Abstract
Skin sensitivity and impaired epidermal barrier function are associated with aging and are at least partly due to increased production of reactive oxygen species (ROS). Transient receptor potential vanilloid1 (TRPV1) is expressed in keratinocytes, fibroblasts, mast cells, and endothelial cells in skin. We investigated in skin biopsies of adult and elderly donors whether TRPV1 expression is involved in the skin aging process. We found that aging skin showed a strongly reduced epidermal thickness, strongly increased oxidative stress, protease expression, and mast cell degranulation and strongly increased TRPV1 expression both in epidermis and dermis. Based on our findings, the aging-related changes observed in the epidermis of the skin level are associated with increased ROS production, and hypothesized alterations in TRPV1 expression are mechanistically linked to this process.
Keywords
Introduction
Skin is the largest organ in the human body, and its main function is to act as a barrier against external stimuli like toxic chemicals, harmful microorganisms, allergens, and ultraviolet light.1–4 It is colonized by more than 100 microbial phylotypes and 1000 different bacterial species; alterations in these communities can cause sensitive skin, a particular condition associated with tightness, pain, itch, inflammation, and impaired epidermal barrier functions.5–8 The perception of skin discomfort increases with age. 9 It has been shown that increased reactive oxygen species (ROS) production may cause or aggravate sensitive skin10–13 by reducing antioxidant activity, damaging proteins, lipids, DNA, and inducing allergic and inflammatory skin diseases, such as atopic dermatitis, urticaria, and psoriasis.14–16 Moreover, high ROS levels in tissues enhance expression of matrix metalloproteinases (MMPs) such as MMP9. 17 Increased proteolytic activity can cause degradation of collagen and other extracellular matrix (ECM) components during skin aging. 18
The transient receptor potential vanilloid1 (TRPV1), initially named capsaicin receptor, or vanilloid 1 receptor, was identified to be expressed on nociceptive sensory nerves endings to mediate sensation of pain and itching.19,20 Pain caused by heat or inflammation and pain caused by cancer is reduced in rodents and humans treated with TRPV1-selective antagonists. Several phase I and II clinical trials of TRPV1 antagonists have been conducted to examine their potential usefulness as analgesics.21–23 David Julius has received the Nobel Prize in Physiology or Medicine in 2021 for the identification of transient receptor potential (TRP) and transient receptor-like protein functions.24,25 The discovery of the molecular transducers underlying the senses of temperature and touch increased greatly our understanding of how the nervous system encodes sensory information. 26
Besides the functional importance of TRPV1 expression in the nervous system, it has been suggested that TRPV1 participates in cutaneous biology as well given its expression in keratinocytes, fibroblasts, mast cells, and endothelial cells.20,27–30 The physiological role of TRPV1 in keratinocytes has been found to be involved in their differentiation and proliferation as well as homeostasis of the epidermal barrier and cutaneous immunological functions.31–33 On the contrary, elevated expr-ession of TRPV1 is associated with atopic dermatitis in mice, and TRPV1 antagonists suppress dermatitis by inhibition of TRPV1 activation.20,34 Basharat et al. 35 showed that TRPV1 is essential for the reduction of ROS production and oxidative stress, pain sensation and inflammation, so TRPV1 has many health benefits. It was also suggested that, whenever TRPV1 is activated, it induces a second message through calcium influx in epidermal keratinocytes. This entry of calcium reduces inflammatory responses, thereby decreasing harmful cutaneous conditions. More recently, Lei et al. 36 reported that TRPV1 signaling ensures skin antiviral competence during wound healing by an unknown mechanism of skin injury activating a TRPV1-mediated innate antiviral immune response. Other TRP channels have been reported to be expressed in nonneuronal cells as well such as TRPM4 in focal adhesions of fibroblasts, 37 TRPV2 in retinal vascular smooth muscle cells, 38 TRPV4 in endothelial cells of the blood brain barrier. 39
To date, TRPV1 functions in nonneuronal cells are not clear. The aim of this study is to verify the presence of TRPV1 in the skin of adults (humans of 50–65 years old) and elderlies (humans of 65 years or older) underlying the possible mechanisms by which this receptor acts on epidermal and dermis cells and other types of cells localized in the skin. We have investigated periocular skin and we evaluated the morphology of skin of the participating donors. First, we analyzed some markers that are involved in oxidative stress, inflammation and protease overexpression, in relation to skin injury. 40 Then, we considered the expression of TRPV1 in skin of the donors for better understanding of the role of this calcium channel protein in skin aging.
Materials and Methods
Human Skin Biopsies
Fourteen cadaveric specimen heads (MedCure, Ams-terdam, the Netherland) were used to obtain the skin biopsies. Specimens (Caucasian; 9 males and 5 fe-males; mean age at death 70 years, ranging from 52 to 83 years) were stored at −20°C, defrosted before the anatomical dissecting session and analyzed at the Anatomical Training Center “Luigi Fabrizio Rodella” of the University of Brescia (Italy). All human cadaveric studies have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Specimens were divided into two groups, adult specimens with an age range between 52 and 65 years (n=7 head specimens) and elderly specimens from donors aged 65 years and older (n=7 head specimens). Medical history revealed no prior head surgical intervention or skin diseases. For all specimens, a 10-mm long × 3-mm wide biopsy was collected from nonlesional skin. The skin biopsies were obtained from the right lateral ocular angle, on the medial end of the zygomatic bone, as shown in Fig. 1A and B. Each skin biopsy was cut sagittally into 2 parts: one half was formalin-fixed (10% in 0.1 M phosphate-buffered solution) for 24 hours, then was rinsed in physiological salt solution, dehydrated in gr-aded ethanol and then embedded in paraffin wax following standard protocols for morphological and immunohistochemical evaluations. 41 The other half of each skin biopsy was frozen and stored at −80°C until homogenization for the detection of ROS and TRPV1 levels by enzyme-linked immunosorbent assay (ELISA) assays.
