Abstract
Molecular cytogenetic aneuploidy testing for pregnant women at increased risk of chromosome abnormality leads to rapid reassurance for those with normal results and earlier decisions on pregnancy management in the case of abnormality. We tested 9080 prenatal samples using a one-tube QF-PCR test for trisomies 13, 18, and 21; the abnormality rate was 5.9%. There were no misdiagnoses for non-mosaic trisomy. A sex chromosome multiplex was developed that detects structural sex chromosome abnormalities as well as aneuploidies. The sex chromosome test was targeted at pregnancies (272) with specific abnormalities suggestive of Turner syndrome; 13.2% showed 45,X, confirmed by follow-up analysis.
Keywords
W
Preparation of samples, formulation of the trisomy multiplex, protocol for QF-PCR, and interpretation of results are as described previously (Mann et al. 2004). Table 1 shows the primer sequences and target loci of the modified sex chromosome multiplex.
All prenatal samples received in this laboratory undergo rapid trisomy testing. Table 2 shows the results from the trisomy multiplex during the period from June 2000 to March 2004. All samples had full karyotype analysis following QF-PCR testing, and there were no misdiagnoses for non-mosaic trisomy. There were 17 mosaic samples detected by QF-PCR and/or karyotype analysis. Of these, QF-PCR identified the abnormal cell line in 13 samples, and karyotype analysis identified the abnormal cell line in 14 samples. We estimate that QF-PCR can identify mosaicism at levels greater than 15%. The current trisomy multiplex is more informative than previous versions, leading to a greatly reduced incidence of uninformative results: in 2003–2004, there was only one sample with one uninformative chromosome. Maternal cell contamination (MCC) was evident as a second genotype but with no fourth allele and with allele peaks in characteristic ratios (Mann et al. 2001). Almost all samples showing MCC were bloodstained amniotic fluid samples. Where the second genotype was low level and did not result in allele peak ratios outside the normal range for the majority genotype, results were interpreted and reported. If allele peak ratios were outside the normal range, samples were reported as “unsuitable.”
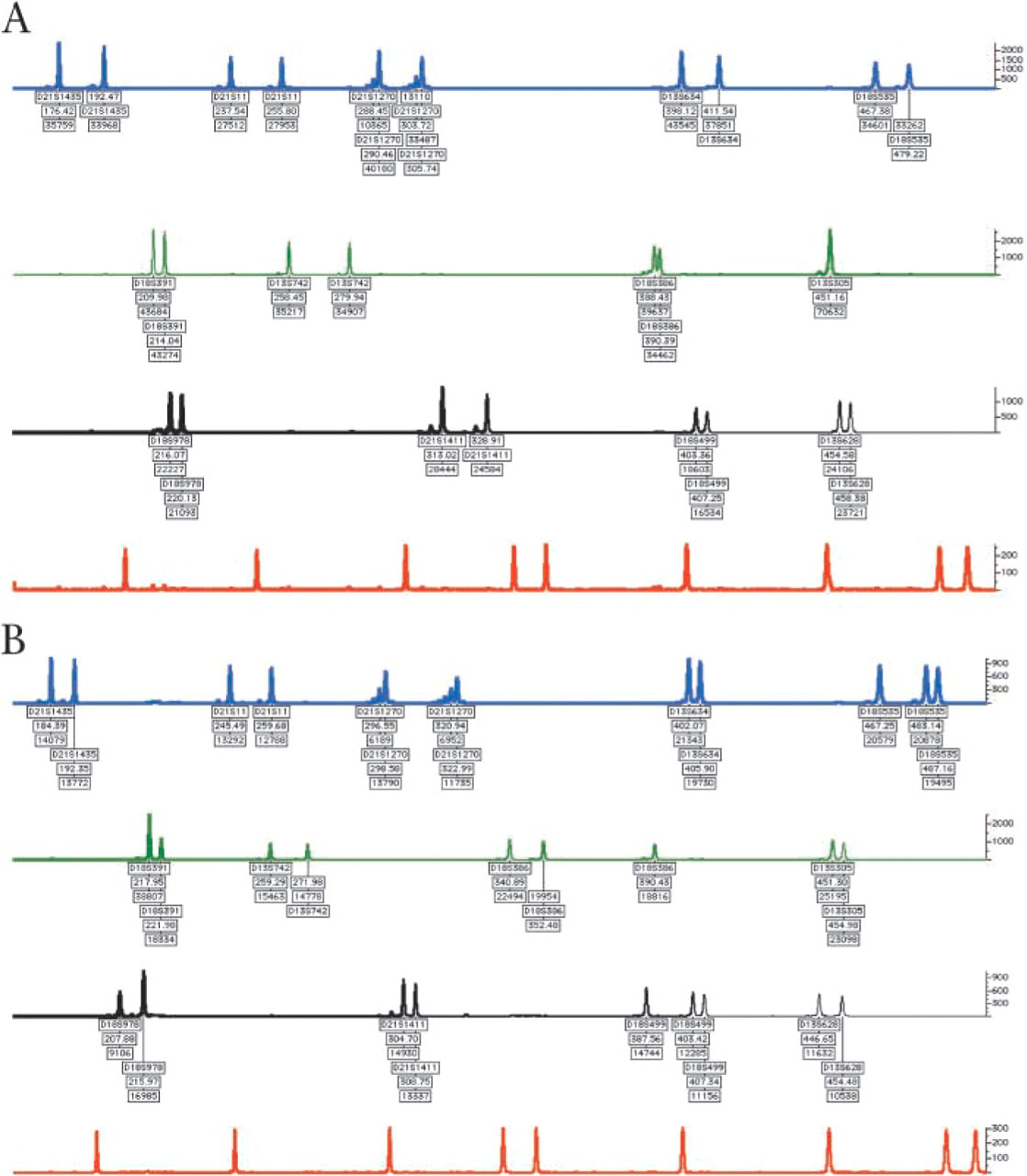
Electrophoretograms of four samples generated using Genotyper software (ABI; Foster City, CA). Arbitary fluorescence shown on the vertical axis. Markers labeled according to locus, length (bp), and peak area. (
