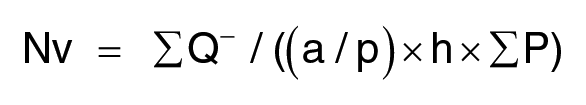Abstract
Gastrointestinal symptoms are common health problems found during aging and neurodegenerative diseases. Trimethyltin-induced rat is known as an animal model of hippocampal degeneration with no data on enteric neurodegeneration. This study aimed to investigate the effect of trimethyltin (TMT) induction on the gastrointestinal tract. A 28-day animal study with male Sprague–Dawley rats (3 months old, 150–200 g) given a single TMT injection (8 mg/kg body weight, intraperitoneal) was conducted. The number of neurons in the colonic myenteric plexus was measured using stereological estimation. Histological scoring of colon inflammation, immunohistochemistry of tumor necrosis factor-α (TNF-α), and quantitative PCR were conducted. This study showed neuronal loss in the colonic myenteric plexus of TMT-induced rat model of neurodegeneration. Minor colon inflammation characterized by inflammatory cell infiltration and slightly higher expression of TNF-α in the colon mucosa were observed in the TMT-induced rat. However, the gut microbiota composition of the TMT-induced rat was not different from that of the control rats. This study demonstrates that TMT induces colonic myenteric plexus neurodegeneration and minor colon inflammation, which suggests the potential of this animal model to elucidate the communication between the gastrointestinal tract and central nervous system in neurodegenerative diseases.
Keywords
Introduction
Aging is characterized by the progressive accumulation of cellular and molecular damages, which leads to impaired physiological function. Thus, aging is a significant risk factor for health problems, including neurodegenerative diseases such as Alzheimer’s and Parkinson’s diseases. In recent years, much evidence has shown that patients with neurodegenerative diseases have concurrent gastrointestinal problems.1,2 Constipation is one of the most frequent gastrointestinal problems observed in patients with neurodegenerative diseases and is considered an early sign of Parkinson’s 3 and Alzheimer’s diseases. 4 Colonoscopy of Parkinson’s disease patients observed colon inflammation and increased mucosal permeability with changes in microbiota composition. 5 Therefore, gastrointestinal problems, particularly those involving the colon, may play a role in neurodegenerative diseases’ pathogenesis and disease course.1,6
The gastrointestinal problems observed in patients with neurodegenerative diseases may be caused by the degeneration of the enteric nervous system (ENS). 7 Bidirectional communication between ENS and the central nervous system (CNS) has been mentioned as a possible passage for transmitting molecules such as prions from the gastrointestinal tract to the brain. 8 This supports the theory that in some cases of neurodegenerative disease, the pathological process starts in the gastrointestinal tracts and is subsequently transmitted to the CNS, possibly through the ENS. 9
Various factors may lead to ENS degeneration, including gastrointestinal inflammation. Tumor necrosis factor-α (TNF-α), a proinflammatory cytokine produced during inflammation, has a neurotoxic effect and induces neuronal apoptosis.10,11 Reactive oxygen species (ROS) produced during inflammation mediate neuronal death. Indeed, several findings observed inflammation and oxidative stress in the intestine of Parkinson’s disease patients. 12 Gastrointestinal inflammation is also associated with an altered composition of gut microbiota. Increasing evidence has shown gut dysbiosis in patients with Parkinson’s disease 13 or Alzheimer’s disease. 14
Animal models are essential in understanding the underlying communication between the gastrointestinal tract and CNS in neurodegenerative disease and aging. In this study, we used rats injected intraperitoneally with a single dose of trimethyltin (TMT; 8 mg/kg body weight) and terminated on day 28 after induction as an animal model of neurodegeneration. 15 TMT is an organotin compound that induces the loss of hippocampal neurons in the CA1 and CA3/CA4 subfield16,17 and the dentate gyrus granule cells.10,18 This model of hippocampal neurodegeneration has been utilized because of its short duration and simple technique. 19 Thus, TMT-induced rat is considered a valuable tool for studying the molecular mechanisms of neurodegenerative diseases. However, no findings have been reported on the effect of TMT on ENS and its possible involvement in the CNS degeneration observed in the TMT-induced rat model of neurodegeneration. Therefore, this study aimed to elucidate whether TMT induces ENS degeneration and colon inflammation and alters gut microbiota composition, all of which may contribute to the CNS degeneration observed in this animal model.
Materials and Methods
Animals
Three-month-old male Sprague–Dawley rats (n=12) weighing 150–200 g were purchased from the Faculty of Veterinary Medicine Institut Pertanian Bogor, Indonesia. All rats were maintained at the animal facility of the Faculty of Medicine, Public Health and Nursing Universitas Gadjah Mada, on a 12:12 hr light–dark cycle. Standard chow diet and drinking water were provided ad libitum. The rats were randomly divided into two groups: the control group and the TMT group. Rats in the TMT group were given a single intraperitoneal injection of TMT (8 mg/kg body weight).15,19 The animals were excluded if they died prematurely. All animal experiments followed a protocol approved by the Ethics Committee of the Faculty of Medicine, Public Health and Nursing, Universitas Gadjah Mada (reference number: KE/FK/0897/EC/2019). The procedures complied with the Animal Research: Reporting of In Vivo Experiments guidelines. All efforts were made to minimize potential animal distress.
Tissue Processing and Hematoxylin Eosin Staining
All rats were sacrificed on day 28 after TMT injection.15,19 Under ketamine anesthesia (100 mg/kg body weight, intramuscular), transcardial perfusion with phosphate-buffered saline (PBS) was performed. The whole colon was excised, and its length was measured (Fig. 1A) before it was cut into 1-cm-long slabs. Colon slabs were sampled in a systematic uniform random sampling manner yielding 6–10 slabs. After a longitudinal cut was made, the colon slabs were put on a filter paper with a marker to indicate orientation (Fig. 1B) and then fixed with 4% paraformaldehyde in PBS. Afterward, the colon segments were processed into 2–3 paraffin blocks per rat and sectioned at a thickness of 3 μm. Two consecutive sections were prepared as a pair of physical disector. The sections were stained with hematoxylin and eosin (Fig. 1C). Digital photographs were obtained using an Olympus CX33 microscope equipped with a camera (Miconos; Yogyakarta, Indonesia).
