Abstract
Highly purified primitive hemopoietic stem cells express BMP receptors but do not synthesize bone morphogenetic proteins (BMPs). However, exogenously added BMPs regulate their proliferation, differentiation, and survival. To further explore the mechanism by which BMPs might be involved in hemopoietic differentiation, we tested whether stromal cells from long-term culture (LTC) of normal human bone marrow produce BMPs, BMP receptors, and SMAD signaling molecules. Stromal cells were immunohistochemically characterized by the presence of lyzozyme, CD 31, factor VIII, CD 68, S100, alkaline phosphatase, and vimentin. Gene expression was analyzed by RT-PCR and the presence of BMP protein was confirmed by immunohistochemistry (IHC). The supportive role of the stromal cell layer in hemopoiesis in vitro was confirmed by a colony assay of clonogenic progenitors. Bone marrow stromal cells express mRNA and protein for BMP-3, -4, and -7 but not for BMP-2, -5, and -6 from the first to the eighth week of culture. Furthermore, stromal cells express the BMP type I receptors, activin-like kinase-3 (ALK-3), ALK-6, and the downstream transducers SMAD-1, -4, and -5. Thus, human bone marrow stromal cells synthesize BMPs, which might exert their effects on hemopoietic stem cells in a paracrine manner through specific BMP receptors.
I
The regulation of hemopoiesis is a complex process, which requires signaling among stromal cells, stem cells, and progenitor cells. Members of TGF-β family signal through two types of serine/threonin kinase receptors and phylogeneticaly highly conserved intracellular transducer molecules SMADs (Korchynsky and ten Dijke 2002; Miyazawa et al. 2002). Various mechanisms precisely regulate BMP signals by positive regulation and negative feedback, both important for gradient formation, which is needed for proper development (Jonsson et al. 1997; Miyazono 2000; Miyazono et al. 2001; Korchynsky and ten Dijke 2002; Martinovic et al. 2002). Moreover, signaling by SMADs is often modulated by crosstalk with other signaling pathways which gives this superfamily a broad array of biological activities (Miyazono et al. 2001).
It is unknown whether BMPs are expressed in adult and differentiated stromal cell layers required to support hemopoiesis and eventually to play a role in regulation of hemopoietic stem cell differentiation. We report that long-term cultures produce BMPs that are required for the maintenance of hemopoiesis.
Materials and Methods
Cell Culture
Normal human bone marrow specimens for long-term culture were obtained from healthy donors by aspiration of the posterior iliac crest during the standard procedure for allogenic bone marrow transplantation. The procedures followed were in accordance with the Helsinki Declaration of 1975 and approved by the Ethical Committee of Medical School University of Zagreb, Croatia. Long-term cultures of human hemopoietic cells were established according to Eaves et al. (1991). Briefly, mononuclear cells were separated on a Ficoll density gradient (Amersham Pharmacia Biotech; Uppsala, Sweden) and cultivated in Myelocult H5100 medium (StemCell Technologies; Vancouver, Canada) with freshly prepared and filter-sterilized hydrocortisone sodium succinate (final concentration of 10−-6 M). The cultures were kept for 4 days at 37C, and after the initiation of the adherent stromal layer were transferred to 33C in a humidified atmosphere with 5% CO2. Half-media were changed once weekly and cultures were maintained up to 8 weeks. Short-term cultures of clonogenic progenitors were prepared at every half-medium change. Cells were seeded in semisolid Methocult H4433 medium (StemCell Technologies) at a concentration of 50,000 cells/well and were cultivated for 14 days. Human clonogenic progenitors were scored according to standard criteria (Rogulj et al. 1999).
Immunohistochemistry
Cells were seeded in eight-well chamber slides at a concentration of 12.5 × 104/ml (5 × 104/well) and incubated in Myelocult H5100 medium for 5 weeks. Cells were fixed in 4% paraformaldehyde in PBS for 5 min, washed three times in PBS, immersed in methanol for 1 min, treated with 3% H2O2 for 5 min, and washed in PBS. Immunostaining was performed using BMP-3 (Vukicevic et al. 1994a), BMP-4 (Santa Cruz Biotechnology; Santa Cruz, CA), and BMP-7 antibody (Vukicevic et al. 1994b) with a biotinylated secondary antibody (Vectastain ABC kit; Vector Laboratories, Burlingame, CA) and horseradish peroxidase-conjugated streptavidin (Vector). Parallel cultures were stained with rabbit anti-human lysozyme, mouse anti-human CD31, mouse anti-human factor VIII, mouse anti-human CD68, rabbit anti-cow S100, rabbit anti-calf alkaline phosphatase, and rabbit anti-vimentin antibodies (DAKO; Glostrup, Denmark). For negative controls the primary antibody was replaced by BSA or the secondary antibody alone, and skin and bone marrow tissue samples were used as positive controls.
