Abstract
Allergic fungal rhinosinusitis (AFRS) shares similarities with eosinophilic chronic rhinosinusitis (ECRS), both characterized by intractable nasal polyps. The key distinction lies in the presence of fungal infection within the nasal cavity. While ECRS nasal polyps are known for significant infiltration of M2 macrophages and eosinophils, as well as an increase in high endothelial venule (HEV)-like vessels, these features are less commonly reported in AFRS. This study compared clinicopathological findings between AFRS (n=10), ECRS (n=12), and non-ECRS (n=10) patients’ nasal polyps using immunohistochemical analysis for CD163 and CD68 to assess the M2/M1 macrophage ratio, and peripheral lymph node addressin (PNAd) and CD34 to evaluate the proportion of HEV-like vessels. AFRS showed a significantly higher number of CD163-positive M2 macrophages and an increased M2/M1 ratio compared with ECRS. However, the percentage of HEV-like vessels and the number of eosinophils infiltrating the nasal polyps were similar in both AFRS and ECRS. The observed increase in M2 macrophages in AFRS nasal polyps is presumed to be induced by fungal infection in the nasal cavity, in comparison with ECRS. These results highlight the distinctive immunological profiles of AFRS and ECRS, emphasizing the role of macrophage polarization in their pathogenesis.
Keywords
Introduction
Fungal infections are well-known contributors to chronic inflammatory diseases. Most such infections manifest on the surface of the human body, notably on the skin and in the oral cavity, and are typically managed using antifungal drugs. However, fungal infections affecting the nasal cavity, known as fungal rhinosinusitis, exhibit poor responsiveness to antifungal medications, often necessitating surgical intervention. Allergic fungal rhinosinusitis (AFRS) represents a subtype of chronic fungal rhinosinusitis characterized by allergic inflammation in response to fungi. Nasal polyps observed in AFRS display resistance to conservative treatments, contributing to the rationale for their surgical management.
The diagnosis of AFRS relies on the presence of fungi in the nasal mucin.
1
Chronic fungal infection within the nasal cavity triggers a systemic allergic reaction to fungi. Elevated levels of fungus-specific IgE are considered significant in the pathology of AFRS, although the specific details remain unclear. Macrophages, which identify β-
Chronic rhinosinusitis (CRS) is broadly categorized into CRS with nasal polyps (CRSwNP) and CRS without nasal polyps (CRSsNP). 4 While this classification is straightforward and widely utilized, CRSwNP encompasses various diseases, including AFRS and eosinophilic chronic rhinosinusitis (ECRS). 5 Despite the ability to differentiate AFRS from ECRS based on the presence of fungi in the nasal cavity, these conditions share several common features, such as persistent nasal polyps and elevated peripheral blood eosinophil counts. Furthermore, both AFRS and ECRS involve eosinophils and macrophages infiltrating nasal polyps, playing a significant role in their pathogenesis.6,7 Nevertheless, there is a limited number of reports that have delved into the distinctions in the pathogenesis between AFRS and ECRS.
In recent years, it has been discovered that there are functionally distinct subtypes of macrophages, and classification is in progress.8,9 M1 macrophages are activated in the early stages of inflammation and secrete cytokines that mobilize Th1 cells. Conversely, M2 macrophages function in a suppressive manner against the immune response, thereby contributing to the persistence of chronic inflammation. 7 According to the latest understanding, macrophages have the plasticity to differentiate into both M1 and M2 depending on the surrounding cellular environment and cytokine stimulation. 10 Studies have reported an increase in the number of M2 macrophages in nasal polyps associated with CRSwNP, which is classified as a type 2 inflammatory disease. 11
We previously reported the involvement of high endothelial venule (HEV)-like vessels expressing peripheral lymph node addressin (PNAd) in eosinophil recruitment within nasal polyps formed in ECRS. 12 Our research demonstrated a correlation between the percentage of HEV-like vessels and the severity of ECRS. 13 HEV-like vessels are also known to be involved in monocyte recruitment, 14 but the contribution of HEV-like vessels in monocyte recruitment in nasal polyps and the difference of M1/M2 macrophage infiltration between ECRS and AFRS have not been reported.
In this study, we conducted a comparative analysis between AFRS and ECRS in terms of the M2/M1 macrophage ratio and the proportion of HEV-like vessels in total vessels using quantitative immunohistochemical analysis. Our findings revealed that the M2/M1 macrophage ratio in AFRS nasal polyps was greater than that observed in ECRS.
