Abstract
Regenerating (REG) gene family belongs to the calcium-dependent lectin gene superfamily and encodes small multifunctional secretory proteins, which might be involved in cell proliferation, differentiation, and carcinogenesis. To clarify REG expression profile in colorectal carcinoma (CRC), the authors examined the expression of REG Iα, Iβ, III, HIP/PAP, and REG IV by immunohistochemistry on tissue microarray. The expression of REG Iα, III, and HIP/PAP was more frequently observed in the CRCs than adjacent non-neoplastic mucosa (p < 0.001), whereas it was the converse for REG Iβ and IV (p < 0.001). The expression of REG Iα, Iβ, III, and HIP/PAP was negatively correlated with the depth of invasion of CRCs (p < 0.05). The REG Iβ and HIP/PAP were less expressed in CRCs with than without venous invasion (p < 0.05). The positive rates of REG Iα and HIP/PAP were significantly higher in CRCs without than with lymph node metastasis (p < 0.05). Mucinous carcinoma more frequently expressed REG IV protein than well- and moderately differentiated ones (p < 0.05). There was a positive relationship between REG Iα, Iβ, III, and HIP/PAP expression (p < 0.05). Survival analysis indicated the REG Iβ or HIP/PAP expression was positively linked to favorable prognosis of carcinoma patients (p < 0.05). This study indicated that aberrant REG expression might be closely linked to the pathogenesis, invasion, or lymph node metastasis of CRCs. REG Iβ and HIP/PAP could be considered reliable markers of favorable prognosis of CRC patients.
Colorectal carcinoma (CRC) is one of the most common cancers in the world, accounting for nearly 10% of all new cases of cancer. Japan has experienced a marked increase in the incidence of colorectal cancer and has recently been listed in the group of countries with the world’s highest incidence rates (Yoshida et al. 2007; American Cancer Society 2005). Malignant transformation of colorectal epithelial cells is a complex process featuring frequent genetic and epigenetic alterations that lead to the activation of oncogenes and inactivation of tumor suppressor genes (Kodama et al. 2000; Zheng et al. 2010a). Knowledge regarding altered expression of these genes during carcinogenesis may not only provide information about the molecular events during the initiation and progression of cancer but may also result in the discovery of biological markers for the evaluation of cancer diagnosis and prognosis.
Yonemura et al. (1984) reported that Reg protein is induced during regeneration of pancreatic islets. Subsequently, many Reg-related proteins have been identified in human and other animals. The Regenerating (Reg) gene family belongs to the calcium-dependent lectin (C-type lectin) gene superfamily, which encodes a group of small multifunctional secretory proteins. Reg family proteins function as acute phase reactants, lectins, antiapoptotic factors, and growth agents and include growth factors for pancreatic β cells, neural cells, and epithelial cells in the digestive system. These proteins are primarily involved in cell proliferation and differentiation, inflammation, diabetes, and carcinogenesis (Zhang et al. 2003). To date, three subtypes of the REG gene have been identified in humans, which include REG I (Iα and Iβ), Reg III (III and HIP/PAP: Hepatocellular carcinoma–Intestine–Pancreas gene/Pancreatitis-Associated Protein), and Reg IV. The five human REG genes (REG Iα, REG Iβ, HIP/PAP, REG III, and REG IV) have a common gene structure that includes six exons and five introns and encode homologous secretory proteins with 158-175 amino acids. The REG genes are located at adjacent sites on chromosome 2p12 with the exception of REG IV (1q12-q21) and are found in the following order from centromere to telomere: 5′-HIP/PAP 3′, 3′ REG Iα 5′, 5′ REG Iβ 3′, 3′ REG III 5′. The cDNAs of REG family genes exhibit significant homology, with 93% homology between REG III and HIP/PAP, 66% between REG III and Iα, 64% between REG III and Iβ, and 38% between REG III and IV. Furthermore, a comparison of amino acid sequences encoded by REG family genes also shows significant homology, with 85% homology between REG III and HIP/PAP, 48% between REG III and Iα, 47% between REG III and Iβ, and 29% between REG III and IV. Previous studies have shown that the members of the human REG gene family share several common features, including sequence homology, tissue expression profiles, and exon-intron junction genomic organization (Nata et al. 2004). However, distinct differences in the functions of these proteins may exist despite their sequence similarity, which has led to further investigation of the roles of REG IV. REG IV has been reported to be a potent activator of the epidermal growth factor receptor (EGFR)/Akt/Activator Protein−1 (AP-1) signaling pathway in colon cancer cells and increases the expression of BCL-2, Bcl-xl, and survivin proteins (Bishnupuri, Luo, Murmu, et al. 2006). REG IV treatment protects normal intestinal crypt cells from irradiation-induced apoptosis by increasing expression of BCL-2, Bcl-xL, and survivin, and REG IV overexpression has been associated with resistance to irradiation-induced apoptosis in CRC cells (Bishnupuri et al. 2010).
A long history of observations has shown that the expression of REG family members, including REG I Iα, Iβ, HIP/PAP, and REG IV, is upregulated at both mRNA and protein levels in cancer, indicating a possible role of these proteins in neoplastic transformation (Rechreche et al. 1999; Violette et al. 2003). Overexpression and high serum levels of REG IV have also been associated with liver metastasis in CRCs (Oue et al. 2007). In the present study, we prepared novel monoclonal antibodies against REG Iα, Iβ, III, HIP/PAP, and REG IV and examined their expression profiles in a large number of CRC samples with a comparison of their expression with the clinicopathological parameters of the carcinomas.
Materials and Methods
Antibody Preparation
The REG peptides were designed by Gentyx according to the principles of low homology, high hydrophilicity, and existence on the surface of protein, as shown in Figure 1a. The synthetic REG peptides were bound to carrier protein and subsequently employed to immunize the BALB/c mice with adjuvant. The single cells isolated from spleen were hybridized with the P3-X63Ag8U1 myeloma cell. The mouse hybridomas were screened by enzyme-linked immunosorbent assay and intraperitoneally injected into BALB/c mice to produce monoclonal antibody. The research protocol was approved by the Ethical Committee of Tohoku University.
