Abstract
Dental pulp elaborates both bone and dentin under pathological conditions such as tooth replantation/transplantation. This study aims to clarify the capability of dental pulp to elaborate bone tissue in addition to dentin by allogenic tooth transplantation using immunohistochemistry and histochemistry. After extraction of the molars of 3-week-old mice, the roots and pulp floor were resected and immediately allografted into the sublingual region in a littermate. In addition, we studied the contribution of donor and host cells to the regenerated pulp tissue using a combination of allogenic tooth transplantation and lacZ transgenic ROSA26 mice. On Days 5-7, tubular dentin formation started next to the preexisting dentin at the pulp horn where nestin-positive odontoblast-like cells were arranged. Until Day 14, bone-like tissue formation occurred in the pulp chamber, where intense tartrate-resistant acid phosphatase-positive cells appeared. Furthermore, allogenic transplantation using ROSA26 mice clearly showed that both donor and host cells differentiated into osteoblast-like cells with the assistance of osteoclast-lineage cells, whereas newly differentiated odontoblasts were exclusively derived from donor cells. These results suggest that the odontoblast and osteoblast lineage cells reside in the dental pulp and that both donor and host cells contribute to bone-like tissue formation in the regenerated pulp tissue.
Keywords
A
Tooth replantation induces at least two types of healing patterns in the regenerated dental pulp: tertiary dentin and bone tissue formation in the regenerated pulp tissue (Kvinnsland et al. 1991; Byers et al. 1992; Rungvechvuttivittaya et al. 1998; Shimizu et al. 2000; Ohshima et al. 2001; Tsukamoto-Tanaka et al. 2006; Hasegawa et al. 2007). Two possibilities are proposed regarding the derivation of bone-forming cells in the replanted pulp: cells migrating from the periodontal tissue and/or resident pulpal mesenchymal cells (Shimizu et al. 2000). To exclude the possibility that the periodontal tissue contributes to this event, we established the animal model of autogenic tooth transplantation of the coronal portion into the sublingual region. In this experiment, both tubular dentin and bone tissue depositions were always induced in the pulp chamber despite the absence of periodontal tissue, suggesting that dental pulp contains two types of competent progenitor cells capable of differentiating into either odontoblast- or osteoblast-like cells (Ogawa et al. 2006). The determination of the healing pattern after tooth replantation may be directly linked to the death or survival of odontoblast lineage cells such as fully differentiated odontoblasts and/or odontoblastic progenitor cells. The proper oxygenated medium is probably decisive for the survival of odontoblast lineage cells. Intentionally prolonged time operating for tooth replantation induces the total death of odontoblast lineage cells before osteoblast lineage cells (Hasegawa et al. 2007). Certain cell populations in the pulp tissue might have the ability to differentiate into bone-forming cells, and their differentiation might take place under pathological conditions in which certain regulatory mechanisms for suppressing bone formation are disturbed. The existence of both odontoblast and osteoblast lineage cells may be supported by the notion that dental pulp is composed of various cell populations, including resident mesoderm-derived and cranial neural crest (CNC)-derived cells (Goldberg and Smith 2004).
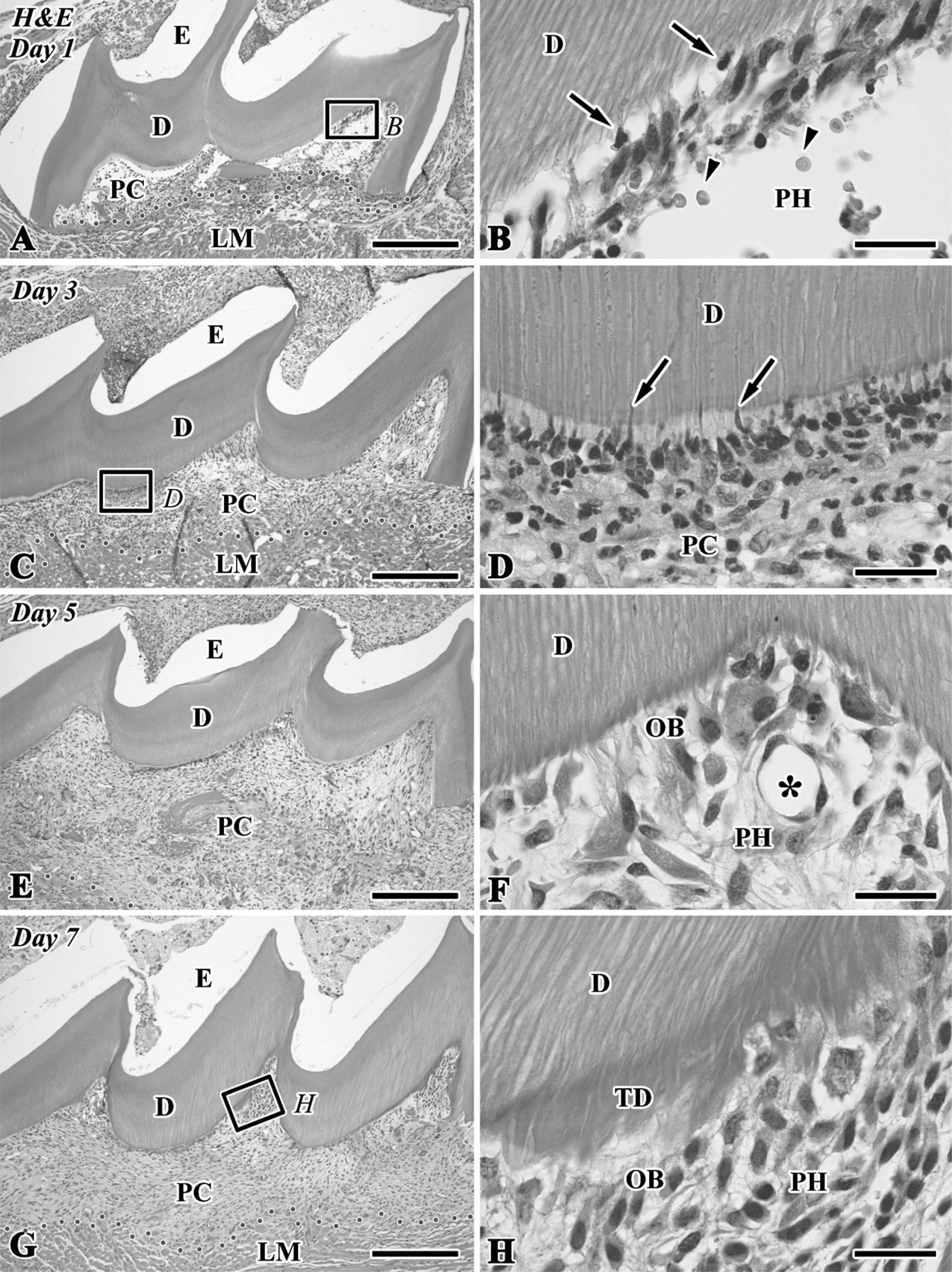
Hematoxylin and eosin (H&E)-stained sections of the transplanted teeth at 1 (