Materials and Methods
Patients and Tissue Samples
Patients who underwent endoscopic sinus surgery (ESS) for CRSwNP at the Department of Otorhinolaryngology and Head & Neck Surgery, University of Fukui Hospital, and its affiliated hospitals between January 2016 and November 2021 were included in the study. Formalin-fixed, paraffin-embedded (FFPE) tissue blocks of surgically resected nasal polyps from these patients were obtained from the hospital’s pathology archives. The diagnosis of AFRS was made based on the criteria by Bent and Kuhn (Table 1).1,15 The diagnosis of ECRS was made according to the Japanese Epidemiological Survey of Refractory Eosinophilic Chronic Rhinosinusitis (JESREC) criteria. 16 Consequently, 10 cases of non-ECRS, 12 cases of ECRS (3 severe, 5 moderate, and 4 mild), and 10 cases of AFRS were included in the analysis (Table 2). This study included adult male and female patients. Of the three severe ECRS cases, one had concomitant nonsteroidal anti-inflammatory drug (NSAID)-exacerbated respiratory disease (NERD). Analysis of human nasal polyp tissues was approved by the Ethics Committee of the Faculty of Medical Sciences, University of Fukui (reference number 20230154, approved on February 9, 2023).
Bent and Kuhn Diagnostic Criteria.

Abbreviation: CT: computed tomography.
Heterogeneous signal intensity in especially ethmoid sinus, and sinus expansion by the fungal mucin, and no existence of fungal invasion.
Univariate Analysis of Clinical Information Relevant to Cases Examined in This Study.

Data are presented as mean ± SD. bp<0.05 indicates that the item differs in some combination between groups.
p<0.001.
Immunohistochemistry
The following monoclonal antibodies served as primary antibodies: PG-M1 (mouse IgG; Dako, Glostrup, Denmark) recognizing CD68, a marker of M1 macrophages;17,18 10D6 (mouse IgG; Leica Biosystems, Newcastle Upon Tyne, UK) recognizing CD163, a marker of M2 macrophages;17–19 QBEND10 (mouse IgG; Immunotech, Marseille, France) recognizing CD34, a marker of vascular endothelial cells; 20 and MECA-79 (rat IgM; BD Biosciences, Franklin Lakes, NJ) recognizing 6-sulfo N-acetyllactosamine attached to extended core 1 O-glycans that constitutes PNAd.21,22 Immunohistochemical staining for CD68, CD163, and CD34 was carried out using the Histofine system (Nichirei Biosciences, Tokyo, Japan) following the manufacturer’s protocol. 23 In addition, staining for PNAd was performed using an indirect method, as previously described. 12
Counting of Eosinophils in Tissue Specimens
In the initial step, we scrutinized hematoxylin and eosin (H&E)-stained tissue specimens using a microscope. Three high-power fields (HPFs) of view were selected, employing ×400 magnification, to identify regions with the highest concentration of eosinophils, commonly referred to as “hot spots.” Subsequently, eosinophils were counted in these three designated areas without access to clinical information. The mean number of eosinophils per HPF was then calculated and considered as the eosinophil count for each specimen. This measurement is essential for diagnosing ECRS according to the JESREC criteria. In this study, we applied a similar measurement to AFRS cases.
Quantification of PNAd-expressing Vessels
In this study, vessels with any portion of the vessel wall stained for PNAd were counted as positive vessels. We first scanned the entire area of tissue specimens immunostained for PNAd and identified three “hot spots” of PNAd-positive vessels at a magnification of ×200. Subsequently, we quantified the number of PNAd-positive vessels in these three specified areas, unaware of any clinical information. Simultaneously, we determined the number of CD34-positive vessels in the corresponding regions. For each sample, the aggregate number of PNAd-positive vessels across the three areas was divided by the total count of CD34-positive vessels to compute the percentage of PNAd-positive vessels among all vessels, denoted as the PNAd+/CD34+ vessel ratio. 13
Counting of Macrophages in Tissue Specimens
Sections immunostained for CD68 and CD163 were scanned with a NanoZoomer S210 (Hamamatsu Photonics, Hamamatsu, Japan) to obtain whole slide images (WSIs). Scanned at ×20 magnification, we first identified a 0.2-mm2 field of view rich in both CD163- and CD68-positive macrophages (“hot spot”). In this area, we then determined the number of macrophages positive for CD163 and CD68 in the same area using NDP.view2 software (Hamamatsu Photonics). The number of CD163-positive cells, excluding obvious monocytes, was defined as the number of M2 macrophages. Similarly, the number of CD68-positive cells was defined as the number of M1 macrophages. The ratio of M2/M1 macrophages ratio was obtained by dividing the number of CD163-positive macrophages by that of CD68-positive macrophages.