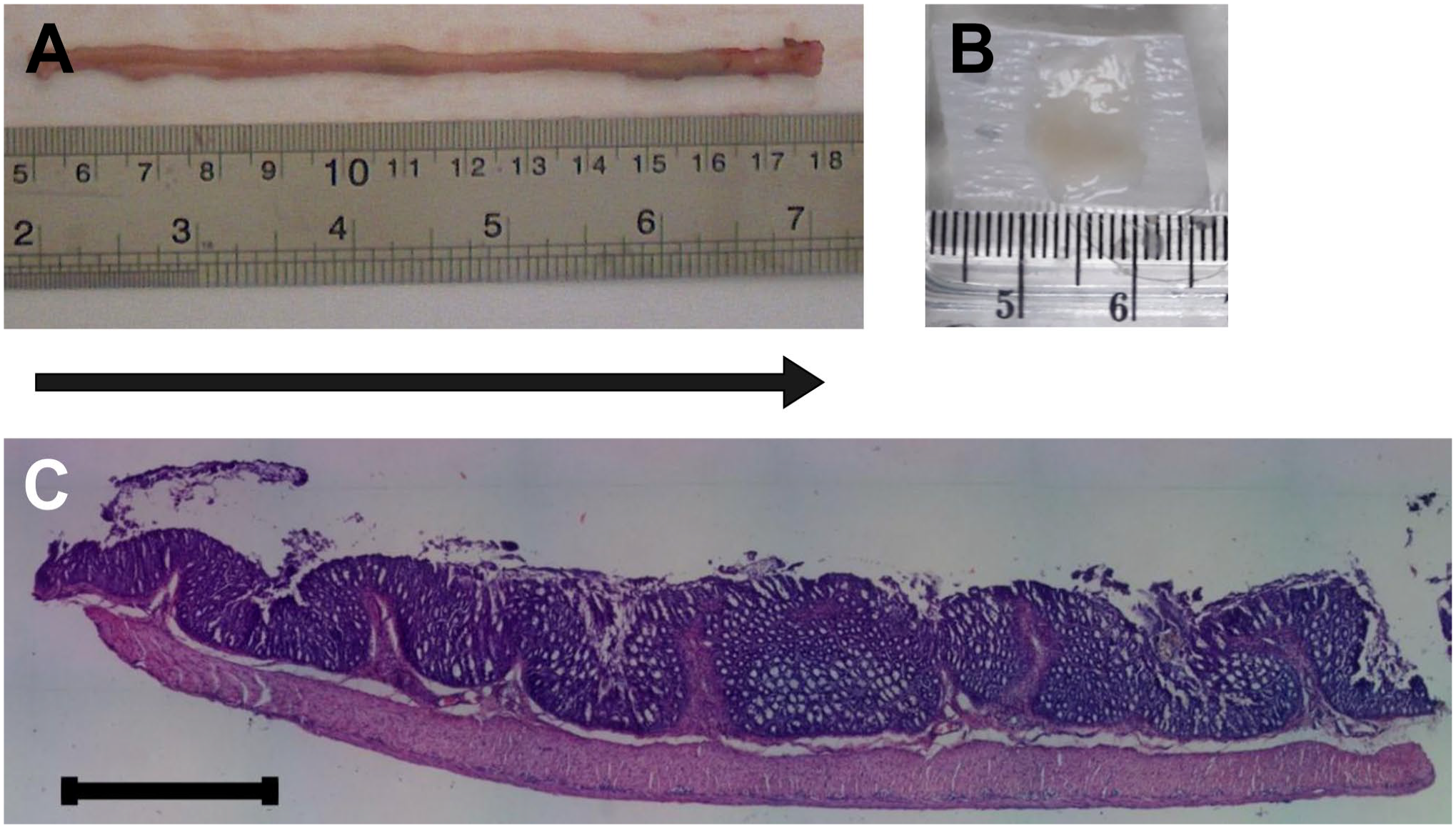
Tissue sampling and hematoxylin and eosin staining. (A) Colon from one rat taken after dissection. The lumen was cleaned, and the whole colon was weighed. The colon was cut into 1-cm slabs that were subsequently sampled in a systematic uniform random sampling manner. (B) One slab of the colon on a piece of paper. The colon slabs were put on paper, and dots were made to indicate the proximal part of the colon. Three to four slabs were embedded in one paraffin block. (C) Representative image of one section of colon stained with hematoxylin and eosin. Arrow: colon axis in Figs. 1A to C. Scale bar: 100 μm.
Estimation of the Total Number of Colonic Myenteric Plexus Neuron
Counting neurons in the myenteric plexus in rat colon was performed using the particle density (Nv) × reference volume (Vref) method in physical disectors with a 3-µm disector height. The myenteric plexus was identified between two layers of muscle layer as an area consisting of a group of neurons. Only one third of the area was photographed on both disector pairs and subsequently used for neuron counting assisted by Image-J FIJI software. 20
The nucleus was used as a counting unit. The neuron was counted if observed in the reference section and not visible in the look-up section (Fig. 2). Back and forth two-way counting was performed when the counted neurons in one rat using one-way counting were less than 100. The sample volume was estimated by point counting with a/p = 0.16 µm2.
