Abstract
Coronavirus disease (COVID-19) is caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). SARS-CoV-2 RNA has been found in the human testis on occasion, but subgenomic SARS-CoV-2 and infectious SARS-CoV-2 virions have not been found. There is no direct evidence of SARS-CoV-2 infection of testicular cells. To better understand this, it is necessary to determine whether SARS-CoV-2 receptors and proteases are present in testicular cells. To overcome this limitation, we focused on elucidating with immunohistochemistry the spatial distribution of the SARS-CoV-2 receptors angiotensin-converting enzyme 2 (ACE2) and cluster of differentiation 147 (CD147), as well as their viral spike protein priming proteases, transmembrane protease serine 2 (TMPRSS2) and cathepsin L (CTSL), required for viral fusion with host cells. At the protein level, human testicular tissue expressed both receptors and proteases studied. Both ACE2 and TMPRSS2 were found in interstitial cells (endothelium, Leydig, and myoid peritubular cells) and in the seminiferous epithelium (Sertoli cells, spermatogonia, spermatocytes, and spermatids). The presence of CD147 was observed in all cell types except endothelium and peritubular cells, while CTSL was exclusively observed in Leydig, peritubular, and Sertoli cells. These findings show that the ACE2 receptor and its protease TMPRSS2 are coexpressed in all testicular cells, as well as the CD147 receptor and its protease CTSL in Leydig and Sertoli cells, indicating that testicular SARS-CoV-2 infection cannot be ruled out without further investigation:
Keywords
Introduction
Since the first cases of infection with severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) were reported in 2019 and a pandemic was declared in 2020,1,2 the medical and research communities have released a massive amount of research in a short period of time to better understand the virus and disease. This was also intended to offer better diagnostic, treatment, and prevention strategies. Yet, aside from a person’s respiratory droplets,3,4 the transmission of SARS-CoV-2 through physiological fluids, including sperm,5–14 has not been well elucidated in the literature.5–7,15–18 As a result, questions about testicular SARS-CoV-2’s viral tropism remain. This is significant for sperm cryopreservation or assisted reproductive technology techniques that use ejaculated sperm in recovered men.
SARS-CoV-2 is an enveloped, single-stranded, positive-sense RNA virus of the coronavirus family.3,19 Its structure consists of four structural proteins, spike (S) protein, membrane (M), envelope (E), and nucleocapsid (N). 20 Protein N is responsible for virus packaging and replication, whereas the S transmembrane glycoprotein is responsible for recognition by the host membrane receptors.21–23 The S protein is composed of two subunits: S1 encodes the receptor binding domain and binds to the host membrane receptor, and S2 encodes the transmembrane portion of the S protein and is responsible for viral membrane fusion with cellular membranes.24–26
SARS-CoV-2 tropism is primarily determined by the S protein, which mediates viral infection by binding to host cell membrane receptors. Despite this, due to the high infectivity of COVID-19, two routes of SARS-CoV-2 infection have been described. 26 The viral RNA enters the cytosol via a fusion pore in the plasma membrane in one route and endocytosis in another.
In the first route, SARS-CoV-2 binds to the angiotensin-converting enzyme 2 (ACE2), a receptor on the host cell’s cell membrane, and enters the cell, where the S protein is primed by the type II transmembrane serine protease transmembrane protease serine 2 (TMPRSS2), which releases the S protein subunit S2 to fuse the viral and cellular membranes. 27 As a result, the virus does not directly enter the cell, but instead it creates a pore through which the RNA and RNA-associated nucleocapsid proteins within the virus’s lumen gain access to the cellular cytosol, triggering infection. Alternatively, the ACE2-bound virus is internalized via clathrin-mediated endocytosis into the late endolysosome, where the S2′ site is cleaved by cathepsins, if the target cell has dispersed receptor and protease, expresses TMPRSS2 insufficiently, or the virus–ACE2 complex does not encounter TMPRSS2. 28 SARS-CoV-2 can also enter host cells via endocytosis, which is mediated by the transmembrane glycoprotein cluster of differentiation 147 (CD147). 29 The viral membrane fuses with the luminal face of the endosomal membrane at this point, allowing RNA transfer to the cytosol using the fusion protease cathepsin L (CTSL), 30 to infect the cell with its genetic material. The main host membrane receptor for SARS-CoV-2 is ACE2, 31 but when ACE2 expression is absent or reduced, 26 SARS-CoV-2 enters the host cell primarily through membrane fusion via CD147. 32 SARS-CoV-2’s activation in TMPRSS2-deficient cells may also be compensated by CTLS, 3 reinforcing the complementary role of receptors and proteases in virus infection. 31
The classic manifestations of COVID-19 are associated with the respiratory system, but SARS-CoV-2 causes a systemic disease with an inflammatory and immune response of the organism,33,34 thus causing deleterious effects on other organs, including the testis. 35 However, it remains unclear whether the injuries are the result of direct cell infection or indirect effects such as systemic inflammatory responses or comorbidities. 36 Several studies have quantified the messenger ribonucleic acid (mRNA) and protein expression of the aforementioned viral receptors and proteases in different cell types of the testicular interstitium [endothelium, Leydig cells (LC), myoid peritubular cells (PTC)] and germinal epithelium, including Sertoli cells (SCs), spermatogonia (SG), spermatocytes, spermatids, and sperm.36–38 However, few studies have focused on characterizing the histological and morphological spatial distribution of these proteins, so this structural aspect remains poorly understood. As a result, we performed a detailed histological and morphological analysis of the distribution of SARS-CoV-2 receptors and their S protein priming-host cell proteases via membrane fusion (ACE2 and TMPRSS2) and endocytosis (CD147 and CTLS) associated with its entry in human testis.
Materials and Methods
Ethical Considerations
The biological material was obtained from archive Pathology tissue paraffin-embedded blocks and was used under strict patient anonymity. The use of hospital archive pathological biological material for research is not subject to the patient’s written informed consent, as patients at the time of hospital treatment have signed an informed consent that allows the use of their pathology biological material for diagnostic and research purposes at any time. The present laboratorial experiments were executed under the joint ethics committee of the hospital and university, Porto University Hospital Center / School of Medicine and Biomedical Sciences (CHUP/ICBAS) approval number 2020-094 (077-DEFI-078-CE). This work did not involve human or animal experiments, and thus, the provisions of the Declaration of Helsinki as revised in Tokyo 2004 do not apply to this work.
Biological Material
Formalin-fixed and paraffin-embedded testicular biopsy specimens were collected from the pathology archives of Vila Nova de Gaia/Espinho Hospital Center in Vila Nova de Gaia, Portugal. The current study included 15 male patients of reproductive age who had open testis diagnostic biopsy for evaluation of male infertility. Testicular tissues were histopathologically examined using hematoxylin and eosin staining, and three histopathological patterns of non-obstructive azoospermia (NOA) testicular biopsy, namely, SC-only syndrome (SCOS), germ cell maturation arrest (MA), and hypospermatogenesis (HP), were selected. At least five random fields of each section were observed under the microscope and evaluated for the area of seminiferous tubules, the thickness of the seminiferous tubules, and Johnson’s score. Briefly, in SCOS, the tunica propria and basement membranes were not thickened, the tubules were normal or slightly decreased in diameter and contained only SCs but no other cells involved in spermatogenesis, and the interstitium contained normal numbers of LCs; in MA, the process of spermatogenesis was arrested at the round spermatids stage, differing from HP by the absence of elongated spermatids (Sd); and in HP, the germinal epithelium was conserved with the presence of all stages of spermatogenesis up to late spermatids; however, its cellularity number was reduced. The current study did not intend to study the virus entry proteins in different testicular pathologies; rather, as spermatogenesis is a complex of closely interconnected cells, these histopathological testicular patterns were selected with the purpose of a clearer individual cellular identification (SCs in SCOS cases; spermatocytes in MA cases; spermatids in HP cases) and assertive localization of receptors, with the ultimate goal of reconstructing the architecture of the normal testis. We recognize that our population is not ideal. The population should ideally comprise fertile normozoospermic males, but for ethical reasons, a testicular biopsy cannot be performed on this group. Yet, successful sperm retrieval has already been reported, 39 regardless of the histopathology of SCOS, MA, or HP, and testicular histopathology is considered a reliable predictor of successful sperm retrieval. 40
Positive control tissues were obtained from formalin-fixed paraffin-embedded blocks derived from the pathology archives of Portuguese Oncology Institute of Porto, in Porto, Portugal: lung for ACE2 detection, human prostate adenocarcinoma for TMPRSS2 detection, human bladder urothelial carcinoma for CD147 detection, and human hepatocellular carcinoma for CTSL detection.
Inclusion and Exclusion Criteria and Number of Samples
Male factor infertility was defined according to World Health Organization. 41 Inclusion criteria were (1) patients with a clinical diagnosis of NOA; (2) age ≤45years; (3) European–Caucasian; (4) no known bacterial infections and antibiotic therapies at the time of surgery and for the preceding 6 months; and (5) no prior testicular surgery. Exclusion criteria included (1) NOA associated with unilateral or bilateral maldescended testis—left varicocele and/or erection/ejaculation disturbance; (2) genetic abnormalities (any type, including abnormal karyotype and Y chromosome microdeletions); (3) known and modifiable hypothalamic/pituitary abnormalities; (4) previous pituitary or testicular surgery; (5) vasectomy or any other genital tract obstruction; and (6) previous testicular tumor, lymphoma, leukemia, or sarcoma.