Statistical Analysis
For comparison between groups in Table 2, the nominal scale was statistically analyzed by Pearson’s χ2 test, and the ordinal scale was statistically analyzed by the one-way analysis of variance (ANOVA). Differences between groups were statistically analyzed by Student’s t-test. Statistical outliers were excluded by the Smirnov–Grubbs test. All statistical analyses were done using JMP 16 software (SAS Institute, Cary, NC). The p values less than 0.05 were considered significant.
Results
Peripheral Blood Eosinophil Counts in AFRS Were Significantly Higher Than in ECRS and non-ECRS
We first analyzed clinical characteristics of patients with non-ECRS, ECRS, and AFRS. As shown in Table 2, the mean age and sex ratio for each group were as follows: 52.9 years and a ratio of 9:1 for the non-ECRS group, 47.3 years and a ratio of 11:1 for the ECRS group, and 41.8 years and a ratio of 7:3 for the AFRS group. All groups had a higher number of males. There was no difference in age and sex ratio among groups. Despite the fact that only non-ECRS patients had a zero incidence of asthma, there was no difference in the prevalence of asthma between groups. While peripheral blood leukocyte counts and total IgE levels did not differ between groups, the percentage of peripheral blood eosinophils in AFRS was significantly greater than that in the other groups.
Multiple Sensitization to Fungi Was Observed in Many AFRS Cases
Antigen-specific IgE levels were determined by ImmunoCAP. 24 If the ImmunoCAP value of an antigen was two or more, the antigen was considered positive. As shown in Table 3, most AFRS cases were sensitized to multiple species of fungi. Similarly, sensitization to non-fungal allergens was identified in most AFRS cases. Across the AFRS cases, total IgE levels varied. Alternaria spp. were the most frequently identified as positive sensitizers among fungi. On the contrary, allergy for Mucor spp. and Malassezia (formerly known as Pityrosporum) spp. were the least seen in these AFRS cases.
ImmunoCAP Results of Patients With AFRS in This Study.

Patients whose data not shown were tested by ImmunoCAP only about fungi. bThe blank means no data about each fungus.
Fungal Hyphae Were Proven by Grocott or Periodic Acid-Schiff Staining in All AFRS Cases