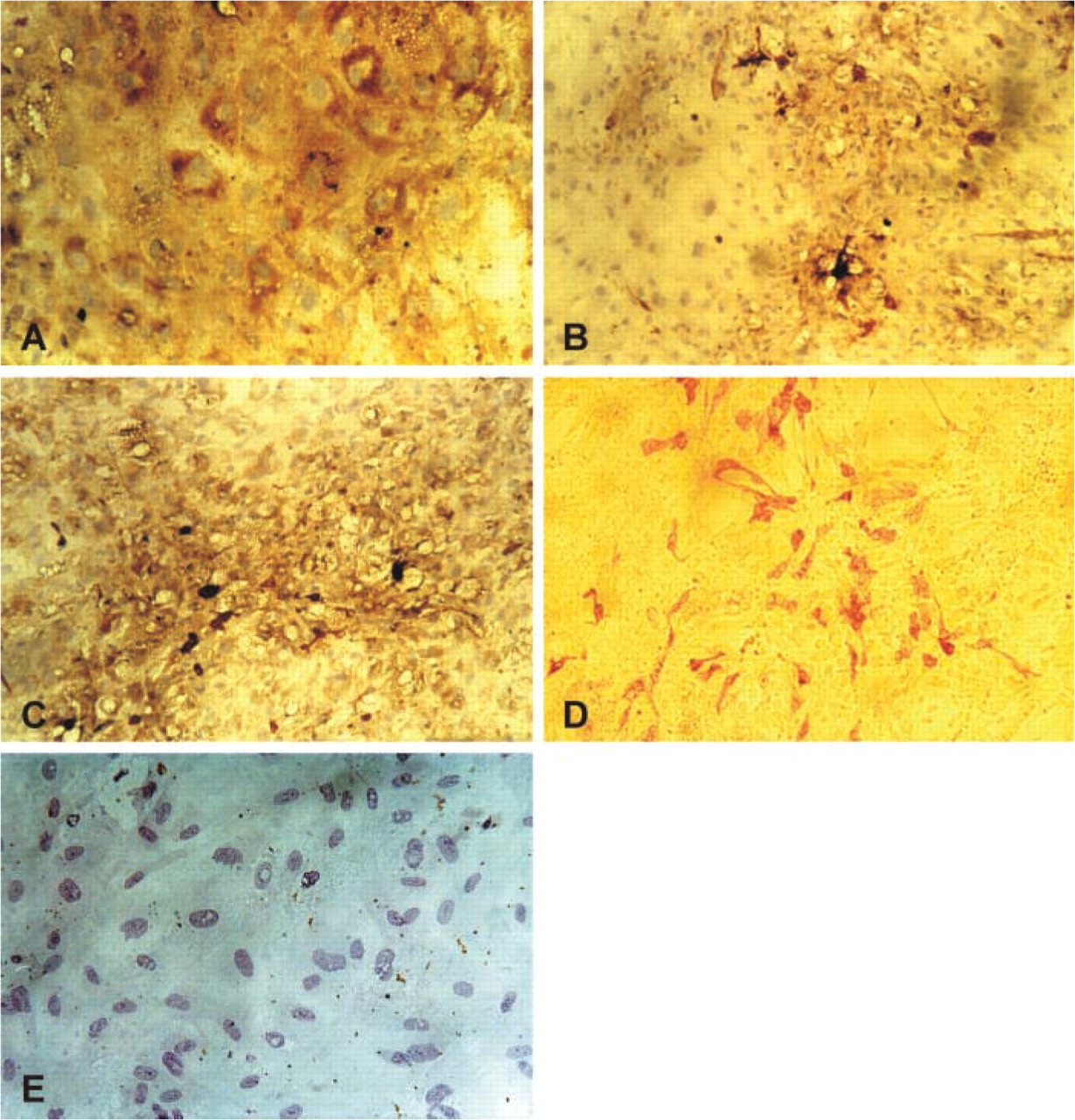
Immunohistochemical staining of human bone marrow stromal cells. Cells were cultured for 5 weeks and stained as described in Materials and Methods for vimentin (
Reverse Transcription PCR Analysis
Total RNA was extracted from freshly isolated bone marrow samples, purified peripheral lymphocytes, and bone marrow samples after 1, 3, 5, and 8 weeks in culture using the guanidine thiocyanate/acid phenol method as indicated by the manufacturer (TRIzol reagent; Gibco BRL, Grand Island, NY). Contaminating genomic DNA was removed with RNase-free DNase (Gibco BRL). cDNA was synthesized with Superscript II RNase H-Reverse Transcriptase as indicated by the manufacturer (Gibco BRL). PCR was performed in the PE GeneAmp 2400 thermal cycler (Perkin-Elmer; Norwalk, CT) using the following primers: GAPDH (5’ ACC ACA GTC CAT GCC ATC AC, 3’ TCC ACC ACC CTG TTG CTG TA); BMP-2 (5’ CAG AGA CCC ACC CCC AGC A, 3’ CTG TTT GTG TTT GGC TTG AC); BMP-3 (5’ TTT CTC TCC TCC CAC ACC, 3’ CAA TCT GAC ATC GCT AAC C); BMP-4 (5’ TTC CTG GTA ACC GAA TGC T, 3’ GGG GCT TCA TAA CCT CAT A); BMP-5 (5’ ACG GAA CCA CGA AAG ACG, 3’ GCC AAC CCA CAT CTA AAG C); BMP-6 (5’ GCA GAA GGA GAT CTT GTC GG, 3’ AGC TGA AGC CCC ATG TTA TG); BMP-7 (5’ TGG CGT TCA TGT AGG AGT TCA G, 3’ ACG CTT CGA CAA TGA GAC GTT C); ALK-2 (5’ TGG AAG ATG AGG AGC CCA AGG T, 3’ GAA GTT CTG CGA TCC AGG GAA G); ALK-3 (5’ CTG CTG CGC TCA TTT ATC, 3’ ACC ATC GGA GGA GAA ACT); ALK-6 (5’ AAG TTA CGC CCC TCA TTC, 