Abstract
We previously reported that cell lines established from human carcinomas and leukemias/lymphomas expressed high levels of an intracellular membrane-bound protein, Haymaker, whereas cell lines derived from non-malignant connective tissue cells and lymphoid cells expressed low levels of this gene product. To determine whether these findings reflect neoplastic transformation or, alternatively, tissue specificity, we examined by immunohistochemical and molecular methods the expression of Haymaker in gynecologic organs with and without tumor. A highly specific, affinity-purified rabbit polyclonal antibody against a 25-mer Haymaker peptide was used for immunohistochemical staining and morphometric analysis of 85 tissue specimens. Immunohistochemical studies demonstrate, for the first time, that Haymaker protein is highly expressed in epithelial cells of the endometrium of the normal uterus and to a somewhat lesser extent in the mucosa of the normal vagina and cervix, but is poorly expressed or absent in cells of the connective tissue and smooth muscle strata of these organs (p < 0.005). Significant differences in Haymaker expression, as assessed by immunohistochemistry, between malignant and normal gynecologic tissues were not observed (p = 0.27). The expression of Haymaker protein does not appear, therefore, to be a marker of malignant transformation of the epithelium of gynecologic organs but rather distinguishes both normal and malignant epithelial cells from normal connective tissue and smooth muscle cells
W
Many normal gene products are overexpressed in tumors and have accordingly been clinically useful as markers for disease. Examples include carcinoembryonic antigen (CEA), alpha-fetoprotein (AFP), Ca125, inhibin, and prostate-specific antigen (PSA) (Gold and Freedman 1965; Smith 1970; Wang et al. 1981; Bast et al. 1983; Sheth et al. 1984). Alternatively, some gene products are translocated to abnormal sites within malignant cells. Although BRCA1 is expressed at normal levels in breast cancer, it is translocated to an abnormal cellular site (Chen et al. 1995). The wild-type BRCA1 gene product normally localizes to the nucleus via two putative signal peptides that are similar to steroid hormone receptor molecules (Arriza et al. 1987; Boulikas 1994). Mutant forms of BRCA1 that occur in breast and ovarian cancer localize, however, to the cytoplasm (Chen et al. 1995). Similar mistransport has been demonstrated for transcription factors involved in oncogenesis, such as c-Fos and SV40 large T antigen (Schneider et al. 1988; Roux et al. 1990; Moll et al. 1991). Haymaker was overexpressed in all malignant cell lines studied thus far. Interestingly, this protein localized to the plasma membrane (as well as an intracellular membrane bound compartment) of malignant cell lines of hematopoetic origin but only to the intracellular compartment in cells of epithelial origin (Das et al. 1997,2001,2005). Although some tumor-associated antigens are associated with tissue-specific tumors, others have a more extensive distribution.
One hypothesis, consistent with our previous findings, is that Haymaker may play a role in the induction or maintenance of the malignant state. Alternatively, the malignant and non-malignant cells examined in our previous work may differ from one another in some other property, thereby excluding a role for Haymaker in the malignant transformation of tissues. For example, many of the cancer cell lines previously studied were derived from epithelia, whereas the non-malignant cells originated from normal connective tissue and peripheral blood. To evaluate these alternative proposals, we determined Haymaker gene expression by immunohistochemistry and RT-PCR in normal and malignant gynecologic organs.
Materials and Methods
Chemicals and Antibodies
Reagents used for electrophoresis were purchased from Bio-Rad, Melville, NY. Titermax was purchased from the CytRx, Atlanta, GA. The A549 and K562 cell lines used in our study were purchased from the American Type Culture Collection, Rockville, MD, and were cultured, respectively, in DMEM and RPMI 1640 medium obtained from GIBCO BRL, Grand Island, NY. Tissue histology arrays were purchased from the Imgenex Company, San Diego, CA. Goat anti-rabbit IgG-alkaline phosphatase, Freund's adjuvant Incomplete, and all other chemicals were obtained from Sigma-Aldrich, St. Louis, MO. RNA isolation and RT-PCR kits and reagents were purchased from Qiagen, Inc., Stanford, CA. RT-PCR primers were obtained from GIBCO BRL. Agarose gel electrophoresis of RT-PCR products was documented using a UV light GelDoc system from UltraViolet Products, Upland, CA. Chemicals for immunostaining were purchased from Chemicon, Temecula, CA.
RT-PCR Studies
Gynecologic malignant and non-malignant tissue was obtained from surgeries performed by the Division of Gynecologic Oncology, Department of Obstetrics and Gynecology, State University of New York Downstate Medical Center at Brooklyn, NY. The above studies were reviewed and approved by the SUNY Downstate Medical Center, Kings County Hospital Institutional Review Board. The endometrium of normal and malignant uteri was removed from the underlying myometrium with a scalpel and immediately frozen with liquid nitrogen spray. The specimens were stored at −80C for subsequent RT-PCR studies. Approximately 60 mg of each tissue sample was mechanically homogenized, and the total RNA was isolated utilizing the RNeasy Kit (Qiagen). A pellet of 1 × 107 A549 cells was obtained from 3- to 4-day cultures, and the cells were lysed with a shredder column prior to isolation of total RNA. Total RNA was treated with RNase-free DNase to eliminate any DNA contamination. RT-PCR was then performed using a QIAGEN OneStep RT-PCR Kit and primers specific for Haymaker and β-actin. The Haymaker forward and reverse primers, LP1- 5′-CTACCACTTCGGGGTCACAT-3′ and XF2-5′-CTCCTCCCTGAGGTTGTCTG-3′, yield a 1.1-kb amplicon. The β-actin primers, 32FRT TCGTCGACAACGGCTCCGGCATGT and 32RRT-5′-CCAGCCAGGTCCAGACGCAGGAT-3′, yield a 519-bp amplicon. Twenty ng of total RNA was used with the β-actin primers, and 100 ng of total RNA was used with the Haymaker primers. RT-PCR was performed as previously described (Das et al. 2001): 30 min at 55C for RT, 15 min at 95C for Taq polymerase activation, 35 cycles of 1 min at 94C, and one cycle for 7 min at 72C. PCR products were then resolved using 1% agarose gels containing ethidium bromide. The gels were photographed under ultraviolet light using the GelDoc system (UltraViolet Products).