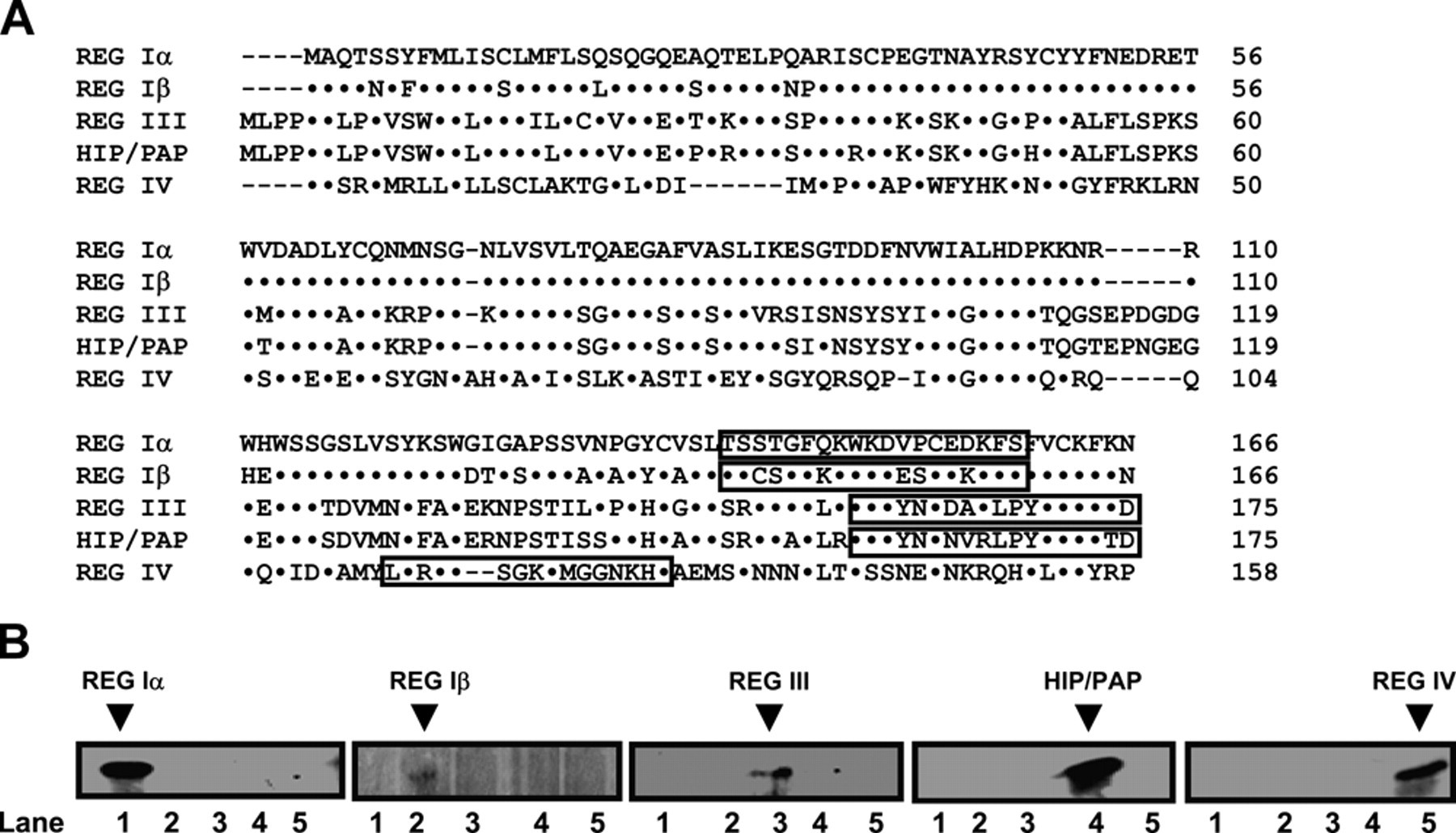
Peptide design and confirmation of the specificity of monoclonal REG antibodies. (a) The REG peptides indicated in the red box were synthesized and used for antibody generation, as these regions were evaluated to be remarkably discriminated by software analysis. (b) The different recombinant REG proteins were loaded and immunoblotted, respectively, using specific monoclonal antibodies screened from mouse hybridomas. Lane 1: REG Iα (19 kDa); lane 2: Reg Iβ (19 kDa); lane 3: Reg III (16 kDa); lane 4: HIP/PAP (16 kDa); lane 5: Reg IV (18 kDa).
Cell Lines and Culture
Colorectal carcinoma cell lines were a kind gift from Prof. Sugiyama, Department of Gastroenterology, Graduate School of Medical and Pharmaceutical Sciences, University of Toyama. The cell lines were maintained in RPMI 1640 (HCT-15, DLD-1, and SW480) and DMEM (WiDr) media supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 µg/mL streptomycin in a humidified atmosphere containing 5% CO2 at 37°C. Cells were harvested by centrifugation, rinsed with phosphate-buffered saline (PBS, pH 7.2), and subjected to total protein extraction in radioimmunoprecipitation assay (RIPA) lysis buffer (50 mmol/L Tris-HCl [pH 7.5], 150 mmol/L NaCl, 5 mmol/L EDTA, 0.5% Nonidet P-40, 5 mmol/L dithiothreitol, 10 mmol/L NaF, and a protease inhibitor cocktail [Nacalai, Tokyo, Japan]).