Previous studies have been unable to exclude the possibility of the host tissue contributing to bone tissue formation in the transplants (Zussman 1966; Luostarinen and Ronning 1977; Yamamura 1985; Takei et al. 1988; Inoue and Shimono 1992; Laino et al. 2005). Isolated pulp tissue implanted in a variety of sites gives rise to an osteo-typical matrix but not to tubular dentin. In our previous study using autogenic tooth transplantation, the presence of surviving pulp tissue was suggested to be necessary for the induction of bone-like tissue formation in transplants with the assistance of osteoclast lineage cells (Ogawa et al. 2006). This notion is supported by the findings that the allograft of teeth without pulp fails to induce hard tissue formation in the transplants (Ogawa et al. 2006). However, we still cannot exclude the possibility that the progenitor cells from the surrounding lingual tissue migrate into the pulp chamber and proliferate to give rise to hard tissue-forming cells. Actually, numerous tartrate-resistant acid phosphatase (TRAP)-positive cells appear from the circulatory system of the host tissue in both the auto- and allograft transplants. Allograft transplantation experiments using GFP or ROSA26 reporter mice are necessary to exclude this possibility. This study aims to clarify the capability of dental pulp to elaborate bone tissue in addition to tubular dentin by allogenic tooth transplantation of the coronal portion into the sublingual region using IHC for 5-bromo-2′-deoxyuridine (BrdU) as a cell proliferation assay, nestin as an odonto-blastic marker (Terling et al. 1995; About et al. 2000b; Ogawa et al. 2006; Hasegawa et al. 2007), osteopontin (OPN) as a marker for osteoblast lineage cells (Haylock and Nilsson 2006), and histochemistry for TRAP as a marker for osteoclast lineage cells (Bonucci and Nanci 2001). Furthermore, we studied the contribution of donor and host cells to the regenerated pulp tissue using a combination of allogenic tooth transplantation and lacZ transgenic ROSA26 mice.
Materials and Methods
Allogenic Tooth Transplantation Into the Sublingual Region
All experiments were reviewed by the Committee on the Guidelines for Animal Experimentation of Niigata University and performed according to the recommendations or under the conditions proposed by the Review Committee. Crlj:CD1 (ICR) mice, 3 weeks old, were used in this study. The upper right first molar was extracted with a pair of dental forceps with modification under anesthesia by an intraperitoneal injection of chloral hydrate (350 mg/kg), and the roots and pulp floor were resected with a surgical knife. The coronal portion of the resected samples without the periodontal tissue was immediately transplanted into the sublingual region after cutting the ventral side of the tongue of the littermates, and the section was sutured with a nylon suture. Furthermore, we performed allogenic tooth transplantation between lacZ transgenic and wild-type ROSA26 mice.
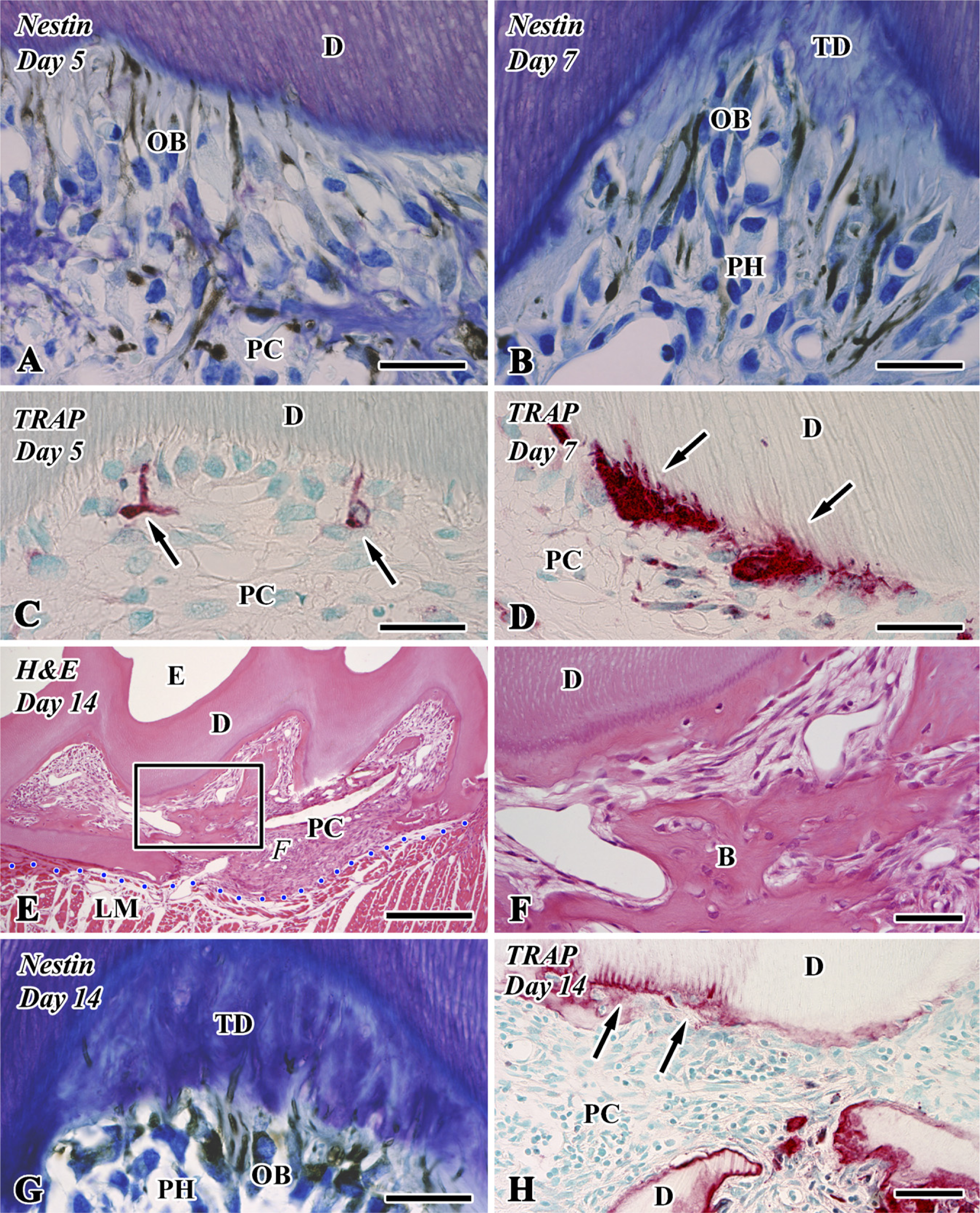
Nestin immunoreactivity (
LacZ Transgenic and Wild-type ROSA26 Mice
The strain of the lacZ transgenic ROSA 26 mice was B6.129S7-Gt(ROSA)26Sor/J. These transgenic mice were purchased from Jackson Laboratory (Bar Harbor, ME). Cells of these lacZ transgenic ROSA 26 mice can be stained by 5-bromo-4-chloro-3-indolyl-β-D-galactopyranoside (X-gal) solution, because the endogenous promoter of the lacZ gene is ubiquitously activated. In contrast, cells of wild-type ROSA26 mice cannot be stained by X-gal solution. In our study, 3-week-old lacZ transgenic and wild-type ROSA26 mice were used as the donor and host, respectively, and vice versa. Our preliminary experiment using lacZ transgenic ROSA26 mice clearly showed that the combination of allogenic tooth transplantation and lacZ transgenic ROSA26 mice can be used to study the contribution of donor and host cells to the regenerated pulp tissue (Kim et al. 2006).