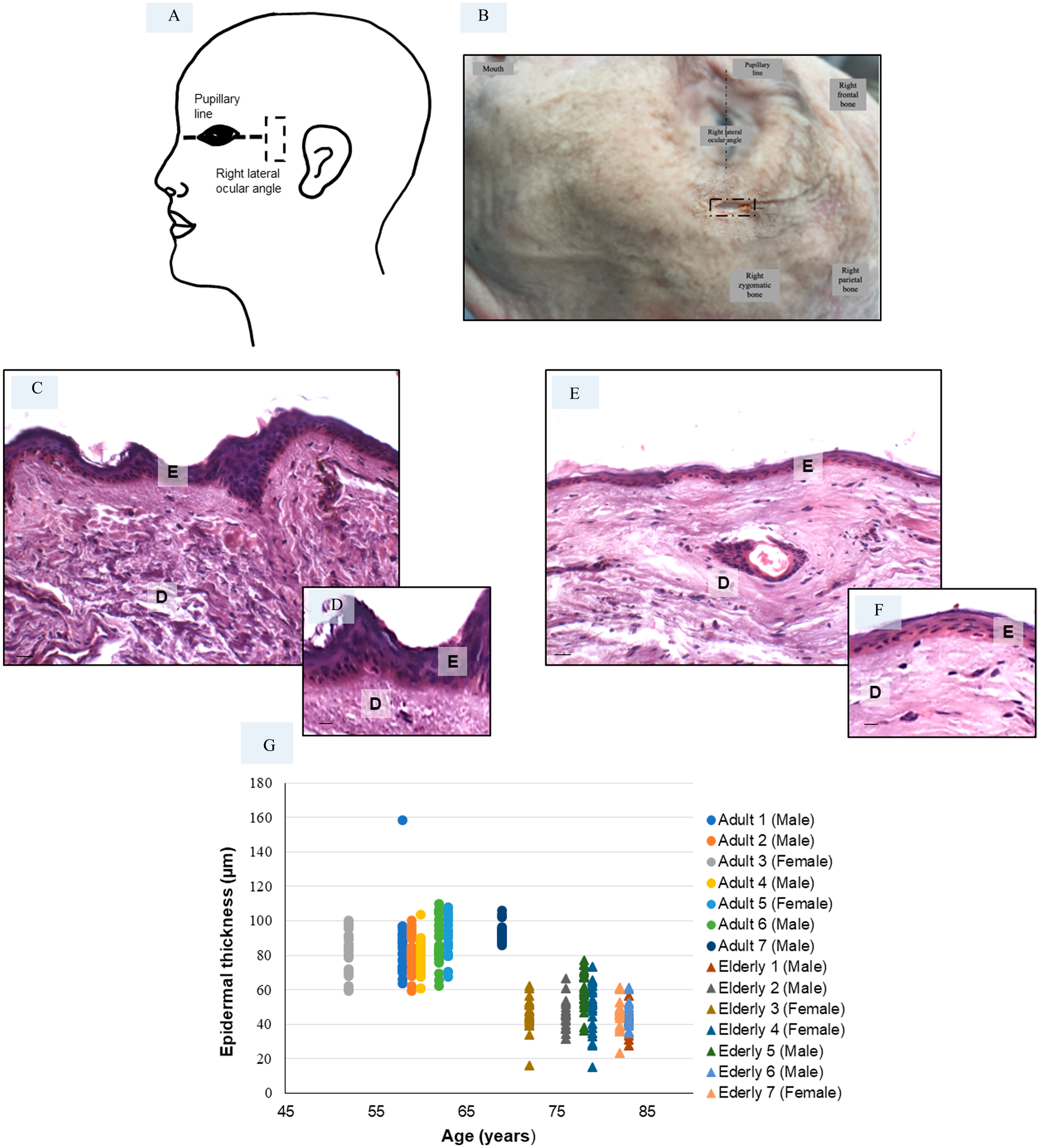
Epidermal thickness measured in skin biopsies—The schematic representation (A) and the photograph (B) represent the incision site (indicated with a dashed black rectangle) where the skin biopsy was taken in all specimens: the right lateral ocular angle has been selected as the site of collection. The other macroscopically recognizable landmarks are indicated in the figure. Haematoxylin and eosin staining of adult specimen (C, D) and elderly specimen (E, F). Bars 20 μm (C, E) and 40 μm (D, F). The plot (G) shows the epidermal thickness distribution of all adult and elderly specimens. Abbreviations: E, epidermis; D, dermis. *p≤0.05 versus adult specimens.
Epidermal Thickness Measurements and Mast Cell Assessment
Serial paraffin sections (5-μm thick) of each sample were cut using a microtome. Alternate sections were deparaffinized, rehydrated, and stained with haematoxylin-eosin to measure epidermal thickness or with toluidine blue for the identification of mast cells and for the visualization of mast cells granules.42,43 The haematoxylin-eosin-stained sections, obtained following standard procedures, 44 were observed using an optical BX50 microscope (Olympus, Hamburg, Germany) at a final magnification of 200×. The thickness of the epidermis of each specimen was calculated in micrometers (μm) by a blind examiner using an image analyzer (Image Pro Premier 9.1; Media Cybernetics, Rockville, MD, USA). The epidermis was measured from the free margin of skin to the dermal papillae and epidermal rete ridge. 45 The analysis was performed in five alternate sections for each skin specimen evaluating five random fields in the same area.
For toluidine blue staining, the skin sections were immersed after deparaffinization and hydration in 0.1% toluidine blue working solution for 5 minutes, washed in distilled water, and dehydrated quickly. 46 The toluidine blue-stained sections were analyzed using an optical BX50 microscope at a final magnification of 400× and 1000×. In detail, an intact mast cell was defined as a mast cell with granulated structures confined within the cell membrane; however, a mast cell was considered to be degranulated when granules were found outside the cell boundaries. 46 The mast cells were identified by their typical metachromasia and showing purple granules in the cytoplasm.42,43,47 The numbers of mast cells were determined by a blind examiner at a magnification of 400× using an optical BX50 microscope. For accuracy of counting, a graticule with 100 fields measuring 0.2 × 0.2 mm (0.04 mm2) surface area was projected over each representative visual field. This counting method was based on the morphometric point-counting technique and the size of overlaying representative visual fields.48–50 At least five representative visual fields of five alternate sections for each skin specimen were analyzed, focusing on the dermis layer. The total mast cells number and the number of intact or degranulated mast cells were expressed as the average number of mast cells per 1 mm2.50,51
Immunohistochemical Evaluation
Alternate sections were deparaffinised, rehydrated, and then were incubated in 3% hydrogen peroxide for 10 minutes at room temperature. To demonstrate the specificity of antibodies, we used a preadbsorption test: 1% bovine serum albumin and 0.05% Tween 20 for 1 hour at room temperature. 52 Skin sections were incubated for 45 minutes at 37°C and for 1 hour at room temperature with the following primary antibodies: rabbit polyclonal anti-TRPV1 (working concentration 1 µg/ml; diluted 1:350; Abcam, Cambrige, UK), 53 rabbit polyclonal anti-MMP9 (working concentration 1 µg/ml; diluted 1:400; Abbiotec, San Diego, CA, USA), rabbit polyclonal anticyclooxygenase-2 (COX-2) (working concentration 0.5 µg/ml; diluted 1:400; Cayman Chemical, Ann Arbor, MI, USA), 54 and mouse monoclonal antisuperoxide dismutase-1(SOD-1) (working concentration 2 µg/ml; diluted 1:100; Santa Cruz Biotechnology, Dallas, TX, USA).55–57 Then, the sections were incubated for 1 hour with specific biotinylated secondary antibodies (Vector Laboratories, Newark, CA, USA) and subsequently conjugated with avidin-biotin peroxidase complex (Vector Laboratories, Burlingame, CA, USA). The reaction products were visualized using 0.33% hydrogen peroxide and 0.05% 3,3ʹ-diaminobenzidine tetrahydrochloride, as chro-mogen (Sigma, St. Louis, MO, USA). Finally, the skin sections were counterstained with haematoxylin, dehydrated and mounted using dibutylphthalate polystyrene xylene. 58 Matched-concentration IgG (Vector Laboratories, Burlingame, CA, USA) isotype control instead of the primary antibody was tested during validation of the antibodies (negative controls).
