Abstract
Mosquitoes mount strong humoral and cellular immune responses against foreign organisms. Two components of the mosquito immune response that have received much attention are the phenoloxidase cascade that leads to melanization and antimicrobial peptides. The purpose of the current study was to use immunocytochemistry and transmission electron microscopy to identify the location of the melanization rate-limiting enzyme phenoloxidase and the antimicrobial peptide defensin in innate immune reactions against
M
The mosquito immune response elicited by invading organisms is robust and involves humoral and cellular components. The humoral component includes the phenoloxidase cascade system of parasite melanization and wound healing (Zhao et al. 1995; Lai et al. 2002), inducible antimicrobial peptides (Lowenberger 2001; Vizioli et al. 2001a), and reactive oxygen and nitrogen intermediates (Luckhart et al. 1998; Lanz-Mendoza et al. 2002; Kumar et al. 2003). The cellular component includes phagocytosis (Hernández-Martínez et al. 2002; Hillyer et al. 2003a,b,2004,2005) and encapsulation (Forton et al. 1985; Christensen and Forton 1986) by hemocytes. Of these, two areas of intense study are phenoloxidase-based melanization and the antimicrobial peptides. Melanization involves a complex series of reactions that include the hydroxylation of tyrosine by phenoloxidase to form 3,4-dihydroxyphenylalanine (dopa) and its sequential conversion to melanin by phenoloxidase and other enzymes (Christensen et al. 2005). This process is used by certain mosquito species and strains as a resistance mechanism against filarial nematodes and malaria (
The yellow fever mosquito,
Materials and Methods
Colloidal Gold Suspension Preparation
Colloidal gold was prepared by the reduction of gold chloride with sodium citrate. Briefly, 200 ml double distilled H2O was placed in an Erlenmeyer flask and heated while stirring with a magnetic bar. When the water was at a near boil, 2 ml of 1% gold(III) chloride trihydrate (HAuCl4 • 3H2O; Sigma, St Louis, MO) was added and mixed for 30 sec. A volume of 2.5 ml of 2.5% sodium citrate (Sigma) was then added and the solution soft boiled for 30 min. After the solution cooled to room temperature, the volume was readjusted to 200 ml and stored at 4C. To determine colloidal gold bead size, a nickel 200-mesh pioloform-coated TEM grid was floated on a 30-μl drop of colloidal gold suspension for 10 min. The grid was then removed, blotted to remove excess liquid, dried, and colloidal gold particles visualized by transmission electron microscopy. Micrographs were taken and bead size determined using Image Pro-Plus software (Media Cybernetics; Silver Spring, MD). Calibration was done using the scale bar of the micrograph. Suspensions were made twice: the first colloidal gold suspension averaged 12.7 nm in diameter (±1.02 nm SD) and was conjugated to mouse anti-rabbit IgG, and the second colloidal gold suspension averaged 14.36 nm in diameter (±1.17 nm SD) and was conjugated to protein A (see below).
Conjugation of Proteins
Protein A (Sigma) and mouse anti-rabbit IgG (Pierce Biotechnology; Rockford, IL) were conjugated to colloidal gold particles by hydrophobic bonding. Initially, concentration and pH adsorption isotherms were done to determine the optimal conditions for conjugation (Albrecht et al. 1993). Briefly, 200 μl of colloidal gold at various pHs (ranging from 5 to 9) was added to 20 μl of Protein A or mouse anti-rabbit IgG at varying concentrations (ranging from 200 μg/ml to 25 μg/ml, in water) and mixed. After 20 min, 100 μl of a saturated NaCl solution was added and mixed. After another 20 min the color of the conjugate was assessed: red/pink was indicative of a stable conjugate and blue/purple was indicative of an unstable conjugate. The colloidal gold pH and protein concentration combination that required the minimum amount of protein to stabilize the colloidal gold, plus 10% excess protein, was used to make the final conjugates. For the final conjugation of mouse anti-rabbit IgG to 12.7 nm colloidal gold, 18 ml of colloidal gold (pH 7.3) was added to 1.8 ml of 75 μg/ml mouse anti-rabbit IgG (in water). For the final conjugation of Protein A to 14.4 nm colloidal gold, 10 ml of colloidal gold (pH 5.9) was added to 1 ml of 50 μg/ml Protein A (in water). Both solutions were individually mixed and allowed to stand for 30 min. The conjugates were then spun at 10,600 × g for 8 min, the supernatant removed, and the loose pellet resuspended in storage buffer (20 mM Tris base, 20 mM NaN3, 150 mM NaCl, 0.1% BSA, pH 7.2) to one tenth the original volume.