Histological Procedure
Materials were collected in groups of five animals at intervals of 1, 3, 5, 7, and 14 days after transplantation (n = 25) in addition to 10 animals at 14 days after allogenic tooth transplantation into the sublingual region between lacZ ROSA26 transgenic teeth and wild-type teeth (n = 10). At each stage, the animals were intraperitoneally injected with BrdU (150 mg/kg) and subsequently perfused with physiological saline transcardially followed by 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) 2 hr after BrdU injection under deep anesthesia. BrdU labeling was omitted in the lacZ transgenic and wild-type ROSA26 mice. The tongues including the transplanted teeth were removed en bloc and immersed in the same fixative for an additional 6 hr. After decalcification in a 10% EDTA disodium salt (EDTA-2Na) solution for 2 weeks at 4C, the specimens were embedded in paraffin, and sagittal sections of transplants with surrounding lingual tissues were cut at 5 μm. The paraffin sections were mounted on Matsunami adhesive silane (MAS)-coated glass (Matsunami Glass; Osaka, Japan) slides and stained with hematoxylin and eosin (H&E).
IHC Analysis
For the immuno-peroxidase procedure, the sections were processed for the Calbiochem BrdU IHC system (EMD Biosciences; Darmstadt, Germany), avidin-biotin peroxidase complex (ABC) method using a rabbit anti-OPN polyclonal antibody diluted to 1:5000 (Cosmo Bio Co.; Tokyo, Japan), and Nichirei Histofine Simple Stain Mouse MAX-PO (Nichirei Biosciences; Tokyo, Japan) using a mouse anti-nestin monoclonal antibody diluted to 1:50 (Chemicon International; Temecula, CA). The sections were counterstained with hematoxylin or 0.05% methylene blue. The brown in BrdU-labeled cells was changed to red and graphically emphasized by using graphic software (Adobe Photoshop CS2 for Windows; Adobe Systems, San Jose, CA). IHC controls were performed by (a) replacing the primary antibodies with non-immune serum or PBS and (b) omitting the streptavidin-peroxidase or the MAX-PO solution. These immunostained sections contained no specific immunoreaction.
Histochemical Analysis
For the histochemical demonstration of TRAP activity, the azo-dye method was used with slight modifications (Tsukamoto-Tanaka et al. 2006). The frozen sections were incubated for 15 min at room temperature in a medium comprising 0.01% naphthol AS-BI phosphatase (Na salt; Sigma Chemical, St. Louis, MO), 0.06% fast red violet LB salt (Sigma Chemical), and 50 mM L-(+)-tartaric acid in 0.2 M acetate buffer (pH 5.3). The sections were counterstained with 0.5% methyl green or 0.05% methylene blue.
Identification of the lacZ Transgenic Cell in the Tissue Section
After decalcification in a 10% EDTA-2Na solution for 2 weeks at 4C, the fixed tongues including the transplanted teeth were washed with 2 mM MgCl2 in PBS for 5 min, rinsed three times with a rinse buffer (2 mM MgCl2, 0.02% NP-40, 0.01% sodium deoxycholate in PBS) for 20 min at room temperature, stained with β-gal staining solution (1 mg/ml of X-gal, 5 mM potassium ferrocyanide, and 5 mM potassium ferricyanide), and incubated at 37C for 1 hr. After X-gal staining, the samples were washed again with PBS for 10 min. The specimens were embedded in paraffin, and sagittal sections of transplants with surrounding lingual tissues were cut at 5 μm. The paraffin sections were mounted on MAS-coated glass (Matsunami Glass) slides and counterstained with nuclear fast red.
Statistical Analysis of Cell Proliferation
The number of BrdU-positive cells in the pulp areas including the pulp horn and chamber of each specimen was calculated. Quantitative analysis was performed in three areas for each sample. The data were obtained from the samples of 20 animals (5 animals per group; the final number of samples was 60 areas), and the grid (211 × 169 μm2) was selected at random in each area (pulp horn and chamber). All data were presented as the means and SD of each group. Furthermore, the number of cells in the pulp horn and chamber among different times after transplantation was compared using Bonferroni's test (one-way ANOVA) by using statistical software (SPSS 14.0J for Windows; SPSS Japan, Tokyo, Japan).
