Abstract
Recent studies suggest that bismuth accumulates in Leydig cells. In addition, a reduced level of serum testosterone and a statistically significant reduction of Leydig cells have been observed. It was therefore hypothesized that Bi has a direct toxic effect on rat Leydig cells. We have now developed a method for double labeling of bismuth and ED-2 (a marker for testicular macrophages). The present data demonstrate that the heavily bismuth-loaded cells in rat testis, originally interpreted as being Leydig cells, are bismuth-loaded macrophages. Consequently, our data suggest a modified hypothesis regarding bismuth-induced interactions between testicular macrophages and Leydig cells.
T
Materials and Methods
Fifteen male Wistar rats, weighing 300 g (Møllegaard Breeding Center; Ejby, Denmark) were divided into two groups. In group 1 10 rats each received one IP injection of 500 mg/kg bismuth subnitrate and were allowed to survive for 2 weeks. In group 2, five rats served as controls. The animals were housed in plastic cages under the following conditions: 12-hr light/dark cycle, 22 ± 2C, and 50 ± 10% relative humidity. Food (Altromin No. 1314; Altromin Spezialfutterwerke, Mannheim, Germany) and tap water were given ad libitum. The study was undertaken in accordance with the Danish and University of Aarhus guidelines for animal welfare.
At the end of the experiment, the rats were anaesthetized with sodium pentobarbital (50 mg/kg body weight) and decapitated. The testes were excised and frozen with gaseous CO2. Thirty-μm cryosections were cut.
Sections were divided into three groups: (a) immunohistochemically stained for macrophages using the anti-ED-2 monoclonal antibody (Serotec; Indianapolis, IN) (Hutson 1990), (b) reacted for Bi using the previously established AMG technique for Bi localization (Danscher et al. 2000), or (c) treated for both macrophage and Bi localization. Control procedures concerning the specificity of bismuth staining were applied as recommended (Danscher et al. 2000) and the procedures for ED-2 IHC were as described by Hutson (1990). All sections were counterstained with toluidine blue.
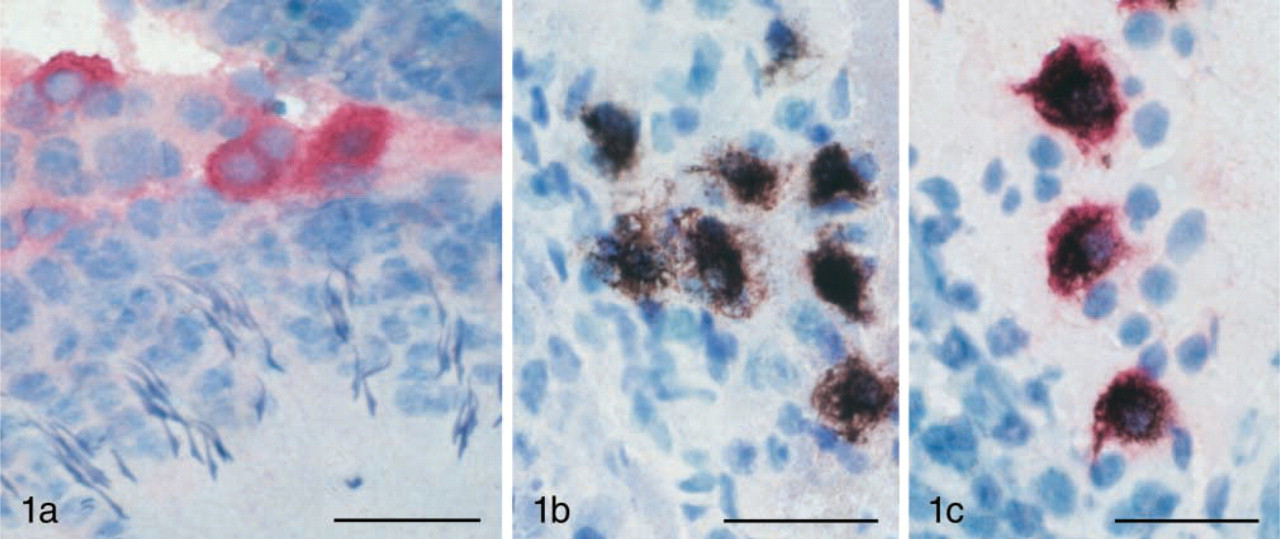
Pictures from an animal treated IP with 500 mg/kg bismuth subnitrate and allowed to survive for 2 weeks. All sections are 30-μm-thick cryosections from rat testis, counterstained with toluidine blue. (
Results and Discussion
Using single-labeling techniques we found both ED-2 positive cells, i.e., macrophages, and bismuth-loaded cells, i.e., presumptive Leydig cells, to be present in the interstitial tissue at a similar density (Figures 1a and 1b). The non-bismuth-exposed animals (controls) did not demonstrate any AMG staining whatsoever, and the proportion of the ED-2-positive cells was similar in the two groups.
Most surprisingly, when sections from bismuth-exposed animals were processed with both ED-2 and AMG (Figure 1c), most of the AMG-positive cells were ED-2-positive as well (mean value 95.7, range 93–98%), indicating that the accumulation of bismuth occurs primarily in macrophages. The number of double-labeled cells was obtained by counting 100 AMG-positive cells in sections from the bismuth-exposed animals. In our previous studies we demonstrated toxic effects of bismuth on Leydig cell viability and steroidogenic capacity, and speculated that bismuth was causing these effects by acting directly on the Leydig cells. The present studies suggest that bismuth may be acting indirectly by way of macrophages, cells that have been shown to influence Leydig cell steroidogenic activity (Yee and Hutson 1985; Hales 1996). Some macrophage-derived factors (IL-1, IL-6, and TNF-α) are inhibitory and one of the factors is stimulatory (25-hydroxycholesterol) (Nes et al. 2000). Because bismuth has been clearly shown to decrease testosterone production (Stoltenberg et al. 2000), it may act by increasing the production of the inhibitory factors or by decreasing the production of 25-hydroxycholesterol. Although the most likely interpretation is that bismuth is acting by interfering with macrophage-Leydig cell interactions, it is also possible that it plays a direct role in inhibiting Leydig cells during its residence in the interstitial fluid before being phagocytosed by macrophages. As the AMG technique generally visualizes heavy metals in lysosomes, it is obviously possible that toxic heavy metals exert their effect on the cells and only later are transported to lysosomes where they can be visualized.
The AMG technique was recently introduced by Danscher et al. (2000) to demonstrate accumulation of bismuth in various cell types. In this study we extend the applicability of this technique by demonstrating that it can be combined with IHC methods to localize bismuth in specific cell types. By combining these two technologies we have found that bismuth is located in testicular macrophages, and we hypothesize that the toxic effects of this compound may be targeted at the paracrine interactions that occur between macrophages and Leydig cells.
Footnotes
Acknowledgements
This study was supported by the Aarhus University Research Foundation, “Aase & Ejnar Danielsens Fond” (MS) and the NICHD (HD34708, JCH).
We wish to thank D. Jensen, A. Meier, H. Mikkelsen, T.A. Nielsen, and K. Wiedemann for excellent technical assistance.