The interindividual variation between two subjects influences the statistical variation of the data far more than any other parameter. 42 A minimum of four subjects should be used to obtain statistically significant results. As a result, five patients were included in the current study for each pathology.
Selection of Antibodies
For the detection of the transmembrane receptor ACE2, we selected the rabbit polyclonal antibody ACE2 (Uniprot ID: Q9BYF1; Human) from Biorbyt (Cambridge, UK; orb638860). Characteristics: immunogen—Keyhole limpet hemocyanin-conjugated synthetic peptide derived from human ACE2: 251-350/805; isotype: IgG; purity: affinity purified by protein A; conjugation: unconjugated.
For the detection of the transmembrane protease TMPRSS2, we selected the rabbit monoclonal antibody TMPRSS2 (clone EPR3862), product code AB109131, from Abcam (Cambridge, UK; ab109131). Characteristics: immunogen—synthetic peptide; hybridoma-based technology for making rabbit monoclonal antibodies; RRID AB_2784992; host/isotype: rabbit/IgG; species reactivity: human; purification: protein A; conjugation: unconjugated.
For the detection of basigin (BSG)/extracellular matrix metalloproteinase inducer/CD147, we selected the recombinant rabbit monoclonal antibody CD147 (clone 125), which has specificity for human CD147/basigin/BSG, from Invitrogen (Rockford, IL; MA5-29060). Characteristics: immunogen—recombinant human CD147 protein; RRID: AB_2784992; host/isotype: rabbit/IgG; species reactivity: human; purification: protein A; conjugation: unconjugated.
For the detection of the endosomal cysteine protease CTSL, we used the recombinant rabbit monoclonal antibody cathepsin H/K/L/V (clone JM10-78) from Invitrogen (MA5-32602). Characteristics: immunogen—synthetic peptide within human cathepsin aa 92-138; RRID AB_2809879; host/isotype: rabbit/IgG; species reactivity: human; purification: protein A; conjugation: unconjugated.
Immunohistochemistry
Four-μm-thick tissue sections, fixed on adhesive slides, were dewaxed with xylene (28975.325; VWR Chemicals, Fontenay-sous-Bois, France) for 5 min and hydrated with ethanol (CAS 64-17-5; Valente & Ribeiro, Lda, Alcanena, Portugal). Subsequently, heat antigen retrieval was performed with citrate buffer pH6 [0.1 M citric acid (7461448; Merck, Darmstadt, Germany) plus 0.1 M trisodium citrate dihydrate (1.06448.0500; Merck)] in a microwave (400 W). Endogenous peroxidases were inhibited with 3% hydrogen peroxide (H3410; Sigma-Aldrich, St. Louis, MO) in methanol (32213; Sigma-Aldrich) for 10 min. After washing in phosphate-buffered saline (PBS; Panreac, Barcelona, Spain), nonspecific background staining was minimized with normal horse serum (PK-8800; Vector Laboratories, Burlingame, CA) for 10 min. Sections were then incubated with primary antibodies overnight. The concentrations used were 1:100 and 1:150 for ACE2, 1:1000 for TMPRSS2, 1:2500 for CD147, and 1:100 for CTSL. For each experiment, a negative control was included by omitting the primary antibody and replaced by PBS. The following day, the sections were washed twice with PBS for 5 min each. The Novolink Polymer Kit (RE7159; Leica Systems, NewCastle, UK) was used to reveal the expression of the antigens. A wash with PBS was performed between each step. After counterstaining with hematoxylin (Merck; 1.09249.1000) and dehydration, the sections were mounted on Coverquick.
Evaluation of Results
The evaluation of staining results was performed independently by a master’s student (M.R.R.) and two senior researchers (R.S.), one of whom has extensive experience in testicular morphology, reproductive medicine, and andrology (M.S.), based on previously defined testicular cell morphological characteristics. 43 Images were taken in a light photomicroscope with a video camera (Olympus BX41; Olympus, Tokyo, Japan). The experiments were carried out in triplicate.
Results
There were no cases of discordant individual labeling in triplicates.
Membrane Fusion Pathway for SARS-CoV-2 Infectivity in the Testis
ACE2 Protein Expression in Human Testicular Cells
ACE2 is a zinc membrane-bound metalloproteinase, a carboxypeptidase, and a negative regulator of the renin–angiotensin system.44–46 This protein has a variety of functions, including transporting amino acids, having catalytic activity with various substrates, and acting as an SARS-CoV-2 receptor. When SARS-CoV-2 is bound, less ACE2 is available for intrinsic actions, which leads to an increase in angiotensin II production and a decrease in angiotensin 1–7 production, reducing the activities of endothelial cells. Superoxide species are created as a result, which leads to oxidative stress and inflammation and causes cell malfunction. 44 The renin–angiotensin system controls male fertility, 47 and the ACE2 receptor has been linked to the regulation of sperm motility 48 and T production by LCs.49,50 The epithelial cells of several tissues, including the testis, contain ACE2.38,51–55
ACE2 IHC was performed in 15 cases (5 of SCOS, 5 of MA, and 5 of HP). Histologically, normal control lung tissue was included for comparison. As expected, ACE2 protein expression was detected in the cytoplasmic membrane of alveolar cells and endothelium (Fig. S1).
In testicular tissue, our findings demonstrated that seminiferous tubule epithelium (SC, SG, ST1, ST2, Sa, Sd) and testicular interstitial cells (endothelium, LC, and PTC) both express the ACE2 protein. While ACE2 stained both the cytoplasmic membrane and the cytoplasm of testicular interstitial cells and SCs, germ cells only showed cytoplasmic membrane staining. Indeed, evident staining was observed in the cytoplasmic membrane and cytoplasm of the endothelium, LCs, PTCs, and SCs; in the cytoplasmic membrane of SG (dark, pale, and B), spermatocytes (primary and secondary), and round spermatids; and in the midpiece of Sd. A slight staining was observed in the cytoplasm of germ cells, mostly in spermatocytes and spermatids when shed off in the lumen of the seminiferous tubules (Figs. 1–4; Table 1).
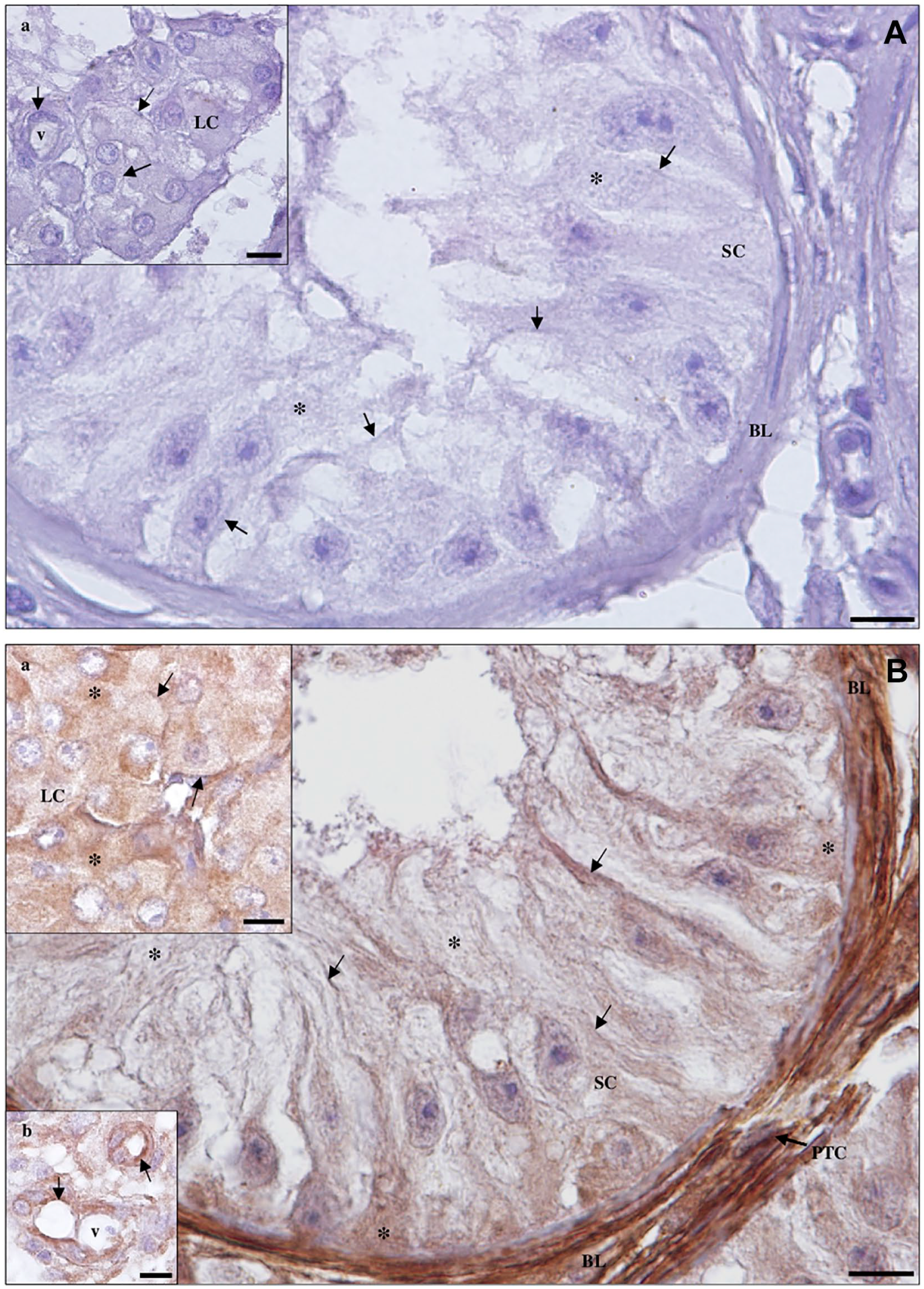
Detection of angiotensin-converting enzyme 2 protein in Sertoli cell (SC)-only syndrome testicular tissue by immunohistochemistry. (A) Negative control. (
Detection of angiotensin-converting enzyme 2 protein in maturation arrest testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): blood vessels (v). Scale bar, 25 µm; (b, inset): Leydig cells (LCs). Scale bar, 20 µm. (B) Staining is observed in the membrane (arrows) and cytoplasm (*) of Sertoli cells (SCs), LCs (b, inset), peritubular cells (PTCs) of the basal lamina (BL) and endothelium (arrow) of v (a, inset), and in the membrane (arrows) of spermatogonia (SG) and primary spermatocytes (ST1). Scale bars, 10 µm.