Subjects
Colorectal carcinoma (n=333) and adjacent non-neoplastic mucosa (NNM, n=331) samples were collected from surgical resection in the Kouseiren Takaoka Hospital (Takaoka, Japan) between 1993 and 2002. The patients with CRC were made up of 184 men and 146 women (18-90 years; mean age = 68.5 years). Among these patients, 129 carcinomas were accompanied by lymph node metastases. None of the patients underwent chemotherapy, radiotherapy, or adjuvant treatment prior to surgery. The patients or their relatives provided consent for the use of tumor tissue for clinical research, and the research protocol was approved by the Ethical Committee of China Medical University. Patients were followed either by consulting case documents or by telephone.
Pathology
All tissues were fixed in 10% neutral formalin, embedded in paraffin, and cut into 4-µm sections. The sections were stained with hematoxylin and eosin (HE) for analysis of histological characteristics. The staging of each colorectal carcinoma was performed according to the Union Internationale Contre le Cancer (UICC) system to indicate the extent of tumor spread (Sobin and Wittekind 2002). The histological architecture of the CRCs was expressed in terms of World Health Organization (WHO) classification (Hamilton and Aaltonen 2000). Tumor size and depth of invasion were also determined. Lymphatic and venous invasion was diagnosed first by HE staining and second by D2-40 immunostaining and EvG staining, respectively, if necessary.
Tissue Microarray
Representative areas of solid tumors and adjacent normal mucosa were identified in HE-stained sections from the selected tumor cases. From these areas, a 4-mm-diameter tissue core was punched out of each donor block and transferred to a recipient block with a maximum of 24 cores using the Tissue Microarrayer (AZUMAYA KIN-1, Tokyo, Japan). Sections with a thickness of 4 µm were consecutively incised from the recipient block and transferred to polylysine-coated glass slides. HE staining was performed on the tissue microarray (TMA) to confirm the presence of tumor or normal tissues.
Western Blot
Denatured proteins or recombinant REG antigens were separated on a 15% sodium dodecyl sulfate (SDS)–polyacrylamide gel and transferred to a Hybond membrane (Amersham, Munich, Germany). The membrane was then blocked overnight in 5% skim milk in Tris-buffered saline containing Tween 20 (TBST; 10 mmol/L Tris-HCl, 150 mmol/L NaCl, 0.1% Tween 20). For immunoblotting, the membrane was incubated for 15 min with mouse anti-human REG Iα, Iβ, III, HIP/PAP, and REG IV antibodies (1:500). Next, the membrane was rinsed with TBST, followed by incubation with anti-mouse IgG conjugated to horseradish peroxidase (DAKO, Carpinteria, CA; 1:1000) for 15 min. All of the incubations were performed in a microwave oven to allow intermittent irradiation, as recommended by Li et al (2002). Bands were visualized using ECL-Plus detection reagents (Santa Cruz Biotechnology, Santa Cruz, CA), followed by exposure to X-ray film (Fujifilm, Tokyo, Japan) or imaging with LAS4000 (Fujifilm).
Immunohistochemistry
Consecutive sections were dewaxed with xylene, rehydrated with alcohol to water, and subjected to antigen retrieval by irradiation in target retrieval solution (TRS; DAKO) in a microwave oven for 15 min (Oriental Rotor Lmt. Co., Tokyo, Japan). Sections were then blocked in 5% bovine serum albumin for 5 min to prevent nonspecific antibody binding. The sections were incubated with mouse anti-human REG Iα, Iβ, III, HIP/PAP, or REG IV antibodies (1:2000) as described above for 15 min, followed by incubation with anti-mouse secondary antibody conjugated to horseradish peroxidase (DAKO; ready for use) for 15 min. All of the incubations were performed in a microwave oven to allow intermittent irradiation as previously described (Kumada et al. 2004). After each treatment, the slides were washed with TBST (3 × 1 min). Bound antibodies were visualized using 3, 3′-diaminobenzidine (DAB). After counterstaining with Mayer’s hematoxylin, the sections were dehydrated, cleared, and mounted. Normal mouse IgG was used instead of the primary antibody as a negative control. As indicated in Figure 2, all of the markers were localized to the cytoplasm. All evaluations were performed blindly by two independent observers (Takano Y and Zheng HC). The percentages of counted cells were scored as follows: 0% to 5%, negative (−), and 6% to 100%, positive (+). Inconsistent data points were reanalyzed by both observers until agreement was reached.
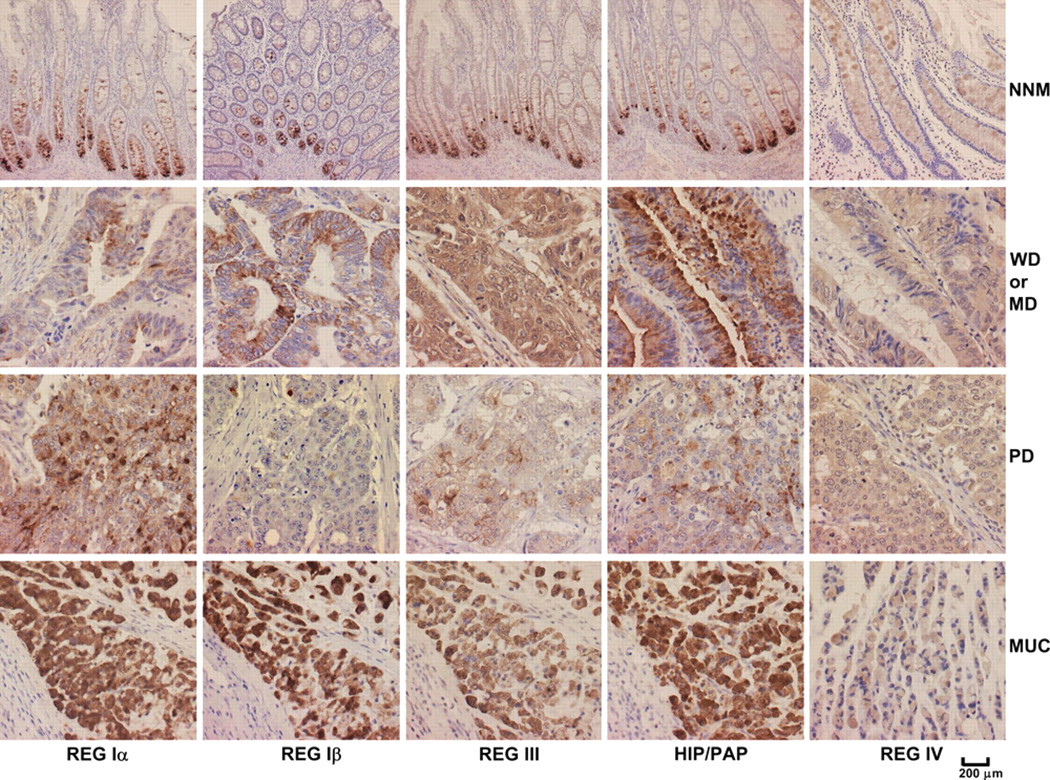
REG protein expression in colorectal samples by immunohistochemistry. Expression of REG Iα, Iβ, III, HIP/PAP, and REG IV was observed in the cytoplasm of positive cells. REG protein expression was analyzed in non-neoplastic mucosa (NNM) samples and in well-differentiated (WD), moderately differentiated (MD), poorly differentiated (PD), or mucinous (MUC) adenocarcinoma of the colorectum.