Results
Histological Analysis
The H&E-stained paraffin sections clearly showed the two types of hard tissue formation (i.e., tubular dentin and bone-like tissue) in the dental pulp chamber of the transplant during the healing process (Figures 1, 2E, and 2F). On Day 1, the pulp chamber was mainly occupied by inflammatory lesions including numerous neutrophils, red blood cells, and fibrin networks. Neutrophils migrated into the degenerating odontoblast layer in the pulp horn (Figures 1A and 1B). On Day 3, a different type of cells appeared along the pulp-dentin border and extended their cellular processes into the dentinal tubules (Figures 1C and 1D). The odontoblast-like cells came to be arranged along the pulp-dentin border and deposited tubular dentin next to the preexisting dentin at the pulp horn on Days 5-7 (Figures 1E-1H). On Day 14, bone-like tissue formation occurred in the pulp chamber independently of the tubular dentin (Figures 2E, 2F, 3A, 3C, and 3D). The distinction between dentin and bone-like tissue was determined by the existence of dentinal tubules in the case of dentin and cell inclusion in the case of bonelike tissue (Figures 1H and 2F) in combination with the immunoreactivity for nestin and OPN (Figures 2B, 2G, 3B, and 3C), although occasionally the mixed features of dentin and bone appeared in the regenerated tissue.
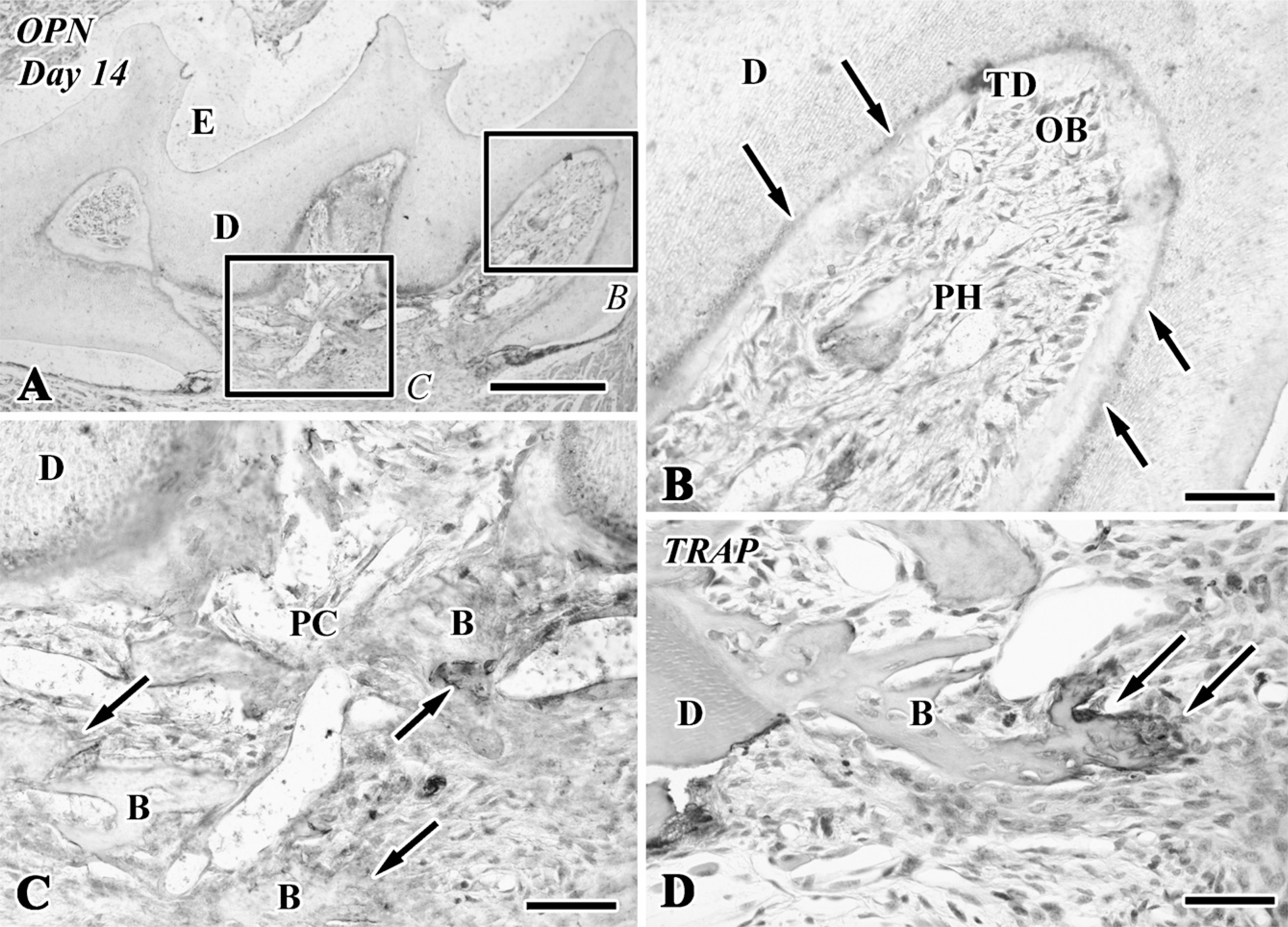
Osteopontin (OPN) immunoreactivity (
Nestin and OPN Analyses
On Days 1-3, although an intense nestin-positive reaction was not recognizable in the pulp chamber (data not shown), the newly differentiated odontoblast-like cells in the pulp horn showed intense immunoreactivity for nestin in their cytoplasm (Figures 2A, 2B, and 2G) and lacked an OPN-immunopositive reaction (Figure 3B) after Days 5-14. The boundary between the preexisting and tertiary dentin always represented an OPN-positive reaction (Figures 3A and 3B). In contrast, the osteoblast-like cells beneath the bone-like tissue matrix showed OPN-positive (Figures 3A and 3C) and nestin-negative reactions (data not shown).