As the confirmation of AFRS diagnosis relies on postoperative pathology, Grocott and periodic acid-Schiff (PAS) staining were applied to all patients’ surgical specimens (Fig. 1). Grocott staining (Fig. 1A) revealed the presence of fungal hyphae within the mucin, indicated by arrows. PAS staining (Fig. 1B) further confirmed the presence of fungal elements in the mucin, as highlighted by the arrows. H&E staining (Fig. 1C) displayed the overall histological architecture of the nasal polyp tissue. A large number of eosinophils (arrowheads) were observed infiltrating the nasal polyp tissue. Arrows indicate fungal hyphae, and insets show their enlarged images. Fungal hyphae were observed in the mucin of all AFRS cases. No fungal invasion within the mucosa of nasal polyp was observed; therefore, there was no case classified into chronic invasive fungal rhinosinusitis.
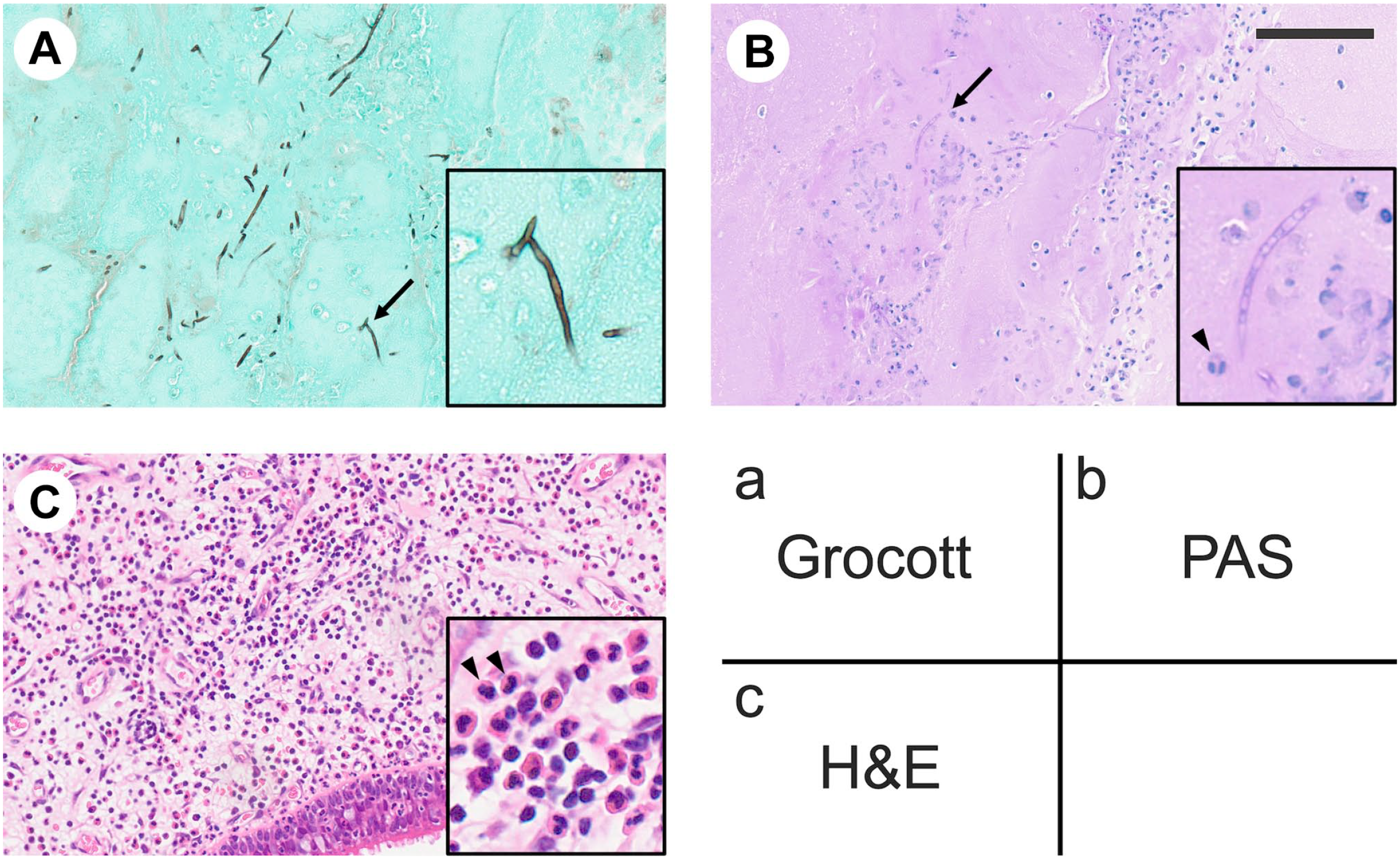
Representative histopathological appearance of mucin and nasal polyps formed in AFRS. Each specimen was stained as indicated in the legend on the lower right. (A) Grocott staining for the mucin. (B) Periodic acid-Schiff (PAS) staining for the mucin. (C) Hematoxylin and eosin (H&E) staining for the nasal polyp. Arrows indicate fungal hyphae, and insets show their enlarged images. Arrowheads indicate eosinophils. Representative cases are shown. Bar = 100 μm.
The Number of Eosinophils Infiltrated Did Not Differ Between ECRS and AFRS
We counted eosinophils infiltrated in nasal polyp as described. As shown in Fig. 2, the number of eosinophils in both ECRS and AFRS was greater than that in non-ECRS with high statistical significance (p<0.001 and p=0.004, respectively). However, no difference was found between ECRS and AFRS (p=0.094).

Box-and-whisker diagram of the number of eosinophils infiltrating nasal polyps in non-ECRS (n=10, white box), ECRS (n=12, light gray box), and AFRS (n=10, dark gray box). The vertical axis represents the count. The horizontal line in each box indicates the median of the group. **p<0.01, ***p<0.001.
The Percentage of PNAd-positive Vessels Did Not Differ between ECRS and AFRS
We next immunostained nasal polyp tissue sections with antibodies against PNAd and the vascular endothelial cell marker CD34. As shown in Fig. 3, all vessels in the tissue were immunolabeled for CD34 (bottom row), whereas only a fraction of these vessels were immunolabeled for PNAd (middle row). The number of both PNAd- and CD34-positive vessels per unit area did not differ between groups (Fig. 4, left and middle panels, respectively). We then calculated the proportion of PNAd-positive vessels among total vessels (the PNAd+/CD34+ ratio) and compared that value among groups. Consequently, the percentage of PNAd-expressing vessels in both ECRS and AFRS was greater than that seen in non-ECRS (Fig. 4, right panel). However, no difference was found between ECRS and AFRS.