Statistical Analysis
Statistical evaluation was performed using Fisher’s exact probability test to compare the positive rates. Kaplan-Meier plots of CRC-specific survival were generated, and comparisons between survival curves were made using log-rank statistics. Cox’s hazards proportional regression was employed for multivariate analysis using a continuous model. p < 0.05 was considered statistically significant. SPSS 10.0 software (SPSS, Inc., an IBM Company, Chicago, IL) was employed for data analysis.
Results
REG Expression in CRC and Adjacent NNM Samples
Although antibody specificity was confirmed by Western blot analysis (Figure 1b), we found no specific and nonspecific bands in DLD-1, HCT-15, SW480, and WiDr cancer cells by Western blot using the antibodies against REG Iα, Iβ, III, HIP/PAP, or REG IV proteins (data not shown). As shown in Figure 2, REG Iα, Iβ, III, HIP/PAP, and REG IV were localized to the cytoplasm. The REG Iα, Iβ, III, and HIP/PAP proteins were primarily expressed in the basal crypt epithelium or some proliferating goblet cells and occasionally in mucinous cells of colorectal NNM tissues. In contrast, REG IV was frequently expressed in mucinous and enteroendocrine cells in NNM tissues. Statistically, expression of REG Iα (55.5% vs 21.9%, p < 0.001, Pearson’s r = 0.346), III (85.4% vs 41.1%, p < 0.001, Pearson’s r = 0.457), and HIP/PAP (31.3% vs 16.7%, p < 0.001, Pearson’s r = 0.171) was more frequently observed in carcinoma compared to NNM tissues, whereas the converse was true for REG Iβ (36.7% vs 53.3%, p < 0.001, Pearson’s r = 0.169) and REG IV (57.9% vs 71.5%, p < 0.001, Pearson’s r = 0.143; Figure 3).
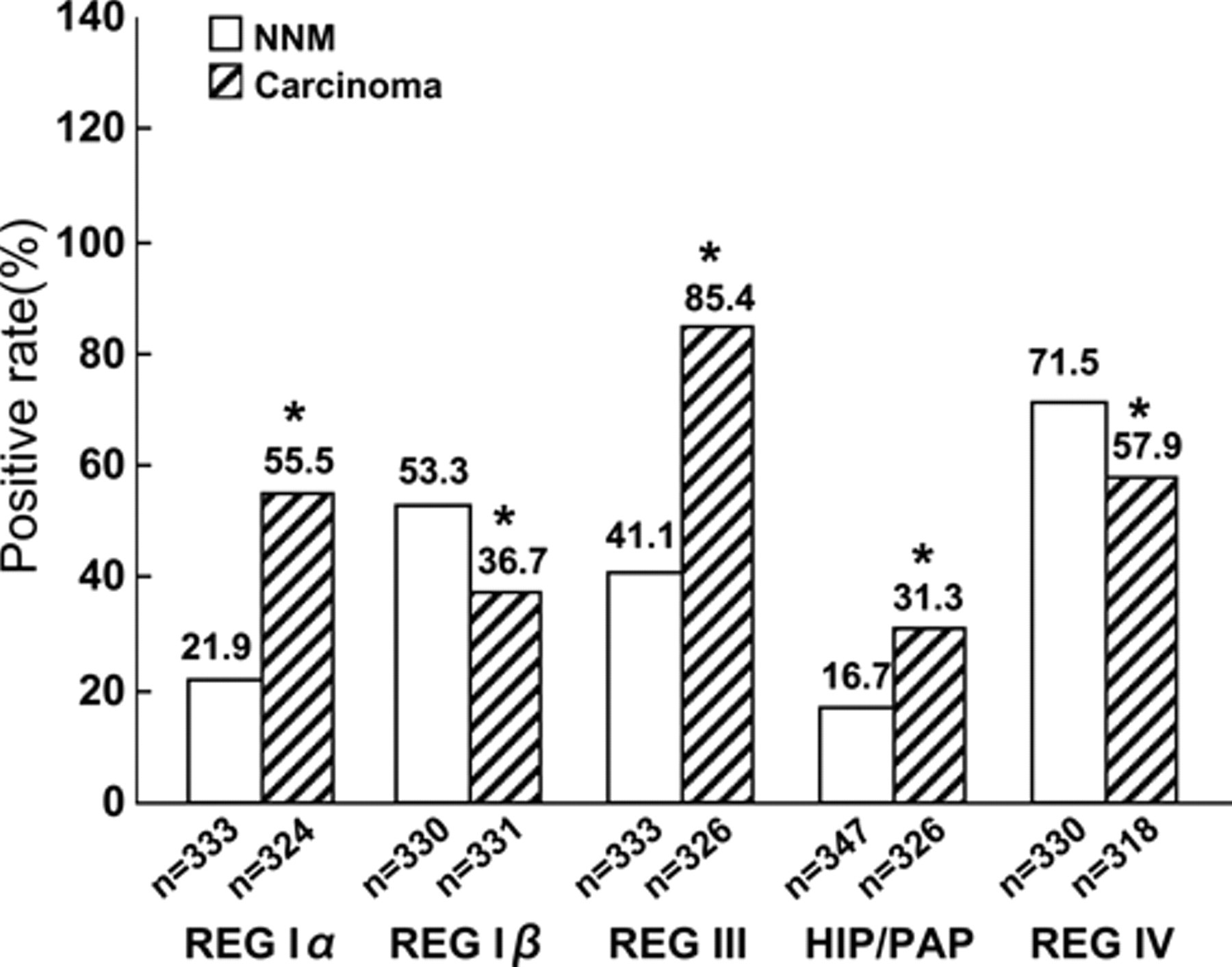
Comparison of positive REG protein expression in colorectal carcinoma and paired non-neoplastic mucosa. *p<0.001 for comparisons with non-neoplastic mucosa (NNM) samples.