3’ TGA TGT CTT TTG CTC TGC); SMAD1 (5’ CGA ATG CCT TAG TGA CAG, 3’ GAG GTG AAC CCA TTT GAG); SMAD4 (5’ AGG TGA AGG TGA TGT TTG, 3’ GCT ATT CCA CCT ACT GAT) and SMAD5 (5’ AGA TAT GGG GTT CAG AGG, 3’ TGT TGG TGG AGA GGT GTA) (Invitrogen Life Technologies; Carlsbad, CA).
Reactions included 5 μl 10× buffer (Promega; Madison, WI), 3 μl MgCl2 (Promega), 1 μl dNTP, 1 μl 3’ primer, 1 μl 5’ primer, 0.5 μl Taq polymerase (Promega), and 1 μl cDNA. After initial denaturation at 94C for 5 min, 32 to 40 cycles of amplification were completed by denaturation for 40 sec at 94C, annealing at temperature specified for each pair of primers for 40 sec, and extension for 60 sec at 72C. To compare the relative quantity of the RT-PCR reactions, the transcription level of GAPDH, a “housekeeping” gene, was used as control. Reactions without cDNA were used as negative control and kidney cDNA as a positive control. Results were visualized by gel electrophoresis in 1% agarose (Seakem GTG; Bioproducts, Rockland, MA) in TAE buffer (Tris-HCl, acetic acid, EDTA, pH 8.0) and stained with ethidium bromide (Sigma-Aldrich; St Louis, MO). Reactions were repeated at least twice.
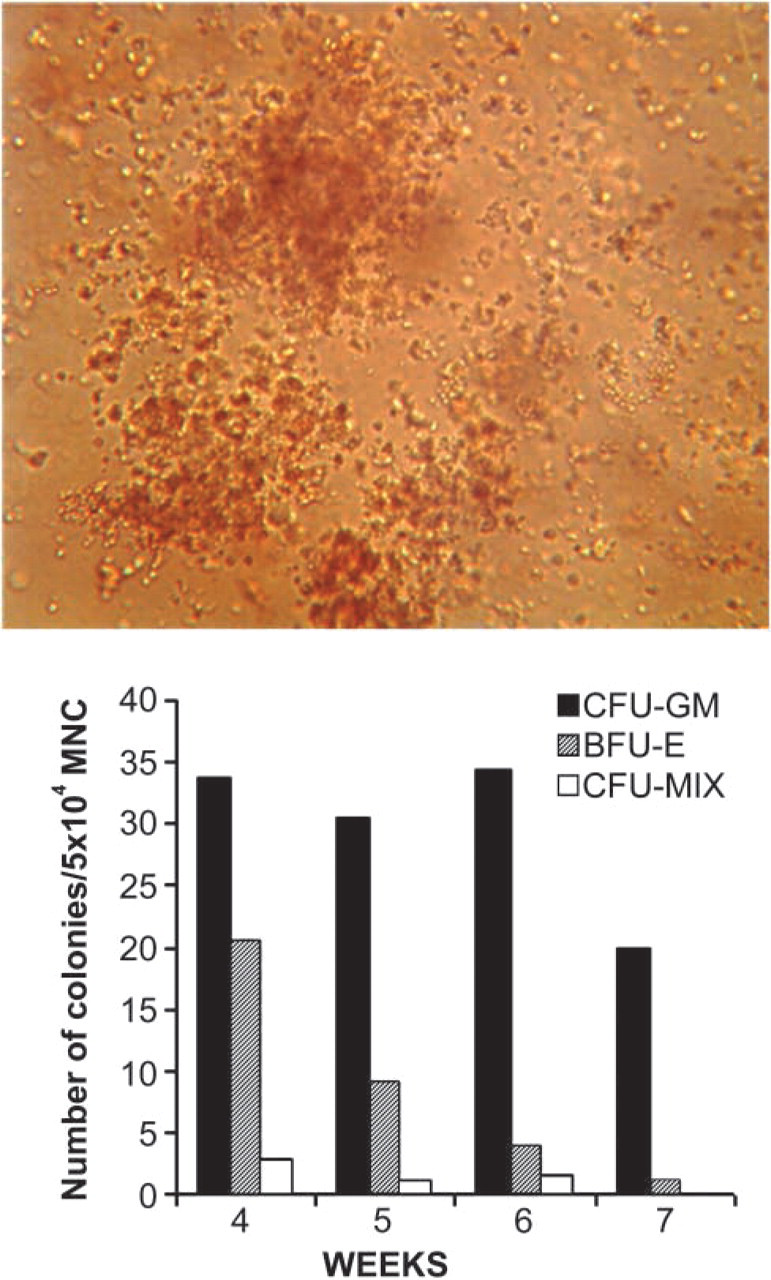
Short-term culture of clonogenic progenitors. Cells were seeded at 50,000 cells/well in semisolid Methocult medium at medium change after 4, 5, 6, and 7 weeks in culture, cultivated for 14 days, and number of clonogenic progenitors was scored according to standard criteria. Magnification ×150.
