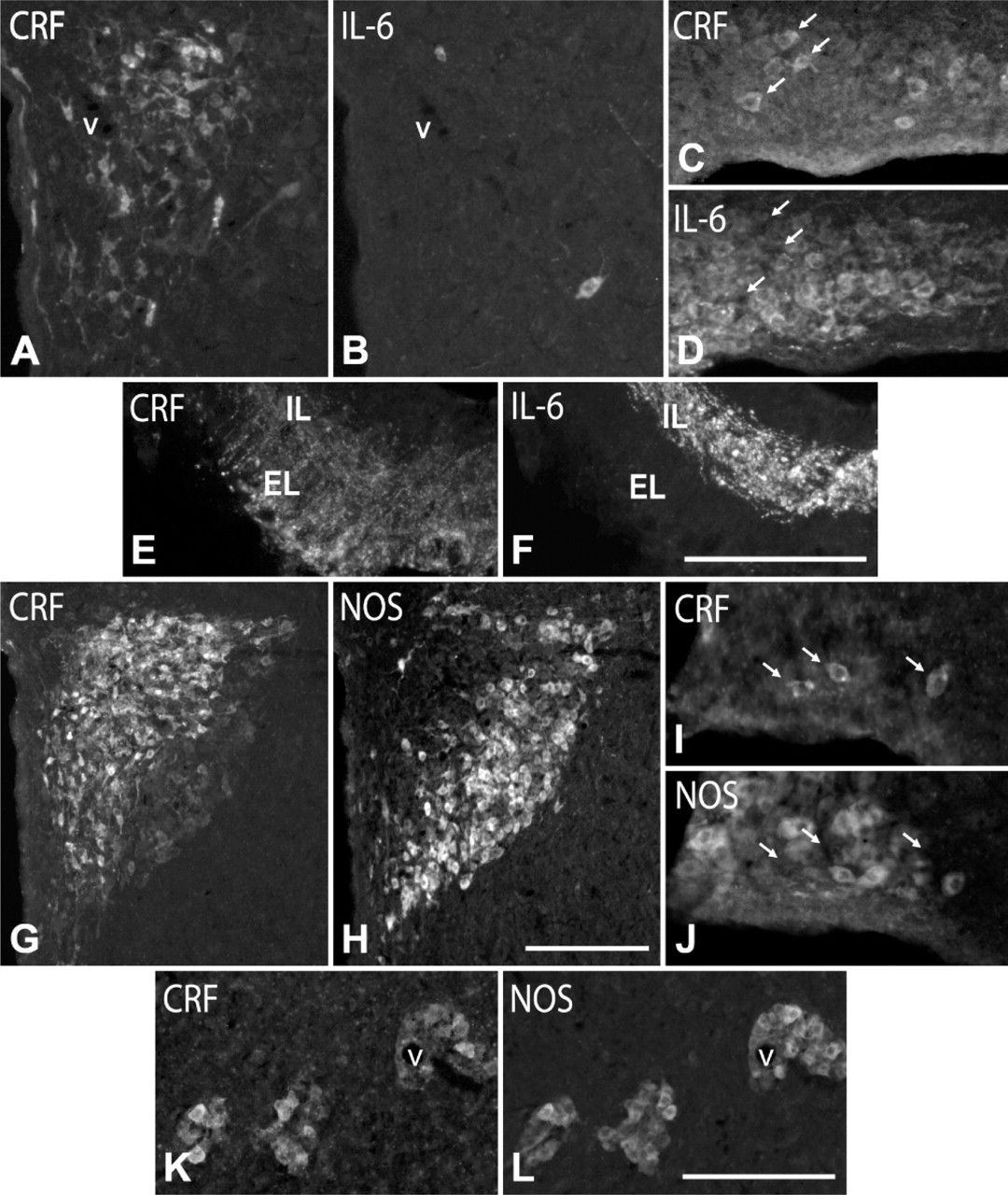
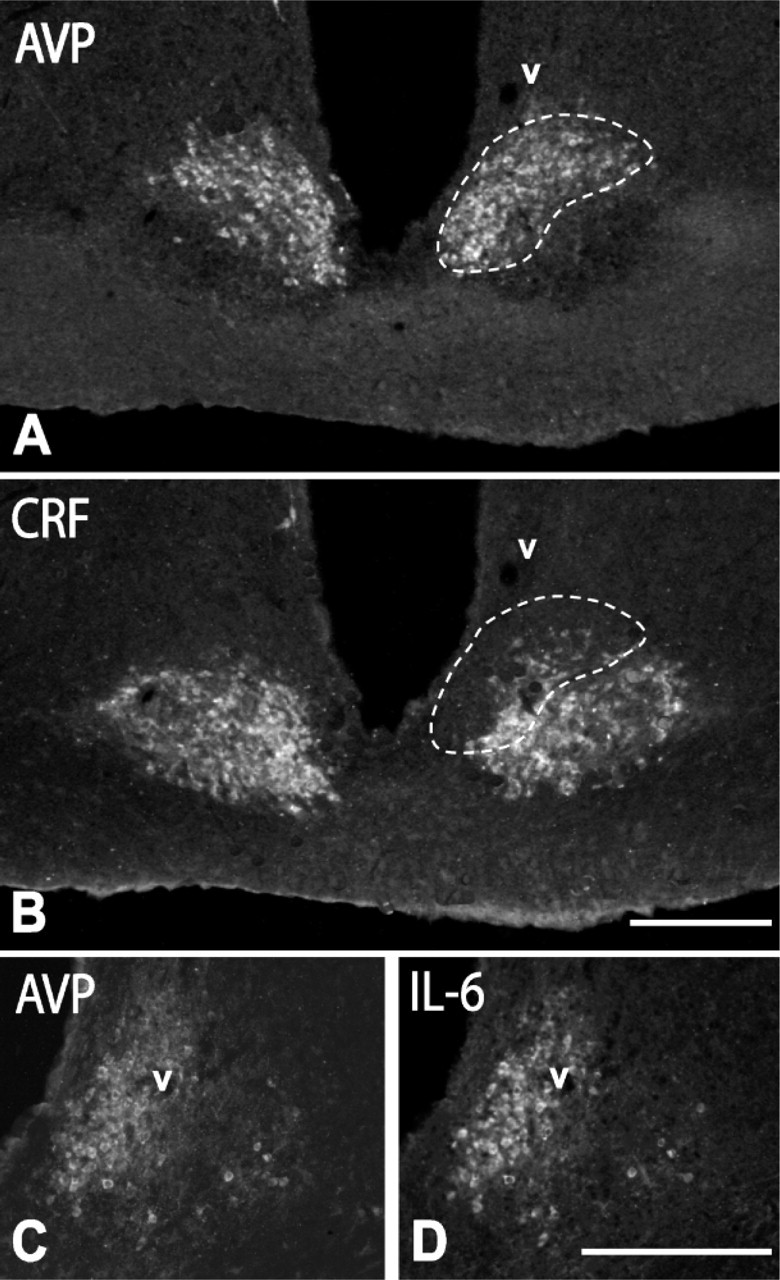
AVP and CRF Immunostaining in Cells and Fibers. Under normal conditions (colchicine-untreated rats), AVP-positive cells were present in the PVN (Figure 1A), SON, and, in a lower number, throughout the lateral hypothalamic region (see Table 1). These neurons were large in size (diameter of 22.6 ± 2.7 μm), and most of them localized in the posterior magnocellular division of the PVN. In the ME, axons showed an intense AVP immunoreactivity (Figure 1C). They formed a dense transversally cut bundle running to the posterior lobe of the pituitary gland in its internal layer, whereas in the external layer, they formed a separate plexus of varicose terminals. No CRF-positive cells were detected in the hypothalamus of these animals (Figure 1B), but CRF immunoreactivity was evident in the terminal plexus of the external zone of the ME (Figure 1D). AVP cells increased in number (approximately 260%; Figure 1E; Figures 2A, 2G) and CRF cells became evident in colchicine-treated rats (Figure 1F; Figure 2H; Table 1). We distinguished three types of AVP cells according to their size and labeling intensity: large (diameter of 22.6 ± 2.7 μm), intensely stained cells, which constituted 39 ± 5% of the total number of AVP cells; large, weakly stained cells (48 ± 6%); and small (diameter of 14.2 ± 2.5 μm), weakly stained cells (13 ± 5%). Large, intensely AVP-stained cells were preferentially localized in the dorsolateral region of the posterior magnocellular division of the PVN, where they were densely clustered (Figure 1E; Figure 2A) and, to a lower degree, in the ventral aspect of the anterior magnocellular divison (Figure 2G; Figure 3A; Figure 4E). Large, weakly stained cells displayed an intermedio-lateral distribution particularly conspicuous just medial and dorsal to the dorsolateral and rostroventral clusters of intensely stained cells (Figures 2A, 2G; Figure 3A; Figure 4E). Small, weakly stained cells were present in the anterior and dorsal parvocellular divisions of the PVN (Figure 2G). In the SON, intensely and weakly stained large cells were intermingled, and in the lateral hypothalamic region, they were also close to each other and arranged in small clusters around vessels (Figure 3C). The terminal plexus in the external zone of the ME lost its AVP immonoreactivity after colchicine injection (Figure 1G; Figure 2E). We also distinguished three types of cells in CRF immunostaining: large, intensely stained cells (51 ± 4%); large, weakly stained cells (38 ± 5%); and small, intensely stained cells (11 ± 2%; see Table 1). Small cells localized preferentially in the anterior and dorsal parvocellular regions of the PVN (Figure 2H), as in AVP, but the distribution pattern of large cells was different from that in AVP. The intensely stained cells localized in the medial aspect of the posterior magnocellular division, just lateral to the small cells, so that both cell populations formed an area occupying the two dorsomedial thirds of the PVN, whereas the weakly stained cells localized in the lateral third of its anterior and posterior magnocellular divisions (Figure 1F; Figure 5G). Intensely and weakly stained large cells were also present around vessels in the lateral hypothalamic region (Figure 5K), and a few cells (approximately 3%) also showed CRF immunoreactivity in SON (Figures 5C, 5I). Double immunohistochemistry for AVP and CRF in colchicine-untreated rats showed that in the external layer of the ME, 50-65% of CRF axons contain AVP (Figures 2K, 2L), and after colchicine injection, approximately 86% of small and weakly stained cells coexpress both peptides (Figures 2I, 2J; Table 1). According to these data, we can distinguish four different cell types: large AVP cells, large CRF cells, large AVP/CRF cells, and small AVP/CRF cells.