Relationship between REG Protein Expression and Clinicopathological or Prognostic Parameters of CRC
As summarized in Table 1, the expression of REG Iα (p < 0.05, Pearson’s r = 0.138), Iβ (p < 0.05, Pearson’s r = 0.163), III (p < 0.05, Pearson’s r = 0.126), and HIP/PAP (p < 0.05, Pearson’s r = 0.181) was negatively correlated with depth of invasion of CRCs. The expression of REG Iβ (p < 0.05, Pearson’s r = 0.122) or HIP/PAP (p < 0.05, Pearson’s r = 0.140) was less frequently expressed in CRCs with venous invasion than in corresponding counterparts. Positive rate of REG Iα (p < 0.05, Pearson’s R = 0.125) or HIP/PAP (p < 0.05, Pearson’s R = 0.121) expression was significantly higher in CRCs lacking l than accompanied with lymph node metastasis. Among WHO histological subtypes, REG IV expression occurred more frequently in mucinous carcinoma than in well-differentiated (p < 0.05, Pearson’s r = 0.174) and moderately differentiated carcinoma (p < 0.05, Pearson’s r = 0.263). A positive relationship was observed between REG Iα and Iβ (p < 0.05, Pearson’s r = 0.232), III (p < 0.05, Pearson’s r = 0.326), or HIP/PAP (p < 0.05, Pearson’s r = 0.492) expression in CRCs. The expression REG Iβ was positively linked to the expression of REG III (p < 0.05, Pearson’s r = 0.122) and HIP/PAP (p < 0.05, Pearson’s r = 0.372) in carcinomas. There was a positive association between REG III and HIP/PAP expression in CRCs (p < 0.05, Pearson’s r = 0.134).
Relationship between REG Expression and Clinicopathological Features of Colorectal Carcinoma
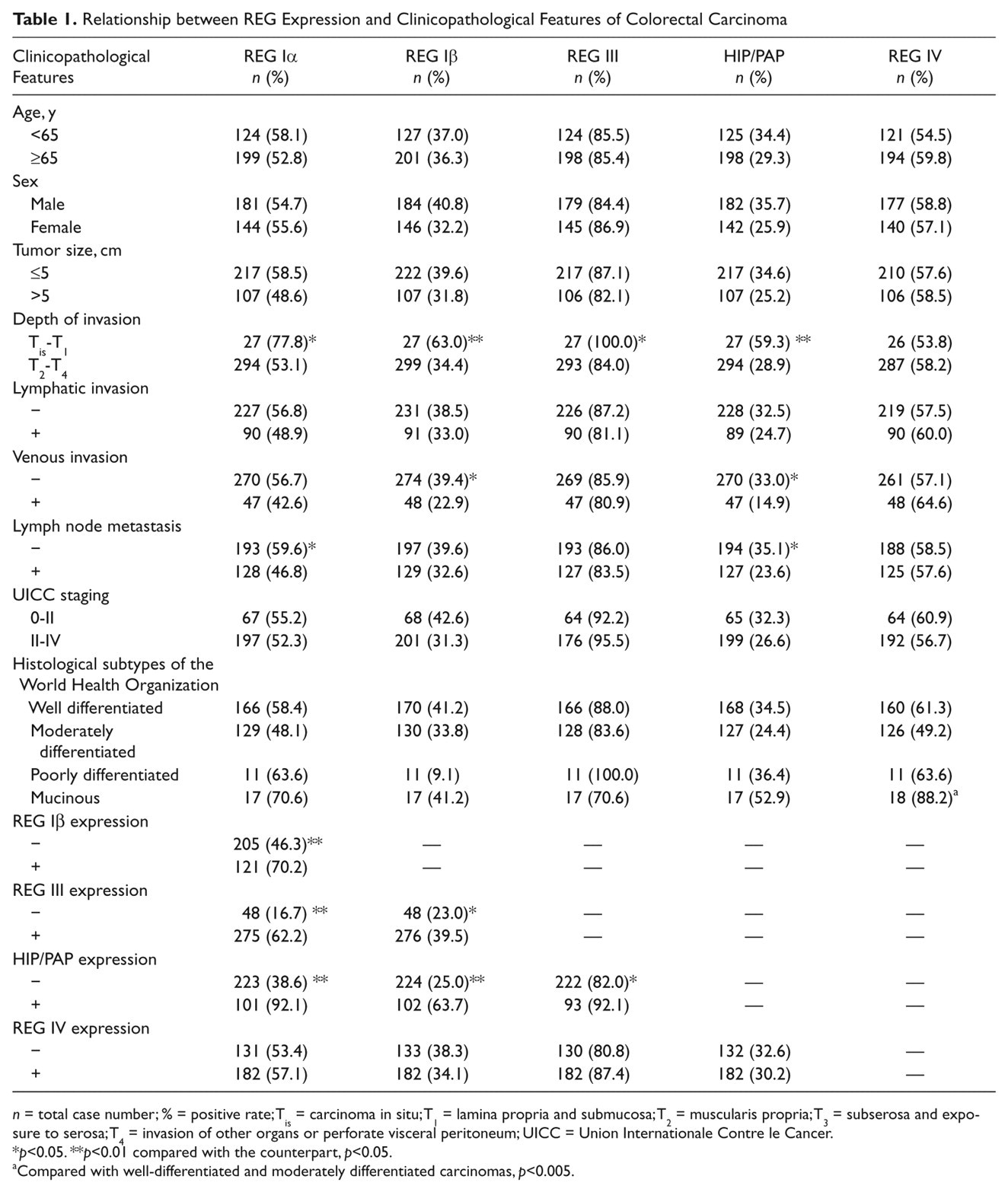
n = total case number; % = positive rate; Tis = carcinoma in situ; T1 = lamina propria and submucosa; T2 = muscularis propria; T3 = subserosa and exposure to serosa; T4 = invasion of other organs or perforate visceral peritoneum; UICC = Union Internationale Contre le Cancer.