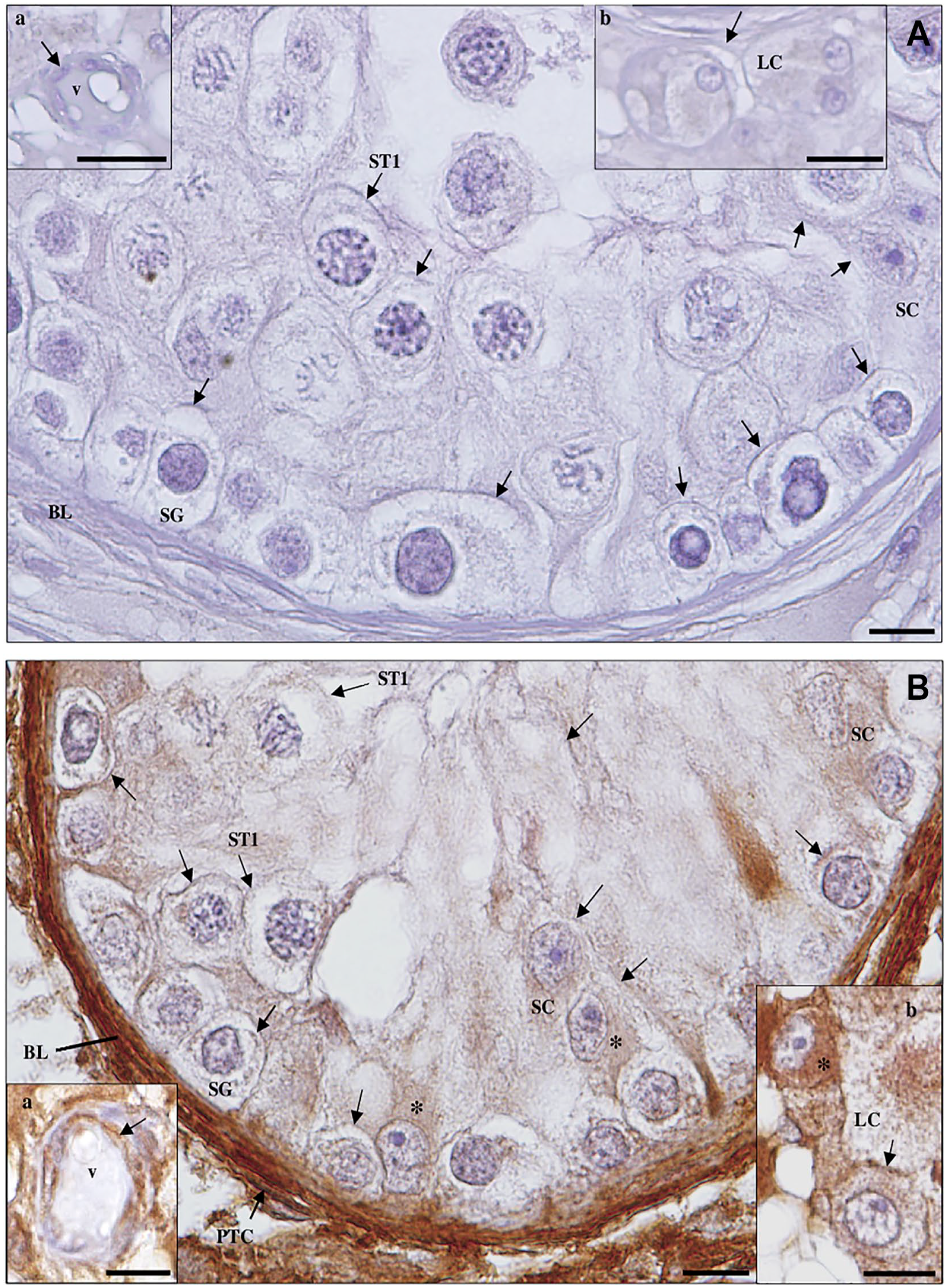
Detection of angiotensin-converting enzyme 2 protein in hypospermatogenesis testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): Leydig cells (LCs); (b, inset): blood vessel (v). (B) Staining is observed in the membrane (arrows) and cytoplasm (*) of Sertoli cells (SCs) and peritubular cells (PTCs) of the basal lamina (BL), in the membrane (arrows) of spermatogonia (SG) and primary spermatocytes (ST1), and in the midpiece of elongated spermatids (Sd). Scale bars, 10 µm.
Detection of angiotensin-converting enzyme 2 protein in hypospermatogenesis (HP) testicular tissue by immunohistochemistry. (A) Staining is observed in the membrane (arrows) and cytoplasm (*) of Sertoli cells (SCs), Leydig cells (LCs; a, inset), peritubular cells (PTCs) of the basal lamina (BL) and endothelium (arrow) of blood vessels (v; b, inset); in the membrane (arrows) of spermatogonia (SG), primary spermatocytes (ST1), secondary spermatocytes (ST2), and round spermatids (Sa); and in the midpiece of elongated spermatids (Sd). Mild staining in the cytoplasm of spermatogonia and moderate staining in the cytoplasm of spermatocytes and spermatids can also be observed, especially when sloughed off in the lumen of the seminiferous tubules. (B) Staining is observed in the midpiece (arrows) of Sd. Scale bars, 10 µm.
Coexpression of ACE2 Receptor and Associated Protease TMPRSS2 in Human Testicular Cells.
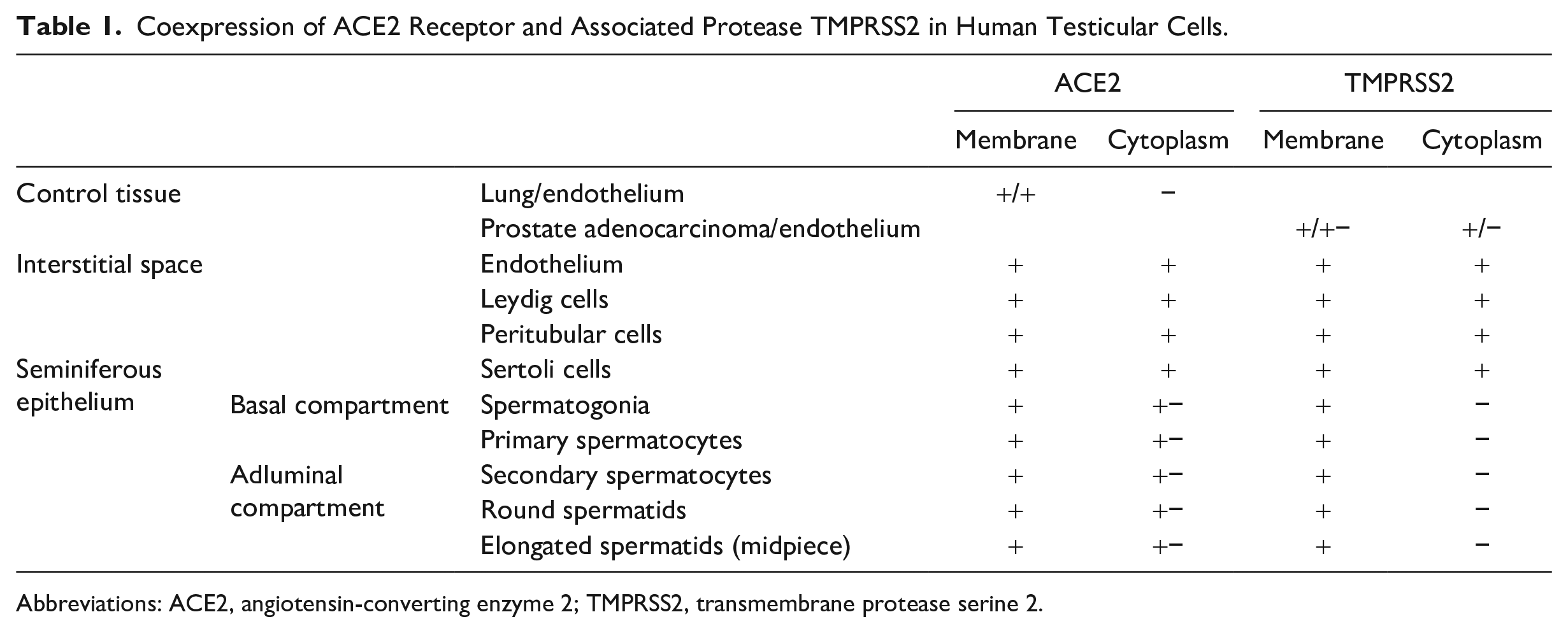
Abbreviations: ACE2, angiotensin-converting enzyme 2; TMPRSS2, transmembrane protease serine 2.