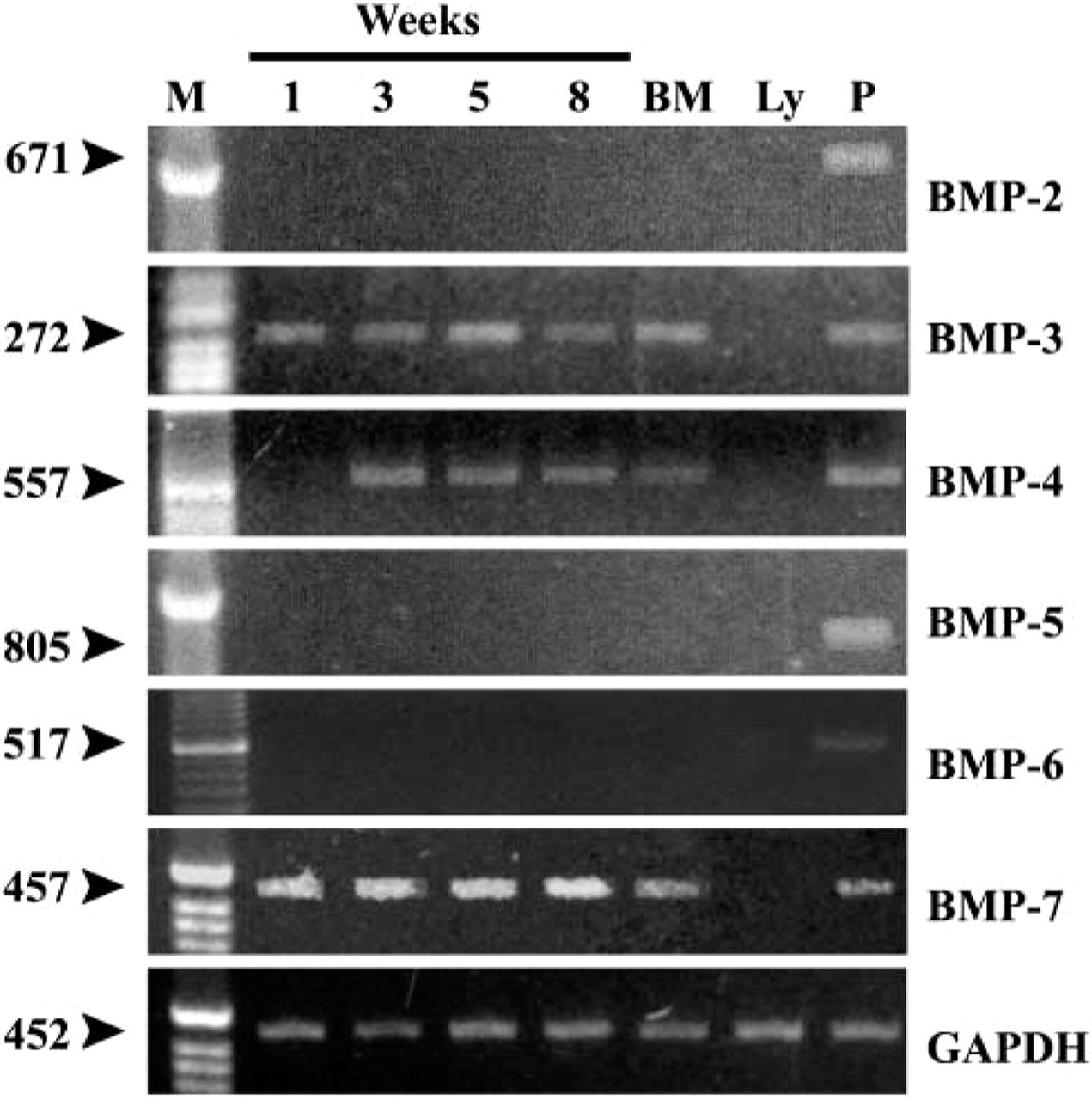
Expression of BMPs in stromal cells from human bone marrow long-term culture. Cells were cultured and gene expression analyses were done as described in Materials and Methods. RNA was isolated from freshly isolated bone marrow (BM), peripheral lymphocytes (Ly), or cultures at designated time points (1, 3, 5, 8 weeks), cDNA was synthesized and semiquantitative RT-PCR performed as described.