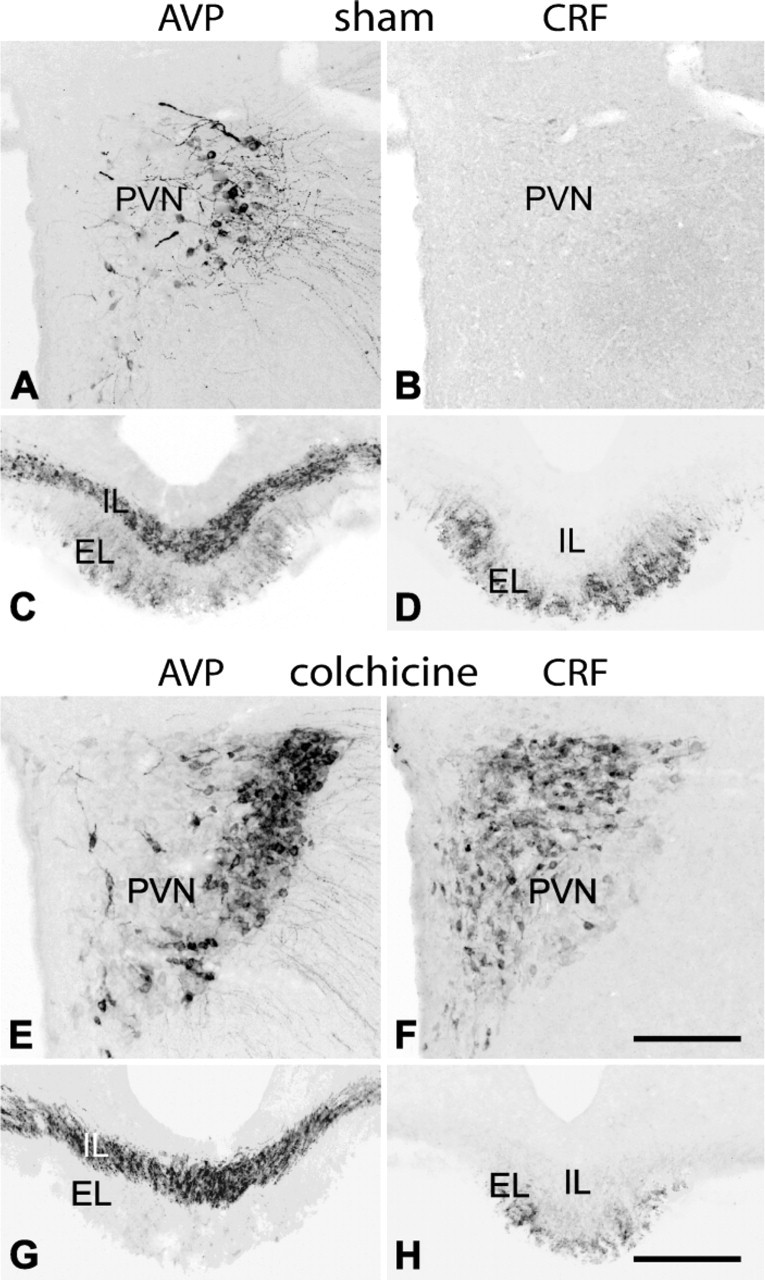
Immunohistochemistry for arginin-vasopressin (AVP) (
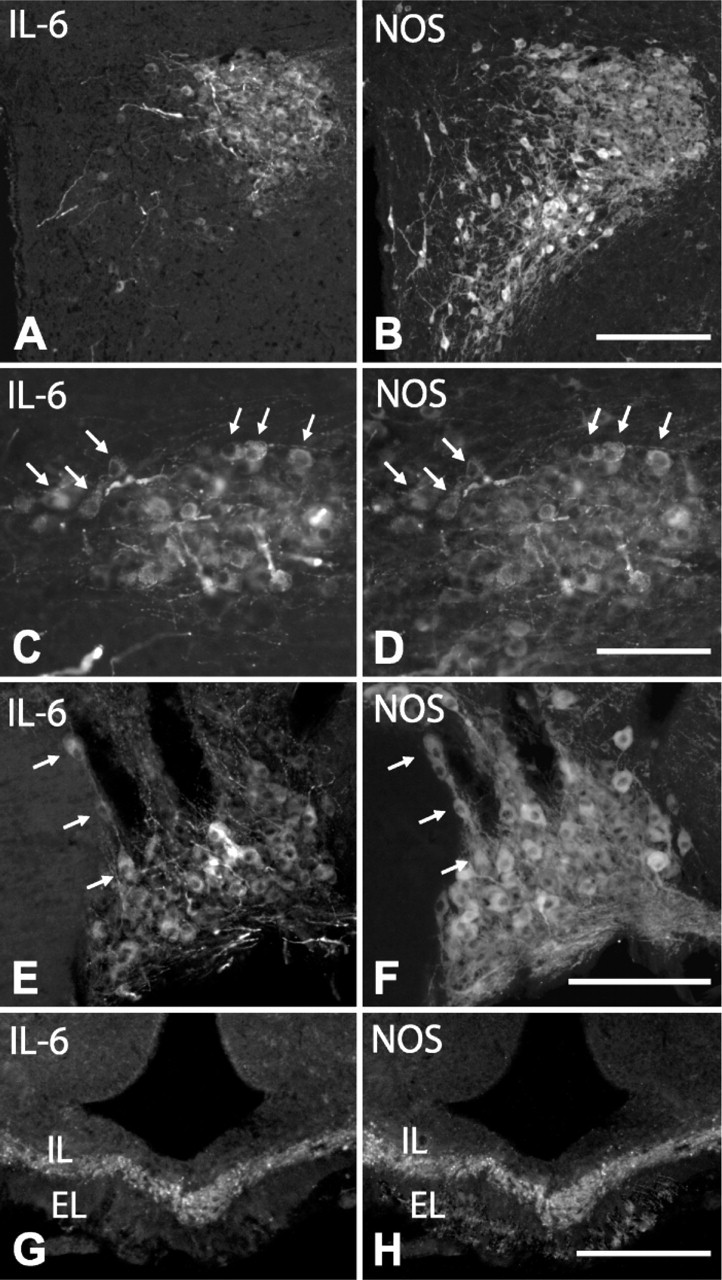
IL-6 and NOS Expression in AVP Cells. Most AVP cells (approximately 96%) contained IL-6 in colchicine-untreated rats. After colchicine injection, we observed that in PVN (Figures 2A-2C; Figures 3A, 3B), SON (Figure 2D), and lateral hypothalamus (Figures 3C, 3D), only large, intensely stained cells, which matched those identified in intact rats, but not small and large weakly stained cells, coexpressed IL-6. In terms of cell count, they corresponded to 36 ± 5% of the total number of AVP cells. The bundle of AVP axons running through the internal zone of the ME showed an intense IL-6 immunoreactivity (Figure 2E), whereas in the external zone, no IL-6 immunoreactive terminals were detected. It should be noted that some neurons sparsely distributed through the PVN, lateral hypothalamic region, and SON contained IL-6 but not AVP (Figure 2D, arrows).