Mosquito Rearing, Immune Challenges, and Sample Collections
Because only female mosquitoes take blood meals and, hence, are the only mosquitoes capable of transmitting diseases through infective bites, the current study focused exclusively on the immune response elicited in females. Female
To immune challenge mosquitoes, cultured bacteria at stationary phase were centrifuged at 2655 × g for 5 min and the supernatant withdrawn. A 0.15-mm steel probe was dipped in the bacterial pellet and inserted through the neck membrane of 1–5-day-old adult female mosquitoes (Hillyer et al. 2003b). At 1 hr and 24 hr postchallenge, hemolymph was collected by volume displacement (perfusion) directly into formaldehyde (Electron Microscopy Sciences; Hatfield, PA) and glutaraldehyde (Electron Microscopy Sciences) fixative as described (Hillyer et al. 2003a). One min after completing mosquito hemolymph collection, the supernatant was transferred to a new tube, allowed to fix for an additional 1 hr, and centrifuged for 20 min at 210 × g. The supernatant was removed and the cellular pellet was embedded in a plug of low-melting-temperature agarose. The agarose plug was then fixed for 20 min, rinsed with buffer, cut into 1.5-mm cubes, dehydrated through 70% ethanol, and embedded in LR-White resin (Polysciences; Warrington, PA). Each sample was comprised of the pooled hemolymph of 50–100 mosquitoes of the same age. A total of six sets of perfusions were done, each yielding at least two samples after cutting the agarose plugs. Furthermore, samples were prepared for 1-, 4-, and 5-day-old mosquitoes at 24-hr postchallenge and for 5-day-old mosquitoes at 1-hr postchallenge.
Labeling and Transmission Electron Microscopy
All labeling experiments were done at room temperature, and all solutions and rinses were made in storage buffer (20 mM Tris base, 20 mM NaN3, 150 mM NaCl, 0.1% BSA, pH 7.2) unless otherwise noted. One hundred-nm-thick sections were cut using a diamond knife on a Reichert Om U3 ultramicrotome (Reichert; Vienna, Austria) and transferred to 200-mesh pioloform-coated nickel grids. Sections were allowed to dry for 10–30 min and rehydrated for 5 min. Sections were then blocked (5% fetal calf serum when using IgG conjugate or 1% BSA when using Protein A conjugate) for 30 min, rinsed three times for 3 min each, incubated in primary antibody [rabbit anti-defensin (Bartholomay et al. 2004) or rabbit anti-phenoloxidase (Lai et al. 2002)] for 2 hr, rinsed three times for 3 min each, incubated in Protein A-colloidal gold or mouse anti-rabbit IgG-colloidal gold for 2 hr, rinsed three times for 3 min each, rinsed three times for 3 min each in water, and allowed to dry. Sections were then counterstained for 5 min in 1% uranyl acetate and for 3 min in Reynold's lead, and viewed with a Philips CM120 TEM (Philips Electron Optics; Eindhoven, The Netherlands) as described (Hillyer and Albrecht 1998).
A total of 10 experiments were done, each including multiple samples, and four of the samples were independently labeled in separate trials at least twice. For all labeling experiments, serial sections for all samples were labeled for both defensin and phenoloxidase. Out of the 10 labeling experiments, 6 were done using mouse anti-rabbit IgG-colloidal gold and 4 with Protein A-colloidal gold. These experiments yielded similar results, with the exception that experiments using IgG-colloidal gold resulted in higher labeling intensities. Controls included labeling cultured bacteria (bacteria never injected into mosquitoes), omission of the primary antibody, and the use of an irrelevant antibody. Control samples were run on three of the labeling experiments with identical results. Finally, labeling experiments for samples collected at 1 hr and 24 hr postchallenge were done within days of each other and with the same reagents. Because they were not labeled on the same day, colloidal gold particles on bacterial melanotic capsules and on other regions of the section were counted, and statistical analysis (Student's
When observing hemolymph samples from bacteria-challenged mosquitoes collected by perfusion, the biological structures present are hemocytes, fat body, cellular debris from lysed cells (predominantly nuclei, mitochondria, and membranes), and bacteria (melanized and unmelanized) (Hillyer and Christensen 2002; Hillyer et al. 2003a). For all labeling experiments, all structures were examined for the presence of colloidal gold particles.