TMPRSS2 Protein Expression in Human Testicular Cells
TMPRSS2 is a type II transmembrane serine protease that regulates cell–cell and cell–matrix interactions. TMPRSS2 is implicated in several physiological and pathological processes, including digestion, coagulation, apoptosis, malignant cell invasion, tissue remodeling, pain, fertility, inflammation, immunity, and respiratory virus activation.26,31,56 The testis is one tissue where TMPRSS2 is found in epithelial cells.36,37,52,54,57
TMPRSS2 IHC was performed in 15 cases (f5 of SCOS, 5 of MA, and 5 of HP). Prostate adenocarcinoma tissue was included for comparison. The TMPRSS2 protein was detected in the cytoplasmic membrane of tumor cells, with discrete staining observed in the endothelium (Fig. S2).
In testicular tissue, evident staining for the ACE2-associated protease, TMPRSS2, was observed in the cytoplasmic membrane and cytoplasm of the endothelium, LCs, PTCs, and SCs; in the cytoplasmic membrane of SG, spermatocytes, and round spermatids; and in the midpiece of Sds (Figs. 5–7; Table 1).
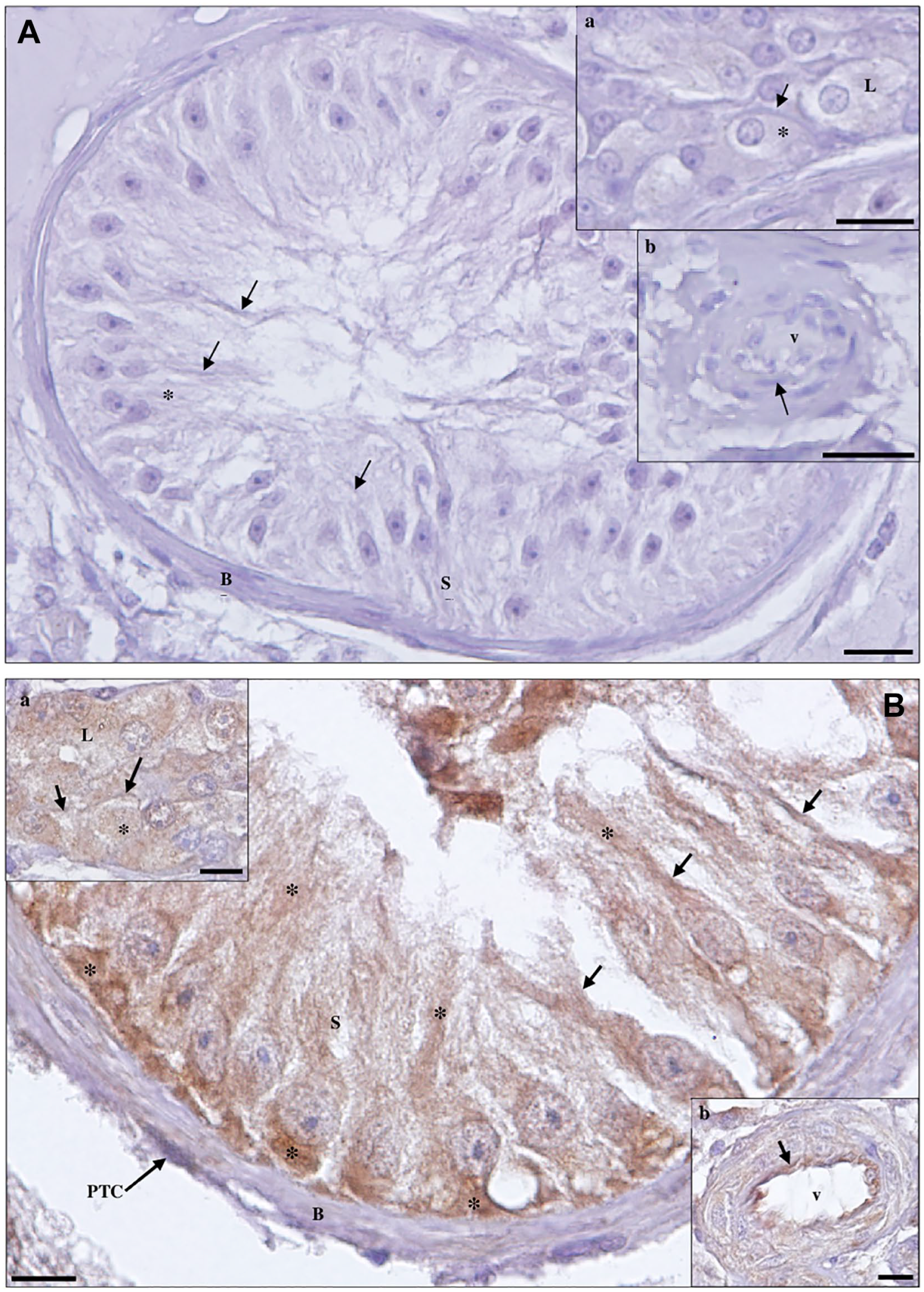
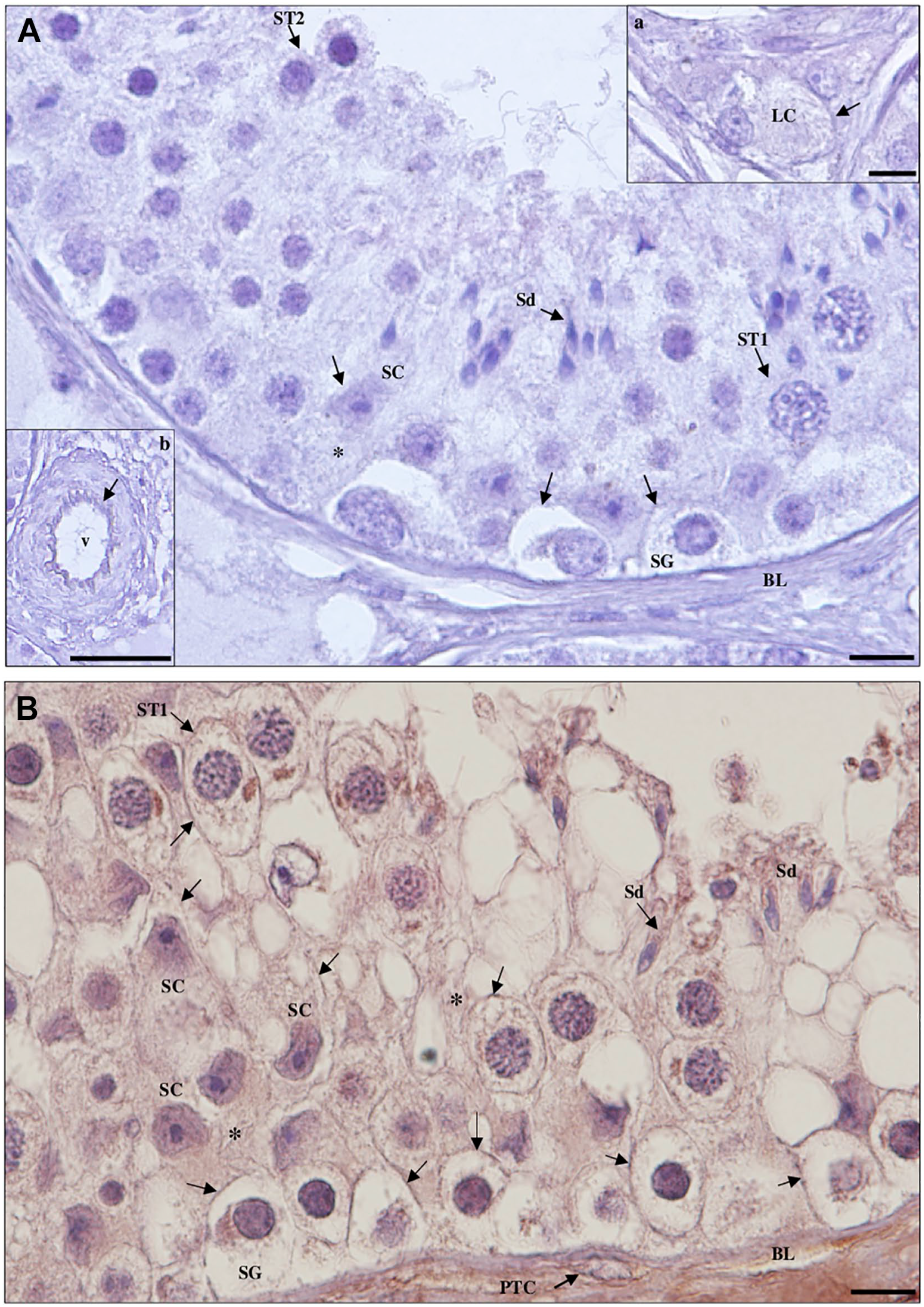
Detection of transmembrane protease serine 2 protein in Sertoli cell-only syndrome testicular tissue by immunohistochemistry. (A) Negative control. Scale bar, 20 µm. (a, inset): Leydig cells (LCs). Scale bar, 20 µm; (b, inset): blood vessel (v). Scale bar, 25 µm. (B) Staining is observed in the membrane (arrows) and cytoplasm (*) of Sertoli cells (SCs), LCs (a, inset), endothelium (arrow) of v (b, inset), and peritubular cells (PTCs) of the basal lamina (BL). Scale bars, 10 µm.