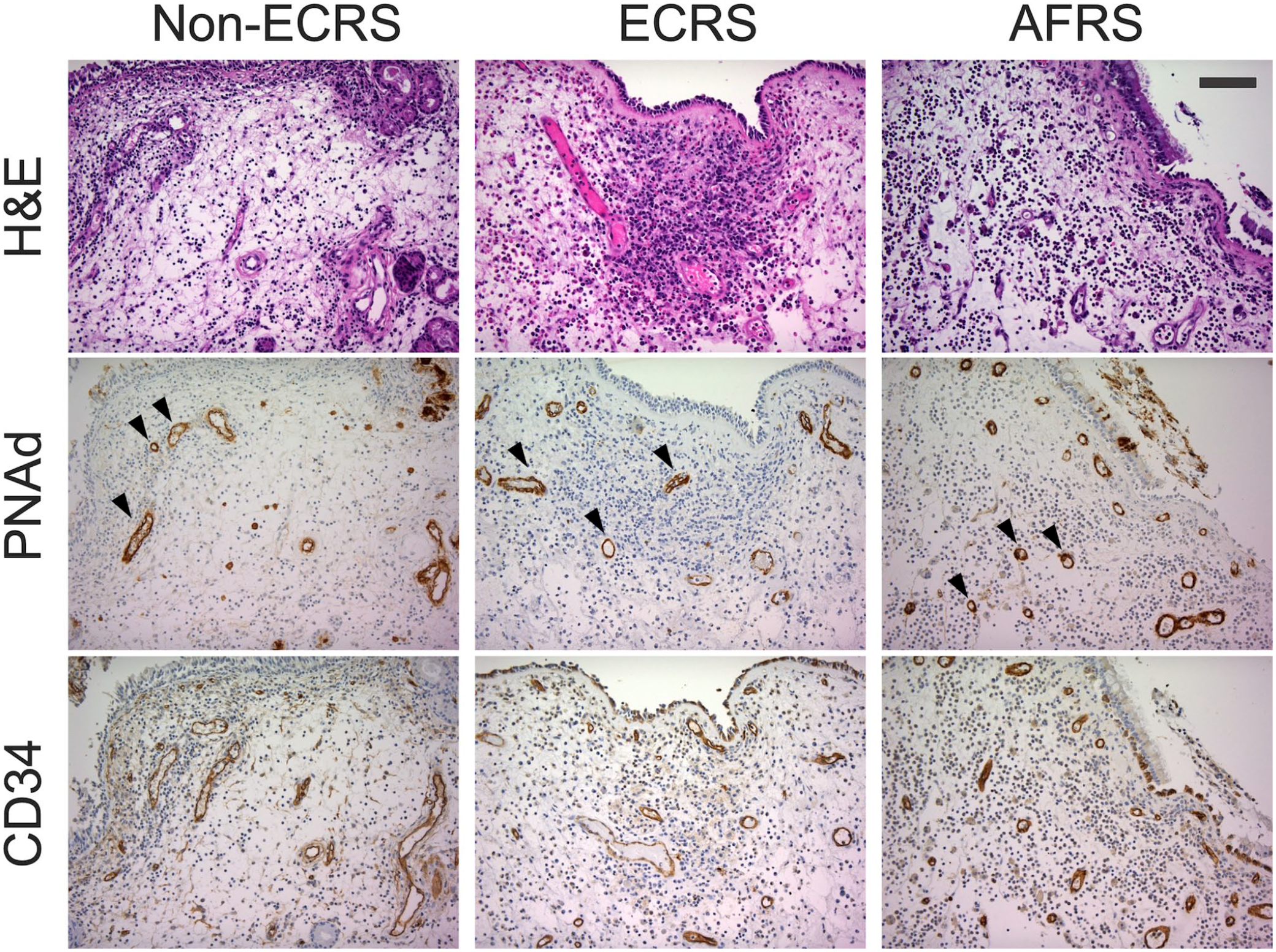
Induction of PNAd-positive vessels in nasal polyps. Serial tissue sections of nasal polyps formed in non-ECRS, ECRS, and AFRS were stained with hematoxylin and eosin (H&E) (top row) or immunostained for PNAd (middle row) or CD34 (bottom row). Signals on tissue sections in middle and bottom rows were visualized with 3,3ʹ-diaminobenzidine (DAB) (brown), and tissues were counterstained with hematoxylin. The arrowheads indicate PNAd-positive (HEV-like) vessels. Representative cases are shown. Bar = 100 μm.