Results
Localization of Phenoloxidase
We have previously reported in naïve adult
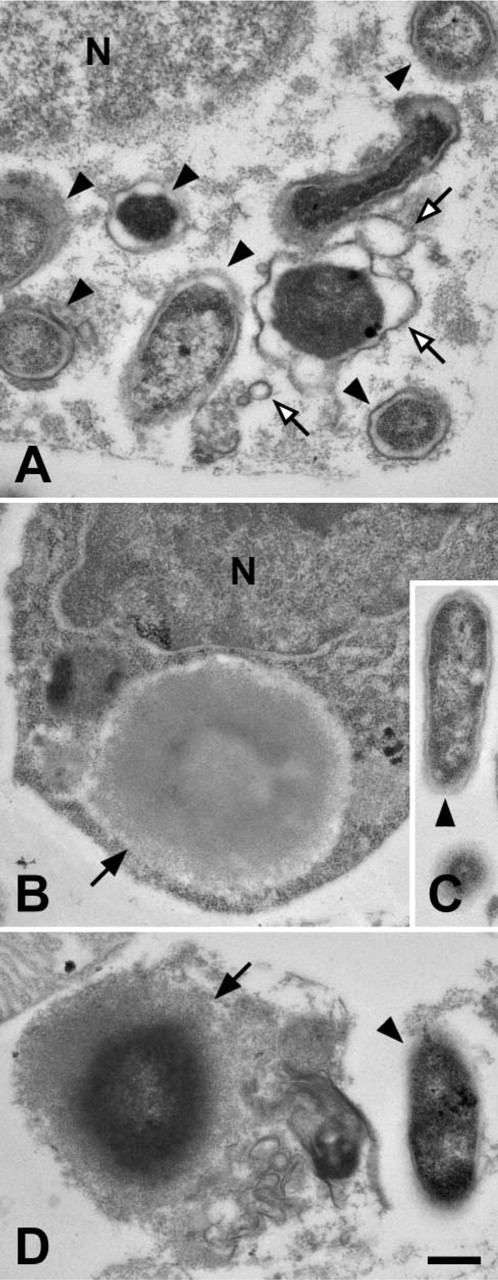
When hemocytes were examined, oenocytoids were the only cell type that contained phenoloxidase independent of bacteria and, similar to naïve mosquitoes (Hillyer and Christensen 2002), phenoloxidase was scattered throughout the cytoplasm and not inside membrane-delimited vesicles (Figure 1D). Phenoloxidase was also detected bound to bacteria that had been phagocytosed by granulocytes (Figure 1A). However, because in naïve and infected mosquitoes phenoloxidase is only present in the cytoplasm of oenocytoids (Hillyer and Christensen 2002; this study), phenoloxidase likely binds bacteria and initiates melanization in the hemolymph prior to phagocytosis. In addition to hemocytes and melanized bacteria, phenoloxidase was also occasionally observed on ruptured membranes (Figure 1B) in a manner similar to that seen in
Localization of Defensin