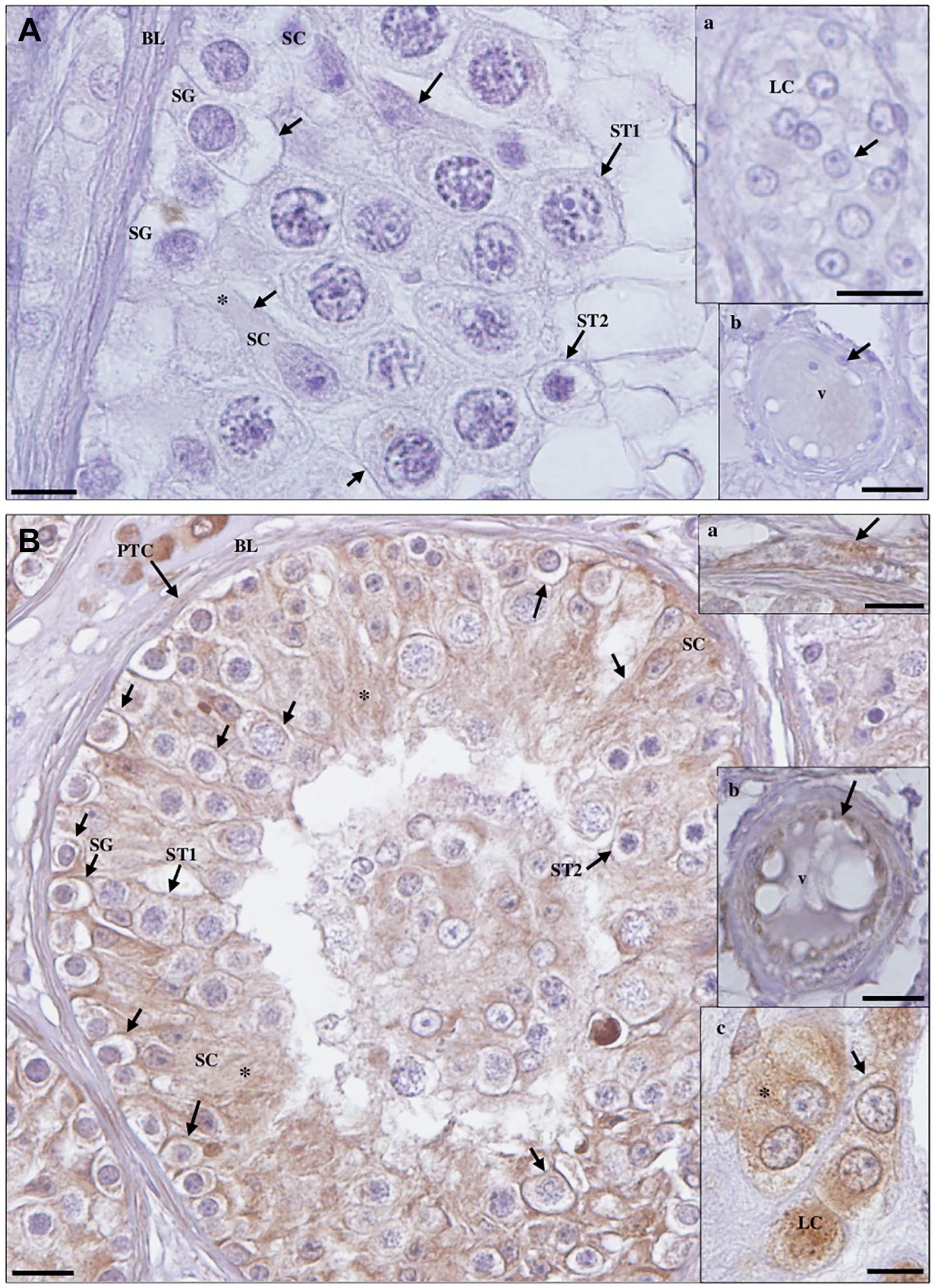
Detection of transmembrane protease serine 2 protein in maturation arrest testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): Leydig cells (LCs). Scale bar, 20 µm. (b, inset): blood vessel (v). Scale bar, 25 µm. (B) Staining is observed in the membrane (arrows) and cytoplasm (*) of Sertoli cells (SCs), LCs (c, inset), peritubular cells (PTCs; a, inset) of the basal lamina (BL) and endothelium (arrow) of v (b, inset), and in the membrane (arrows) of spermatogonia (SG), primary spermatocytes (ST1), and secondary spermatocytes (ST2). Scale bars, 10 µm.
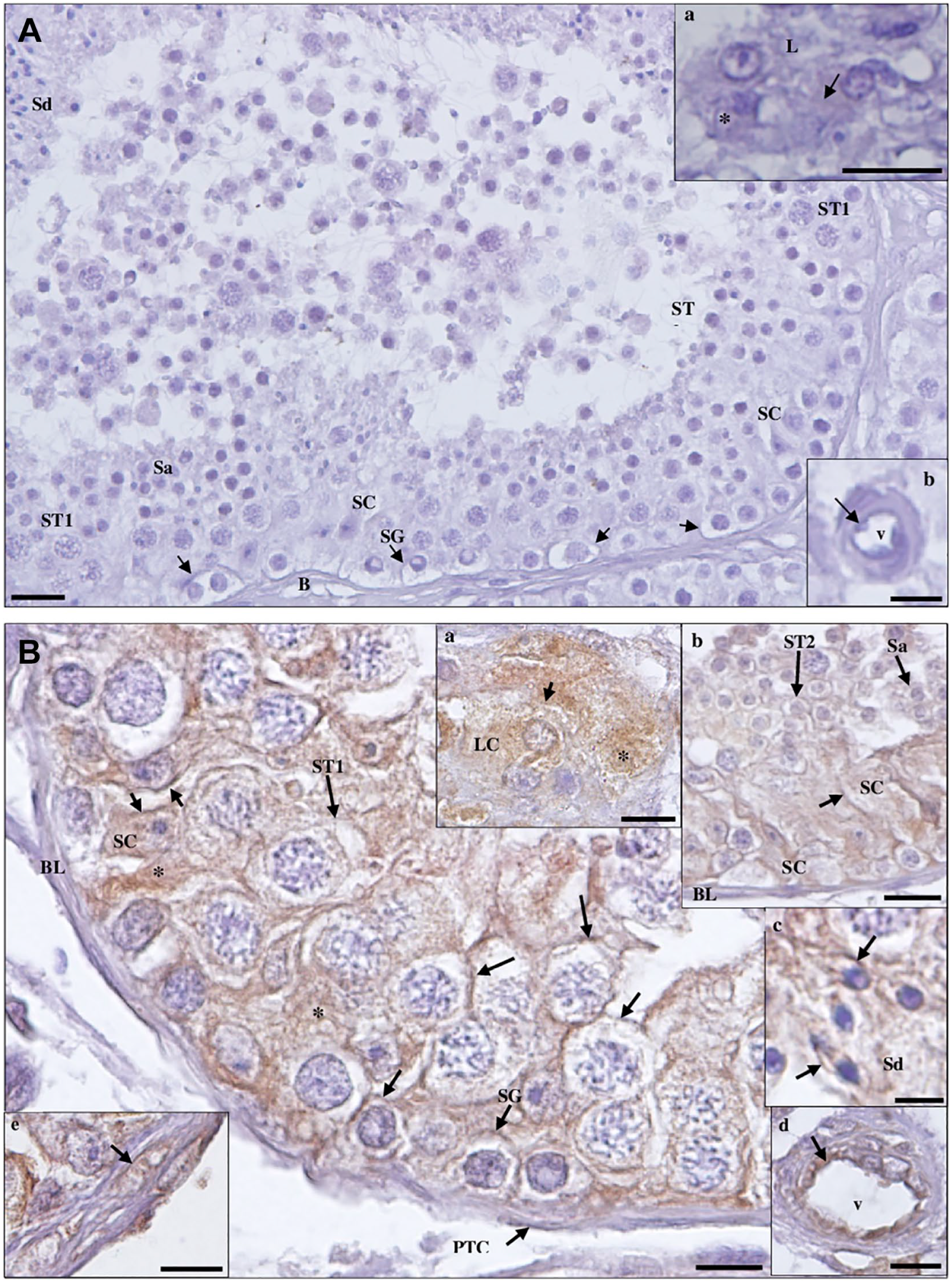
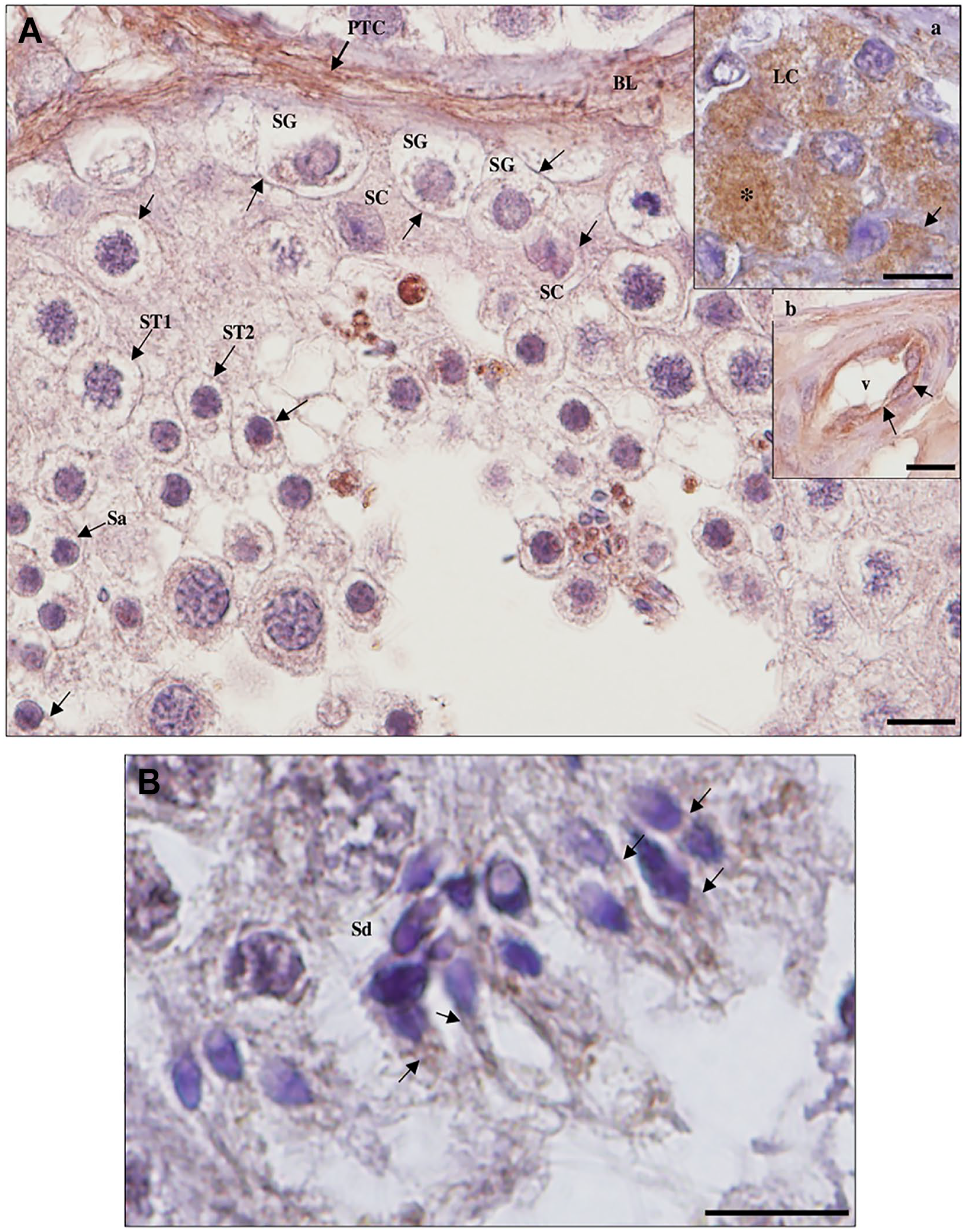
Detection of transmembrane protease serine 2 protein in hypospermatogenesis (HP) testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): Leydig cells (LC); (b, inset): blood vessel (v). Scale bars, 20 µm. (B) Staining is observed in the membrane (arrows) and cytoplasm (*) of Sertoli cells (SC), LCs (a, inset), peritubular cells (PTC; e, inset) of the basal lamina (BL), and endothelium (arrow) of v (d, inset); in the membrane (arrows) of spermatogonia (SG), primary spermatocytes (ST1), secondary spermatocytes (ST2; b, inset; scale bar, 20 µm), round spermatids (Sa; b, inset); and in the midpiece of elongated spermatids (Sd; c, inset; scale bar, 5 µm). Scale bars, 10 µm.
Coexpression of ACE2 Receptor and TMPRSS2 Protease in Human Testicular Cells
After being recognized by ACE2 and before fusing with the host cell, the SARS-CoV-2 S protein is primed by TMPRSS2. Data that immunohistochemically visualized the expression patterns of ACE2 and TMPRSS2 proteins in human testis revealed no specific expression at any stage of spermatogenesis; thus, the protein and protease were found to be coexpressed in all testicular cells, both in the interstitial space and in the seminiferous tubules.