The immunostained sections were observed with an optical BX50 microscope at a final magnification of 200× and 400×. Five random fields of a total of five nonconsecutive sections per skin biopsy were analyzed by a blind examiner, and the immunostaining for each primary antibody was calculated.
We quantified the positive immunostaining for each primary antibody investigated in this study as pixels by using a computer-assisted image analysis system (Image Pro Premier 9.1). 59 We performed white balancing and background subtraction for all evaluated representative visual fields before applying a pixel quantification algorithm to calculate the positive diaminobenzidine tetrahydrochloride-stained pixels per area.60,61
The immunostaining for each primary antibody was expressed as arbitrary units and their mean values are presented as bar-whisker graphs for each evaluated parameter.
ROS and TRPV1 ELISA Assays
The half of each biopsy stored at −80°C was adequately processed for the quantitative measurement of ROS and TRPV1 levels using specific sandwich ELISA kits (ROS ELISA kit of Novatein Biosciences, Woburn, MA, USA; TRPV1 ELISA kit of MyBioSource, San Diego, CA, USA), according to the manufacturer’s instructions. Samples were processed in duplicate and the optical density, for both ELISA kits, was measured at 450 nm. A small fragment of each skin biopsy was separated and homogenized with fresh lysis buffer in a glass homogenizer on ice. The resulting suspension was sonicated with an ultrasonic cell disrupter until the solution appeared to be clear. Then, the homogenates were centrifuged, the pellets were discarded, and the supernatants were used for ROS and TRPV1 analysis.
The used ROS ELISA assay evaluated reactive chemical species containing oxygen (including peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen), and the ELISA kit recognizes both natural and recombinant human ROS. The sensitivity or minimum detectable dose of human ROS was determined to be 20 pg/ml.
The ROS and TRPV1 skin levels are presented as pg/ml for the ROS assay and ng/ml for the TRPV1 assay.
Statistical Analysis
Results were expressed as the mean ± standard deviation. Data for multiple variable comparisons were analyzed by one-way analysis of variance (analysis of variance–corrected Bonferroni test). P values ≤0.05 were considered significant for all statistical analysis of this study.
Results
Epidermal Thickness
The morphometrical analyses of the right lateral ocular angle skin biopsies (Fig. 1A and B) revealed a significant difference between the periocular epidermal thickness of adult (81 ± 13 µm) versus elderly (48 ± 12 µm) donors. Interindividual differences among the specimens analyzed in each group were observed, but differences between donors were much smaller than the aging-related differences. In Fig. 1, the periocular skin of a representative adult specimen (Fig. 1C and D) and a representative elderly specimen (Fig. 1E and F) are shown, and the morphometric evaluation of epidermal thickness is shown in Fig. 1G.
Oxidative Stress Evaluation in Skin
In order to investigate whether oxidative stress affect mast cell degranulation in skin aging-related alterations and whether endogenous antioxidants are sufficient to counteract skin cell stress injury, we evaluated levels of both ROS and SOD-1, a ubiquitously expressed antioxidant enzyme. 62 We observed that skin ROS levels were more pronounced in elderly specimens when compared to adult specimens (Fig. 2A). Furthermore, cutaneous expression of SOD-1 was weak in adult specimens (Fig. 2B and C) and significantly higher in elderly specimens (Fig. 2D and E). The SOD-1 immunopositivity was observed in the cytoplasm of cells of epidermis and dermis. The negative controls of SOD-1 immunohistochemistry were similar in both adult and elderly skin samples, and Fig. 2F shows a representative elderly skin specimens. These observations were confirmed by the immunomorphometric analysis, as summarized in Fig. 2G.