Results
Identity and Clonogenicity of Long-term Cultured Bone Marrow Stromal Cells
The identity of heterogeneous stromal cells was confirmed by immunostaining with commonly used markers: factor VIII and CD31 for endothelial cells; lysozyme, CD68, and S100 for macrophages; vimentin for fibroblasts; and alkaline phosphatase for committed preosteoblasts (Figure 1).
To confirm the supportive role of bone marrow stromal cells in hemopoiesis in vitro, colony assay of clonogenic progenitors in standard short-term culture was performed. The number of different colonies was evaluated 14 days after initiation of the culture. The number of colony-forming units granulocyte/macrophage remained constant until week 6 and than declined by one third of the maximum level. Burst forming unit erythrocyte declined throughout the investigation period, as expected. The number of mixed colonies was low but within the normal range, and they were present until week 6 (Figure 2).
Bone Marrow Stromal Cells Produce BMPs
To detect mRNA for different BMP family members, we performed a semiquantitative RT-PCR analysis using specific primers for BMP-2, -3, -4, -5, -6, and -7, as well as their specific receptors, activin-like kinases (ALK-2, -3, and -6) and transducer molecules SMAD-1, -4, and -5. Samples were analyzed after 1, 3, 5, and 8 weeks in culture. The results revealed the presence of mRNA for BMP-3, BMP-4, and BMP-7. The message for BMP-3 (Figure 3) was expressed throughout the investigation period as well as in freshly isolated bone marrow (BM) but not in the lymphocytes