Quantification of AVP and CRF cells expressing IL-6 and NOS in the rat hypothalamus
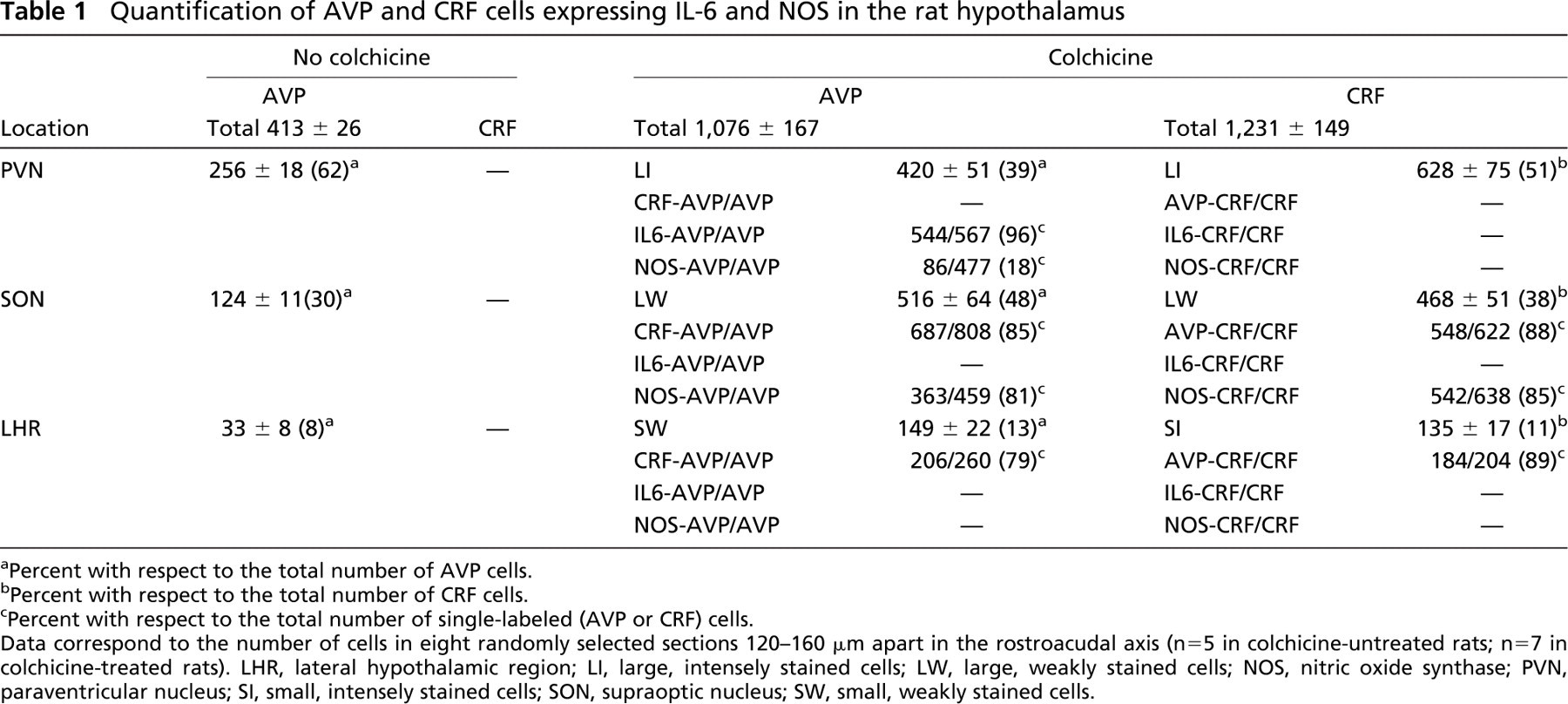
Percent with respect to the total number of AVP cells.
Percent with respect to the total number of CRF cells.
Percent with respect to the total number of single-labeled (AVP or CRF) cells.
Data correspond to the number of cells in eight randomly selected sections 120-160 μm apart in the rostroacudal axis (n = 5 in colchicine-untreated rats; n=7 in colchicine-treated rats). LHR, lateral hypothalamic region; LI, large, intensely stained cells; LW, large, weakly stained cells; NOS, nitric oxide synthase; PVN, paraventricular nucleus; SI, small, intensely stained cells; SON, supraoptic nucleus; SW, small, weakly stained cells.