Since the introduction of a separate sex chromosome multiplex, 7794 prenatal samples (1753 CVS and 6041 AF samples) have been received and, of these, 272 (187 CVS and 85 AF samples) had referrals indicative of 45,X and were tested using either the sex chromosome multiplex previously published (Donaghue et al. 2003) or the modified multiplex described here (Figures 1C and 1D). Of the 187 CVS tested, 29 (15.5%) had QF-PCR traces indicative of monosomy X; of the 85 AF samples tested, 7 (8.2%) were indicative of monosomy X. These results were confirmed by FISH analysis prior to reporting and were consistent with the follow-up karyotype analysis. All but two 45,X samples in this sample set were detected by the application of these selection criteria for testing. The two that did not fulfil the selection criteria were referred with nuchal measurements of 4.6 mm and 2.9 mm. No structural abnormalities or other sex chromosome aneuploidies were detected by the QF-PCR assay.
Our center applies sex chromosome testing only to pregnancies referred with characteristics suggestive of 45,X (Turner syndrome). This is because QF-PCR with primers for loci on the sex chromosomes will, in addition to 45,X, detect other sex chromosome aneuploidies such as 47,XXY (Klinefelter syndrome) and 47,XYY. These aneuploidies are associated with mild clinical phenotype and establishing their presence in pregnancies is of debatable value. In previously published multiplexes, the presence of the Y chromosome has been established by amplification of the amelogenin locus (Cirigliano et al. 1999; Donaghue et al. 2003). However, deletion of this locus in a proportion of the population has been reported (Steinlechner et al. 2002) with associated possibilities of misdiagnosis of a normal male as a Turner syndrome female. The multiplex reported here has, in addition to primers on the short and long arms of the X chromosomes, primers for the SRY locus on the short arm of the Y chromosome and two loci on the long arm and, thus, has the potential to diagnose structural abnormalities of the Y chromosome, such as i(Yp). In addition, there are other sex chromosome abnormalities, such as i(Xq), which are associated with a phenotype similar to Turner syndrome. The sex chromosome multiplex described here will detect i(Xq) and other structural abnormalities.