Production of Polyclonal Antibody
The complete open reading frame (ORF) of the Haymaker gene was determined by Das and colleagues (Das et al. 2001). A 25-mer peptide encoded within the Haymaker ORF (S43 to G67) was used to generate a monospecific polyclonal antibody in rabbits as previously described (Das et al. 1997,2001). This protocol was approved by the SUNY Downstate Medical Center Animal Care and Use Committee. Briefly, the synthetic peptide, 250 μg (purchased from New England Peptide, Inc., Gardner, MA) was mixed with Titermax, 1:1 (v/v) and injected subcutaneously into a rabbit at four sites according to the manufacturer's instructions. Subcutaneous booster injections of 100-μg peptide in incomplete Freund's adjuvant were given every other week. Serum was collected 1 week after the third booster injection and weekly thereafter. The anti-25-mer antibodies were affinity purified from the rabbit sera using CNBr-Sepharose 4B beads (Sigma-Aldrich) linked with BSA-25-mer peptide conjugate. As shown in our previous work, the affinity-purified antibody had a titer of 1:10,000 and a protein concentration of 153 ng/ml (Das et al. 2001). To evaluate the specificity of our anti-Haymaker antibody, lysed K562 cells were used as a source of Haymaker antigen. The K562 cell line was chosen because our previous work had demonstrated abundant Haymaker expression in this line (Das et al. 1997). The antiserum reacted with a single 38.5-kDa protein on Western blots of the lysates of K562 cells, and passage of the polyclonal rabbit anti-25-mer sera through a column containing covalently bound 25-mer peptide removed all 38.5-kDa binding activity (Das et al. 2001). Furthermore, the affinity-purified anti-25-mer Haymaker antibody reacted with recombinant Haymaker protein synthesized in both bacterial and in vitro transcription-translation systems (Das et al. 2005).
Immunohistochemistry
A sampling of gynecologic malignant and non-malignant tissue was selected from among the cases diagnosed by the Departments of Obstetrics and Gynecology and Pathology at the Kings County Hospital Center, Brooklyn, NY. All tissue samples examined had been fixed in formalin and embedded in paraffin. Additional tissues were examined via the use of tissue histology arrays purchased from the Imgenex Company. Hematoxylin and eosin (H and E)-stained slides were available for all of the tissues we examined for Haymaker expression via immunohistochemistry. Prior to immunostaining, H and E sections were examined for each sample for confirmation of the pathologic tissue diagnosis.
The immunostaining was performed using the streptavidin-biotin-peroxidase method (Hsu et al. 1981). Briefly, the following technique was used. A 5-μm-thick section was cut from each paraffin block and placed onto slides. The specimens were then deparafinized in xylene and rehydrated in graded alcohol and then distilled water. The endogenous peroxidase was consumed by immersing the sections in 0.3% hydrogen peroxide in absolute methanol for 20 min. Pressurized steam heat was applied to the slides while in a 10-mM citrate buffer solution, pH 6.0, for 30 min. Nonspecific secondary antibody binding was blocked via incubation in 10% normal goat serum for 10 min. Affinity-purified rabbit anti-25-mer Haymaker antibody at a dilution of 1:250 was then applied for 60 min. The primary antibody was then washed off with PBS. The slides were then incubated with biotinylated goat anti-rabbit-IgG antibody (1:30), followed by streptavidin-peroxidase (1:30) for 20 min each. DAB was then applied and reacted with the sections for 10 min. After washing in PBS, the sections were lightly counterstained with hematoxylin, washed in xylene, and then coverslips were applied.
All immunohistochemical sections were evaluated by two of the authors, independently (CAA and MEB), and a consensus was reached when opinions differed. Semi-quantitative analyses were recorded as an assessment of the percentage of cells that stained with the anti-Haymaker antibody and the intensity of staining. The categories 0%, 1-5%, 6-25%, 26-50%, 51-5%, and >75% were used to characterize the proportion of cells that expressed the Haymaker antigen. For those specimens that expressed Haymaker, the intensity of staining was graded on a +1, +2, or +3 scale.
Statistical Methods
All statistical analyses were performed using SPSS 12.0 (SPSS, Inc., Chicago, IL). Non-parametric analysis was used because of the small sample size and an inability to assume that the data follow a normal distribution. The Mann-Whitney test was used to test independent samples, and the Wilcoxon signed ranks test was used to test paired samples. Specifically, the Mann-Whitney test was used to compare the percent staining as well as the intensity of staining between subjects with non-malignant cells of epithelial origin and those with malignant cells of epithelial origin. The above test was also used to compare the percent staining and intensity of staining between subjects with non-malignant cells of stromal/mesodermal origin and those with malignant cells of stromal/mesodermal origin. The Mann-Whitney test was used to compare both the percent and intensity of staining within subjects, comparing epithelial cells to stromal/mesodermal cells for both malignant and non-malignant cells in a given individual. A type I error of 5% was used to determine statistical significance.