With respect to the expression pattern of NOS in AVP cells, a low-magnification view of the PVN and SON in colchicine-untreated animals revealed that many NOS-positive cells colocalized with those immunoreactive for AVP (Figure 4) or IL-6 (Figures 6A-6F); however, at a higher magnification, we saw that in some regions, particularly in the anterior third of the PVN, only a few (3 ± 2%) large, intensely AVP- or IL-6-stained cells coexpressed NOS (Figures 4A, 4B), whereas in other regions, such as the dorsolateral region of the posterior magnocellular division of PVN and SON, 32 ± 3% of the cells contained NOS (Figures 4C, 4D; Figures 6A-6F). When weakly stained cells became evident after colchicine injection, we observed that approximately 80% of large, weakly AVP-stained cells (immunonegative for IL-6 and immunopositive for CRF) in the PVN and around vessels in the lateral hypothalamic region coexpressed NOS (Figure 2M; Figures 4E, 4F). Therefore, in colchicine-treated animals, approximately 48% of the total number of AVP cells express NOS. NOS activity was also detected in both the varicose plexus of the external layer and the bundle of transverse fibers of the internal layer in the ME of intact rats (Figures 6G, 6H). Numerous NOS-positive, AVP-negative cells were present throughout the PVN and in neighboring regions; however, because the distribution of NOS neurons in the hypothalamus has been extensively studied using NOS immunohistochemistry and NADPH-diaphorase histochemistry (Bredt et al. 1990; Arevalo et al. 1992; Calka and Block 1993; Miyagawa et al. 1994; Sanchez et al. 1998; Nylen et al. 2001; Xiao et al. 2005), we have omitted this description here.
IL-6 and NOS Expression in CRF Cells. Immunofluorescence for CRF and IL-6 in colchicine-treated rats revealed that although CRF and IL-6 cells colocalized in different hypothalamic regions, no cells coexpressing both markers were found (Figures 5A-5D). In addition, CRF localized in the axonal plexus of the external layer of the ME, whereas IL-6 was largely restricted to the axon bundle in the internal layer, with only a discrete overlap at the border between the layers (Figures 6E, 6F), supporting the idea that most CRF cells do not contain IL-6.
With respect to NOS, a low-magnification view of the distribution pattern of CRF and NOS cells in the PVN showed a fairly complementary distribution, because intensely stained CRF cells occupy the dorsomedial half and NOS cells occupy the ventrolateral half in the posterior magnocellular division (Figures 5G, 5H). However, at a higher magnification, we saw that the lateral third of this division also contained large, weakly stained CRF cells and that most of them (83 ± 6%) expressed NOS (Figure 2N; Figures 5G, 5H). Furthermore, most (91 ± 5%) CRF cells around vessels in the lateral hypothalamic region contained NOS (Figures 5K, 5L), whereas those in the SON were NOS immunonegative (Figures 5I, 5J). CRF and NOS colocalized in most fibers (78 ± 6%) forming the plexus of the external layer of the ME in colchicine-untreated rats (Figure 2O). NOS was also present in the bundle of the internal layer, but its axons did not contain CRF.
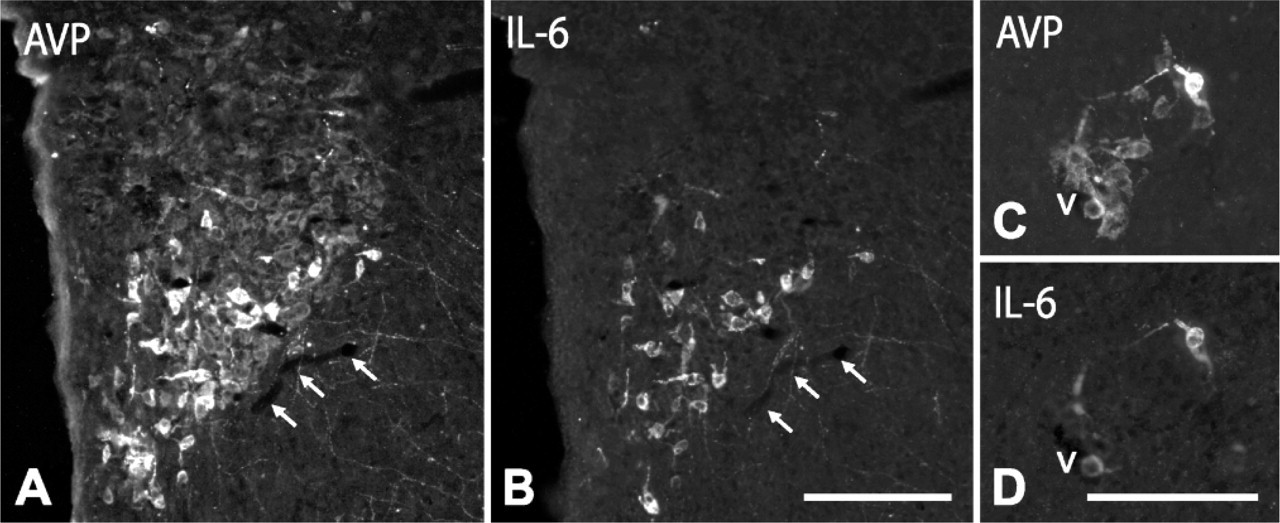
Double labeling for AVP (
Suprachiasmatic Nucleus
As shown in immunohistochemistry for AVP, CRF, and IL-6 (Figure 7), the rat SCN is a well-delimited nucleus composed of densely packed small cells (diameter of 9.3 ± 2.7 μm), which, according to their topographical distribution and neurochemical profile, may be divided into two different cell populations. One of these, localized dorsomedially, close to the ventricular wall, is immunoreactive for AVP (Figure 7A), and the other, localized ventrolaterally, is immunoreactive for CRF (Figure 7B). Double immunofluorescence for AVP and IL-6 revealed that approximately 65% of AVP cells were immunoreactive for IL-6 (Figure 2F; Figures 7C, 7D). However, although there was a certain overlap between AVP/IL-6 cells and CRF cells at the border between the dorsomedial and ventrolateral regions, no AVP/CRF or IL-6/CRF double-labeled cells were found. With respect to NOS, a few immunoreactive cells were detected in the neighboring regions, but not in the SCN.