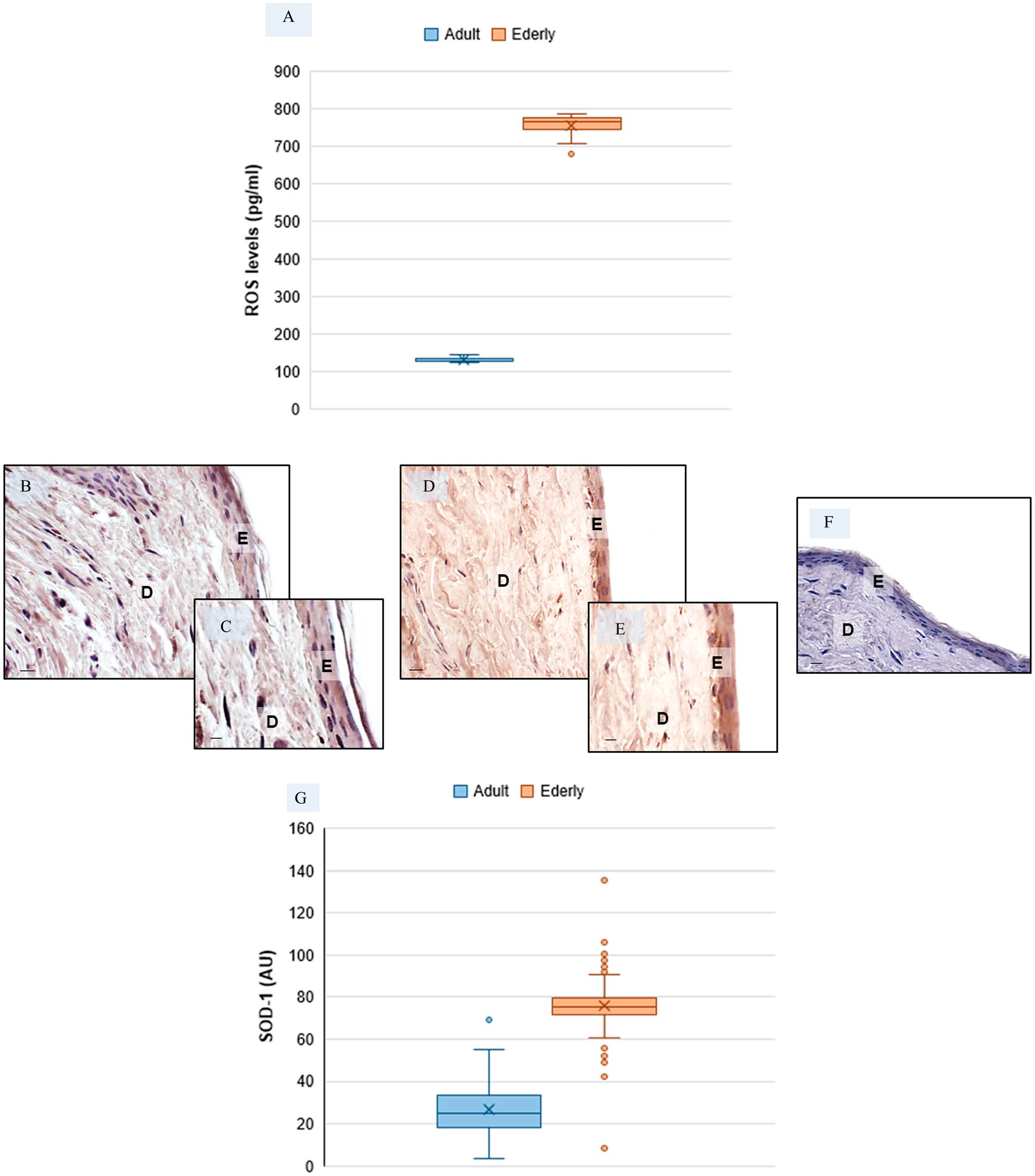
Evaluation of oxidative stress in skin biopsies—Skin reactive oxygen species (ROS) level evaluation in adult and elderly specimens (a). Representative skin photomicrographs of superoxide dismutase-1 (SOD-1) immunostaining of adult specimens (B, C) and of elderly specimens (D, E). SOD-1 negative control—elderly skin specimens (f). Bars 40 μm (B, D, F) and 80 μm (C, E). The plot (G) summarizes SOD-1 immunomorphometric measurement. Abbreviations: E, epidermis; D, dermis; AU, arbitrary units. *p≤0.05 versus adult specimens.
MMP9 Expression in Human Skin
To stress the role of ROS in skin aging, we also evaluated MMP9 expression, which is a major protease involved in degradation of collagen and other ECM components producing cutaneous injury.
The immunohistochemical analysis showed that the cutaneous expression of MMP9 is rather weak in adult specimens (Fig. 3A to D) and significantly higher in the dermis of elderly donors (Fig. 3E to H). The negative controls of MMP9 immunohistochemistry were similar in both adult and elderly skin specimens, and Fig. 3I shows a representative elderly skin specimens. The MMP9 immunopositivity was observed in the cytoplasm of cells. Figure 3J shows the immunomorphometric analysis of MMP9.
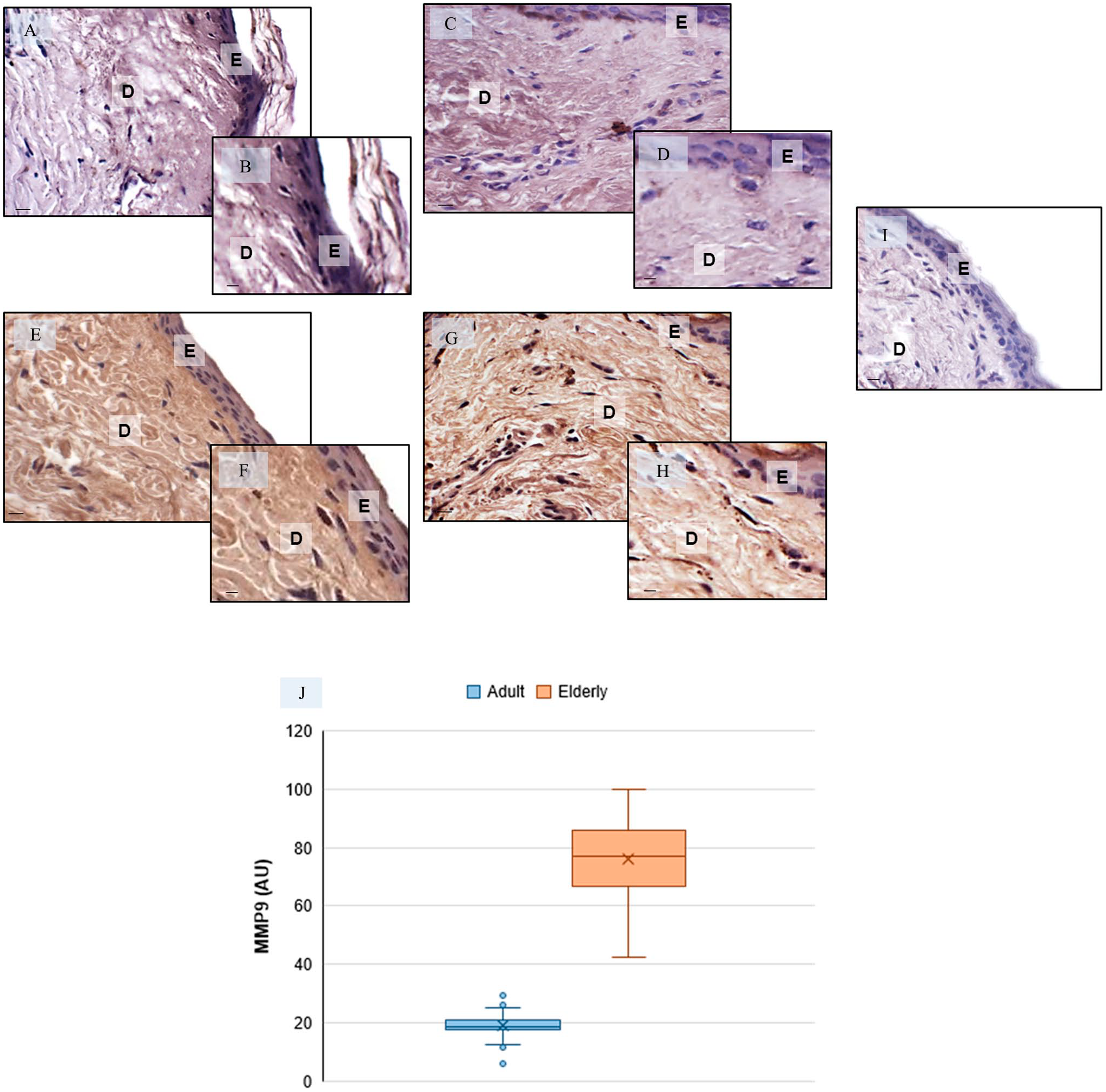
Metalloproteinase9 (MMP9) skin evaluation—Representative skin photomicrographs of MMP9 immunostaining of adult specimens (A–D) and elderly specimens (E–H). MMP9-negative control—elderly skin specimens (I). Bars 40 µm (A, C, E, G, I) and 80 µm (B, D, F, H). The plot (J) summarizes the MMP9 immunomorphometrics. Abbreviations: E, epidermis; D, dermis; AU, arbitrary units. *p≤0.05 versus adult specimens.