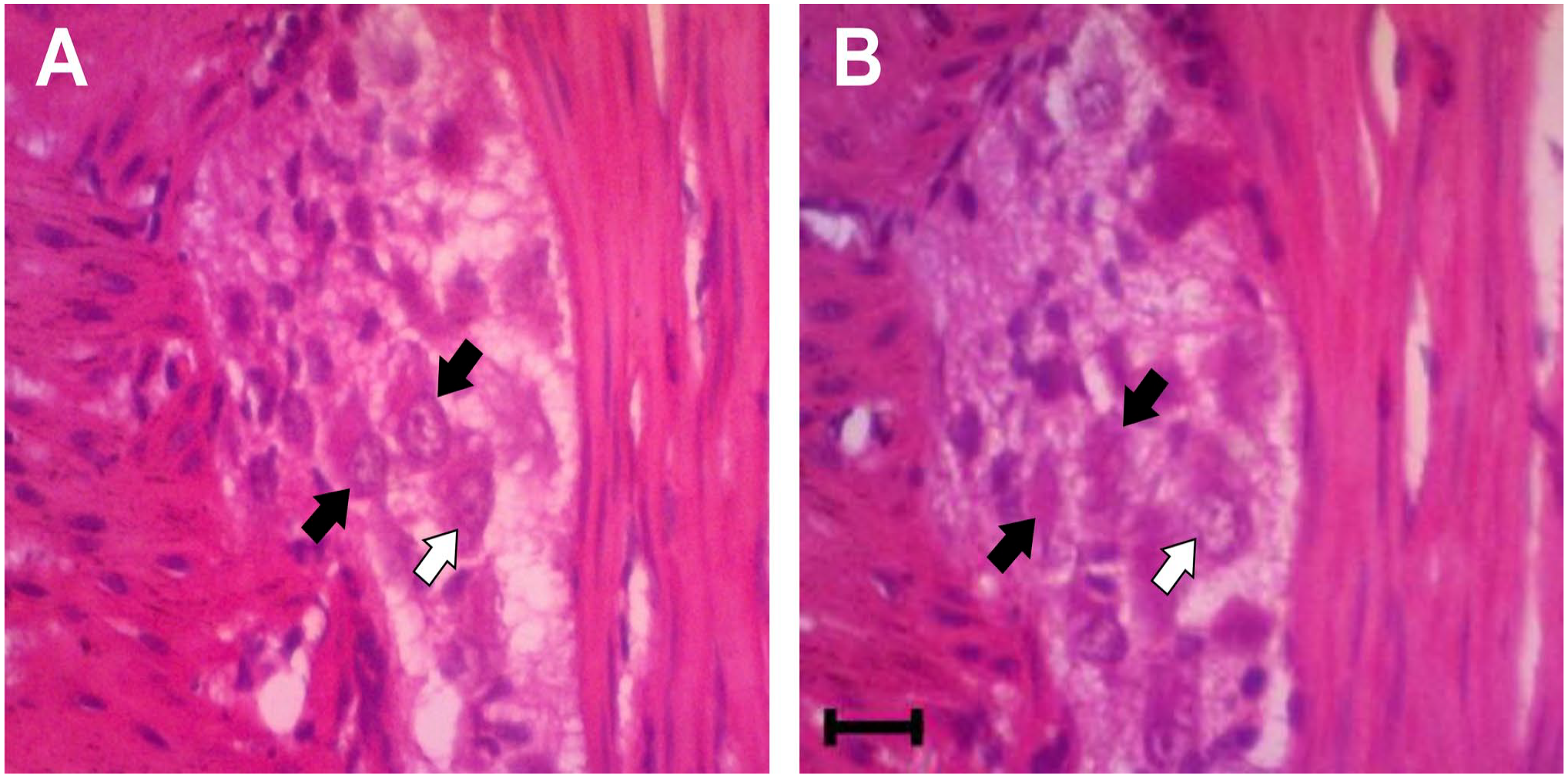
The physical disector principle is used in counting the colonic myenteric plexus neuron. (A) Example of the reference section. (B) Example of the look-up section. The nucleus was used as a counting unit. Black arrows indicated the counted neurons as their nuclei were visible in the reference section but not visible in the look-up section. The white arrow indicated the uncounted neuron, as its nucleus was visible in both the reference and look up sections. Scale bar: 20 μm.
Vref was estimated by obtaining a fraction of the area with myenteric plexus (a/p of 0.0009 µm2) divided by the whole area of the colon (a/p of 0.04 µm2). The weight of each colon was used to obtain a Vref of the whole colon. The colon volume is assumed based on a colon weight of 1 mg = volume of 1 mm3. 21 The number of neurons in a defined sampling volume was counted, and the following formula 22 calculated the Nv.
∑Q– was the number of counted neurons, a/p was the area of the counting frame, h was the distance between the two sections, and ∑P was the number of points hitting the reference space. The total number of colonic myenteric plexus neurons was finally determined by multiplying Nv with the total Vref.
The efficiency and accuracy of the stereological method were measured by the coefficient of error. A stereological method was considered optimum when the value of observed coefficient of error (OCE) < 10%, OCE = ½ observed coefficient of variation (OCV), and 0.2 < OCE 2 /OCV 2 < 0.5.
Inflammation Scoring and Histological Analysis
Inflammation scores were determined by two blinded observers based on previously used criteria. 23 Briefly, the following criteria were used for calculation: (1) depth of inflammation (0 = none, 1 = mucosa, 2 = submucosa, 3 = transmural); (2) crypt damage (0 = none, 1 = basal 1/3 damage, 2 = basal 2/3 damage, 3 = crypt loss, surface epithelium present, 4 = crypt and surface epithelium lost); (3) injury extent (0 = 0%, 1 = 1–25% 2 = 26–50%, 3 = 51–75%, 4 = 76–100%); (4) inflammatory cell infiltration (0 = none; 1 = light, 2 = medium, 3 = severe). The inflammation score was derived from the sum of the score for each criterion above.