Membrane Fusion Pathway for SARS-CoV-2 Infectivity in the Testis
CD147 Protein Expression in Human Testicular Cells
A putative alternative receptor for SARS-CoV-2 is CD147,26,58 which can be present in two isoforms, membrane-bound or soluble.59,60 CD147 (BSG) is a highly glycosylated type I transmembrane protein involved in the activation of extracellular matrix metalloproteinases.26,59 In addition to germ cell migration, survival/apoptosis, and intercellular junctions,59,61,62 CD147 is also implicated in sperm motility and acrosome reaction. 60 CD147 is also involved in embryonic development and tumor development. 62 According to descriptions, numerous tissues’ epithelia, including the testis’, have CD147.36,52,54,57
CD147 IHC was performed in 15 cases (5 of SCOS, 5 of MA, and 5 of HP). Bladder urothelial carcinoma tissue was included for comparison. The CD147 protein was detected in the cytoplasmic membrane of tumor cells, with discrete staining in the endothelium (Fig. S3).
In testicular tissue, evident staining was observed in the cytoplasmic membrane of LCs, SCs, SG, spermatocytes, round spermatids, and in the midpiece of Sds. No staining was observed in the endothelium or PTCs (Figs. 8–12; Table 2).
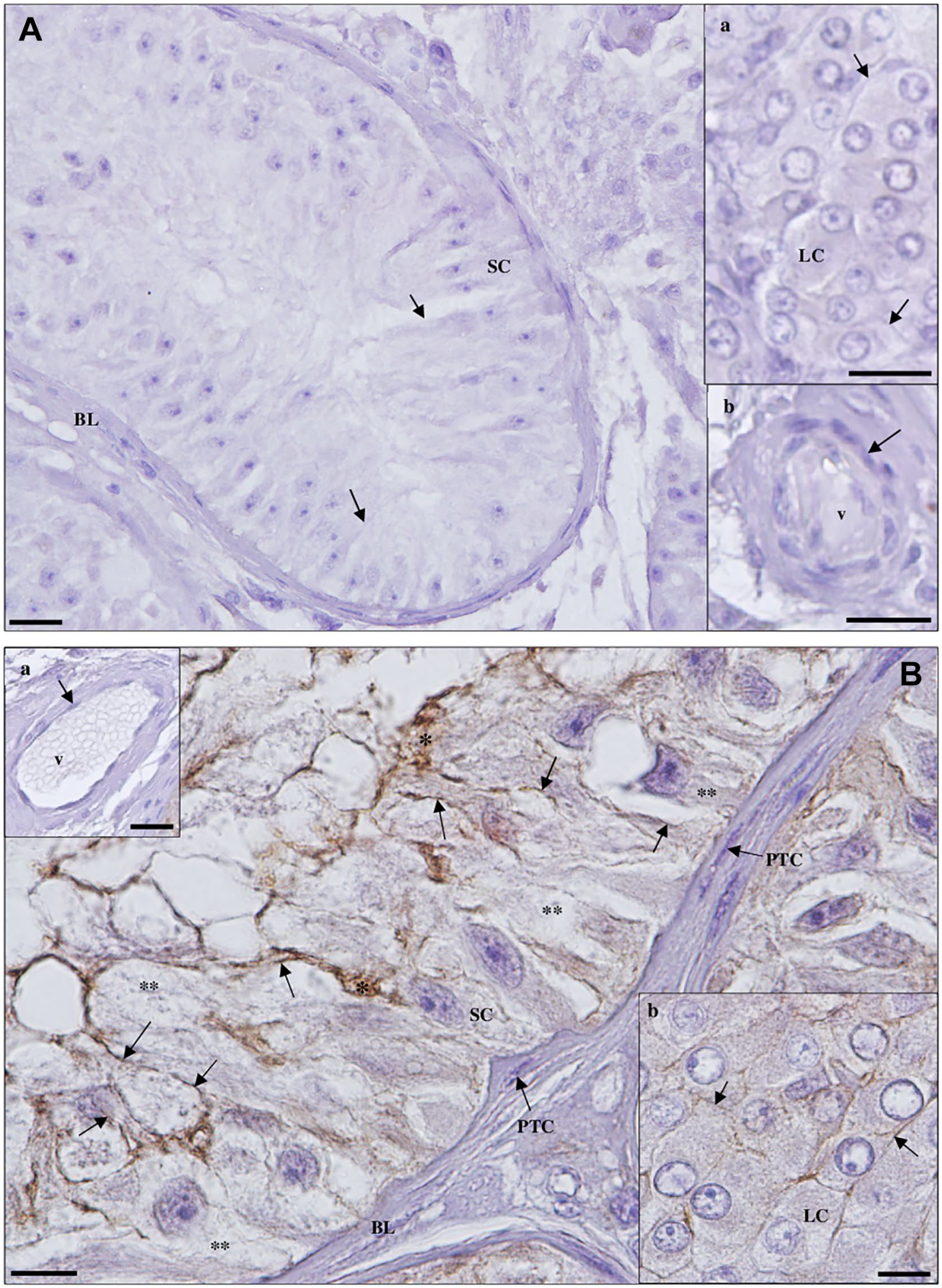
Detection of cluster of differentiation 147 protein in Sertoli cell-only syndrome testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): Leydig cells (LCs); (b, inset): blood vessel (v). Scale bars, 20 µm. (B) Staining is observed in the membrane (arrows) of Sertoli cells (SCs) and LCs (b, inset). No staining is observed in peritubular cells (PTCs) of the basal lamina (BL) and endothelium (arrow) of v (a, inset; scale bar, 20 µm). Note the densely stained extracellular material (*) outside the SC cytoplasm (**). Scale bars, 10 µm.