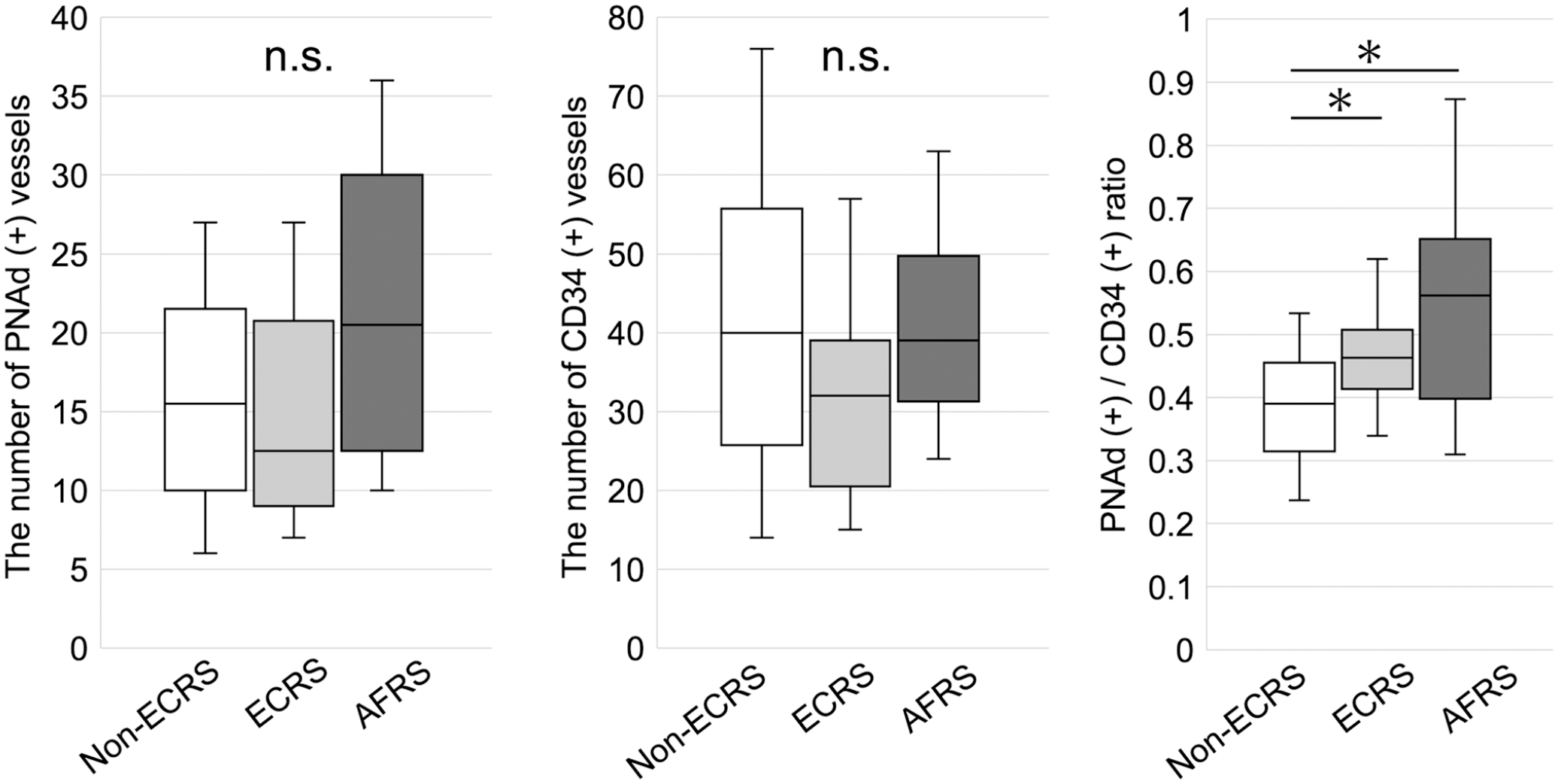
Box-and-whisker diagram of the number of PNAd- and CD34-positive vessels and the percentage of PNAd-expressing vessels in nasal polyps formed in non-ECRS (n=10, white box), ECRS (n=12, light gray box), and AFRS (n=10, dark gray box). The horizontal line in each box indicates the median of the group. NS, not significant; *p<0.05.
The M2/M1 Ratio in AFRS Was Significantly Greater Than That in ECRS
We next immunostained nasal polyp tissues for CD163 and CD68 to determine the number of M2 and M1 macrophages infiltrating nasal polyps formed in non-ECRS, ECRS, and AFRS. As shown in Fig. 5, in non-ECRS, infiltration of CD163- and CD68-positive cells was relatively sparse, but in ECRS and AFRS, nasal polyps were infiltrated with a large number of CD163- and CD68-positive macrophages. Interestingly, while the number of CD68-positive M1 macrophages did not differ among the three groups (Fig. 6, middle panel), the number of CD163-positive M2 macrophages in AFRS was significantly greater than that in non-ECRS (p<0.001) and ECRS (p=0.003) (Fig. 6, left panel). Moreover, the M2/M1 ratio in AFRS was significantly greater than that in non-ECRS (p=0.003) and ECRS (p=0.026) (Fig. 6, right panel). However, the M2/M1 ratio did not differ between non-ECRS and ECRS (p=0.057).