p<0.05. **p<0.01 compared with the counterpart, p<0.05.
Compared with well-differentiated and moderately differentiated carcinomas, p<0.005.
Analysis of Univariate and Multivariate Survival of CRC Patients
Follow-up information was available for 279 CRC patients ranging from 0.9 months to 12.1 years (median = 66.2 months). Figure 4 shows the survival curves stratified according to REG expression status. Univariate analysis using the Kaplan-Meier method indicated that the cumulative survival rate of patients with positive REG Iβ or HIP/PAP expression was clearly higher than that without their expression (p < 0.05), which was not true for REG Iα, III, and IV expression (p > 0.05). Multivariate analysis using Cox’s proportional hazard model indicated that lymphatic and venous invasion, lymph node metastasis, and UICC staging (p < 0.05) but not age, sex, tumor size, depth of invasion, or REG expression (p > 0.05) were independent prognostic factors for patients with CRC (Table 2).
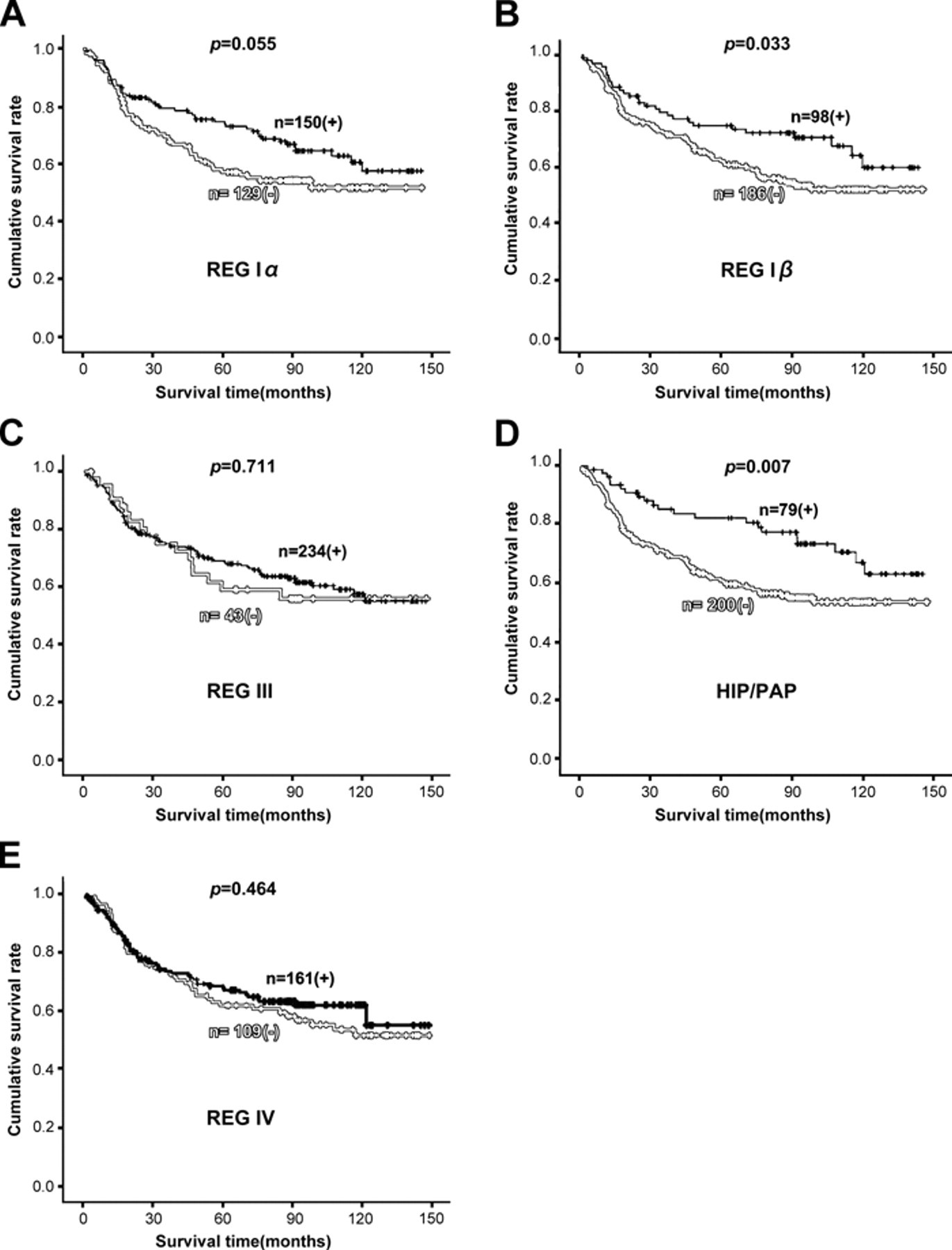
Correlation between the REG protein expression status and the prognosis of patients with colorectal carcinoma (CRC). Kaplan-Meier curves showing the cumulative survival rate of patients with colorectal carcinomas grouped by the REG Iα (a), REG Iβ (b), REG III (c), HIP/PAP (d), and REG IV (e) expression status.