from the peripheral blood. IHC of the bone marrow stromal cells was performed after 5 weeks in culture using a specific BMP-3 antibody raised in rabbits against the specific peptide from the N-terminal part of mature BMP-3. Figure 4 shows the presence of BMP-3 protein in cytoplasm of bone marrow stromal cells. The message for BMP-4 (Figure 3) appeared at week 3 and retained the same expression level until week 8. Freshly isolated bone marrow also expressed BMP-4 message. IHC using a BMP-4-specific antibody showed positive staining of the cell cytoplasm (Figure 4). Strong expression of mRNA for BMP-7 (Figure 3) was present from the first week of culture and remained at the same level until the eighth week. BMP-7 message was also expressed in fresh BM samples but not in peripheral lymphocytes (Ly). Positive staining for BMP-7 protein was shown using a specific BMP-7 antibody raised in rabbits (Figure 4).
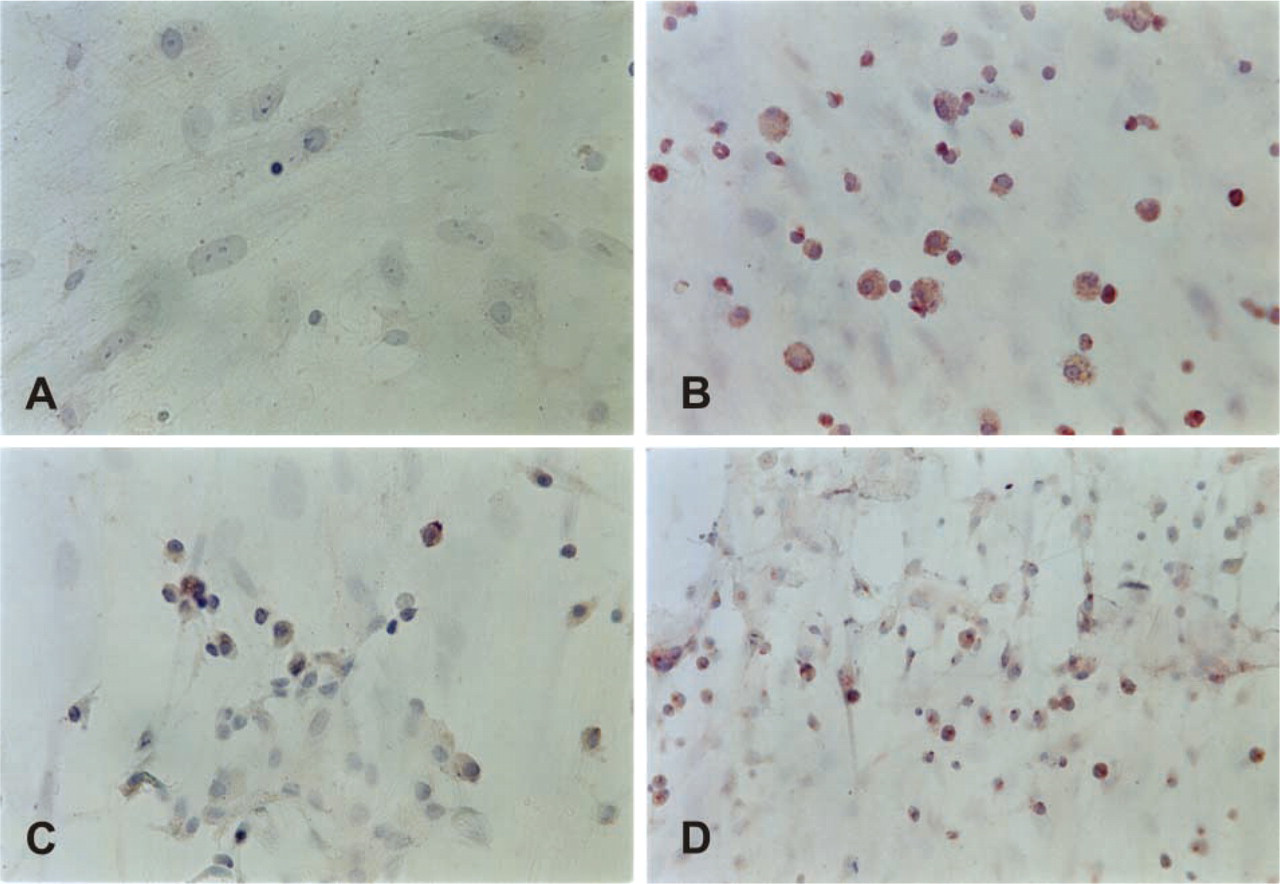
Immunostaining of human bone marrow stromal cells. Cells were cultured for 5 weeks (
Accordingly, we therefore tested the expression of mRNA for BMP-2, BMP-5, and BMP-6, but no detectable expression was found throughout the entire investigation period. When the positive results were compared, mRNA for BMP-7 showed the highest expression.
Expression of BMP Receptors and Signaling Molecules by BM Stromal Cells