TRPV1 Expression in Human Skin
In contrast to its role in pain, itch, or heat sensation in sensory nerves, the role of TRPV1 in skin has thus far rarely been investigated. Cutaneous TRPV1 expression is affected by aging as shown in Fig. 4. In detail, TRPV1 expression was very weak in the epidermis and dermis in adult specimens (Fig. 4A to D) when compared to that in elderly specimens which expressed TRPV1 strongly in the epidermis and moderately in the dermis (Fig. 4E to H). The negative controls of TRPV1 immunohistochemistry were similar in both adult and elderly skin specimens, and Fig. 4I shows a control reaction in a representative elderly skin specimens. These observations were confirmed by the immunomorphometric analysis as summarized in Fig. 4J. Furthermore, the TRPV1 ELISA evaluation, shown in Fig. 4K, supported the above-reported data.
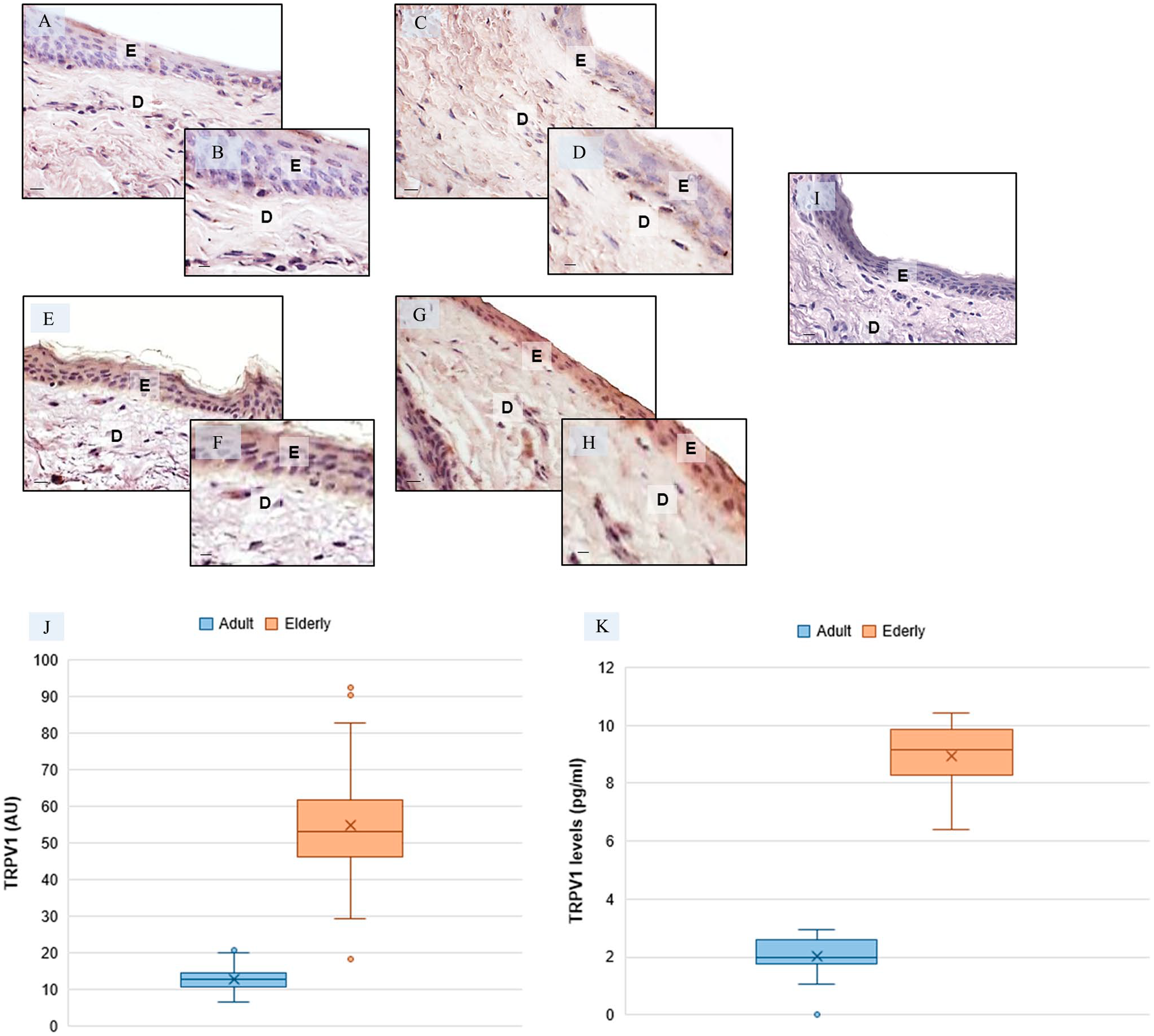
Transient receptor potential vanilloid1 (TRPV1) skin evaluation—Representative skin photomicrographs of TRPV1 immunostaining in adult specimens (A–D) and in elderly specimens (E–H). TRPV1-negative control—elderly skin specimens (I). Bars 40 µm (A, C, E, G, I) and 80 µm (B, D, F, H). The plots summarize the TRPV1 immunomorphometric measurement (J) and the skin TRPV1 ELISA evaluation (K). Abbreviations: E, epidermis; D, dermis; AU, arbitrary units. *p≤0.05 versus adult specimens.