Multivariate Analysis of Clinicopathological Variables for the Survival of Patients with Colorectal Carcinoma
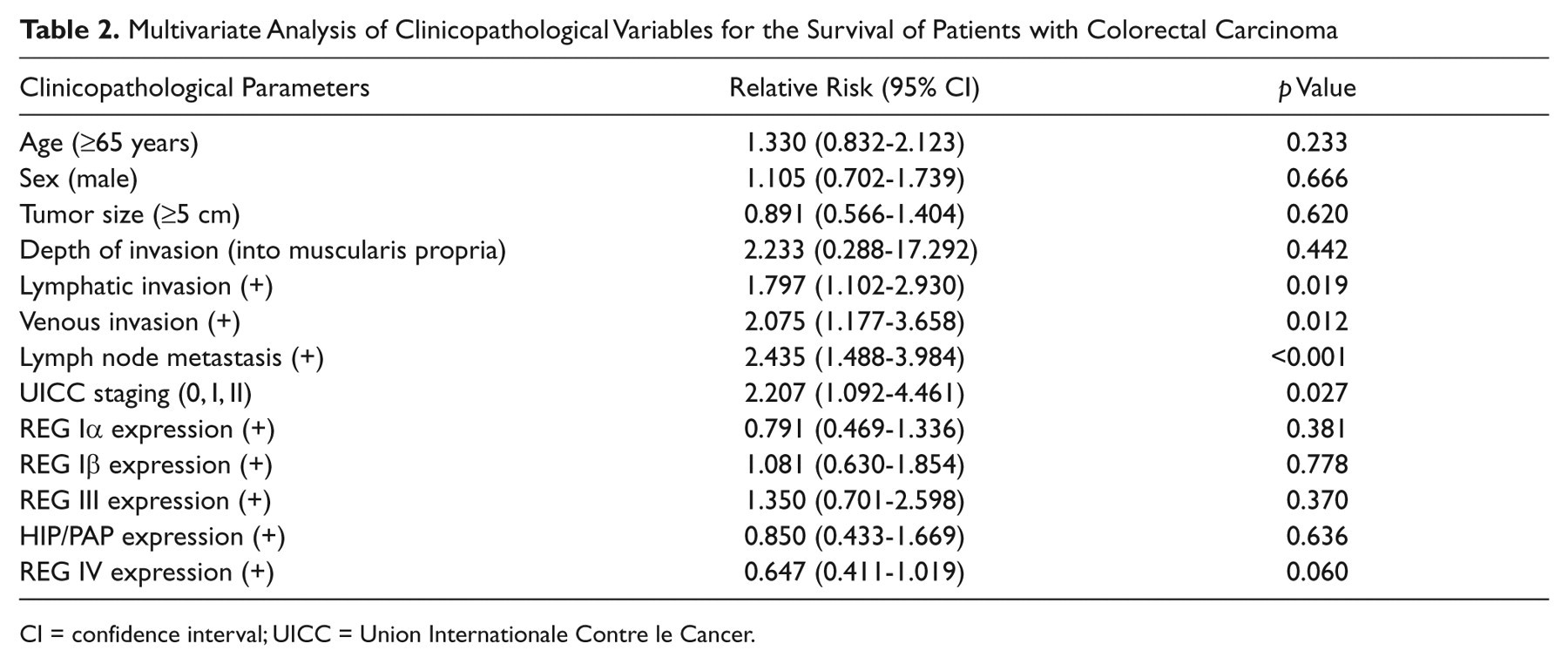
CI = confidence interval; UICC = Union Internationale Contre le Cancer.
Discussion
Despite many attempts to associate the expression of various molecules with the clinicopathological parameters of CRCs, including cell proliferation and regeneration, cell adhesion, resistance to apoptosis, mucin production, angiogenesis, and differentiation, our knowledge of colorectal carcinogenesis and subsequent progression remains limited. In the present study, for the first time we prepared monoclonal antibodies against each member of the human REG family to analyze their expression profiles and correlated them with tumorigenesis, aggressive behaviors, and prognosis of CRCs. To confirm their specificity and sensitivity, we performed Western blot and found that no cross-reaction occurred in these human REG proteins. No signals were detected in carcinoma cell lines, suggesting the antibodies might not react with other proteins. However, we have not detected colorectal carcinoma samples to confirm the conclusion, which is a limitation of our work and will be solved using more cell lines and tissue samples in the future.