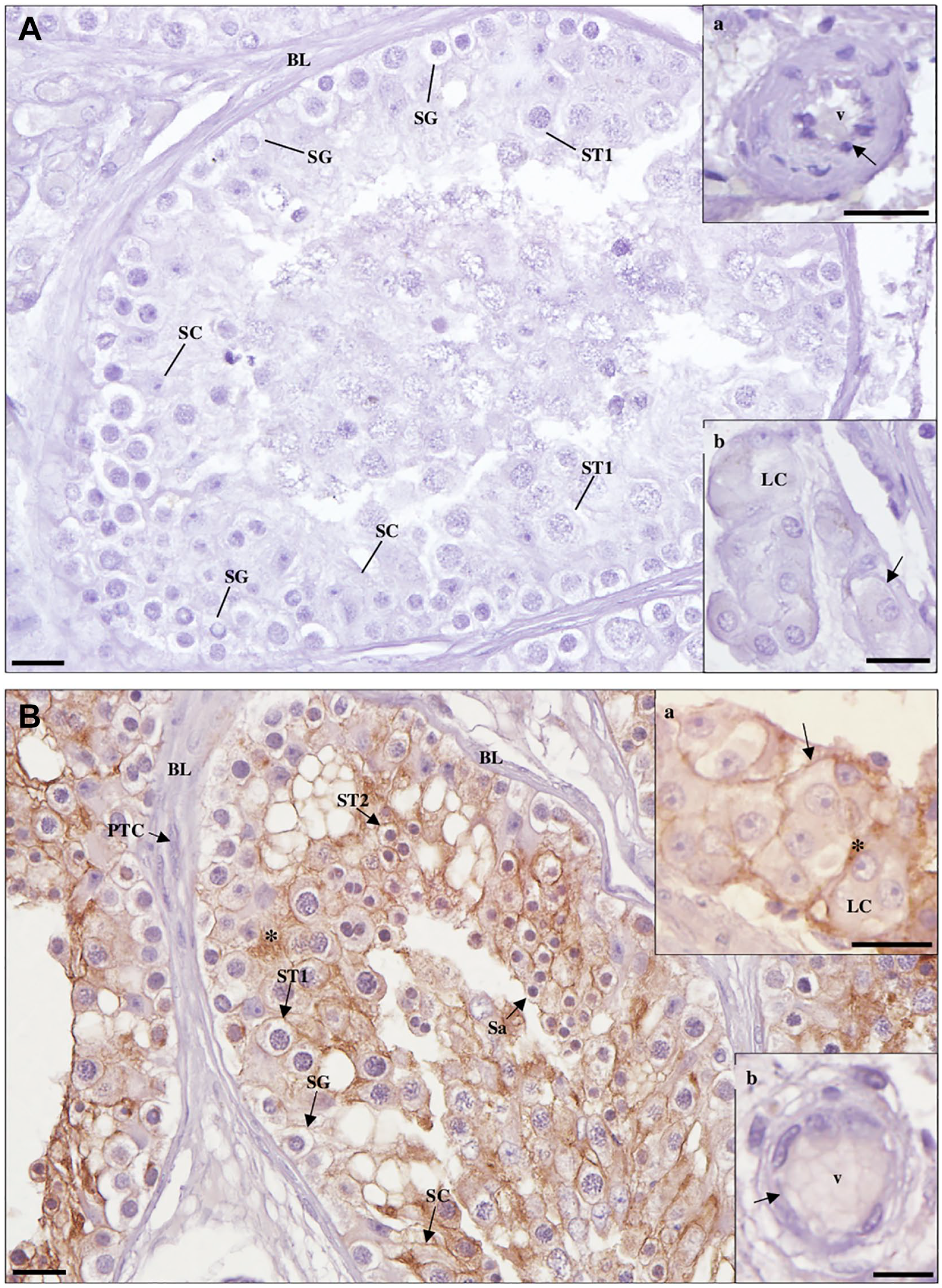
Detection of cluster of differentiation 147 protein in maturation arrest testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): blood vessel (v); (b., inset): Leydig cell (LC). (B) Staining is observed in the membrane (arrows) of Sertoli cells (SCs), spermatogonia, primary spermatocytes (ST1), secondary spermatocytes (ST2), round spermatids (Sa), and LC (a, inset). No staining is observed in peritubular cells (PTCs) of the basal lamina (BL) and endothelium (arrow) of (b, inset). Note the densely stained extracellular material (*). Scale bars, 20 µm.
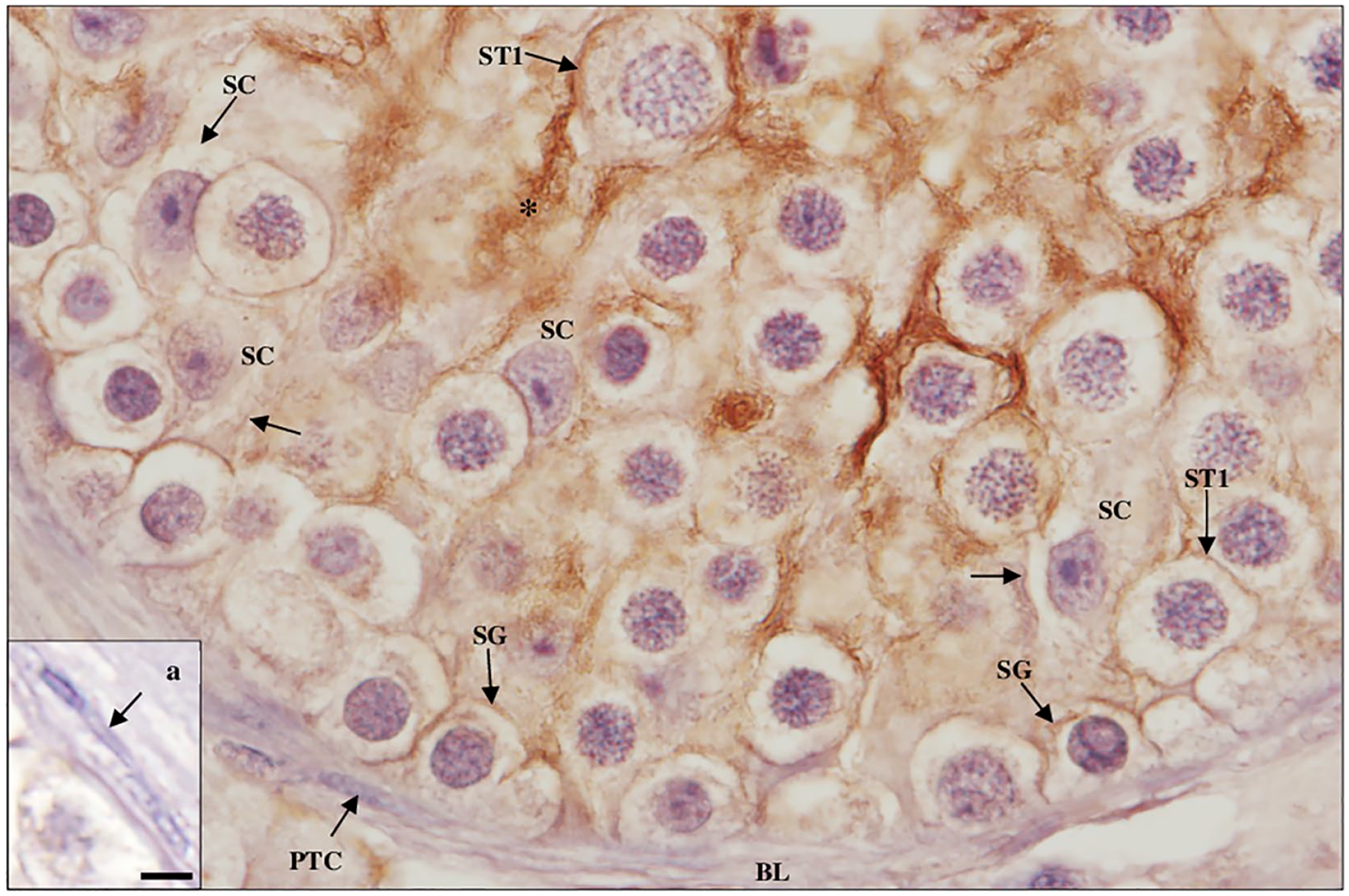
Detection of cluster of differentiation 147 protein in maturation arrest testicular tissue by immunohistochemistry. Staining is observed in the membrane (arrows) of Sertoli cells (SCs), spermatogonia (SG), and primary spermatocytes (ST1). No staining is observed in peritubular cells (PTCs; a, inset; scale bar, 5 µm) of the basal lamina (BL). Note the densely stained extracellular material (*). Scale bar, 10 µm.