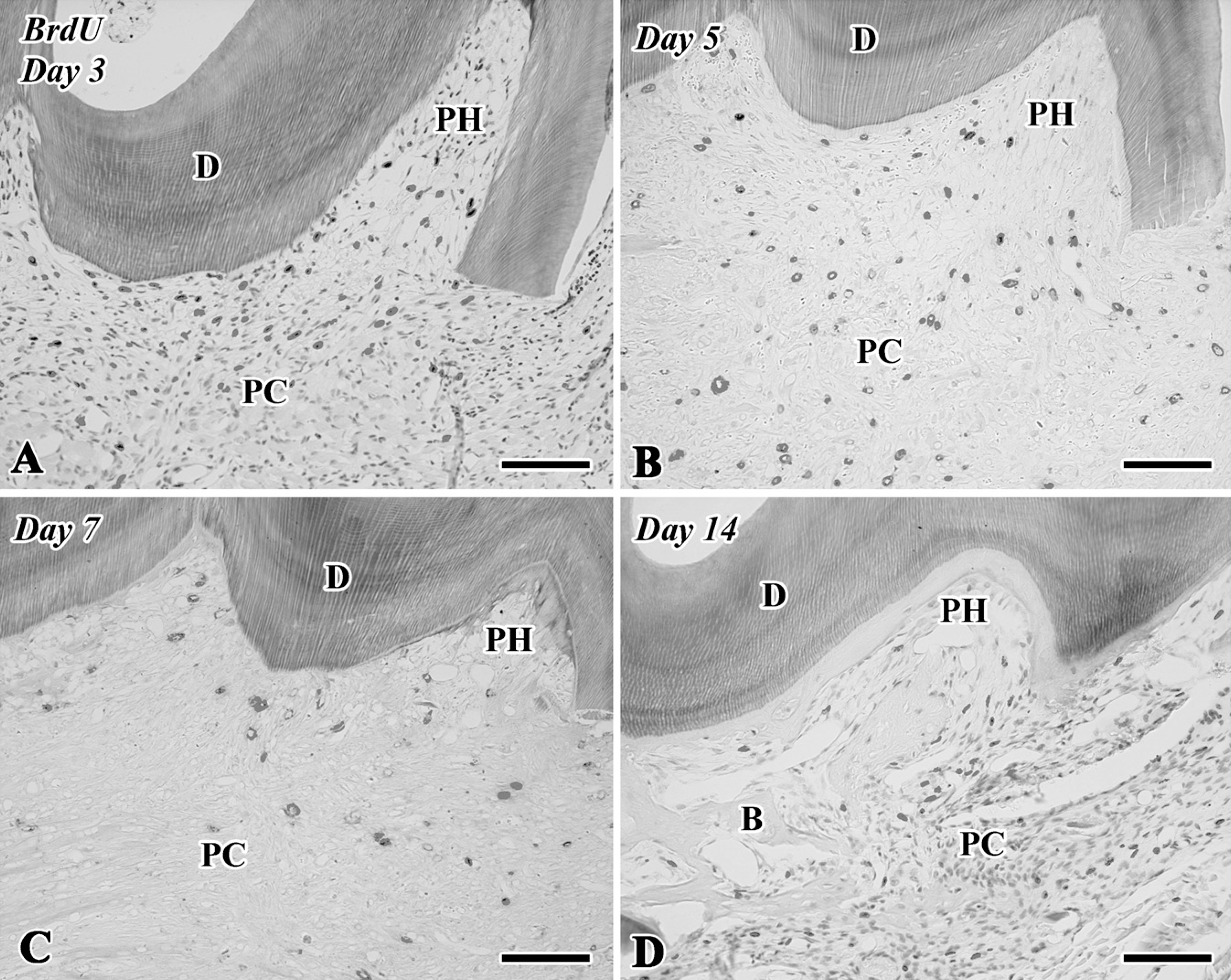
5-Bromo-2′-deoxyuridine (BrdU)-labeled sections of the transplanted teeth at 3 (
TRAP Analysis
Tooth transplantation caused the appearance of intense TRAP-positive reactions in the pulp chamber after Day 5 (Figure 2C). TRAP-positive cells were occasionally situated at the pulp-dentin border, elongated their cellular processes into the dentinal tubules (Figure 2D), and remained around the bone matrix until Day 14 (Figures 2H and 3D).
Cell Proliferation Assay by BrdU Labeling
Although few BrdU-positive cells were recognizable in the pulp chamber on Day 1 (data not shown), a large number of BrdU-positive cells were observed in both the pulp horn and chamber on Day 3 (Figures 4A and 5). On Days 5-14, the BrdU-positive cells were significantly decreased in number in the pulp chamber (Figures 4B-4D and 5). The number of BrdU-labeled cells was statistically analyzed separately in two areas such as the pulp horn and chamber (Figure 5).
Allogenic Transplantation Between lacZ Transgenic and Wild-type ROSA26 Mice
The blue-stained cells after X-gal staining corresponded to the lacZ transgenic cells from the lacZ transgenic mice (Kim et al. 2006), although stainability differed depending on the types of cells and their cellular activity. In tooth transplantation using lacZ transgenic ROSA 26 mice as the donor and wild type as the host, newly differentiated odontoblast-like cells beneath the dentin matrix contained granular depositions with the blue color. Osteoblast-like cells also showed the blue color in their cytoplasm in addition to intense stainability in polynuclear giant cells and osteoclast-like cells, although osteocytes and red blood cells in blood vessels showed negative reactions (Figures 6A-6D). In the experiment using lacZ transgenic ROSA 26 mice as the host and wild type as the donor, blue-stained cells were recognized in the osteoblast- and osteoclast-like cells, mesenchymal cells, and red blood cells in blood vessels in contrast with odontoblast-like cells and osteocytes with negative reactions (Figures 6E-6H). The results of X-gal stainability in the above two types of experiments are summarized in Table 1. To confirm the specificity of X-gal staining in the osteoclast-like cells, we performed both X-gal staining and TRAP histochemistry in the periodontal tissue of the wild-type ROSA26 mice. TRAP-positive osteoclast-like cells showed blue color in their cytoplasm, suggesting that X-gal staining was unable to determine the lacZ transgenic osteoclast-like cells in our experiment (Figure 7).