Results
A paraffin block was prepared with cultured A549 cells and used as a positive control, because these cells are known to have a high level of Haymaker expression (Das et al. 2001). Figure 1A shows an extensive intracellular granular staining pattern with the anti-25-mer Haymaker antibody in most A549 cells. To demonstrate the specificity of the anti-25-mer antibody, 100 μmg of the 25-mer Haymaker peptide was added to 230 μl of the anti-Haymaker antibody solution. This solution was then applied to a section from the A549 cell paraffin block. Addition of the 25-mer Haymaker peptide completely abrogated the staining of the cells (Figure 1B). This observation is consistent with previous studies demonstrating depletion of reactivity following passage of the antibody over Haymaker peptide affinity columns, as assessed by Western blot analysis. Further evidence of the specificity of the affinity-purified anti-25-mer antibody was demonstrated by its reactivity with recombinant Haymaker as well as with the native 38.5-kDa molecule and the lack of reactivity with other proteins in Western blots of tumor cell lines or normal cells (Das et al. 2005).
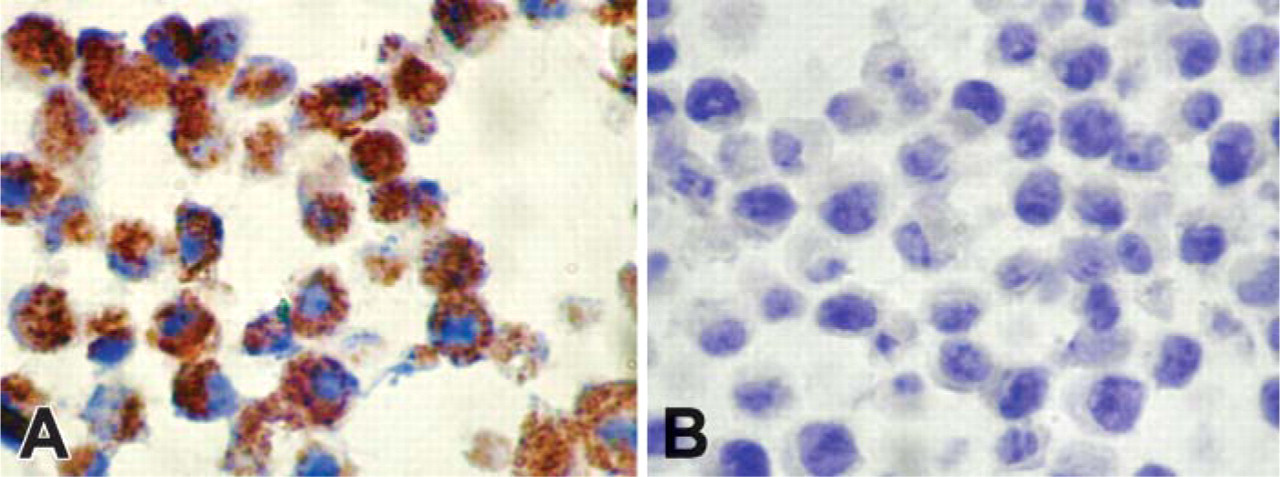
Specificity of anti-25-mer Haymaker antibody. (
A total of 85 gynecologic tissue specimens were examined by immunohistochemistry. These specimens consisted of 10 sections from ovarian carcinoma, 7 from normal ovarian organs, 23 endometrial carcinomas, 2 malignant mixed mullerian tumors, 14 normal endometrial sections, 7 normal myometrial sections, 12 cervical carcinomas, 4 normal cervical sections, 2 normal vaginal sections, and 4 normal lymph nodes. Of the 85 patient specimens that were examined, there were 21 paired specimens in which tumor was matched to normal adjacent tissue from the same patient. The remaining 43 specimens were unmatched (25 with tumor and 18 normal). All epithelial tissues reacted with the anti-25-mer peptide antibody. Examples of the typical staining patterns of endometrial tissues with the anti-25 mer Haymaker antibody are shown in Figure 2. We found the anti-25-mer antibody staining to be granular in nature and confined to the apical and basolateral areas of the cytoplasm of positive cells. The connective tissues of the stroma and the smooth muscle cells of the myometrium were usually devoid of stained cells.