Immunohistochemistry
Immunohistochemical staining of colon tissues was conducted using the Abcam Mouse and Rabbit Specific HRP/DAB IHC Detection Kit (ab236466, Abcam; Waltham, Massachusetts) according to the manufacturer’s instructions. Briefly, tissue sections were deparaffinized and rehydrated through a series of descending ethanol solutions. Afterward, heat-induced antigen retrieval within the Tris-EDTA buffer using microwaves for 5 min was performed. After that, the tissue sections were incubated overnight at 4C with rabbit polyclonal anti-TNF-α antibody (1:200, bs-2081R, Bioss; Woburn, Massachusetts). After washing in 0.5-M Tris buffer, tissue sections were incubated at room temperature for 15 min with goat antirabbit Horseradish Peroxidase (HRP) conjugate. Tissue sections were then incubated with 3,3'-diaminobenzidine solution and counterstained with Mayer’s hematoxylin according to the manufacturer’s instruction. To demonstrate the immunoreaction specificity, negative and positive controls were performed for all immunoreactions. The adjacent tissue section was used for negative controls with the primary antibody omitted. Spleen tissue sections were used as the positive control.
DNA Extraction and Real-time Quantitative PCR of Cecal Content
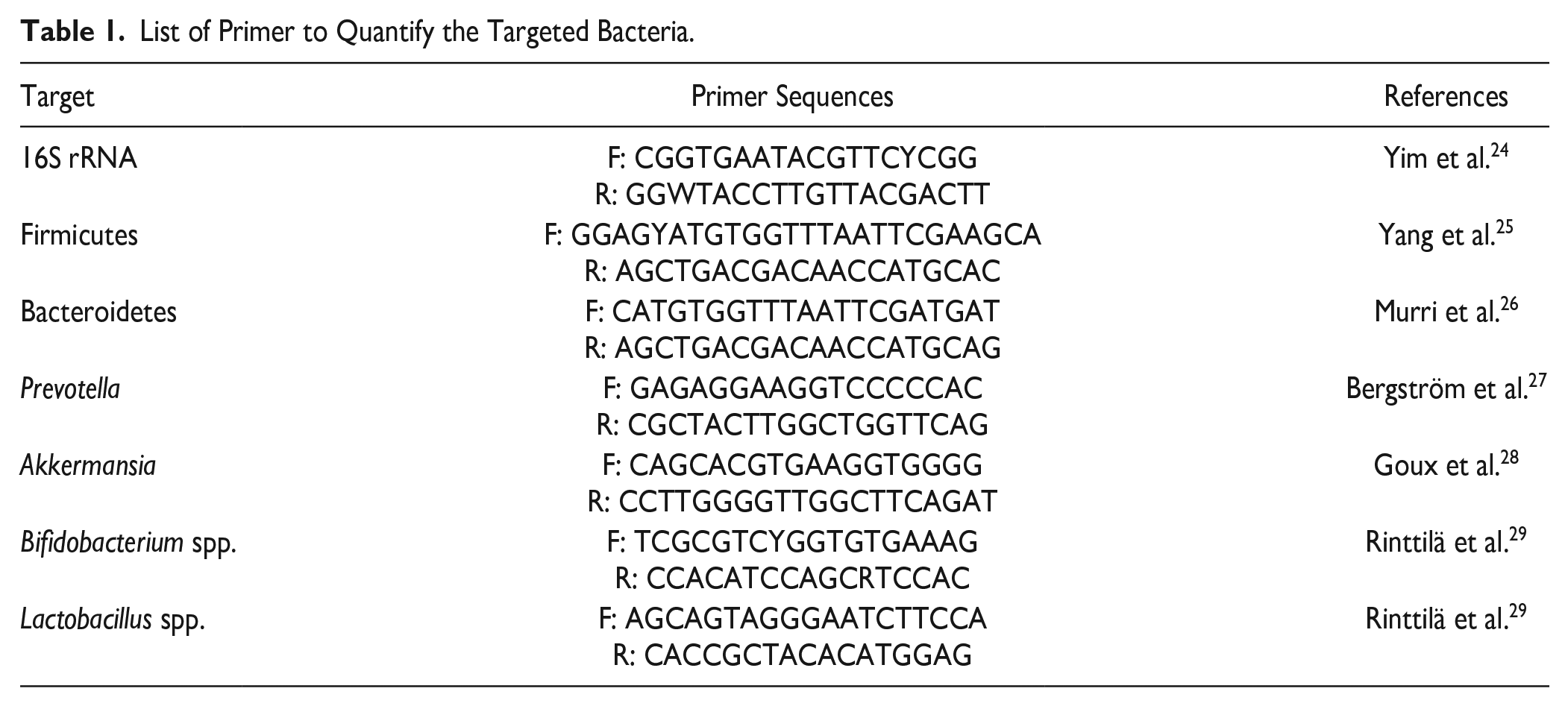
Cecal contents were collected and thoroughly frozen in liquid nitrogen. They were then stored at −80C until DNA extraction was performed. Total genomic DNA from the cecal content was extracted using a FavorPrep Stool DNA Isolation Mini Kit (Favorgen; Ping Tung, Taiwan) according to the manufacturer’s instruction. The DNA samples were analyzed to calculate the relative copy of bacterial genes. Specific primer pairs (Table 1) were used to quantify the targeted group of bacteria. Real-time quantitative PCR (qPCR) was performed using a SensiFAST SBYR Lo-Rox kit (Bioline, Meridian Bioscience; Memphis, Tennessee) on ABI 7500 Fast Real-Time PCR System (Applied Biosystem, Waltham, Massachusetts). The thermocycling program consisted of initial denaturation at 95C for 2 min, denaturation at 95C for 5 sec, and 40 cycles at 95C for 5 min, and primers annealing at 60C for 10 sec and 72C for 20 sec. The relative copy of bacterial genes per sample was calculated using the ∆∆CT analysis.
List of Primer to Quantify the Targeted Bacteria.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 8.0 (GraphPad; San Diego, California). Nonparametric data were analyzed by the Mann–Whitney test. Parametric data were analyzed using the Student’s t-test. p<0.05 was considered to be statistically significant.