Defensin is one of the most studied antimicrobial peptides in mosquitoes. Because it has been widely speculated that defensin functions by lysing bacteria, we originally expected to see defensin bound to unmelanized bacteria. This was not the case. Similar to phenoloxidase, at 24 hr postchallenge, defensin localized to
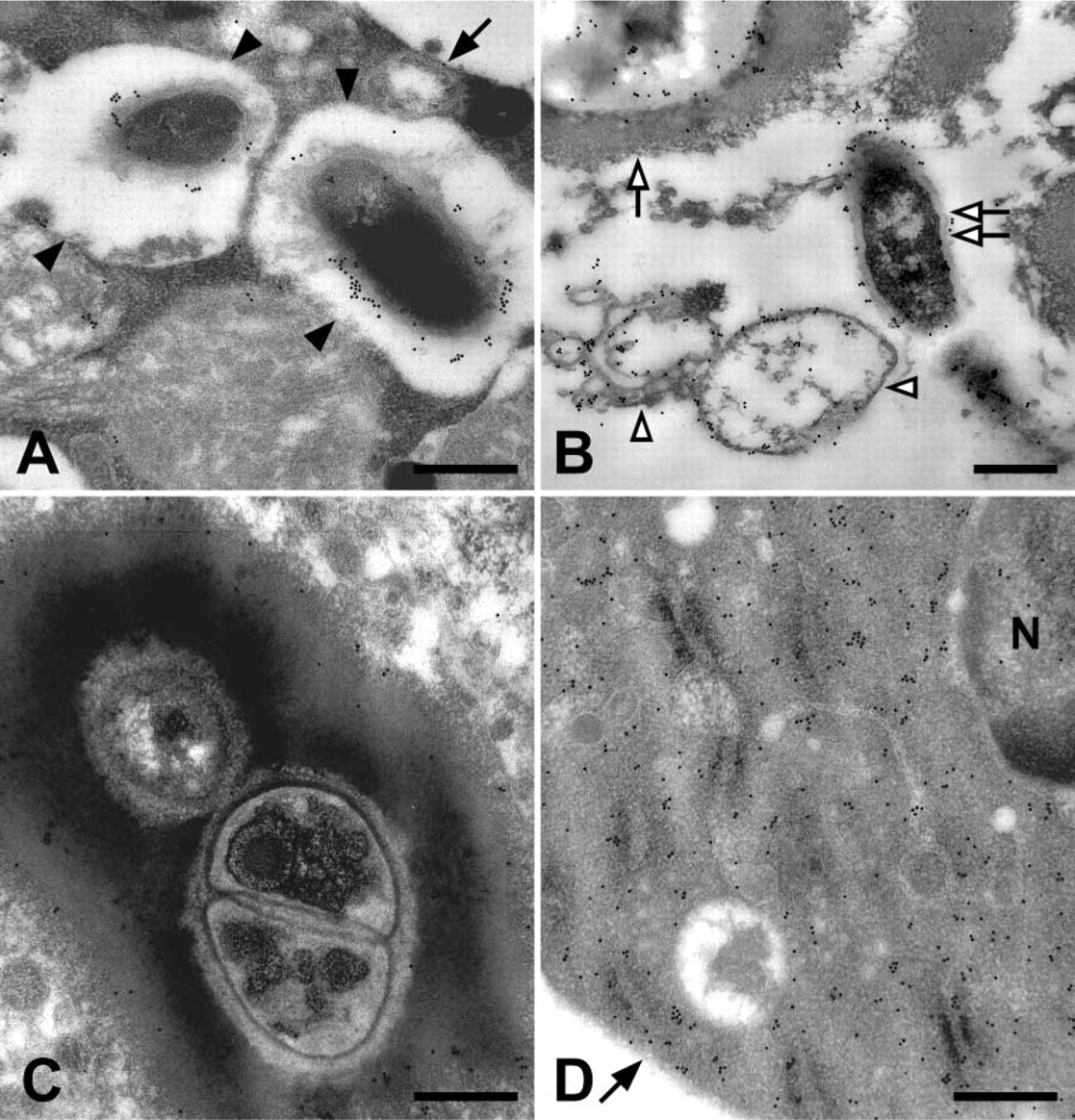
Colloidal gold labeling of mosquito hemolymph tissues for phenoloxidase. (
When hemocytes were examined, defensin was occasionally found inside membrane delimited vesicles of granulocytes (Figures 2B and 2E). In most instances, the defensin molecules in these vesicles were attached to phagocytosed bacteria (Figure 2B), but in rare occasions defensin was also observed in these vesicles in the absence of bacteria (Figure 2E). However, given the rarity of this event and that these membrane-delimited vesicles are resorptive in nature (Hillyer et al. 2003b,2004), it is unlikely that granulocytes produce defensin. Also, defensin was not present in oenocytoids (Figure 2F) or adipohemocytes (not shown).