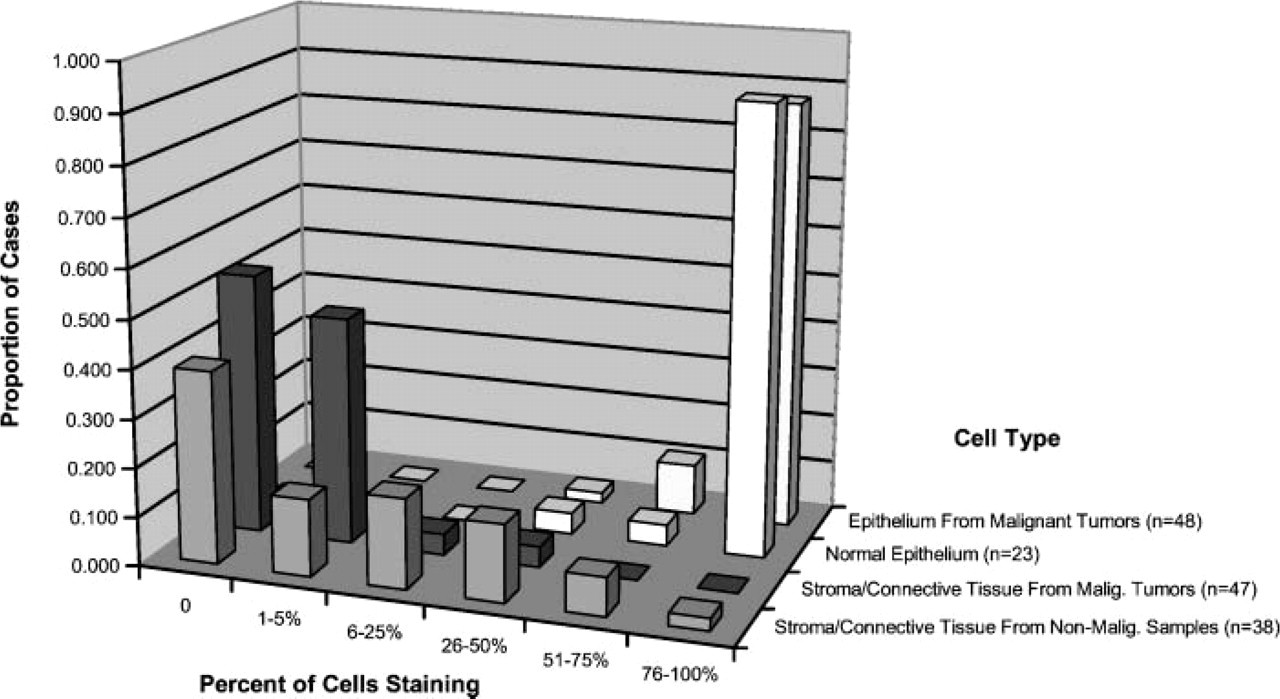
Haymaker expression appeared greater in the columnar epithelial cells of the uterus as compared with the stratified squamous epithelium of the mucosa of the vagina and cervix. However, the squamous cells of the vagina and cervix stained to a much greater degree than the underlying connective tissue stroma and muscularis. The proportion of epithelial cells that reacted with the anti-25-mer Haymaker antibody is shown in Figure 3. In no case (benign or malignant) did less than 25% of the epithelial cells stain; in 2 cases, between 26% and 50% of cells stained; in 6 cases, 51 to 75% of cells stained; and in the remaining 59 cases, more than 75% of the epithelial cells stained for Haymaker protein. A high proportion of all of epithelial cells of the mucosa stained, regardless of whether they were of benign or malignant tissue origin. The proportion of carcinoma cells reacting with the anti-Haymaker antibody was not significantly different from that of non-malignant epithelial cells from the same patients (p = 0.180). The overall proportion of epithelial cells reacting with the antibody in the benign samples was not significantly different from that in malignant samples (p = 0.270).
In contrast to the ubiquitous epithelial staining seen across all the various samples examined, few cells in the connective tissue stroma or smooth muscle of these specimens reacted with the anti-25-mer Haymaker antibody (Figure 4). Only 17 of the 80 samples had more than 5% of stromal cells staining for Haymaker. Eleven of these cases with significant stromal staining were in normal endometrium, two were in normal myometrium, two were in the stroma of an endometrial carcinoma, and two were in the sarcomatous portion of malignant mixed mullerian tumors. The vast majority of samples showed less than 5% of stromal cells staining. However, there was a trend toward an increase in stromal staining in the benign tissue as compared with the malignant tumors. This was largely due to 11 benign cases in which endometrial stroma showed 6-75% of cells staining for Haymaker. This trend reached statistical significance among the paired samples [tumor vs normal in same patients (p < 0.001)] but did not reach significance on examination of the independent unpaired samples [tumor vs normal in different patients (p = 0.181)].
By far the greatest difference in Haymaker expression was observed between epithelial cells and cells of the connective tissue and muscularis (Figure 5 and Figure 6). When the different histologic tissue types were pooled and examined with respect to benign versus malignant tissue origin, significant differences between the proportions of benign and malignant epithelial cells of the mucosa that reacted with the anti-Haymaker antibody were not observed (Figure 5). A higher proportion of the epithelial cells expressed Haymaker, as compared with the stromal cells (p < 0.001) from the paired gynecological samples with tumor. The epithelial cells also stained with much greater intensity than the stromal cells (p < 0.001). Similar findings were seen in the non-malignant samples, where the proportion of epithelial cells staining and the intensity of staining were markedly higher in the epithelium than in the connective tissue or smooth muscle (p = 0.004 for proportion of cells and p = 0.005 for intensity).
RT-PCR Studies
Relative levels of Haymaker gene transcripts were examined in normal and malignant endometrium using highly specific primers for Haymaker (Figures 7A and 7B). The level of β-actin RT-PCR product was similar in all samples tested, indicating that equivalent amounts of total RNA were used for the gene-specific amplification of Haymaker (Figures 7C and 7D). However, RT-PCR with Haymaker primers of RNA from three of the five normal endometrial samples failed to yield a product (Panel A, Lane 1 and Panel B, Lanes 5 and 6). The other RNA samples from normal patients yielded a moderate amount of amplified product (Panel A, Lane 2 and Panel B, Lane 7). In contrast, all five of the RNA samples from the patients with carcinoma yielded amplified Haymaker-specific product. Grade 2 endometrial adenocarcinoma (Panel B, Lane 1) gave the most intense signal, whereas one of the Grade 3 endometroid adenocarcinomas (Panel A, Lane 4) had the least intense signal. These data suggest that Haymaker transcripts are readily detected in the endometrium of uteri bearing epithelial tumors and to a lesser extent in the endometrium of normal uteri.