Results
Lower Number of Colonic Myenteric Plexus Neurons in TMT-induced Rat Model of Neurodegeneration
The colon length was 10–16 cm, averaging 14.17 ± 2.56 cm. Colon weight was 1319–2402 mg, averaging 1685.17 ± 387.33 mg. To obtain Vref, 100–200 points were counted in the whole colon and ganglionic areas of the histological section. The volume fraction of ganglionic area to total colon area was 0.005–0.008. The calculated volume of the ganglionic area (Vref) was 6201.99–13,271.86 μm3. One third of each rat’s ganglionic area in the myenteric plexus was sampled and yielded 50–70 ganglionic samples for neuron count. With a 3-μm disector height, 112–194 neurons (Q–) were counted in each rat.
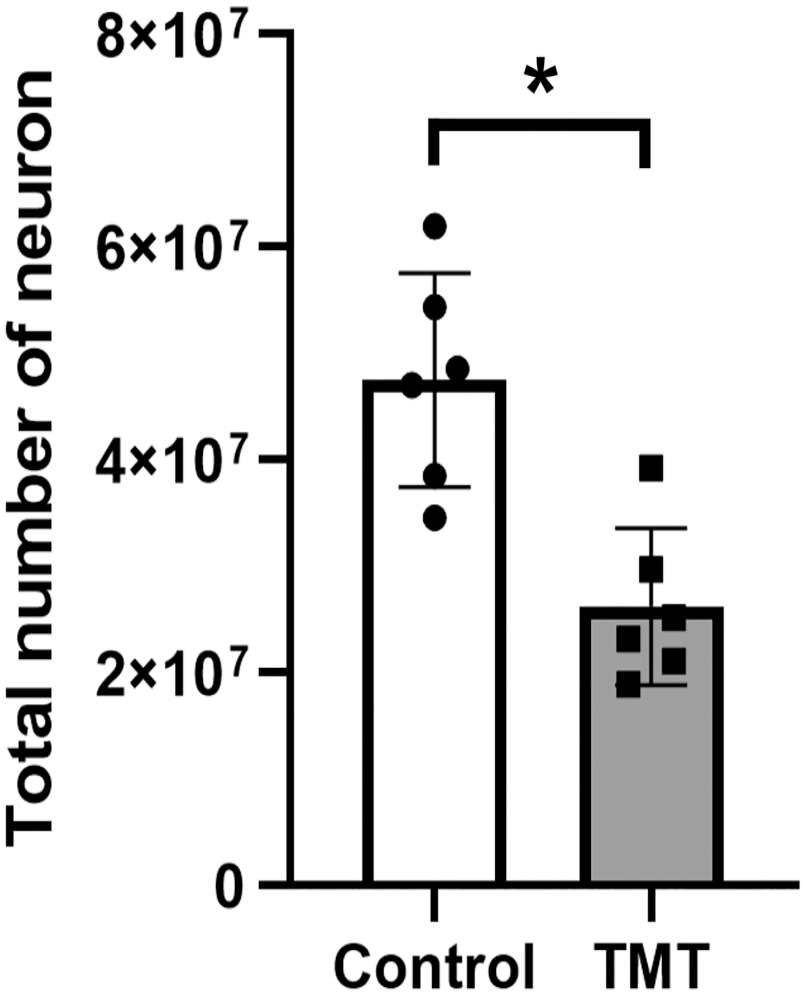
The stereological estimation of the number of myenteric plexus neurons in the rat colon is shown in Fig. 3. The total number of myenteric plexus neurons was significantly higher in the control rats (47,493,841 ± 10,062,166; p<0.005) and more than twice the neuron number in the TMT-induced rat (26,201,475 ± 7,374,521). The OCE was relatively high at 15–31% (average 22%), and the OCV was similarly high at 31%. However, the OCE 2 /OCV 2 value of the total neurons in the myenteric plexus was 0.5, within the targeted value of 0.2–0.5.
Trimethyltin Caused Minor Colon Inflammation
Histological analysis showed minor colon inflammation in the colon of TMT rats and the normal control rats. The main inflammatory sign found in the colon of TMT rats was the infiltration of inflammatory cells. The infiltration was mainly observed in the mucosa of the colon and rarely reached the submucosa. However, the observed colon inflammation was not evenly distributed along the colon length but distributed randomly. As shown in Fig. 4A, an intact crypt and area relatively free from inflammatory cell infiltration was observed in control rats. On the other hand, crypt damage and more abundant inflammatory cell infiltration were more prominent in TMT rats (Fig. 4B). Higher inflammation score was also observed in TMT rats (Fig. 4C).