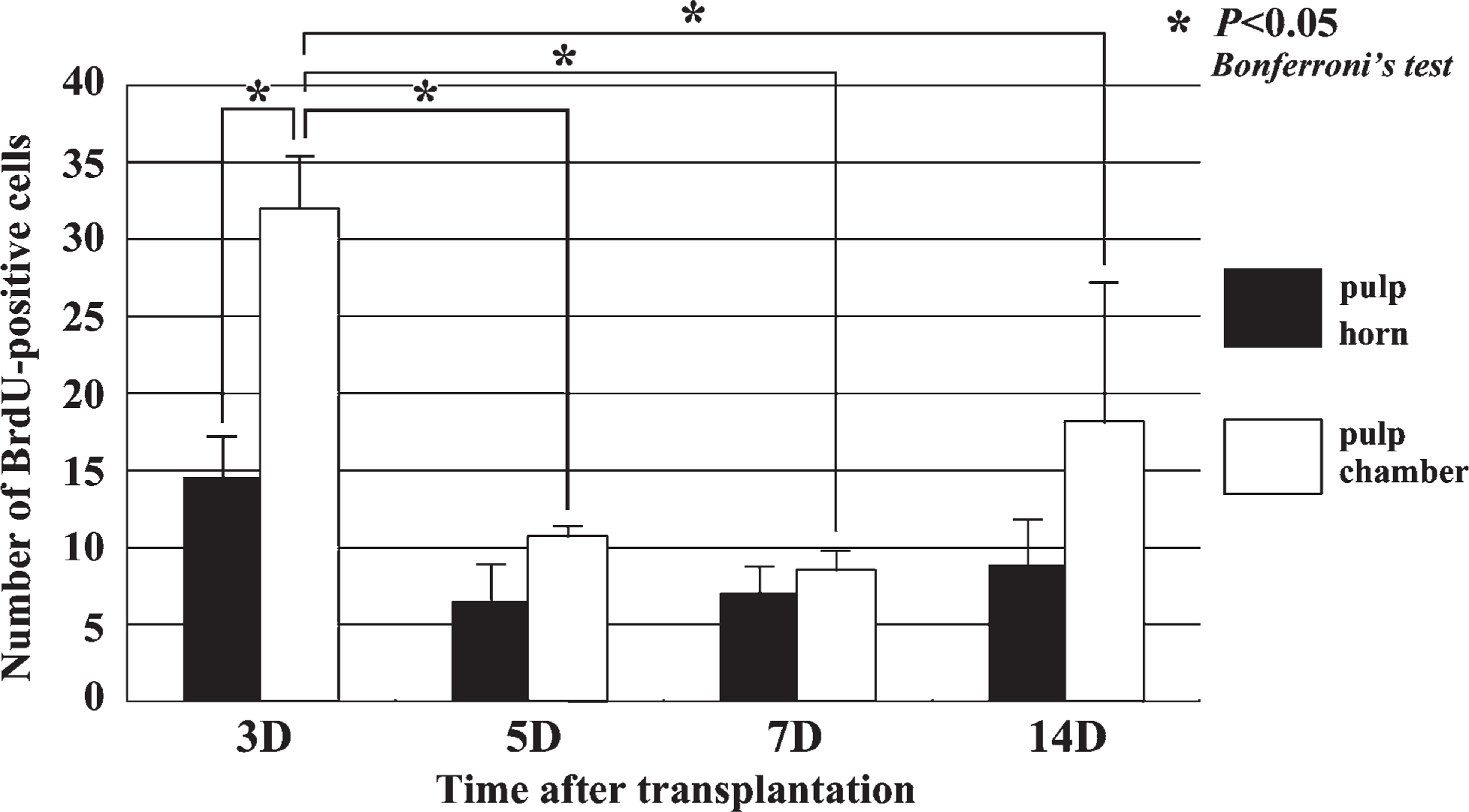
Quantitative analysis of cell proliferation in the dental pulp of replanted teeth at 3, 5, 7, and 14 days after tooth transplantation. BrdU-positive cells in the pulp chamber are significantly increased in number at Day 3 compared with the other stages.
Discussion
An experimental mouse model for allogenic transplantation of tooth crown into the sublingual region was established in this study. This model is useful for clarifying the capacity of dental pulp differentiation in the regenerative process after tooth transplantation, because the tooth root and periodontal tissues including root dentin, periodontal ligament, and cementum have already been removed from the donor transplant before allograft into the sublingual region. In contrast, the experimental model for conventional tooth replantation or transplantation includes the commitment of the periodontal tissue for pulp regeneration. Actually, the vascular supply from the periodontal tissue reaches the coronal portion of dental pulp until 5 days after tooth replantation (Shimizu et al. 2000). Another advantage of this model is that immunological rejection does not occur in the case of the use of littermates, despite allogenic tooth transplantation. The obtained results on the histological changes occurring in this study were almost the same as those in autogenic tooth transplantation (Ogawa et al. 2006). The summarized pulpal changes in the regenerative process are as follows: odontoblasts showed the degenerative features and inflammatory reactions including neutrophil infiltration and hemorrhage occurred in the pulp chamber on Day 1, cell proliferation in the pulp chamber became most active on Day 3, nestin-positive and OPN-negative newly differentiated odontoblast-like cells were arranged along the pulp-dentin border to deposit the dentin matrix in the pulp horn on Days 5-7, and bone-like tissue occurred in the pulp chamber apart from the continuous deposition of the dentin matrix on Day 14. Thus, this observation in allogenic tooth transplantation confirms that the dental pulp contains two types of competent progenitor cells capable of differentiating into either odontoblast- or osteoblast-like cells, as shown in autogenic tooth transplantation.