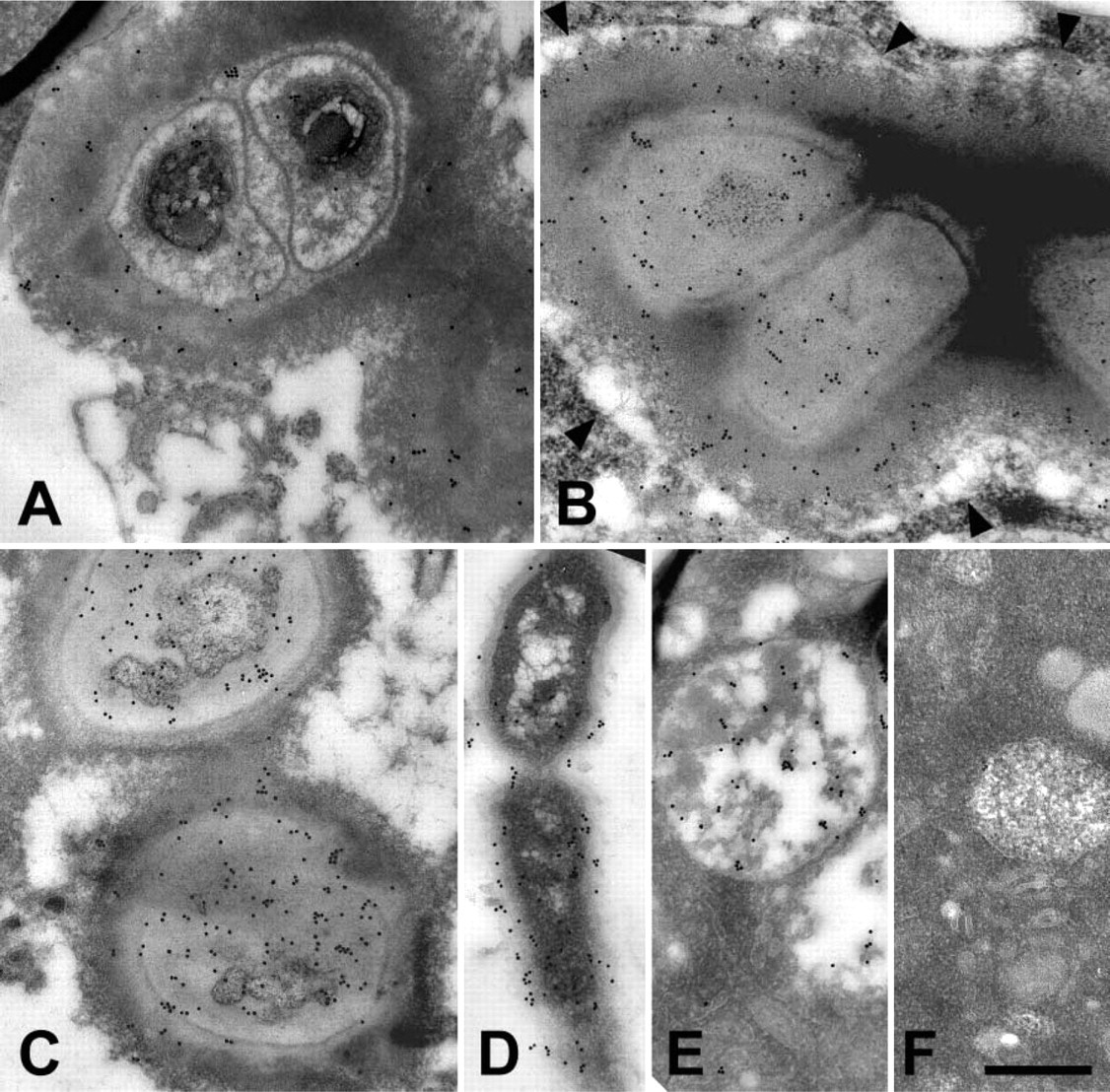
Colloidal gold labeling of mosquito hemolymph tissues for defensin. (
Because defensin is not detectable in naïve mosquitoes and becomes measurable for the first time several hours following challenge (Lowenberger et al. 1999b; Bartholomay et al. 2004), we labeled, in parallel, samples collected at 1 hr and 24 hr following bacterial challenge. Defensin was present in melanotic capsules at both 1 hr and 24 hr postchallenge (Figure 3). The overall labeling pattern was similar for both timepoints. However, the intensity of labeling at 24 hr postchallenge was 3.6 times higher than at 1 hr postchallenge (Figure 3). When comparing samples labeled at 1 hr postchallenge vs 24 hr postchallenge, background for both was negligible and statistically similar (
Co-localization of Phenoloxidase and Defensin
Prior to the initiation of this work, our working hypothesis was that phenoloxidase would localize to melanized bacteria and defensin would localize to unmelanized bacteria. Surprisingly, both