Herein, we found that the REG Iα, Iβ, III, and HIP/PAP proteins were primarily expressed in the basal crypt epithelium, some proliferating goblet cells, and occasionally in mucinous cells of colorectal NNM samples. In inflammatory bowel disease, intact REG Iα protein was expressed by a metaplastic Paneth granular cell population (Dieckgraefe et al. 2002). Furthermore, the REG Iα treatment was shown to reduce apoptosis of colorectal epithelial cells exposed to hydrogen peroxide (Dieckgraefe et al. 2002). These findings suggest that REG proteins may play critical roles in cell proliferation and resistance to apoptosis in colorectal epithelial cells (Dieckgraefe et al. 2002; Bishnupuri, Luo, Korzenik, et al. 2006). In this study, we also found that REG IV was frequently expressed in the mucinous cells of NNM samples, consistent with previous reports showing its preferential expression in mucin-rich carcinomas, such as mucinous or SRC carcinoma (Zheng et al. 2010b; Sentani et al. 2008). Furthermore, overexpression of REG Iα, III, and HIP/PAP and reduced expression of REG Iβ and IV were observed in our CRC cases. These observations suggest that either increased or decreased expression of these proteins may be involved in the malignant transformation of colorectal epithelial cells. Consistent with this observation, HIP/PAP expression has been shown to be elevated in liver tumors at the transcriptional level and was not detected in adjacent non- neoplastic areas or in normal liver (Lasserre et al. 1999; Cavard et al. 2006). Macadam et al. (2000) demonstrated that a higher percentage of tumors expressed REG I and PAP mRNA compared to paired normal mucosa in CRCs. Rechreche et al. (1999) reported that REG Iα, Iβ, and HIP/PAP mRNAs were commonly upregulated in CRCs. Furthermore, they also showed REG Iα and HIP/PAP overexpression at the protein level, in agreement with our findings. Another group found that REG IV mRNA was more highly expressed in most CRCs compared to normal colon tissue (Violette et al. 2003), whereas stronger immunoreactivity against REG IV protein in colorectal NNM compared to CRC samples has been reported (Cavard et al. 2006). Expression of some REG proteins may be repressed under normal conditions, but autocrine and/or paracrine factors, such as interleukin (IL)−1, IL-6, or tumor necrosis factor α (TNF-α), may stimulate their expression in cancer cells (Rechreche et al. 1999). In contrast to the upregulation of its mRNA, the downregulation of REG Iβ protein has been observed, potentially due to the regulation at the translational level or to the differences in detecting approaches.
To analyze the clinicopathological significance of REG proteins’ expression, we examined the in situ expression of the proteins and compared their expression with clinicopathological parameters of CRC. We found a negative correlation between REG Iα expression and depth of invasion or lymph node metastasis of CRCs. REG Iβ was less frequently expressed in CRCs without venous invasion. HIP/PAP expression was inversely linked with depth of invasion, venous invasion, and lymph node metastasis of CRCs. To get better details of the percentage distribution of immunohistochemistry, we performed Spearman rank correlation analysis using the four-tie system (0%-5%, −; 6%-25%, +; 26%-50%, ++; 50%-100%, +++) and found the same results as described in the Results section. Similar results for REG Iα, Iβ, III, and HIP/PAP expression patterns and their negative associations with invasion or lymph node metastasis could be attributed to their similar gene structures and potentially similar functions (Zhang et al. 2003; Nata et al. 2004). Similarities in function may underlie the positive relationship between REG Iα, Iβ, III, and HIP/PAP expression in CRCs. However, no relationship between REG IV expression and the aggressiveness-related parameters of tumors was identified, consistent with our previous study (Zheng et al. 2010b). These findings indicate that some members of the REG protein family, excluding REG IV, might be considered effective markers of less invasive CRC behavior and could explain the favorable prognosis of patients with REG Iβ or HIP/PAP expression. Furthermore, multivariate analysis demonstrated that lymphatic and venous invasion, lymph node metastasis, and UICC staging, but not REG expression, were independent factors in CRCs, indicating that REG Iβ or HIP/PAP expression may serve as a prognostic indicator for patients with CRC, albeit not independent. However, Ohara et al. (2008) reported that REG IV is significantly associated with longer relapse-free survival of clinically localized patients with prostate cancer as an independent prognostic factor. In contrast, no significant relationship was found between expression of the REG Iα, Iβ, and PAP proteins and the clinical outcome of patients, Duke’s state of tumors, or tumor localization, size, or differentiation degree in a study from Rechreche et al. (1999). These paradoxical observations may be attributable to differences in detecting approaches and sample cases.
Several lines of evidence indicate that REG IV expression is associated with the intestinal mucin phenotype in human carcinomas (Zheng et al. 2010b; Oue et al. 2005). In this investigation, REG IV was more frequently expressed in mucinous carcinoma in comparison to well-differentiated and moderately differentiated carcinomas, suggesting that it may be specific for mucinous carcinoma, consistent with previous findings (Oue et al. 2005; Sentani et al. 2008; Yamagishi et al. 2009; Zheng et al. 2010b). In particular, a positive correlation between REG IV and MUC2 or MUC5AC expression was observed by both statistical analysis and double immunostaining (Sentani et al. 2008; Yamagishi et al. 2009). The expression pattern of REG IV in CRCs is in agreement with the similar distribution pattern of MU5AC, MUC2, and REG IV in the neoplastic goblet cells of appendiceal mucinous cystadenoma and pseudomyxoma peritonei (Heiskala et al. 2006). REG IV overexpression in colorectal mucinous carcinoma further supports a significant association with mucin production, suggesting that the protein might be regarded as good biomarkers for colorectal mucinous carcinomas.
In the present study, a large number of colorectal carcinoma cases were screened by TMA, which takes advantage of high-throughput, identical immunohistochemical conditions; economy of samples; antibodies; and time (Zheng et al. 2006; Zheng et al. 2008). Although 4-mm-diameter needles were used for samples and representative regions were carefully selected with the reference of HE slides, avoidance of selection bias proved difficult. Therefore, one limitation of the present study is the difficulty in separating the edge and center of colorectal carcinomas when establishing TMA.
In summary, aberrant REG expression might be linked to pathogenesis, invasion, or lymph node metastasis of CRC. HIP/PAP and REG IV expression could be considered a marker of colorectal mucinous adenocarcinoma. The functions of these REG proteins in colorectal carcinogenesis and progression should therefore be analyzed in further investigations.
Footnotes
Acknowledgements
We thank Toshiro Sugiyama for his gift of colorectal carcinoma cell lines.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This study was supported by Shenyang Outstanding Talent Foundation of China; Shenyang Science and Technology Grant (1091175-1-00); Liaoning BaiQianWan Talents Program; Scientific and Technological Projects for Overseas Returned Persons, Ministry of Personnel; Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry; Natural Scientific Foundation of China (No. 81001093); grant-in-aid for Scientific Research from the Ministry of Education, Culture, Sports and Technology of Japan (20659109; 21790624); Japanese Smoking Research Foundation; Japanese Uehara Memorial Foundation; and Research Grants for JSPS Postdoctoral Fellows.