Discussion
Our current concept of the peptidergic profile of hypothalamic nuclei comes from immunohistochemical studies carried out on different mammals since the early eighties (Kiss et al. 1984; Sawchenko et al. 1984a, b; Sawchenko and Swanson, 1985; Whitnall et al. 1985; Alonso et al. 1986; Piekut and Joseph 1986; Sawchenko 1987; Whitnall 1988; De Goeij et al. 1992; Mouri et al. 1993; Kikusui et al. 1997; Matz and Hofeldt 1999). According to these studies, CRF is preferentially expressed in parvocellular cells and AVP (and oxytocin) in the magnocellular cells in the PVN. In addition, subpopulations of parvo- and magnocellular cells coexpress both CRF and AVP (or oxytocin).
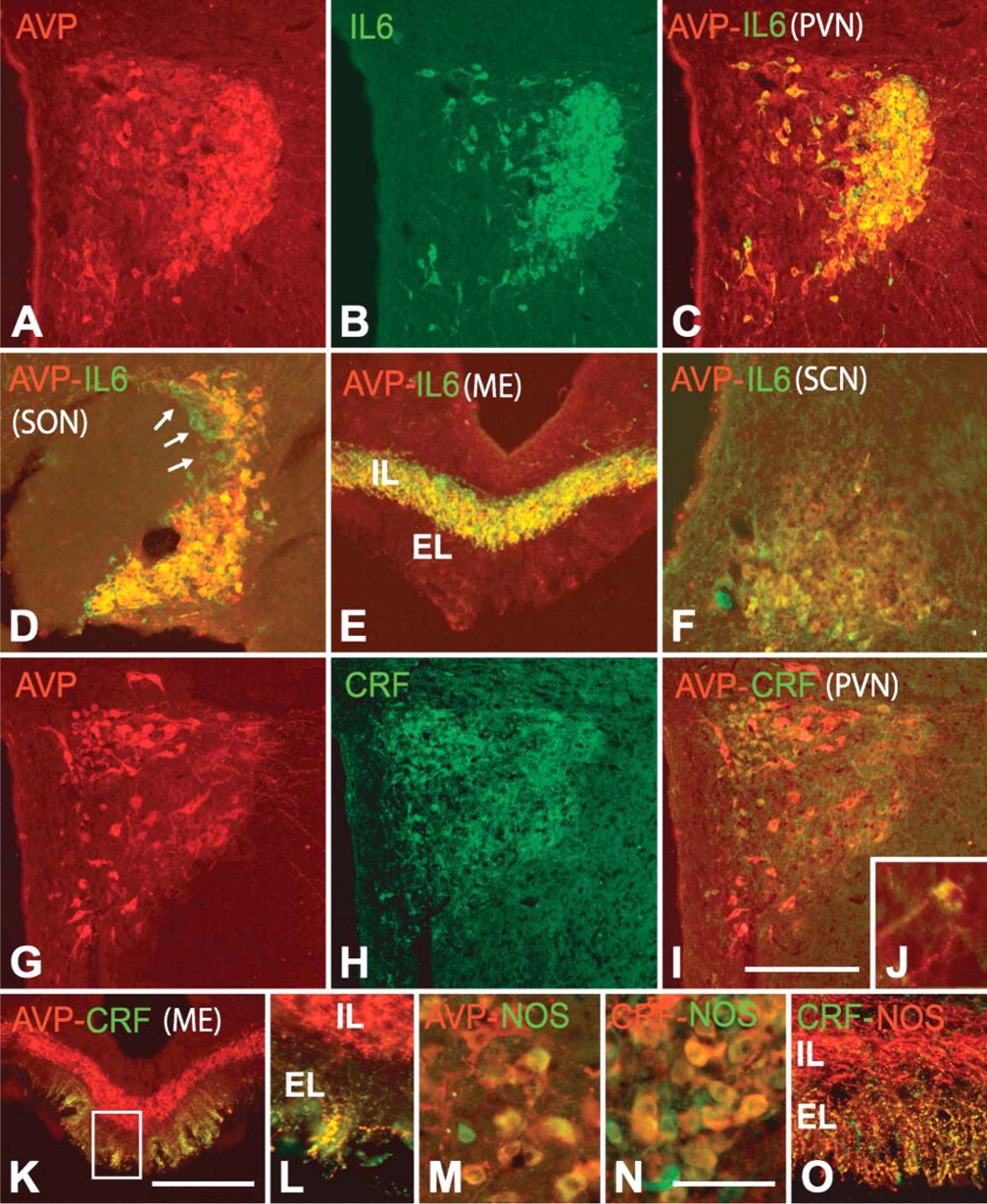
Double labeling for AVP and NOS in the PVN of an intact rat (
Double labeling for IL-6 and NOS in the PVN (
Double labeling for AVP and CRF (
Our results confirm these findings, and enable us to distinguish subpopulations of AVP- and CRFergic hypophysiotropic cells according to their IL-6 and neuronal NOS coexpression patterns (Figure 8). Although we have not used triple labeling, the combination of different pairs of antibodies together with cytoarchitectural criteria reveal five different types of hypophysiotropic cells. Within AVP cells: large, intensely stained AVP cells coexpressing IL-6; and large, intensely stained AVP cells coexpressing IL-6 and NOS. Within CRF cells: large, intensely stained CRF cells immunonegative for AVP, NOS, and IL-6; large cells weakly stained for CRF and AVP, immunopositive for NOS and immunonegative for IL-6; and small cells intensely stained for CRF and AVP and immunonegative for IL-6 and NOS. In addition, we identified two different cell populations in the SCN, one in its dorsomedial region, which expresses AVP and IL-6, and the other in its ventromedial region, which is immunoreactive for CRF.