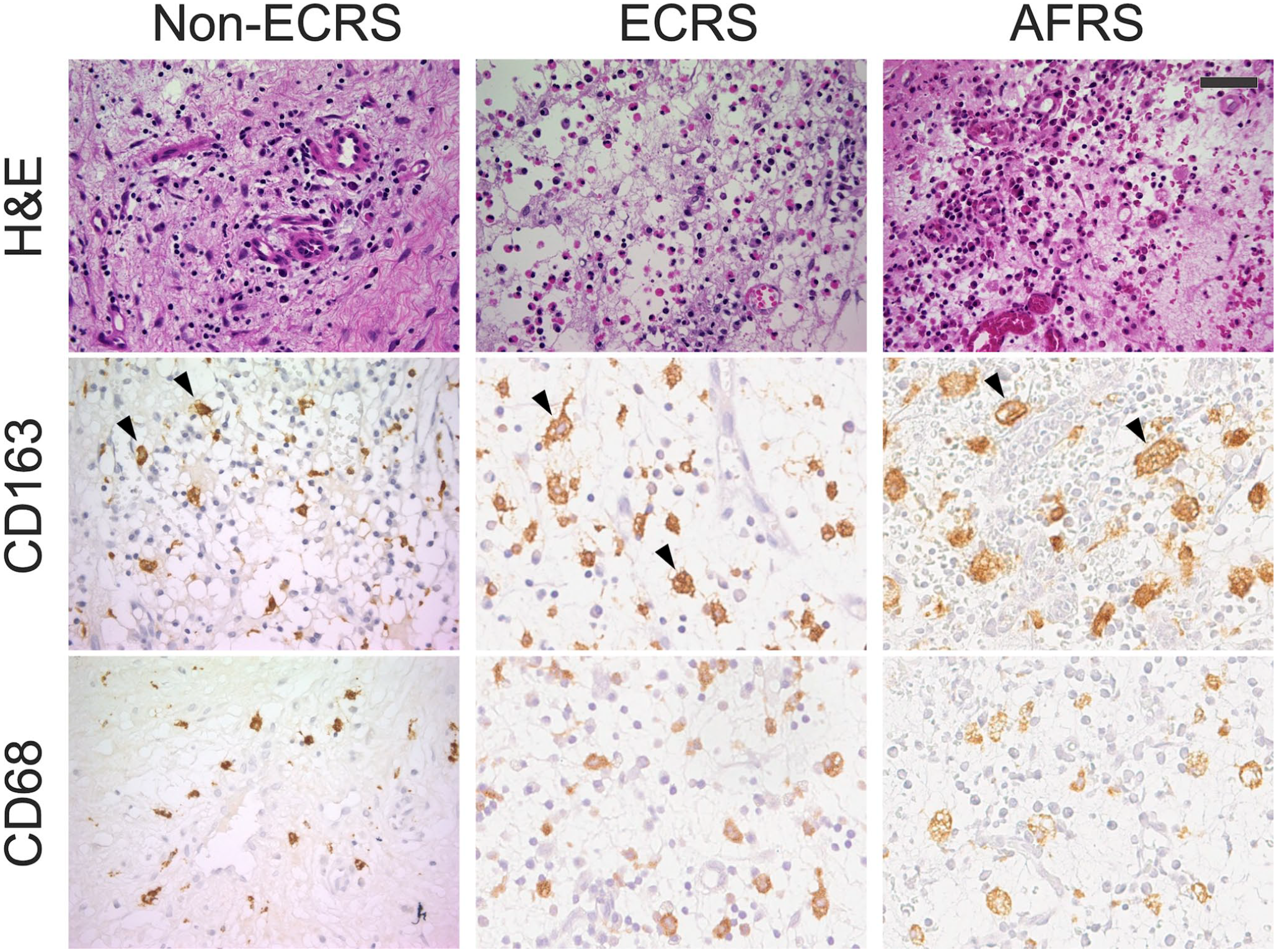
Infiltration of CD163-positive and/or CD68-positive macrophages in nasal polyps. Serial tissue sections of nasal polyps formed in non-ECRS, ECRS, and AFRS were stained with hematoxylin and eosin (H&E) (top row) or immunostained for CD163 (middle row) or CD68 (bottom row). Signals were visualized with 3,3ʹ-diaminobenzidine (DAB) (brown), and tissues were counterstained with hematoxylin. The arrowheads indicate macrophages. Representative cases are shown. Bar = 50 μm.