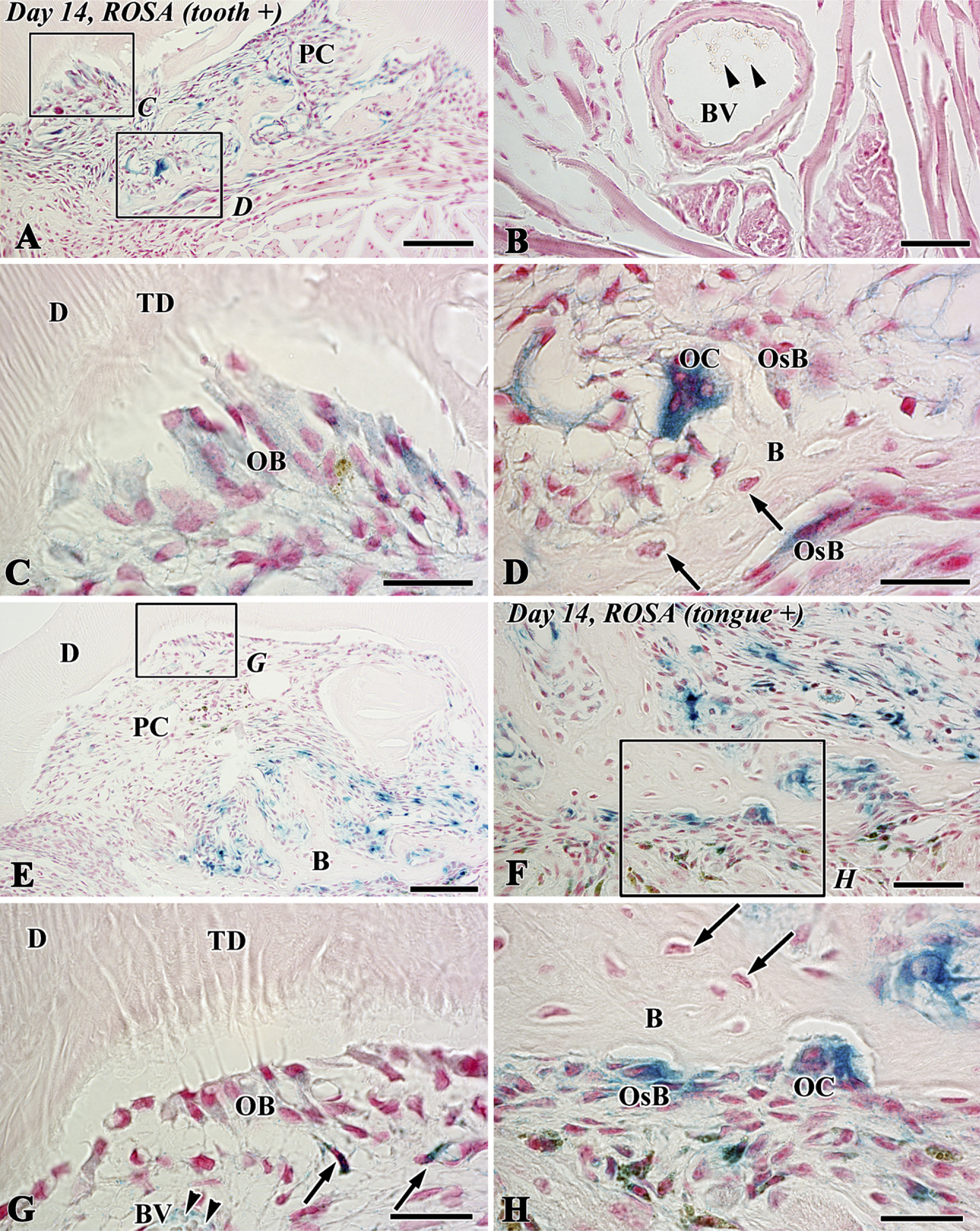
X-gal staining in sections of the transplants at 14 days after allograft of the tooth of LacZ transgenic ROSA26 mice into the sublingual region of wild-type mice (
X-gal stainability in each cell in the dental pulp after tooth transplantation using lacZ transgenic ROSA26 and wild-type mice
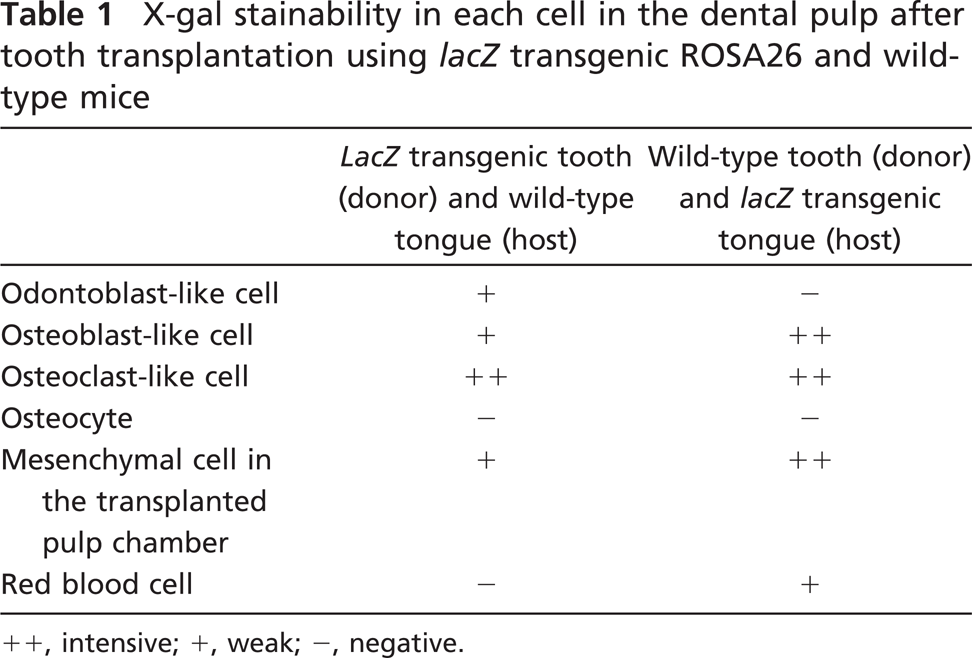
++, intensive; +, weak; −, negative.
With respect to the capacity of dental pulp differentiation, it is well established that undifferentiated mesenchymal cells exist in the dental pulp and have the ability to differentiate into odontoblast-like cells, which are responsible for reparative dentin formation after tooth injury (Yamamura 1985; Ohshima 1990; Tziafas 1995; About et al. 2000a; Nakakura-Ohshima et al. 2003; Ohshima et al. 2003). Other studies have shown that isolated pulp tissue contributes to bone tissue formation in transplants (Zussman 1966; Luostarinen and Ronning 1977; Yamamura 1985; Takei et al. 1988; Inoue and Shimono 1992; Laino et al. 2005). In contrast, the pulpal mesenchymal cells from postnatal mouse incisors can give rise to odontoblasts and osteoblasts and functionally produce matrix when transplanted into kidney capsules (Braut et al. 2002, 2003). Thus, it is reasonable to suppose that dental pulp is composed of various progenitor populations including odontoblast and osteoblast lineage cells and/or one multipotent stem cell, which gives rise to differentiation of both the odontoblasts and osteoblasts, and that certain regulatory mechanisms for determining the fate of these different lineage cells may exist in the microenvironment or niche. One important regulatory factor may be the existence of scaffolds such as preexisting dentin, because the differentiation of odontoblast-like cells always occurred beneath the preexisting dentin, and new dentin formation never occurred apart from the preexisting dentin matrix in this study. This notion is supported by evidence that signals similar to those involved in physiological dentinogenesis (Ruch et al. 1995) might be sequestered within the dentin matrix and released under pathological conditions (Tziafas et al. 2000; Smith and Lesot 2001; Goldberg and Smith 2004). Another important regulatory factor may be the type of cells appearing along the pulpdentin border before the differentiation of hard tissue-formative cells. The close relationship between the appearance of intense TRAP-positive cells and the induction of bone-like tissue formation was shown in this study. The bone tissue formation occasionally occurred even beneath the preexisting dentin, where TRAP-positive osteoclast lineage cells appeared and extended their cellular processes into the dentinal tubules (Figures 2D and 2F). Thus, the appearance of osteoclast lineage cells may be involved in the induction of bone-like tissue formation in the allogenic transplanted tooth and the autografted tooth.