Mast Cells Assessment in Skin
The cytoplasm of intact mast cells is loaded with metachromatic purple granules43,47 and were found in the dermis of all skin specimens. The adult specimens contained mainly intact mast cells and only few degranulated cells (Fig. 5A and B), whereas degranulated mast cells were observed in the dermis level of elderly specimens (Fig. 5C and D). The background connective tissue was stained in shades of blue. Total numbers of mast cells (both intact and degranulated forms) were higher in elderly skin specimens as compared to adult skin specimens. In detail, numbers of degranulated mast cells were significantly higher in elderly samples (13.2 ± 3 mast cell/field) than in adult skin specimens (2.6 ± 2.0 mast cell/field). In contrast, number of intact mast cells were not much different in dermis of adult specimens (1.3 ± 1.0 mast cell/field) compared to elderly specimens (1.6 ± 1.0 mast cell/field). The above-reported observations are summarized in Fig. 5E and F and underline that mast cells degranulation is significantly correlated with age.
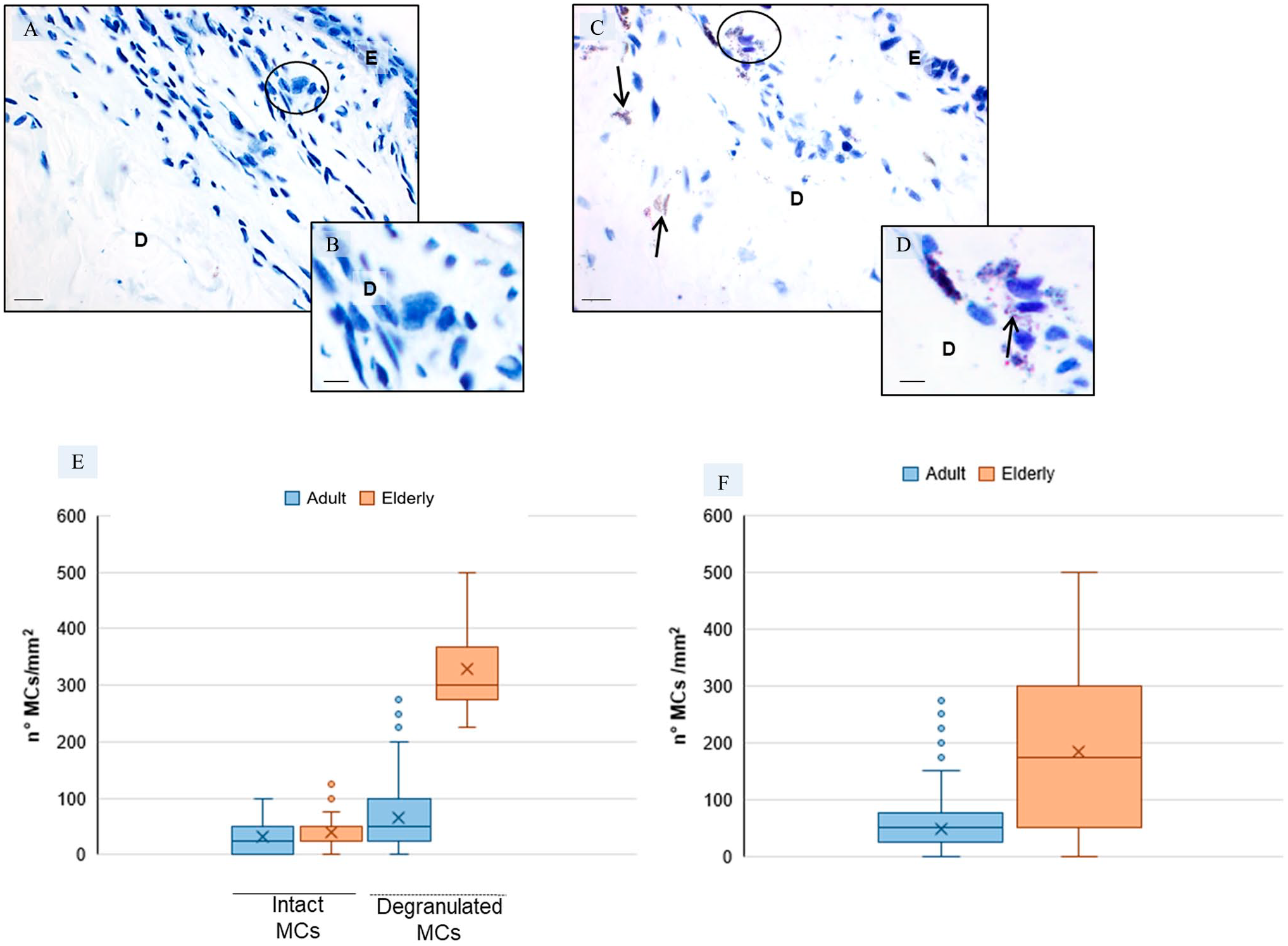
Mast cell assessment in the dermis—Representative skin photomicrographs after toluidine blue staining of adult specimens (A, B) and elderly specimens (C, D). The black circles indicate intact (A) and degranulated (C) mast cells; the arrows show the mast cells granules. Bars 40 µm (A, C) and 80 µm (B,D). *p≤0.05 versus adult specimens. Plot (E) summarizes the number of intact and degranulated mast cells per mm2 in adult and elderly specimens and plot (F) summarizes the total number of mast cells (intact plus degranulated mast cells) per mm2 in adult and elderly specimens. Abbreviations: E, epidermis; D, dermis; MCs, mast cells.
COX-2 Expression in Human Skin
Besides regulation of MMPs, the expression of pro-inflammatory markers is upregulated during aging. 63 Therefore, we investigated the expression of COX-2, a rate-limiting inducible enzyme in the production of prostaglandins. 64 COX-2 was strongly expressed in dermis of periocular skin of the elderly specimens as compared with adult specimens, as is shown in Fig. 6A to H. The negative controls of COX-2 immunohistochemistry were similar in both adult and elderly skin specimens, and Fig. 6I shows a representative control adult skin specimens. These observations were confirmed by the immunomorphometric analysis that is summarized in Fig. 6J.