localized to melanized bacteria. Because of this finding, serial sections of hemolymph samples collected at 24-hr postchallenge were cut and one labeled with defensin and the other with phenoloxidase. On many occasions, defensin and phenoloxidase colocalized to the same
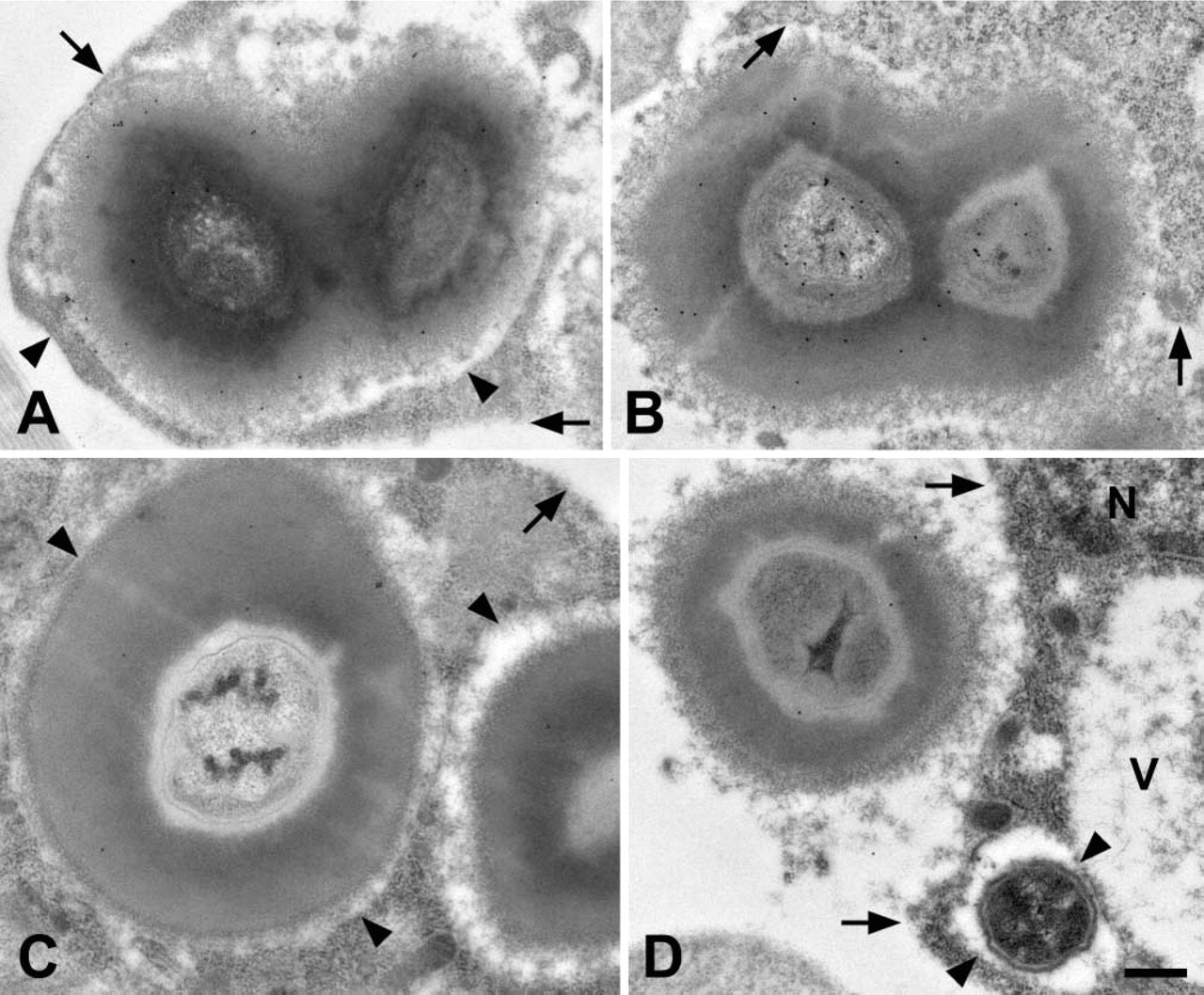
Comparison of defensin labeling at 1 hr and 24 hr postchallenge. Samples collected at 24 hr postchallenge (
Evaluation of Control Samples
Labeling of defensin and phenoloxidase in melanization reactions is specific. When identical labeling experiments were carried out on embedded cultured bacteria (bacteria never injected into mosquitoes), no significant labeling was observed (Figure 5;
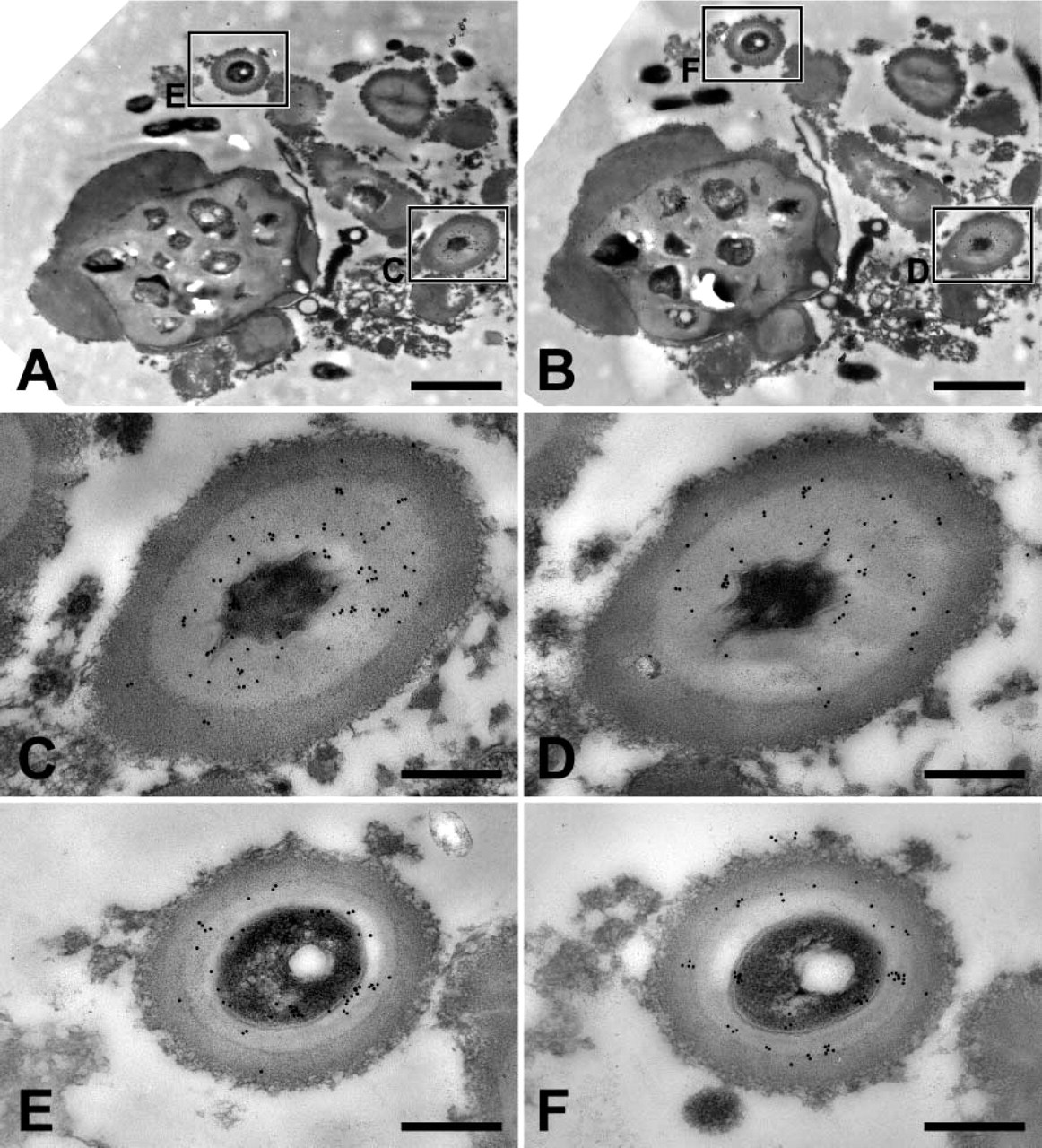
Colocalization of phenoloxidase and defensin in melanization reactions. (
Discussion
To date, much work has been directed toward characterizing the enzymes involved in melanization reactions (Zhao et al. 1995; Beerntsen et al. 2000). In a similar manner, much work has been