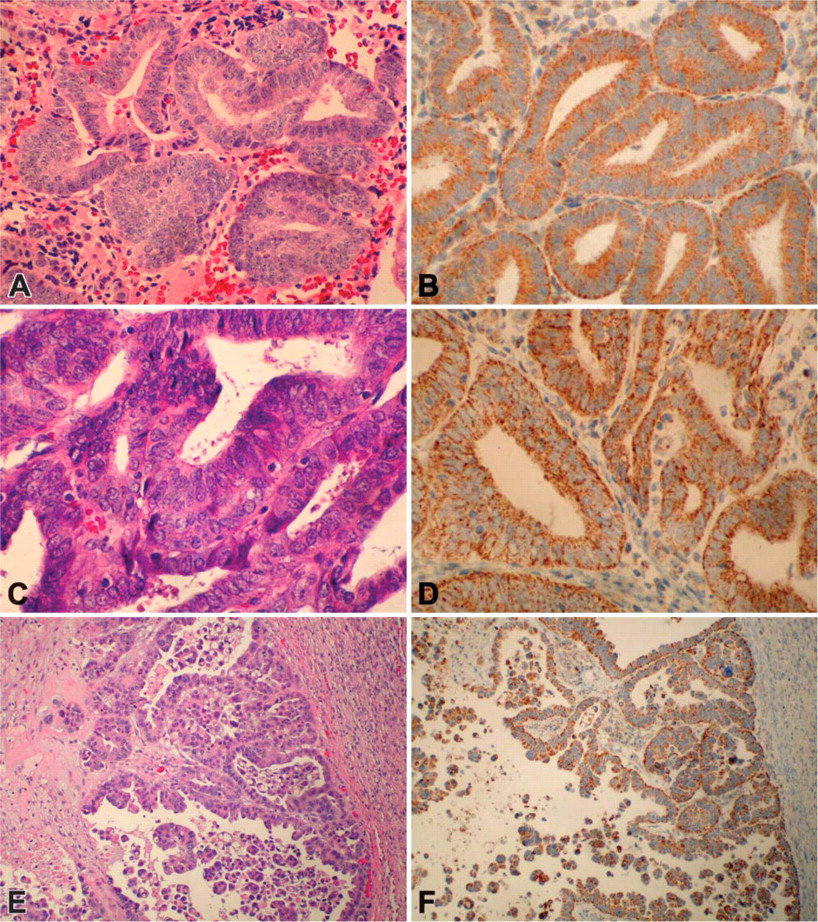
Representative staining reactivity with anti-25-mer Haymaker antibody. (
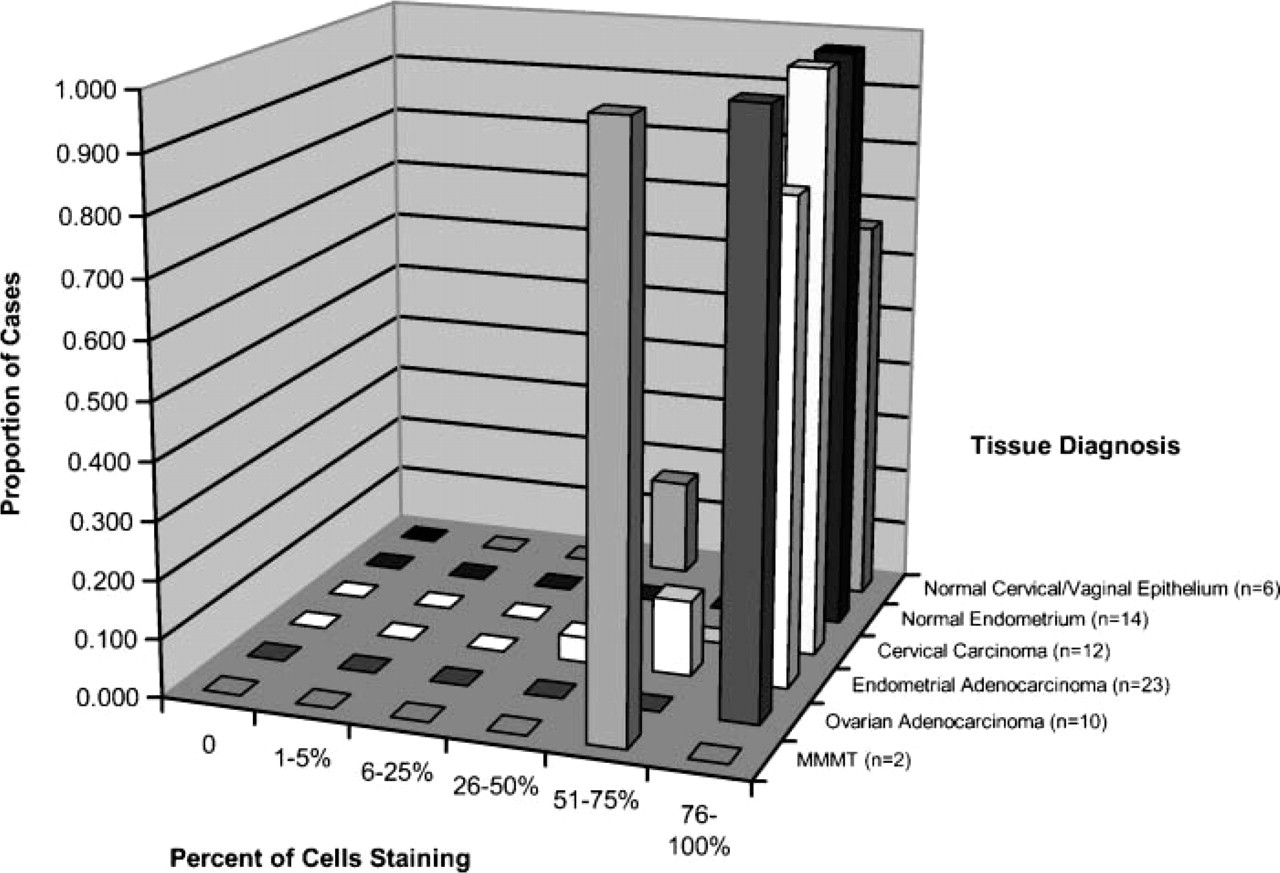
Percent of epithelial cells reacting with anti-Haymaker antibody.