Details of primers used in the QF-PCR sex chromosome multiplex
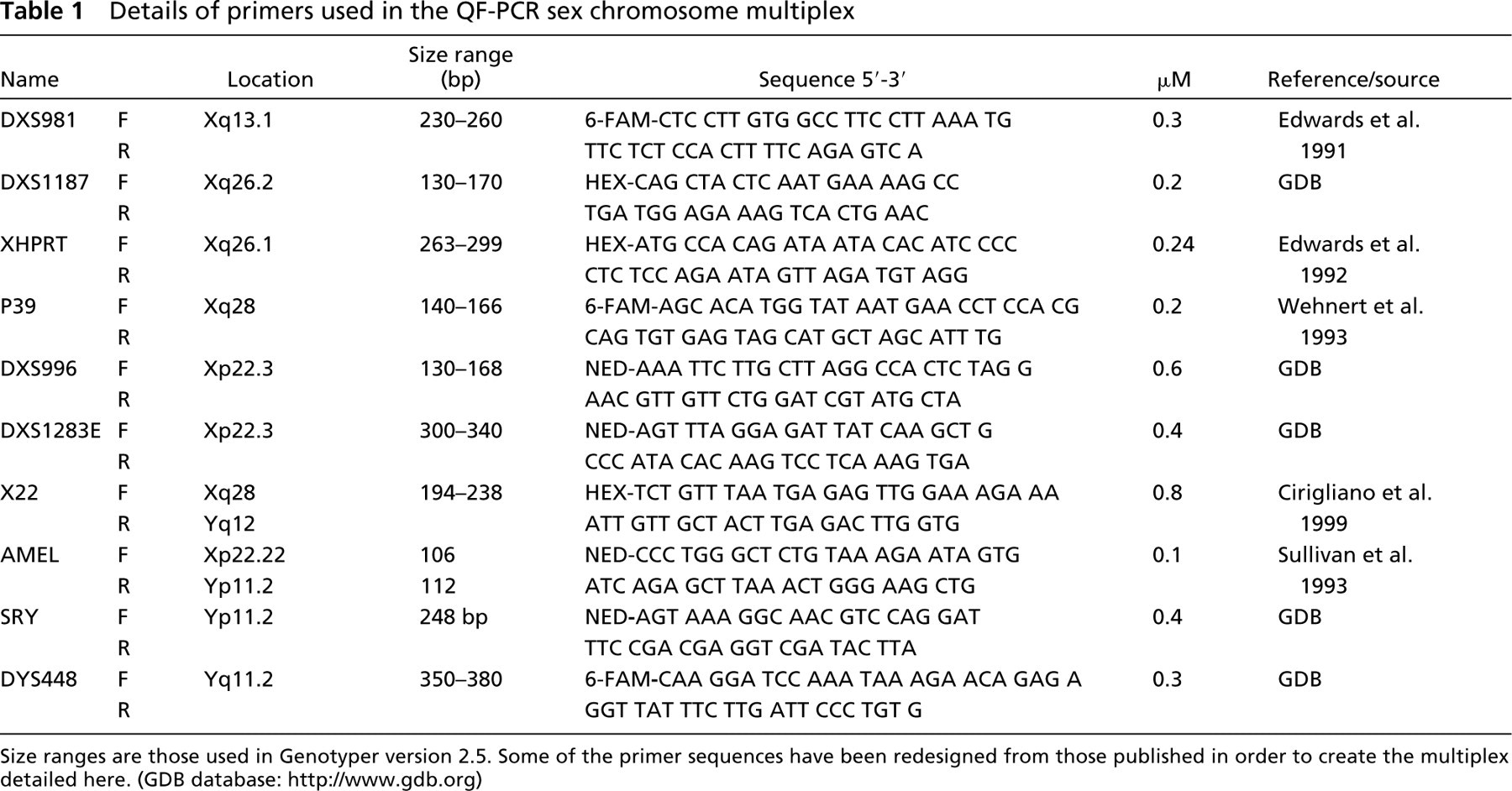
Size ranges are those used in Genotyper version 2.5. Some of the primer sequences have been redesigned from those published in order to create the multiplex detailed here. (GDB database: http://www.gdb.org)
Summary of the prenatal results using the QF-PCR trisomy assay (June 2000 to March 2004)

The targeted approach to sex chromosome testing (Donaghue et al. 2003) detected all but two cases of non-mosaic 45,X in the sample set reported here. All rapid results indicating 45,X were confirmed by FISH due to the small possibility (1:907) (Donaghue et al. 2003) that the result may represent a normal female with X chromosomes homozygous for the markers tested. The QF-PCR trace from such a case would be indistinguishable from a 45,X female. In the samples not targeted for QF-PCR sex chromosome testing, karyotype analysis found two cases of 47,XXX, four cases of 47,XXY, one case of 47,XYY, one balanced (X;8) translocation, one unbalanced (X;9) translocation, and one terminal deletion of the long arm of the X chromosome. It has been suggested that QF-PCR could be used as a stand-alone test for women referred for prenatal testing because of an increased risk of Down syndrome (Leung et al. 2003). In the event of this change of policy, a rapid test that did not detect chromosome abnormalities of unknown or negligible significance (such as 47,XYY) would alleviate counseling difficulties and parental anxiety.
QF-PCR for the detection of common chromosomal trisomies and sex chromosome aneuploidy has been introduced as a validated service into a number of European centers (Schmidt et al. 2000; Cirigliano et al. 2001; Levett et al. 2001; Mann et al. 2001). These centers use different multiplexes and different approaches to sex chromosome testing; however, QF-PCR appears to be a robust generic approach to rapid prenatal diagnosis and has advantages over other published approaches (Mann et al. 2004). The data set presented here confirms that QF-PCR is reliable, accurate, and robust, with no misdiagnoses for non-mosaic trisomy 13, 18, or 21, or triploidy.
Footnotes
Acknowledgements
The authors thank the other members of the prenatal team for their contribution and Alysia Hallam for technical assistance.