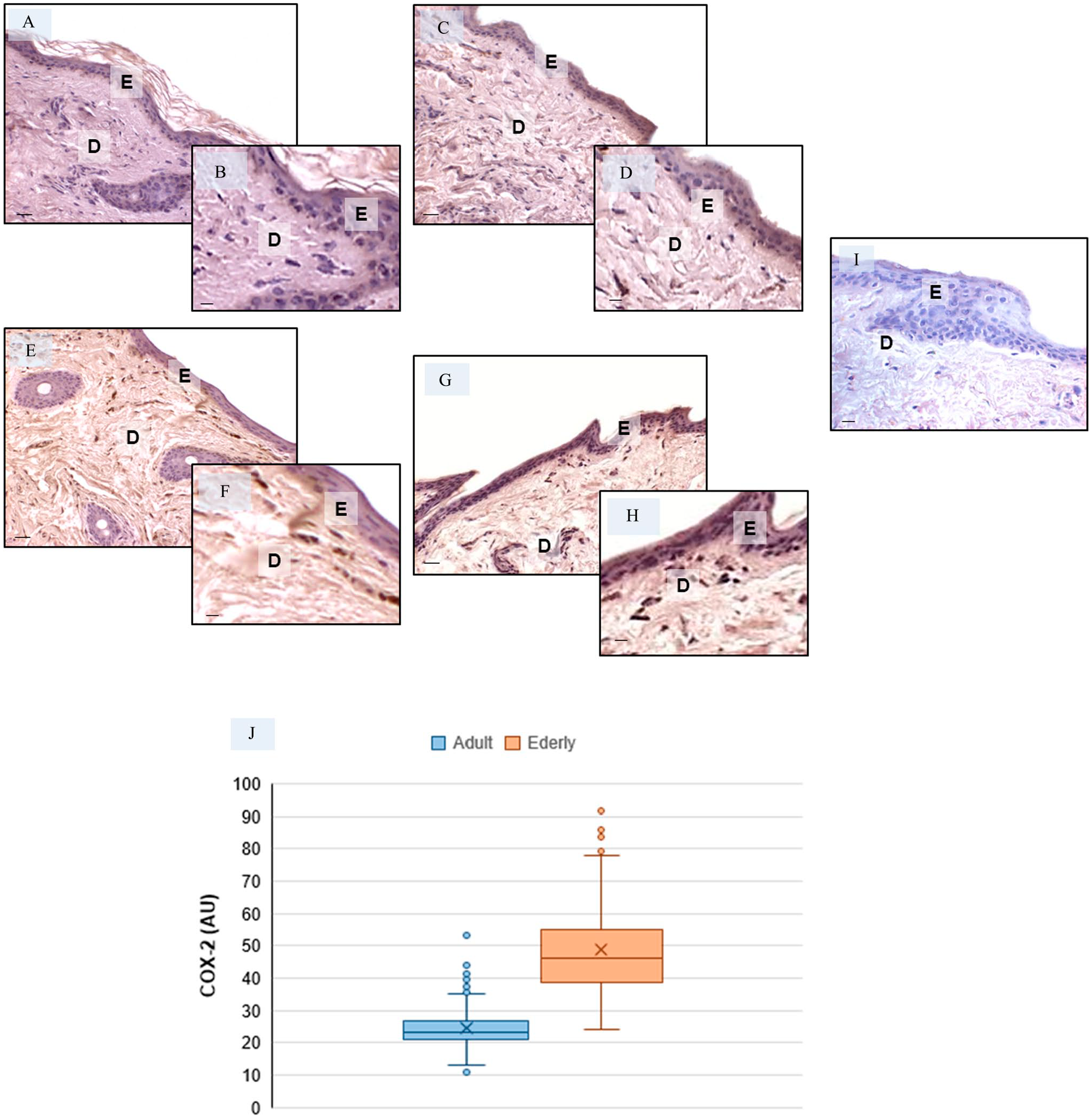
Evaluation of COX-2 expression in skin—Representative skin photomicrographs of COX-2 immunostaining in adult specimens (A–D) and elderly specimens (E–H). COX-2-negative control—adult skin specimens (I). Bars: Bars 60 µm (A, C, E, G, I) and 90 µm (B, D, F, H). The plot (J) summarizes the COX-2 immunomorphometric measurements. Abbreviations: E, epidermis; D, dermis; AU, arbitrary units. *p≤0.05 versus adult specimens.
Discussion
The skin is the interface between body and environmental factors, and it acts as the first line of defense against many insults, including microbes.36,65 It plays a fundamental role in innate antimicrobial host immunity. 66 Moreover, with the aging process of the skin, the three layers of skin (epidermis, dermis, and hypodermis) undergo degenerative changes, and alterations of the dermis are the most obvious.67–70 The hypodermis is the deepest layer of the skin and is present between dermis and underlying tissues underneath such as muscle. It consists mainly of adipocytes and ECM; therefore, we did not include hypodermis in this study. Regardless of the type of skin, wrinkles and reduced elasticity are typical phenomena of skin aging as is also atrophy of the dermis.71–73 These changes cause modifications of the content of bacterial species in the skin leading to a vicious circle with increased barrier destruction.34,74,75 Moreover, it is known that skin changes are also due to ROS production leading to elevated oxidative stress at the cellular level. ROS have important targets in many cell types affecting signaling events prior to the effects of other substances or ion mobilization. 14
With respect to skin morphology during aging, we observed:—a significant reduction in epidermal thickness;—increased ROS levels and increased SOD-1, MMP9, and COX-2 expression.
These first findings allowed us to continue our study on TRPV1 expression showing a clear increase in TRPV1 immunopositivity in keratinocytes during aging. After this assessment, we investigated whether TRPV1 is also expressed in other cell types in the dermis and whether expression is elevated in elderly donors.