Figure 5 shows the RT-PCR results obtained using specific primers for the BMP type I receptor, activin-like kinase-3, which was expressed strongly during the first few weeks of culture in all samples, as well as in bone marrow and peripheral lymphocytes (Figure 5). Similar results were obtained for human ALK-6 primers, except for peripheral lymphocytes. In contrast, no expression of ALK-2 transcripts in human bone marrow stromal cells was found (Figure 5).
Among signaling molecules associated with the BMP pathway, SMAD-1 was present in all samples except freshly isolated bone marrow and lymphocytes (Figure 6). Message for a common mediator for TGF-β super-family members, SMAD-4, was expressed in all samples explored except for peripheral lymphocytes, as well as the message for SMAD-5, which increased towards week 8 (Figure 6).
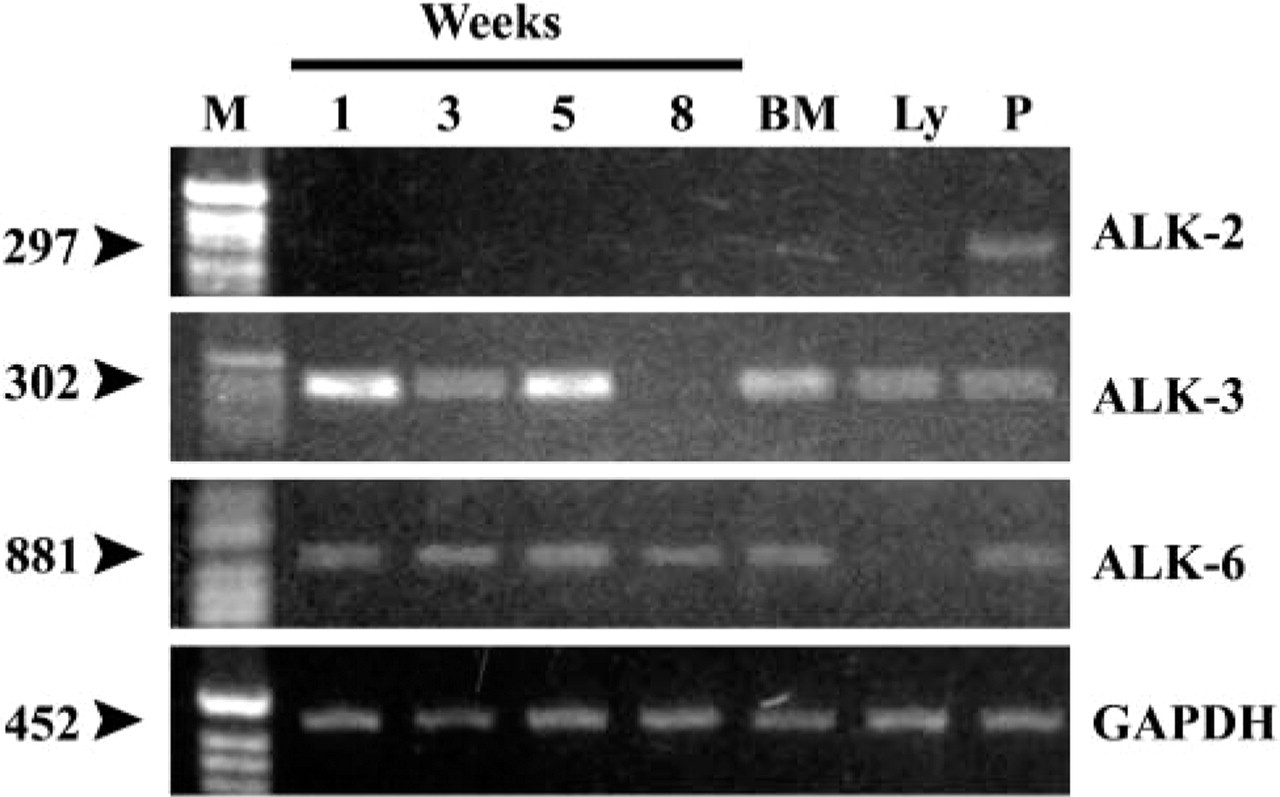
Expression of type I receptors (activin-like kinases 2, 3, and 6) in stromal cells from human bone marrow long-term culture. Cells were cultured and gene expression analyses were done as described in Materials and Methods. RNA was isolated from freshly isolated bone marrow (BM), peripheral lymphocytes (Ly), or cultures at designated time points (1, 3, 5, 8 weeks), cDNA was synthesized and semiquantitative RT-PCR performed as described.
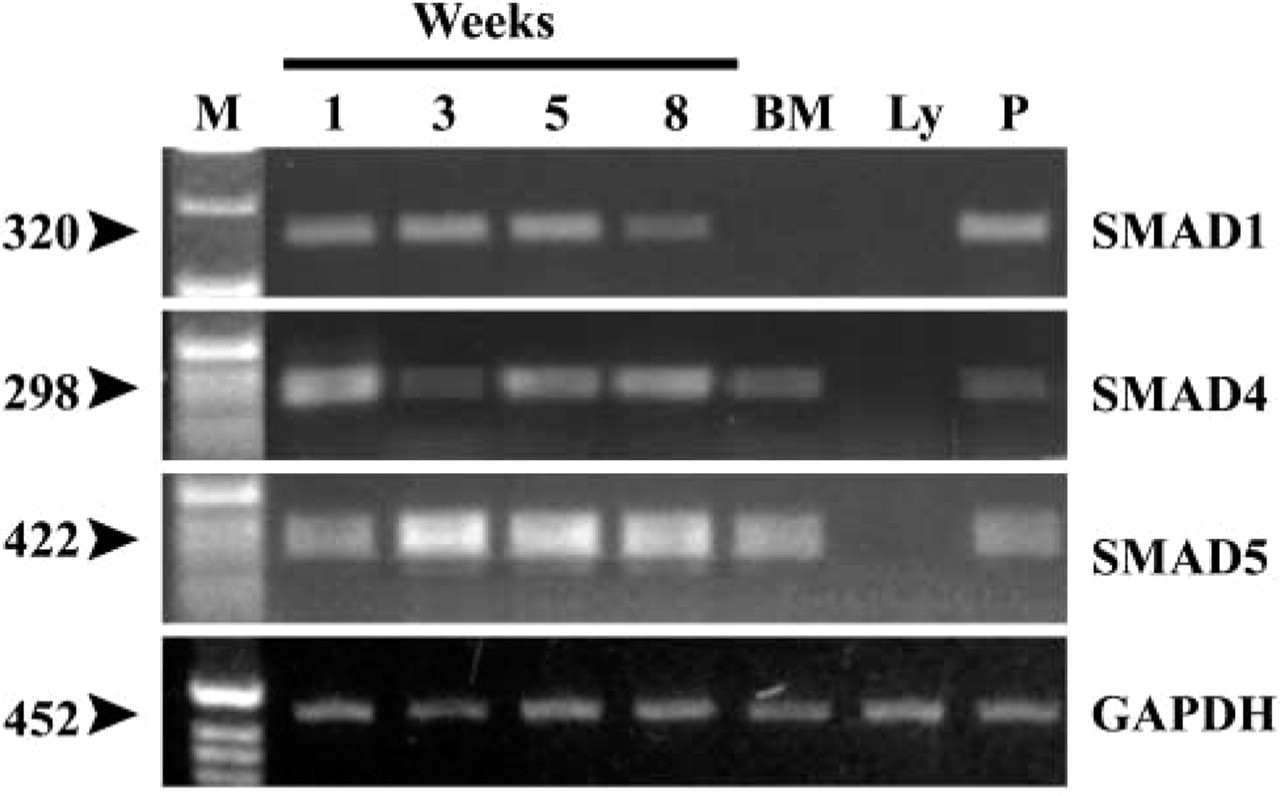
Expression of SMAD transducers in stromal cells from human bone marrow long-term culture. Cells were cultured and gene expression analyses were done as described in Materials and Methods. RNA was isolated from freshly isolated bone marrow (BM), peripheral lymphocytes (Ly), or cultures at designated time points (1, 3, 5, 8 weeks). cDNA was synthesized and semiquantitative RT-PCR performed as described.