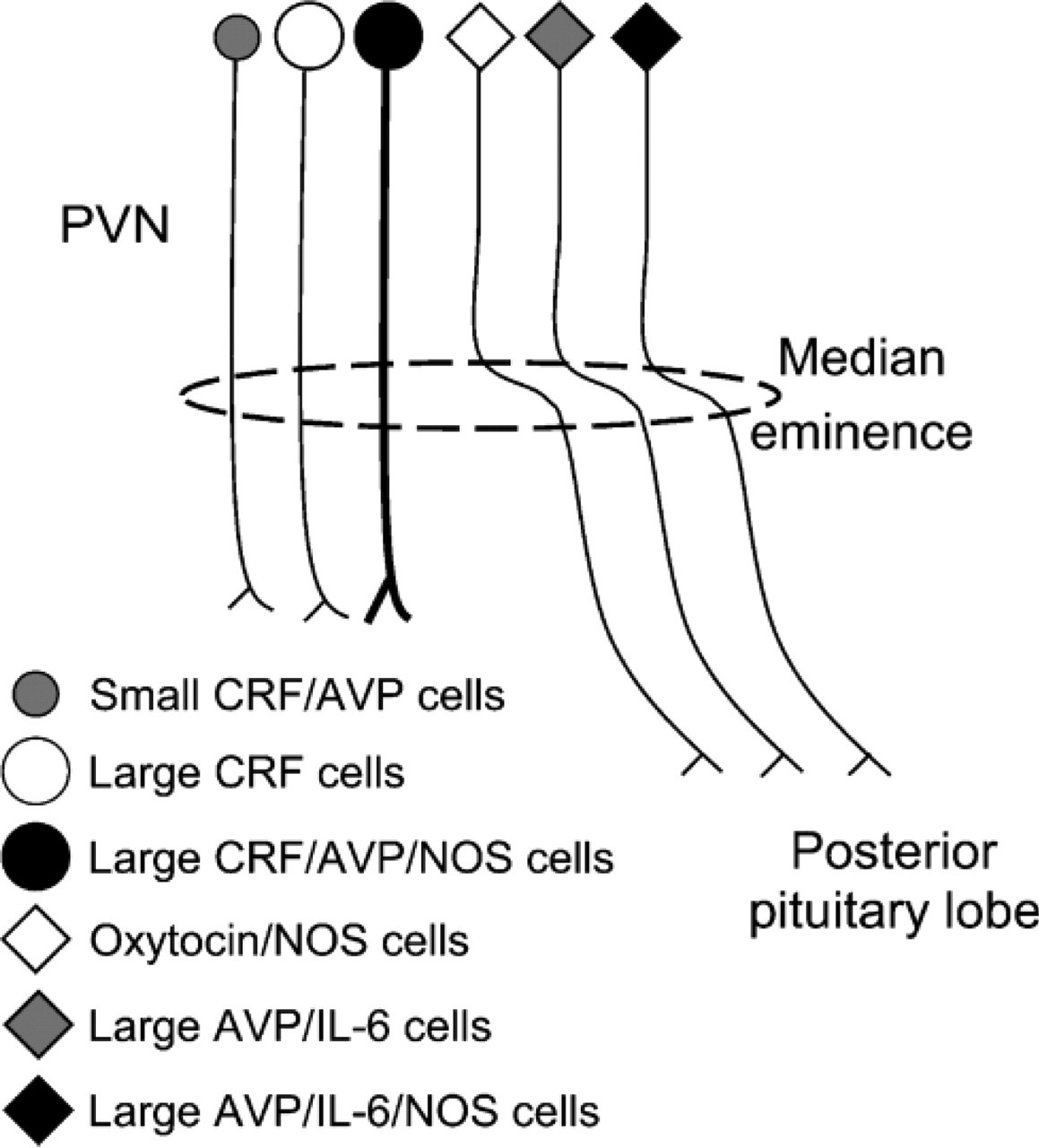
Schematic drawing summarizing the different populations of AVP and CRF hypothalamic cells according to the expression of NOS and/or IL-6 and their projections. PVN, paraventricular nucleus. In light of previous studies (Miyagawa et al. 1994; Hatakeyama et al. 1996; Yamada et al. 1996; Nylen et al. 2001; Xiao et al. 2005), oxytocin/NOS cells have also been included.
In light of previous morphological studies combining tract-tracing methods (Armstrong and Hatton 1980; Ju et al. 1986; Taniguchi et al. 1988) or partial hypo-thalamic lesion (Vandersande et al. 1977; Antoni et al. 1990) with immunohistochemistry, and the study in parallel of axons in the ME, the fact that large, intensely AVP-stained cells coexpress IL-6 and that axons running through the internal layer of the ME coexpress AVP and IL-6 indicates that they correspond to the AVPergic cells recently described by Ghorbel et al. (2003), which corelease AVP and IL-6 in the posterior pituitary lobe. However, our data reveal that they actually represent a subpopulation of no more than 40% of the total number of AVP cells, that 18% of them also coexpress NOS, and that two other subpopulations that do not express IL-6, small AVP/CRF cells (13%) and large, AVP weakly stained cells immunoreactive for CRF and NOS (47%), become evident after colchicine injection. In any case, these percentages should be taken as tentative, because they have been partially obtained from colchicine-treated rats, and the effect of colchicine can differ from one cell type to another and from one animal to another.