TRPV1 immunopositivity increased in keratinocytes, fibroblasts, and mast cells in elderly donors. It is known that TRPV1 is a nonselective cation channel with a prevalence of permeability to divalent and monovalent cations, such as calcium ions (Ca2+). These ionic channels mediate the receptor-induced response of a cell to external stimuli which can increase or decrease the selective permeability of TRPV1 channels to ions altering cell membrane potential leading to depolarization.76–78 The influx of Ca2+ could modify the biology of dermis cells, in the same way as occurs in fibroblasts and mast cells. 77 Moreover, Ca2+-influx can cause their degranulation.28,79 Interestingly, we observed an incr-eased number of mast cells in the dermis of elderly donors. These cells were prevalently degranulated in comparison with those observed in adult donors. So, we postulated that the degranulated cells are at least partially responsible for the inflammation status in skin cells in elderly donors, as determined as increased COX-2 expression. Our findings are in agreement with the data reported by Lee et al. 77 indicating that TRPV1 increases COX-2 expression. It is known that COX-2 is an important mediator of inflammation linked to changes in the Ca2+-influx in the cells synthetizing prostaglandins and lipid mediators. 80
In conclusion, we propose that the changes in skin during aging and in skin diseases, caused by, among others, ROS production, cause modifications in TRPV1 expression in keratinocytes, fibroblasts, and mast cells, leading to a negative feedback loop among these cells and their products. We hypothesize that all these phenomena contribute to the development of sensitive skin, itching, and related conditions. The above-reported hypothesis is summarized in Fig. 7.
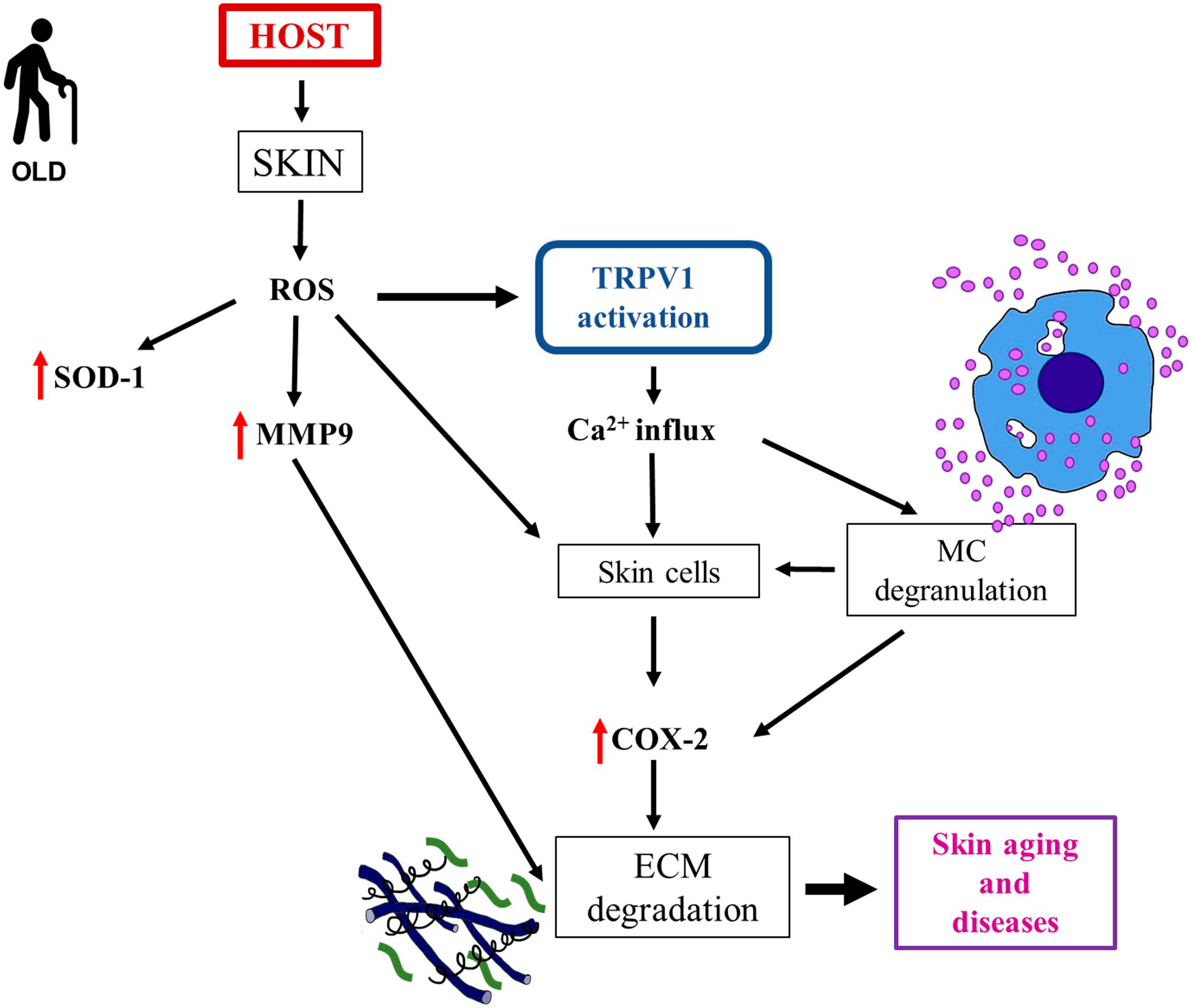
Aging-related injury in skin—Schematic representation of how host, environmental, and extrinsic factors act on the skin introducing aging-related alterations and leading to possible dermatological diseases. Superoxide dismutase-1 (SOD-1) increases its expression in both dermis and epidermis; it acts as an antioxidant molecule by playing a protective role against ROS production. Abbreviations: MC, mast cell; ECM, extracellular matrix; TRPV1, transient receptor potential vanilloid1; ROS, reactive oxygen species; SOD-1, superoxide dismutase1; MMP9, metalloproteinase9; COX-2, cyclooxygenase-2; Ca2+, calcium2+.
This study has limitations that must be considered. Traditional small-sample anatomical dissection studies have inherent limitations insofar as they are not representative of skin all over the body. The objective study of large skin areas and samples of different face areas is required to effectively confirm the data obtained in this study. Moreover, the authors are aware that this research requires a larger sample size, and inclusion of more age categories, gender, and exposition or not to the sun.
Despite these limitations, we are convinced that this study can open new scientific research avenues to preserve barrier epithelial integrity during aging and in dermatological diseases.
Footnotes
Acknowledgements
The authors sincerely express their highest gratitude to the donors of their bodies to science. They thank Giuliani S.p.A. (Milan, Italy) for the precious support to this research project. Moreover, they thank Professor Borsani Elisa from the University of Brescia (Italy) for the essential support in obtaining skin biopsies.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: conception and design of the work (RR, FR); acquisition, analysis, and interpretation of data (CF, GF, MG, DP); drafting and revising the manuscript for important intellectual content (GF, RR); editing and the revision of the manuscript (CJFvN); and all authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Giuliani S.p.A. (Milan, Italy). CJFvN was supported by the Slovenian Research Age-ncy (ARRS P1-0245 and J3-2526 research grants).