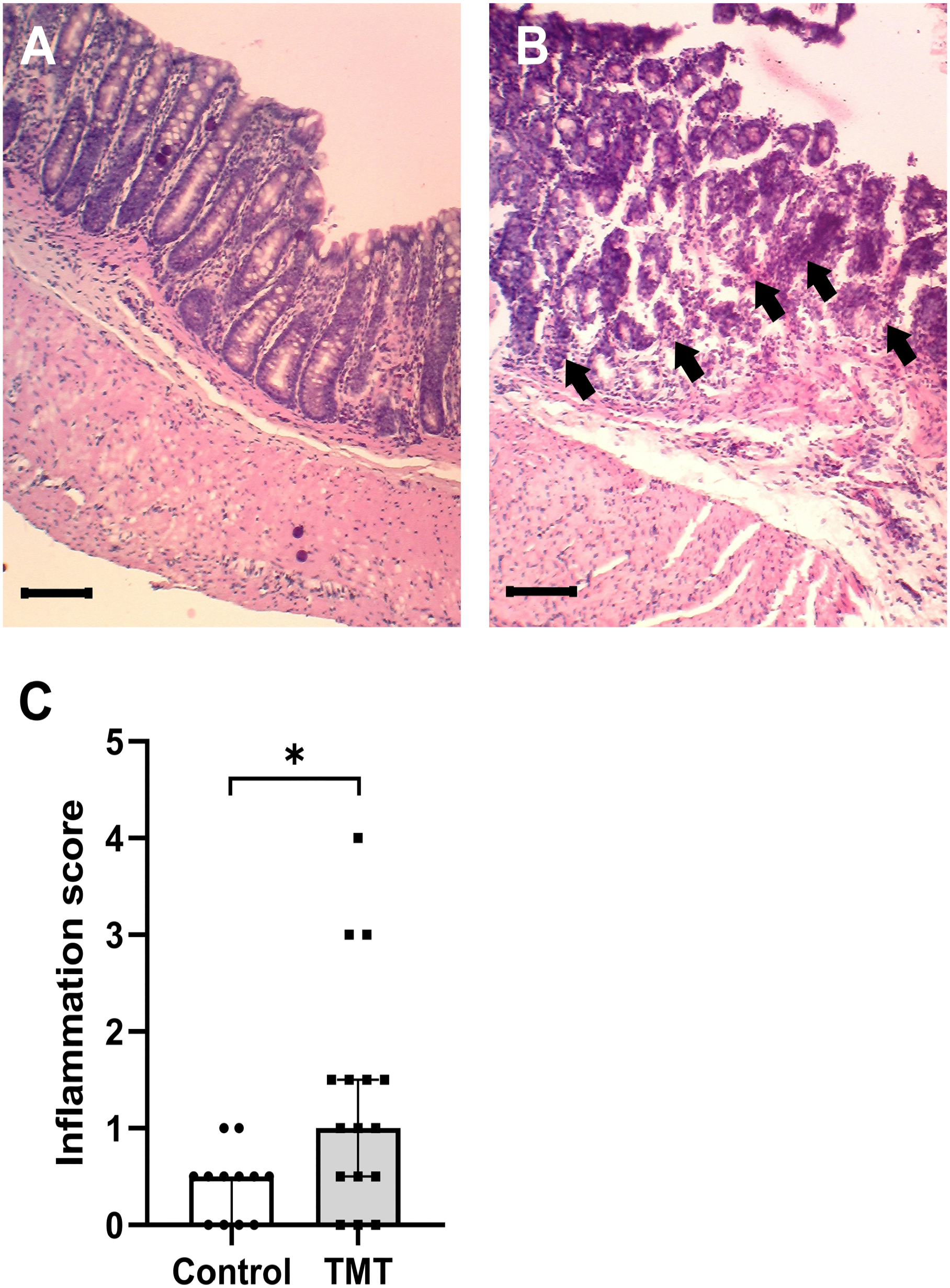
TMT induces colon inflammation. (A) Representative images of hematoxylin and eosin staining in the colon of control rats. (B) Representative images of hematoxylin and eosin staining in the colon of TMT rats. Crypt damage and inflammatory cell infiltration are indicated by arrows. Scale bar: 100 μm. (C) The total inflammation score of TMT and control rats. TMT, trimethyltin; CI, confidence interval. Data are presented as median with 95% CI, n=6 per group, *p<0.05.
Trimethyltin Induced Slightly Higher Expression of TNF-α in the Mucosa of Colon
The expression of TNF-α, one of the proinflammatory cytokines that reportedly mediates TMT-induced neuronal death, 12 was determined by immunohistochemistry. The TMT and control rats both expressed TNF-α. In the mucosa of the colon, a slightly higher expression of TNF-α was observed in TMT rats than that in control rats (Fig. 5A and B). The TNF-α expression was also observed in the colon’s myenteric plexus and muscle layer, both in the TMT and control rats, with a similar intensity (Fig. 5C and D). Similar to the inflammation feature found in TMT rats, the TNF-α was unevenly expressed in each TMT rat, in which several mucosal areas and myenteric plexuses had high expression of TNF-α, but several other areas had low expression.