Discussion
In previous studies using Western blot, flow cytometry, and RT-PCR analysis, we demonstrated that Haymaker expression was significantly greater in cell lines established from malignant epithelial and hematopoetic tissues, compared with cell lines derived from non-cancerous connective tissues and from freshly isolated normal lymphocytes (Das et al. 2001). The concordance of elevated levels of both protein and specific mRNA in the malignant cell lines compared with these non-malignant cells suggested the possibility of tran-scriptional upregulation of the Haymaker gene in human cancer. The cell lines examined in our previous work may, however, have differed in a property other than neoplastic transformation, such as tissue specificity. Accordingly, in the current investigation, we directly measured Haymaker expression in biopsy specimens of normal and malignant gynecologic organs, where it was possible to evaluate these alternative proposals using immunohistochemical techniques. This first report of the distribution of Haymaker protein in human tissues revealed that Haymaker is highly expressed in the epithelium of the endometrium of both cancerous and normal uteri and to a somewhat lesser extent in the epithelium of malignant and benign cervix and vagina. The cells of the underlying connective tissue stroma exhibited little Haymaker expression. Similarly, the smooth muscle cells of the uterus, cervix, and vagina were largely devoid of Haymaker.
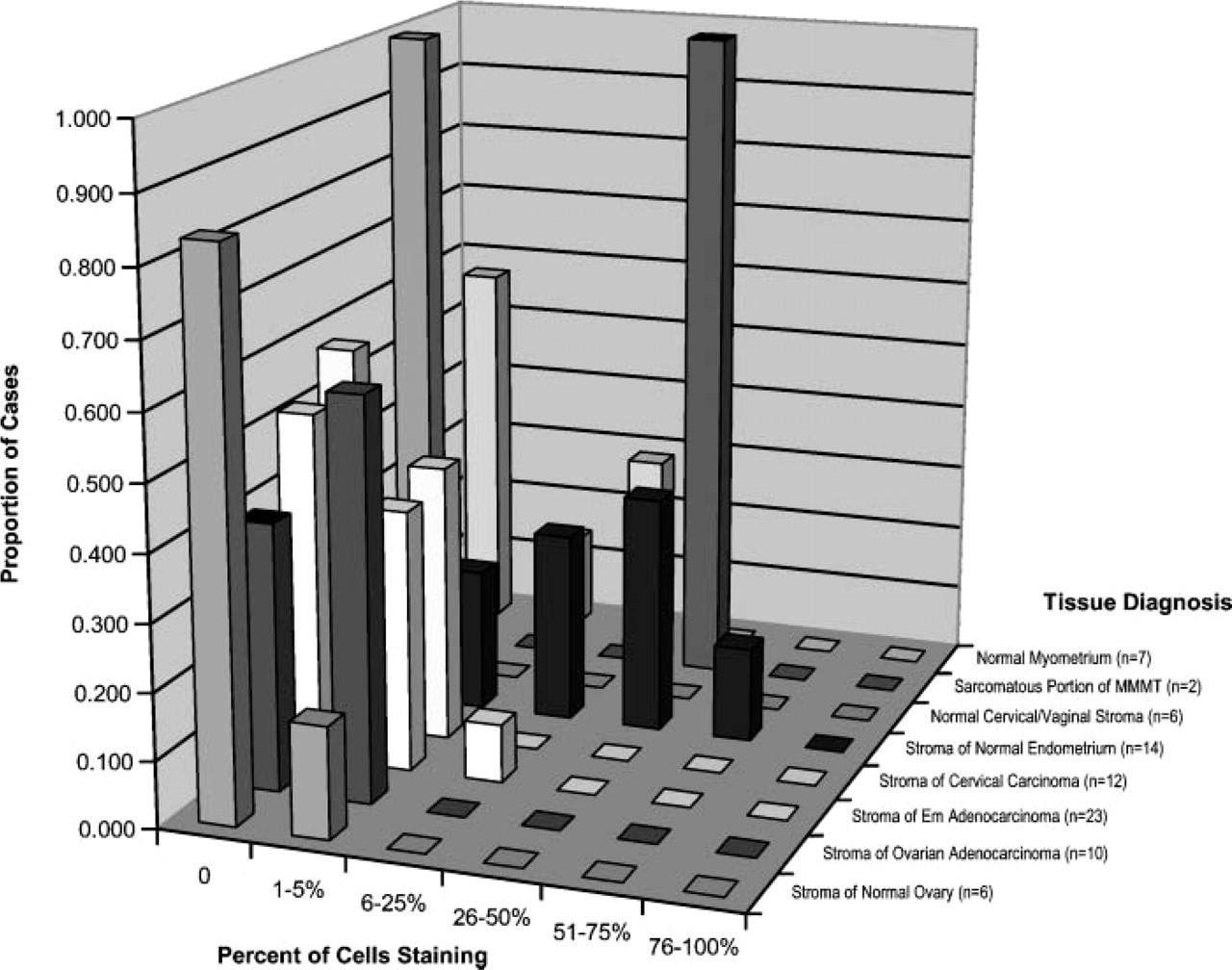
Percent of connective tissue and myometrial cells reacting with anti-Haymaker antibody.