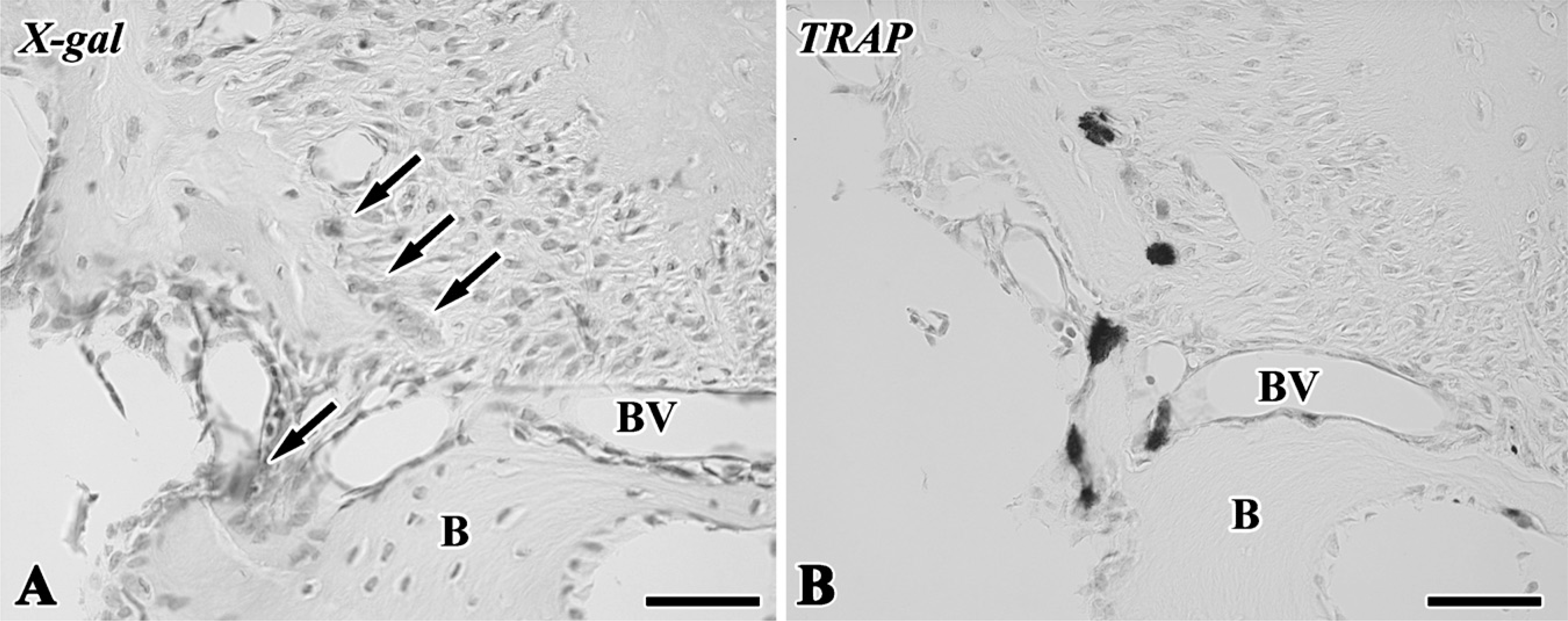
X-gal staining (
A recent study has shown that odontoblast and osteoblast lineage cells reside in the dental pulp in autogenic tooth transplantation (Ogawa et al. 2006). Regarding the origin of osteoblast lineage cells, we still cannot exclude the possibility that progenitor cells from the surrounding lingual tissue migrate into the pulp chamber and proliferate to give rise to hard tissue-forming cells. These allograft transplantation experiments using ROSA26 reporter mice clearly showed that both donor and host mesenchymal cells differentiated into osteoblasts with the assistance of osteoclast lineage cells, whereas newly differentiated odontoblasts were exclusively derived from donor pulpal cells. The findings are supported by a recent study describing allogenic tooth transplantation experiments using GFP reporter rats, although the previous study failed to clarify the capacity of dental pulp differentiation because the donor-transplanted tooth contained the periodontal tissue (Zhao et al. 2007). However, we have not been able to determine the origin of osteoclast lineage cells, because X-gal staining was unable to determine lacZ transgenic osteoclast-like cells in our experiment, in which TRAP-positive osteoclast-like cells in the periodontal tissue of wild-type ROSA mice showed LacZ-positive reactions in their cytoplasm. This osteoclast pitfall was mentioned in a recent study (Kopp et al. 2007). It is reasonable to suppose that osteoclast lineage cells are derived from the circulatory system in the host tissue, because TRAP-positive osteoclast-lineage cells first appear 5 days after transplantation, and they are associated with blood vessels. In contrast, the osteocytes in both donor and host tissues showed LacZ-negative reactions, probably because of their decreased cellular activity. Thus, the results suggest that the odontoblast and osteoblast lineage cells reside in the dental pulp and that both donor and host cells contribute to the bone-like tissue formation in the regenerated pulp tissue. With advances in stem cell biology and emerging concepts of tissue engineering (Langer and Vacanti 1993), biological teeth (Sharpe and Young 2005) may become an alternative for replacing missing teeth. The idea is to cultivate stem cells with odontogenic induction signals through epithelial-mesenchymal interactions, thereby programming the stem cells to adopt dental lineage and, with the help of scaffold/extracellular matrix, to become part of the tooth (Yen and Sharpe 2008). Thus, exact knowledge of the capacity of dental pulp differentiation would provide useful information for future regenerative treatment of the dental pulp and the application of this dental pulp capacity to biological teeth. Further studies are needed to clarify the relationship between the biological properties of dental pulp and the localization and differentiation capacity of DPSCs.
Footnotes
Acknowledgements
This work was supported in part by grants from KAKENHI (B) (16390523 and 19390462 to HO) and KAKENHI (C) (18592232 to KN-O) from MEXT and the Japan-Korea Joint Research Project from JSPS and KOSEF (F01-2005-000-10210-0).
The authors thank Shin-ichi Kenmotsu for technical assistance.