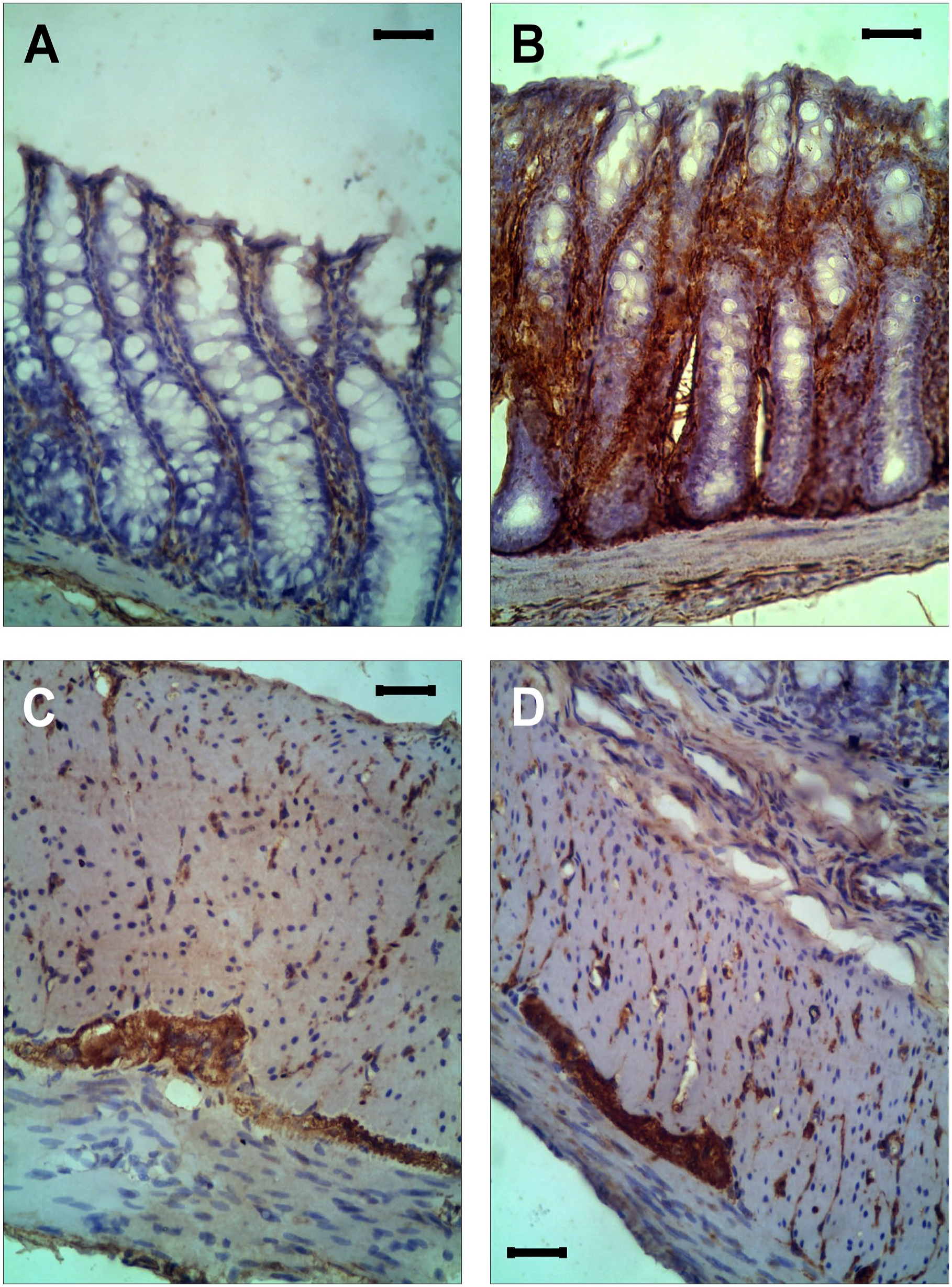
Immunohistochemical staining of TNF-α expression in the colon’s mucosa, muscle layer, and myenteric plexus. Representative images of TNF-α expression in the mucosa of the colon in control (A) and TMT rats (B). Representative images of TNF-α expression in the muscle layer and myenteric plexus of the colon in control (C) and TMT rats (D). Scale bar: 20 μm. TMT, trimethyltin; TNF-α, tumor necrosis factor-α.
On Day 28 After TMT Induction, the Gut Microbiota Composition Was Not Affected
To investigate the effect of TMT on gut microbiota composition, qPCR analysis of cecal contents obtained from the control and TMT rats was performed. The relative copy of Firmicutes (Fig. 6A), Bacteroidetes (Fig. 6B), Prevotella (Fig. 6C), Akkermansia (Fig. 6D), Bifidobacterium spp. (Fig. 6E), and Lactobacillus spp. (Fig. 6F) was not different between TMT and control rats. Our data revealed that on day 28 after TMT induction, the gut microbiota composition was similar to the normal control.
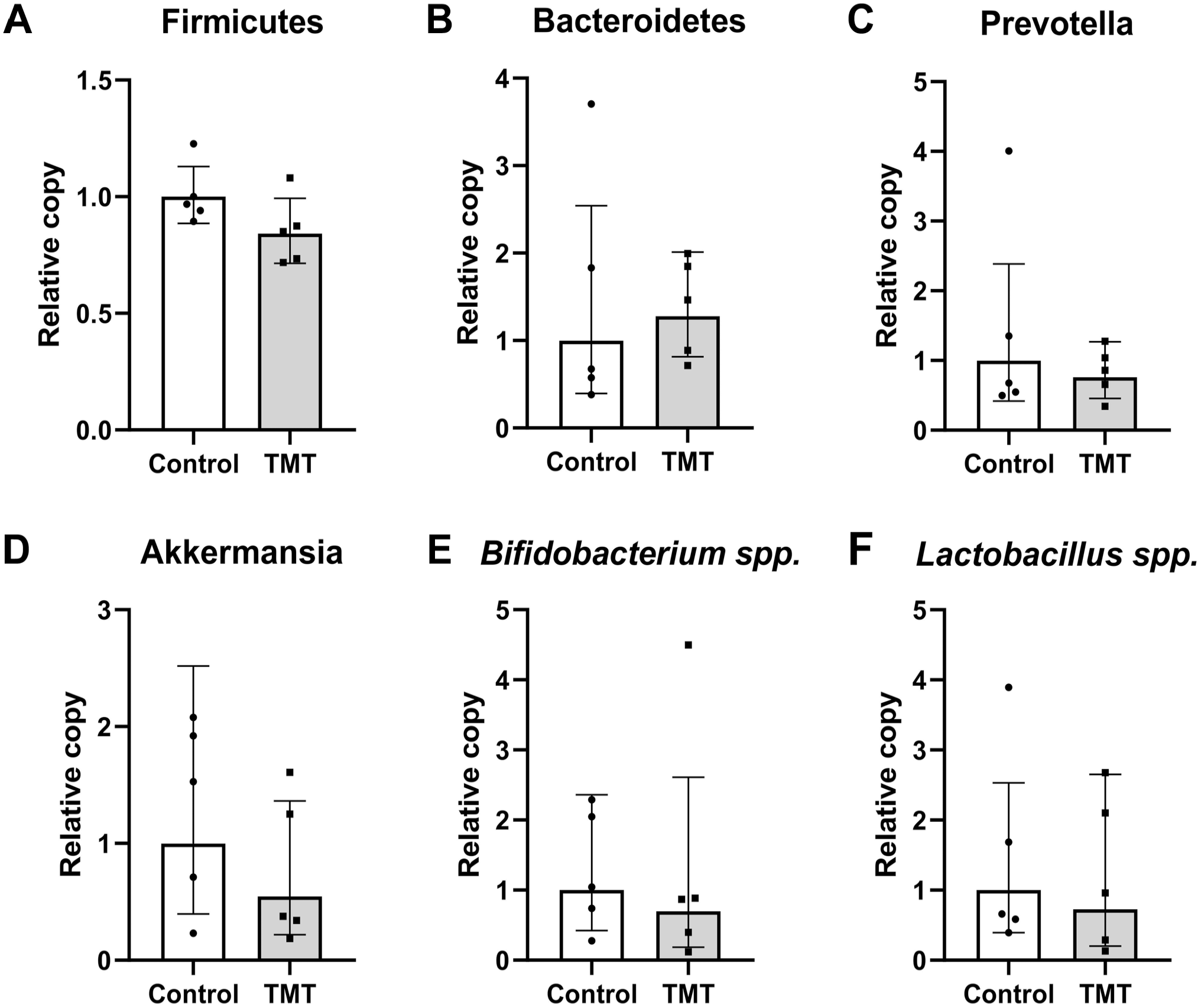
Gut microbiota composition in TMT and control rats. The relative copy of Firmicutes (A), Bacteroidetes (B), Prevotella (C), Akkermansia (D), Bifidobacterium spp. (E), and Lactobacillus spp. (F) in cecal contents. TMT, trimethyltin. Data are presented as mean ± SD, n=6 per group, *p<0.05.