Percent of cells reacting with anti-Haymaker antibody in epithelial versus non-epithelial and benign versus malignant tissues.
These findings are consistent with our previous observations, in that tumor cell lines of epithelial origin, such as human lung adenocarcinoma (A549), human breast carcinomas (T47-D and MD231), human kidney carcinoma (DU145), and human colorectal carcinoma (COLO), highly expressed Haymaker protein. In contrast, the normal cells and non-malignant cell lines studied previously were not of epithelial origin but rather of lymphoid or connective tissue origin; i.e., peripheral blood lymphocytes, embryonic fibroblast line CCD 27-SK, and fetal lung fibroblast line FHS expressed little Haymaker protein (Das et al. 2001). Thus, the previous work and current work on the level of Haymaker expression demonstrate an association with tissue type (i.e., epithelium compared with smooth muscle and connective tissue) rather than with the malignant state. Interestingly, RT-PCR analysis suggested that Haymaker transcripts were more readily detected in uteri bearing epithelial-derived tumors than in normal uteri. These latter results are consistent with our immunohistochemistry studies, when one considers that the bulk of the cells (>90%) in the cancerous uterus are of epithelial origin rather than connective tissue origin. In contrast, detection of Haymaker transcripts would be less likely in normal uteri, because the endometrium consists of about only 20% epithelium.
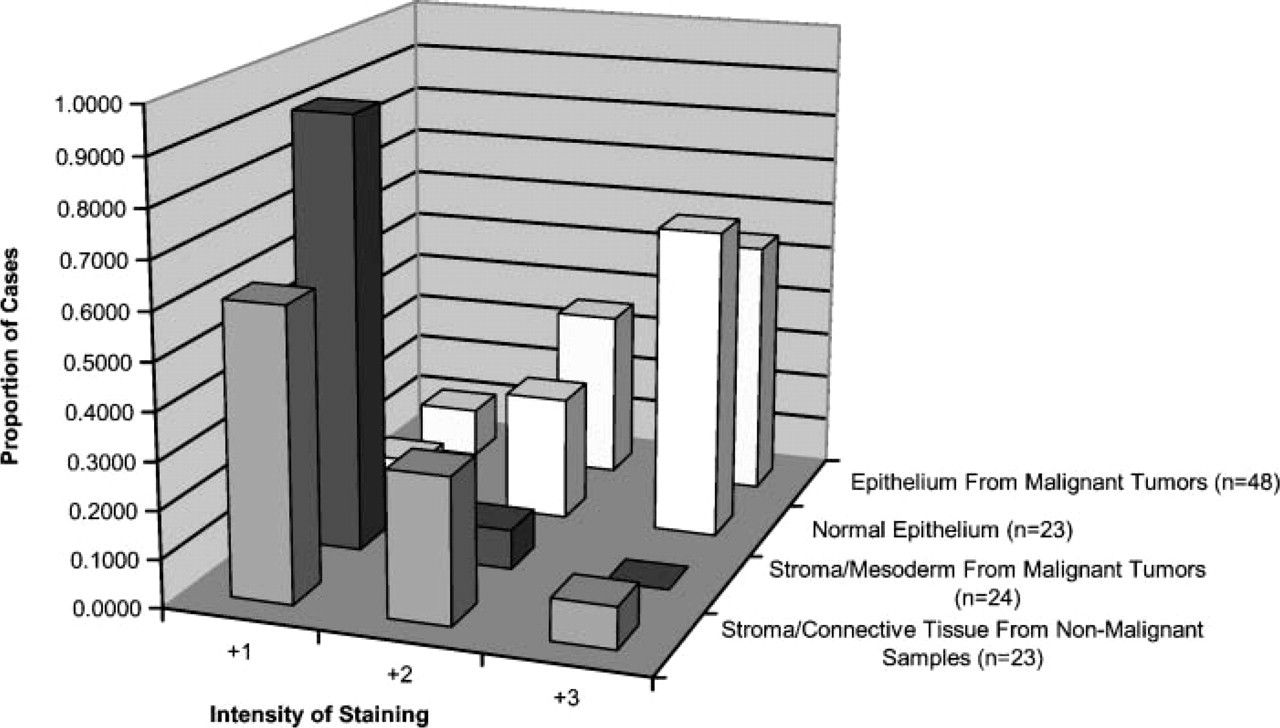
Relative reactivity of anti-Haymaker antibody staining in epithelial versus non-epithelial and benign versus malignant tissue.