Box-and-whisker diagram of the number of CD163- and CD68-positive macrophages and the CD163/CD68 (M2/M1) macrophage ratio in nasal polyps formed in non-ECRS (n=10, white box), ECRS (n=12, light gray box), and AFRS (n=10, dark gray box). The horizontal line in each box indicates the median of the group. NS, not significant; *p<0.05, **p<0.01, ***p<0.001.
Discussion
In this study, we illustrated that the quantity of M2 macrophages, but not M1 macrophages, in AFRS was significantly higher than that observed in ECRS. In addition, we demonstrated that the M2/M1 ratio in AFRS was significantly elevated compared with ECRS. These findings suggest that nasal polyps formed in AFRS exhibit a heightened macrophage presence, with a notable shift in the macrophage phenotype toward M2.
M2 macrophages play a role in tissue remodeling and repair in chronic inflammation.
10
However, recent reports have highlighted increased M2 macrophages as a cause of various chronic diseases such as vasculitis and systemic lupus erythematosus (SLE).
25
As macrophages and dendritic cells have receptors for β-
In contrast to earlier reports indicating an increased presence of M2 macrophages in nasal polyps associated with ECRS,7,27 our present findings revealed no significant difference between non-ECRS and ECRS cases. This discrepancy could be attributed, in part, to the relatively small number of severe ECRS cases included in our study. Another potential factor may involve the transition from M1 to M2. It is suggested that monocytes migrate into peripheral tissues and subsequently undergo differentiation into M1, M2, or other macrophages, each with distinct functions. 10 M1 macrophages are hypothesized to have the capability to transform into M2 macrophages within peripheral tissues, 28 although the underlying mechanism remains unclear. Our observations of M2 macrophages in nasal polyps, even in non-ECRS and ECRS cases, suggest that the presence of fungi in the nasal cavity might not be a prerequisite for the increased presence of M2 macrophages in nasal polyps. However, it is plausible that the presence of fungi could potentially influence the transition from M1 to M2 macrophages.
The involvement of PNAd-expressing vessels in the recruitment of inflammatory cells in nasal polyps remains unclear. However, it is presumed that eosinophils and monocytes may migrate to tissues through PNAd-expressing vessels.12,14,29 In this context, the PNAd/CD34 vessel ratio did not differ between ECRS and AFRS, but the number of M2 macrophages infiltrating nasal polyps was increased by the presence of fungi in the nasal cavity. In our previous study, similar to this finding, the PNAd+/CD34+ vessel ratio did not change in severe ECRS even when complicated by NERD, but the degree of eosinophil infiltration was significantly increased. 13 Exploring the similarities and differences between the pathogenesis of NERD and fungal inflammation may shed light on the function of PNAd-expressing vessels in the recruitment of eosinophils and monocytes in nasal polyps.
There are various options for choosing antibodies that differentiate M2 macrophages from M1 macrophages. While certain studies designate M1 macrophages as CD68-positive/CD163-negative cells and M2 macrophages as CD68-positive/CD163-positive (double positive) cells, 30 in our current study, similar to another report, 18 we defined M2 and M1 macrophages using CD163 and CD68, respectively. Double immunostaining for CD68 and CD163 was performed on the current cases, confirming that a portion of CD163-positive macrophages was doubly positive for CD68 and CD163 (data not shown). Nevertheless, it should be noted that the outcomes of this study might vary if re-evaluated using different combinations of antibodies.
When there is an elevation in fungus-specific IgE levels in cases of CRS, consideration should be given to the possibility of AFRS. 31 Despite Aspergillus spp. being the primary causative agents of AFRS, specific IgE elevations against Alternaria spp. were the most prevalent in the present cohort. 32 Investigating the distinctions in macrophage response to various fungal species present in the nasal cavity is a subject for future studies. The possibility that some of the ECRS cases examined in this study were AFRS cases cannot be ruled out. This is because it is virtually impossible to identify fungi by examining the entire surface of the nasal mucosa. Fungal infection in the nasal cavity induces the infiltration of various inflammatory cells into nasal polyps. The observed increase in M2 macrophages in AFRS nasal polyps is presumed to be induced by fungal infection within the nasal cavity, in comparison with ECRS.
Footnotes
Acknowledgements
We are grateful to Hisataka Kato and Maiko Yamanaka for technical assistance, and Ryan Sullenberger for critical reading of the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
EK performed the immunohistochemistry. EK, MF, and MK designed the study and drafted the manuscript. AM and NY helped digitalization of stained specimens. YM provided surgical samples of AFRS patients. MS and SF revised it critically for important intellectual content. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid for Young Scientists 24K19746 (to EK) and a Grant-in-Aid for Scientific Research (B) 21H02702 (to MK) from the Japan Society for the Promotion of Science.