directed toward identifying mosquito proteins that have antimicrobial activity in vitro (Lowenberger 2001). Nevertheless, our understanding of these two humoral processes is far from complete. Melanization involves a complex series of reactions that involve the sequential conversion of dopa to melanin by the enzymes phenoloxidase, dopa decarboxylase, dopachrome conversion enzyme, and others (Zhao et al. 1995). This enzymatic cascade is well understood, and silencing genes that produce these enzymes result in decreased melanization efficiency (Shiao et al. 2001; Infanger et al. 2004). However, the molecules responsible for triggering these site-specific melanization reactions have not been well characterized, although recently a thioester-containing protein and a glucan-binding protein have been shown to be necessary for the successful melanization of
In
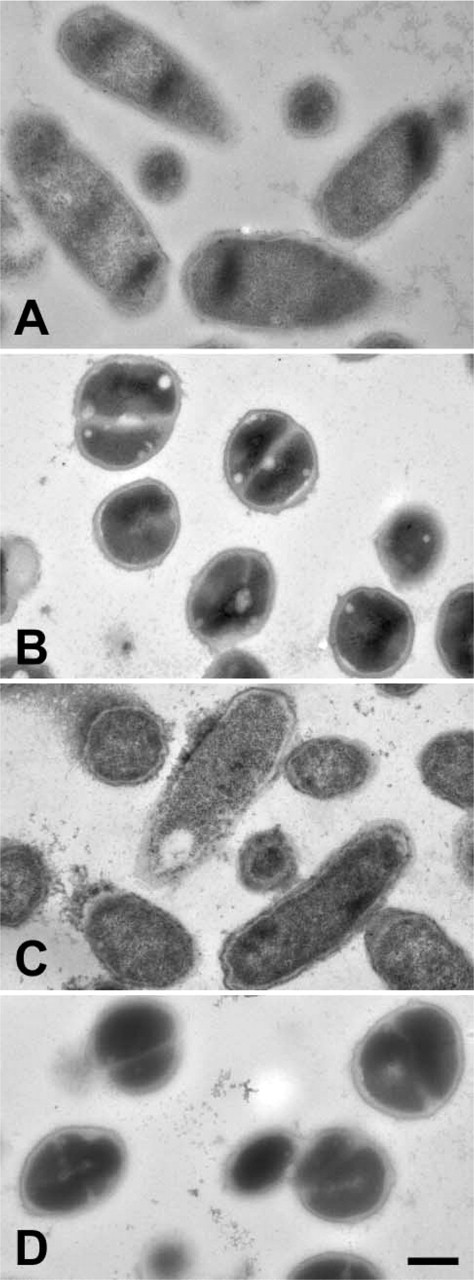
Phenoloxidase and defensin labeling of cultured bacteria (bacteria never injected into a mosquito). (
Labeling of mosquito immune reactions in the absence of primary antibody. No significant labeling of
Furthermore, in the current study, the cellular immunolocalization of phenoloxidase was as expected: in bacteria-challenged mosquitoes, phenoloxidase was found exclusively in oenocytoids. This is in accord with our previous studies in
In summary, these data show that defensin and phenoloxidase colocalize in melanization immune responses and suggest that the antimicrobial peptide defensin may be involved in the mosquito melanization response against bacteria. These data argue for further studies into the potential role of defensin in melanization innate immune responses in mosquitoes.
Footnotes
Acknowledgements
This work was supported by NIH Grant F31 AI-50252 to J.F.H. and NIH Grants AI-19769 and AI-46032 to B.M.C.
We thank S.L. Schmidt for mosquito rearing. We also thank C.C. Chen and J. Vizioli for providing the anti-phenoloxidase and anti-defensin antibodies, respectively. Useful discussions with A.J. Nappi are also greatly appreciated.