Discussion
TMT has been established as a substance that causes neurodegeneration, particularly in the hippocampus.10,16,18 However, its effect on the colonic myenteric plexus was unknown. Considering the bidirectional communication between ENS and CNS, and the possibility that problems in the gastrointestinal tract precede CNS degeneration,8,9 it is essential to investigate the effect of TMT on ENS.
In this study, we reported the loss of neurons in the colonic myenteric plexus after TMT induction. This finding suggests that the neurodegeneration induced by TMT is not restricted to the brain but also involves the ENS, specifically the colonic myenteric plexus. The degeneration of ENS is observed in aging and also age-related neurodegenerative diseases, including Alzheimer’s and Parkinson’s diseases.1,2 More importantly, ENS degeneration probably precedes brain neurodegeneration and afterward contributes to brain pathology through the gut–brain axis. 30 Whether ENS degeneration observed after TMT induction precedes and correlates with brain neurodegeneration needs to be elucidated.
Various factors may be involved in causing neuronal loss; one of them is inflammation. As inflammation was observed in the brain of the TMT-induced rat model of neurodegeneration,10,16,31 we investigated whether inflammation occurs in the colon after TMT induction. Our findings showed inflammation that randomly spread along the colon of TMT-induced rat model of neuron degeneration. This colon inflammation was characterized by inflammatory cell infiltration, crypt damage, and slightly higher expression of TNF-α in the mucosa of the colon. The minor inflammation observed in this study may suggest that the resolution of colon inflammation is in progress. Resolution of inflammation was also reported in the brain of rats injected with TMT. 31 However, neuronal loss caused by colon inflammation may persist for a long time even after the inflammation has resolved, as observed in animal models of colitis. 32
In the present study, TNF-α expression was slightly higher in the mucosal colon of TMT rats. TNF-α may induce cellular apoptosis 33 and necroptosis 34 by binding to its receptor, the TNFR1. The binding of TNF-α to its receptor may lead to ROS production 33 and NLRP3 inflammasome activation, 35 which eventually activate the apoptosis pathway. The expression of TNFR1 by neurons 36 suggests the role of TNF-α in the loss of colonic myenteric plexus neurons after TMT induction.
In recent years, accumulating evidence suggested a strong connection between colon inflammation, gut dysbiosis, and neurodegeneration. 37 Intimate communication and interaction occur between gut microbiota with cells in the intestine, including immune cells, neurons, and intestinal epithelial cells.38,39 However, this study did not observe any differences in the composition of gut microbiota between TMT and control rats, even though colon inflammation and neuronal loss in colonic myenteric plexus were observed after TMT induction. This finding suggested that TMT did not influence gut microbiota composition or that the gut microbiota resumed their normal composition on day 28 after TMT induction. The resilience of commensal gut microbiota during inflammation 40 was reported and may contribute to our result. Commensal gut microbiota responds and performs adaptation upon inflammation to survive the inflammatory niche. 40 On the other hand, normal gut microbiota may support the resolution of inflammation. 41 High interindividual variation in each rat group and the limited variety of the examined bacteria also may affect the result of the present study. A further study that includes more examined bacteria, especially in the genus and species level, and the use of metagenomic sequencing will elaborate more information regarding the gut microbiota composition in the TMT-induced rat model of neurodegeneration.
Primarily, TMT induction is used to model hippocampal neuron degeneration. Our study demonstrates that neurodegeneration caused by TMT is not limited to the hippocampus but also in the ENS, specifically in the colonic myenteric plexus. Because enteric nerve degeneration may precede neurodegeneration in CNS, as observed in Parkinson’s and Alzheimer’s diseases, this animal model has the potential to be used in elucidating the underlying mechanism of communication between the gastrointestinal tract and CNS in neurodegenerative diseases. The contribution of animal models, including rotenone-induced rats, 42 6-hydroxydopamine (6- OHDA)-induced rats, 43 α-syn preformed fibril-injected mice, 44 and transgenic mice such as Thy-1-αSyn 45 and APP/PS1 mice, 46 in clarifying the connection between the gastrointestinal tract and CNS has been tremendous. Therefore, the TMT-induced rat model of neurodegeneration may contribute to further elucidating the connection between the gastrointestinal tract and CNS. Further characterization of the TMT-induced rat neurodegeneration model is necessary to ensure its application in elucidating the underlying communication mechanism between the gastrointestinal tract and CNS in neurodegenerative diseases.
Footnotes
Acknowledgements
We thank the Department of Histology and Cell Biology Faculty of Medicine, Public Health and Nursing Universitas Gadjah Mada staff for valuable discussions. We also thank Dr. Titis Nurmasitoh for her support and advice during animal handling.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: study design (DES, RS), data collection (DES, HAZ, MFR, S, DS, YAAS), data analysis (DES, HAZ, RS), writing the first draft of the manuscript (DES, RS), manuscript review (DES, HAZ, DKP, YAAS, RS).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Dana Masyarakat Grant from the Faculty of Medicine, Public Health and Nursing, Universitas Gadjah Mada (number: 211/UN1/FKKMK/PPKE/PT/2021).