Discussion
This study demonstrates that human bone marrow stromal cells synthesize and produce different BMP molecules, which might exert their effects on hemopoietic stem and/or progenitor cells in a paracrine manner via specific BMP receptors. Expression of mRNA for BMP-3, BMP-4, and BMP-7 was detected in bone marrow stromal cells from the first to the eighth week of culture, and the BMP protein was confirmed using specific BMP antibodies. Previous studies have shown that mRNA levels of BMP-2 increased with time in human bone marrow stromal cells cultured in medium inducing osteogenic differentiation (Frank et al. 2002). Human marrow-derived mesenchymal stem cells transduced with a retroviral vector carrying the simian virus 40 large T-antigen expressed BMP-4 mRNA (Nishioka et al. 2003). Unlike Bi et al. (1999), who found the expression of BMP-2 in rat bone marrow stromal cells in culture, the expression of BMP-2, BMP-5, and BMP-6 by human stromal cells in this study was not recorded. In addition to different receptor tyrosine kinases already shown to be expressed by these cells (Satomura et al. 1998), we demonstrated that human bone marrow stromal cells express BMP type I receptors, activin-like kinase (ALK)-3, and ALK-6, as well as their downstream transducers SMAD-1, -4, and -5. Besides this paracrine mechanism, an autocrine regulatory mechanism of tissue homeostasis is also proposed.
Differentiation of hemopoietic stem cells into lineages depends on stromal cells and their specific signals. It has been shown that stromal cell layer possesses the ability to support B-lymphopoiesis for up to 9 months (McGinnes et al. 1991). Treatment of isolated stem cell populations with soluble BMPs induced dose-dependent changes in proliferation, clonogenicity, cell surface phenotype, and multilineage repopulation capacity after transplantation into non-obese diabetic/severe combined immunodeficient (NOD/SCID) mice (Bhatia et al. 1999). Treatment of purified cells with BMP-2 or -7 at high concentrations inhibited proliferation and supported the maintenance of the primitive CD34 + CD38 – phenotype. Low concentrations of BMP-4 induced proliferation and differentiation of CD34 + CD38–Lin– cells, while at higher concentrations it extended the time of repopulation capacity in ex vivo culture (Bhatia et al. 1999). BMP-4 activates ectodermal and mesodermal markers in human embryonic stem cells (Schuldiner et al. 2000). Furthermore, BMP-4 was shown to stimulate expression of GATA-2, a DNA-binding protein that regulates hemopoiesis-specific gene transcription in Xenopus laevis (Maeno et al. 1996). Exogenous BMP-6 was found to block the VLA4/VCAM-1 adhesion pathway mediating the adhesion of primitive hemopoietic stem cell to marrow stroma, thus enabling mobilization of CD34+ cells, which may have a potential therapeutic use in patients with myeloid leukemia lacking BMP expression (Ahmed et al. 2001). BMP-6 significantly reduces IL-6 and IL-11 production from the marrow stroma (Ahmed et al. 2001) and stimulates their common receptor gp130, implying its possible role as a therapeutic agent in multiple myeloma. It also promotes chondrogenesis in a subpopulation of small and rapidly self-renewing marrow stromal cells (Sekiya et al. 2001,2002). Exogenously added BMPs may evoke elaboration and release of hemopoietic cells from the bone marrow. However, endogenously produced members are responsible for differentiation of hemopoietic cells and possibly for determination of the mesenchymal cell fate and their commitment to the specific lineage. Recently, it was reported that members of TGF-β family, their receptors, and their second messengers may play a role in the development of certain types of neoplasms (Miyazono et al. 2003). Loss of functional cell surface TGF-βR type I correlates with insensitivity to TGF-β in chronic lymphatic leukemia (DeCoteau et al. 1997). The current use of BMPs in reconstruction of the skeleton provides us with hope that BMPs might have therapeutic indications in patients with acute leukemias and lymphomas.
The results of this study contribute to understanding of the mechanism that enables the bone marrow stromal cell population to support production and maintenance of hemopoietic cells.