The finding that NOS displays the same labeling pattern as AVP and IL-6 in the internal layer of ME agrees with the presence of NOS in AVP/IL-6 cells. However, although the thickness and labeling intensity in the ME was similar for the three markers (NOS, AVP, and IL-6), only 35% of AVP/IL-6 cells showed NOS activity. This discrepancy could be explained by the fact that most oxytocinergic cells (another PVN magnocellular subpopulation whose axons reach the posterior pituitary lobe through the internal layer of the ME) also contain NOS (Nylen et al. 2001; Xiao et al. 2005) and consequently contribute to the NOS labeling intensity of this bundle.
With respect to CRF, we identified three cell types: magnocellular cells intensely stained for CRF, which are immunonegative for AVP, NOS, and IL-6; magnocellular cells weakly stained for CRF and AVP and also containing NOS; and parvocellular CRF cells, which coexpress AVP. In addition, we observed that CRF axons localize in the external layer of the ME, and that many of them contain AVP and NOS. These findings are consistent with previous studies reporting that CRF axons running to the anterior pituitary lobe occupy a ventral position in the ME (Rho and Swanson 1987, 1989; Kawano et al. 1988; Antoni et al. 1990; Weiss and Cobbett 1992; Lennard et al. 1993; Reyes et al. 2005), that the external zone of the ME expresses NOS activity (Sagar and Ferriero 1987; Ceccatelli et al. 1993), and that approximately 50% of CRF terminals in the external zone of the ME contain AVP (Whitnall and Gainer 1988). Taken together, these findings suggest that in the posterior pituitary lobe, constitutive IL-6 is synthesized by AVP cells, and NO by both oxytocin and AVP cells; and that in the hypophysial portal vascular system, NO is synthesized by AVP/CRF cells. However, in light of previous studies reporting that a subpopulation of large CRF cells also contains oxytocin (Dohanics et al. 1990; Imaki et al. 2001) and that NOS is expressed in oxytocinergic cells (Miyagawa et al. 1994; Hatakeyama et al. 1996; Yamada et al. 1996; Nylen et al. 2001; Xiao et al. 2005), it is possible that CRF/oxytocin cells also participate in the putative modulatory role of NO in the hypothalamic-pituitary system (Duvilanski et al. 1995; Ceccatelli 1997; Rivier 2001).
We also found AVP- and CRF-immunoreactive (−ir) cells in the SCN. These cells are densely packed and display a compartmental distribution, with AVP-ir cells localized in the dorsomedial region and CRF-ir cells in the ventrolateral region. This labeling pattern is partially supported by previous studies reporting that AVP cells are the main component of the dorsomedial (or shell) region of the SCN (Hofman et al. 1996; Herzog et al. 2000; Abrahamson and Moore 2001; Moore et al. 2002). In addition, as described for AVP hypophysiotropic cells projecting to the posterior pituitary lobe, we found that SCN AVP cells contain IL-6, indicating that IL-6 may be released from different hypothalamic AVPergic cells, and consequently, may be involved in different regulatory processes, possibly including the control of circadian rhythms.
On the other hand, although the ventrolateral (or core) SCN is mainly composed of vasoactive intestinal polypeptide- and gastrin-releasing polypeptide-immunoreactive cells (Hofman et al. 1996; Herzog et al. 2000; Abrahamson and Moore 2001; Moore et al. 2002), the presence of CRF-ir cells is supported by previous immunohistochemical and bioassay studies in the rat (Hashimoto et al. 1982; Daikoku et al. 1985; Hornby and Piekut, 1989) and goat (Kikusui et al. 1997). However, this finding should be interpreted cautiously, because the protein fragments used to obtain the CRF antibodies display structural homology with the peptide histidine isoleucine (PHI) (Sawchenko 1987; Antoni and Linton 1989), which has been detected in the ventrolateral SCN (Stopa et al. 1988; Denis et al. 1993). Therefore, a cross-reaction between CRF antisera and PHI cannot be ruled out.
With respect to NOS expression in the SCN, our data agree with those reporting that in the rat, SCN neurons do not contain NOS (Decker and Reuss 1994; Reuss et al. 1995; Wang and Morris 1996; Chen et al. 1997) and suggesting that the source of NO in the signal transduction of retinal glutamatergic inputs in the SCN (Ding et al. 1994; Watanabe et al. 1994; Weber et al. 1995; Amir and Edelstein 1997; Mitome et al. 2001; Golombek et al. 2004) is neighboring neurons or nonneuronal elements. In this respect, the finding that different parameters of circadian activity were not modified in neuronal NOS knockout mice (Kriegsfeld et al. 1999) and that SCN astrocytes express endothelial NOS (Caillol et al. 2000) suggested that this isoform could be the source of NO in the SCN. However, other studies have shown that endothelial NOS is not necessary for maintaining circadian rhythms (Kriegsfeld et al. 2001) and that the inducible isoform of NOS can be constitutively expressed in the adult SCN (Starkey et al. 2001). Taken together, these data suggest that in the SCN, NO can be synthesized by different NOS forms and can arise from different cell types.
In conclusion, the labeling patterns of IL-6 and NOS found here indicate that IL-6 is coexpressed with AVP in hypophysiotropic cells projecting to the posterior pituitary gland and in the dorsomedial region of the SCN, and that NOS is coexpressed with AVP and IL-6 in a subpopulation of AVPergic cells projecting to the posterior pituitary gland and with AVP and CRF in magnocellular cells projecting to the external layer of the ME. In light of previous functional studies, IL-6 can exert an autocrine/paracrine modulatory effect on posterior pituitary secretion and SCN AVPergic targets, and NO can exert a similar effect on the secretion of both pituitary lobes.
Footnotes
Acknowledgments
This work was supported by the Gobierno Autónomo de Canarias (grant no. PI042004/074), the Fundación Canaria de Investigación y Salud (grant no. PI 86/04), and the Ministerio de Educación y Ciencia de España (grant no. BFU2004-05756).