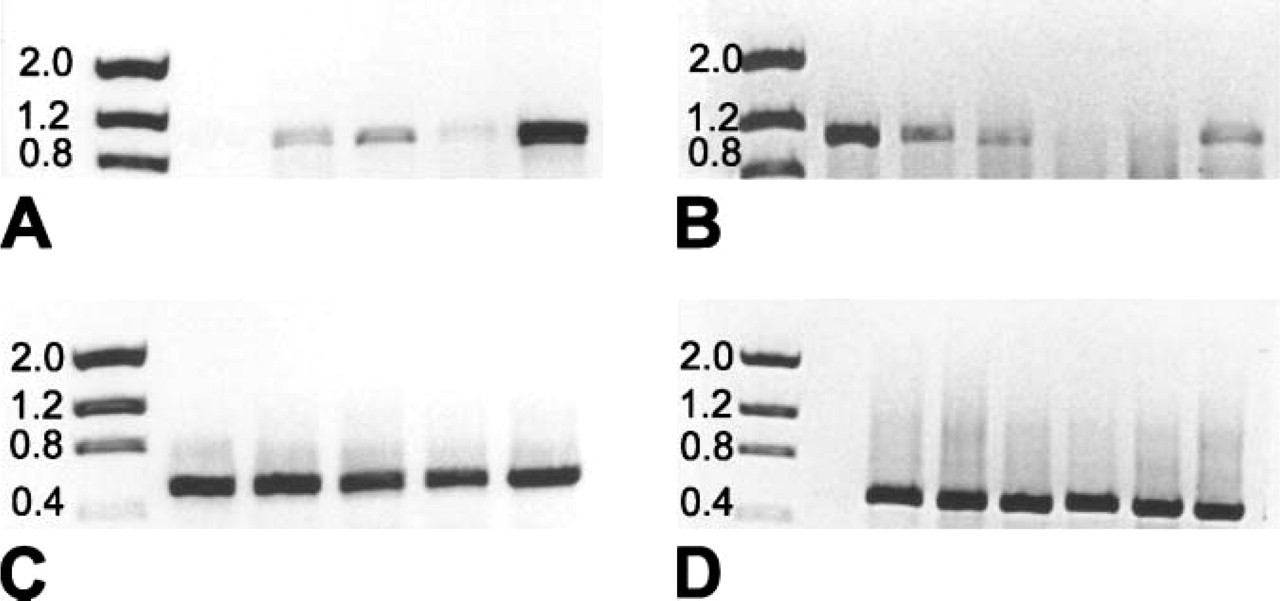
Detection of Haymaker mRNA by RT-PCR in malignant and non-malignant endometrial tissue. (
There are a number of possible explanations for the differential expression of Haymaker protein in epithelial cells compared with connective tissue and muscle cells. One possibility is that Haymaker expression may be related to the rate of cell proliferation. Other markers of cell proliferation, such as Ki-67 antigen and proliferating cell nuclear antigen (PCNA) (Takasaki et al. 1981), are commonly used in immunohistochemical studies to evaluate cell proliferation. Although the epithelial cells clearly have a higher rate of proliferation than smooth muscle cells, fibroblasts of the connective tissue stroma and fibroblast cell lines replicate at rates similar to those of epithelial cells. Further immunohistochemical studies on various tissues that correlate Haymaker expression with markers of cell cycle or cell proliferation, such as Ki-67 antigen (Gerdes et al. 1983) and PCNA (Takasaki et al. 1981), may fully resolve this matter. A more likely possibility, however, is that elevated Haymaker expression is related to cellular function. For example, epithelial cells of the uterus perform a secretory function, in contrast to the contractile function of the smooth muscle of myometrium. Haymaker protein may play a role in the secretory function of the former cells. Such a proposal is consistent with our previous biochemical data demonstrating localization of this molecule to membranes of an as yet undetermined intracellular vesicle (perhaps a secretory vesicle) (Das et al. 2001,2005). This proposal is also consistent with the granular pattern of staining we observed herein. Clearly, further studies beyond the focus of the current report are required to resolve this issue.
In our initial report, we provided evidence that Haymaker was expressed on the plasma membrane of certain cell lines derived from leukemias but was not expressed on the surface of either normal lymphocytes or malignant cell lines of epithelial origin. Further investigations clearly demonstrated that Haymaker mRNA was abundant not only in the plasma membrane positive cell lines but also in the surface negative tumor-derived epithelial lines. Additional studies revealed that Haymaker protein was highly expressed in all tumor cell lines but not in the non-epithelial cell lines of normal origin. Although some reports have suggested that Haymaker might be localized to mitochondria and may function as the mammalian homolog of yeast TOM40 (Baker et al. 1990; Suzuki et al. 2000), we have previously provided biochemical evidence that this molecule is not found in mitochondria under normal physiologic conditions (Das et al. 2001,2005). Haymaker did not segregate with other mitochondrial markers, and normal cells that have functional mitochondria do not express this protein. The current immunohistochemical study supports our previous observations by demonstrating the lack of staining of smooth muscle cells with a highly specific anti-Haymaker peptide antibody. The latter tissue has abundant mitochondria and thus should be highly reactive with the anti-Haymaker antibody if Haymaker were localized to this organelle.
In summary, Haymaker functions in certain leukemia/lymphoma cell lines as a cell surface target molecule for cytolytic action by non-activated natural killer cells (Das et al. 1997,2005). The function of Haymaker in normal epithelial cells is presently unknown; however, it may comprise part of a channel in an organelle other than mitochondria, inasmuch as it is a member of the Porin family of proteins (www.sanger.ac.uk/Software/Pfam/) and is clearly an intracellular integral membrane protein in epithelial cells (Thinnes 1992).
Footnotes
Acknowledgements
The authors appreciate the financial support of the Department of Obstetrics and Gynecology, and the Transplant Immunology and Immunogenetics Program, State University of New York Downstate Medical Center.