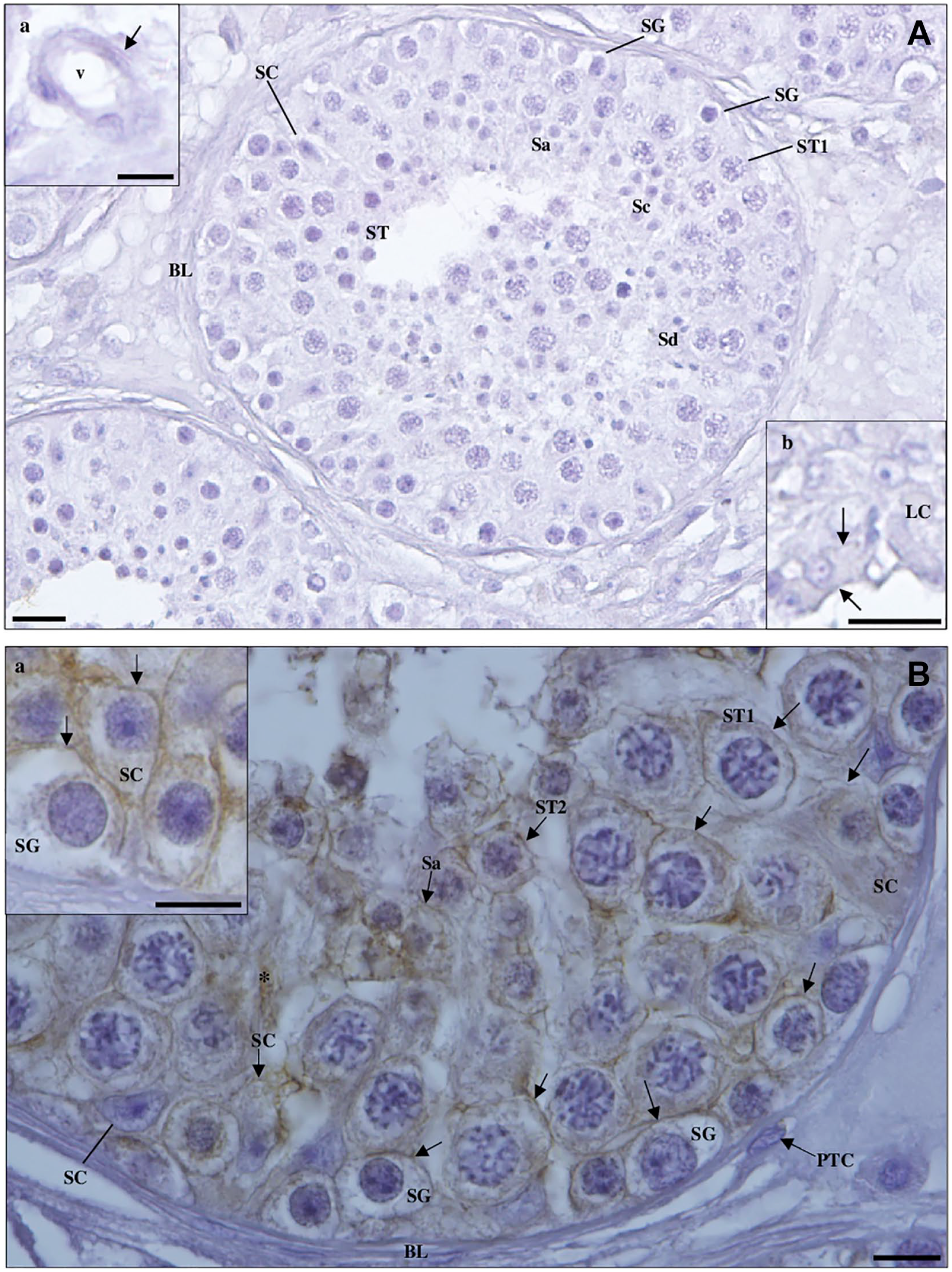
Detection of cluster of differentiation 147 protein in hypospermatogenesis (HP) testicular tissue by immunohistochemistry. (A) Negative control. (a, inset): blood vessel (v); (b, inset): Leydig cell (LC). Scale bars, 20 µm. (B) Staining is observed in the membrane (arrows) of Sertoli cells (SCs; a, inset), spermatogonia (a, inset), primary spermatocytes (ST1), secondary spermatocytes (ST2), and round spermatids (Sa). No staining is observed in peritubular cells (PTCs) of the basal lamina (BL). Note the densely stained extracellular material (*). Abbreviations: Sc, elongating spermatids; Sd, elongated spermatids. Scale bars, 10 µm.
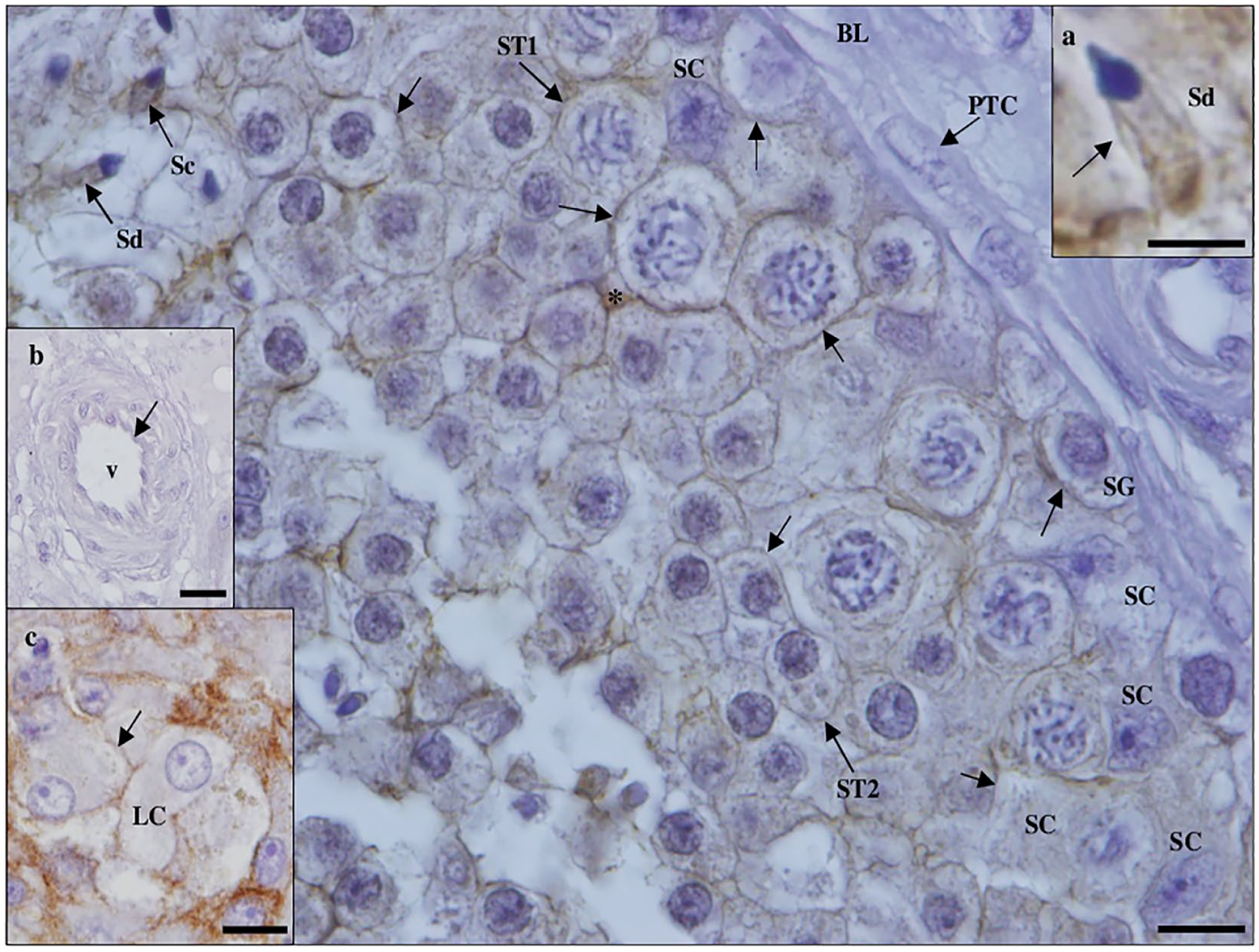
Detection of cluster of differentiation 147 protein in hypospermatogenesis testicular tissue by immunohistochemistry. Staining is observed in the membrane (arrows) of Leydig cells (LCs; c, inset; scale bar, 20 µm), Sertoli cells (SCs), spermatogonia, primary spermatocytes (ST1), secondary spermatocytes (ST2), and in the midpiece of elongating (Sc) and elongated spermatids (Sd; a, inset; scale bar, 5 µm). No staining is observed in peritubular cells (PTCs) of the basal lamina (BL) and endothelium (arrow) of blood vessels (v; b, inset; scale bar, 20 µm). Note the densely stained extracellular material (*). Scale bar, 10 µm.
Coexpression of CD147 Receptor and Associated Protease CTSL in Human Testicular Cells.
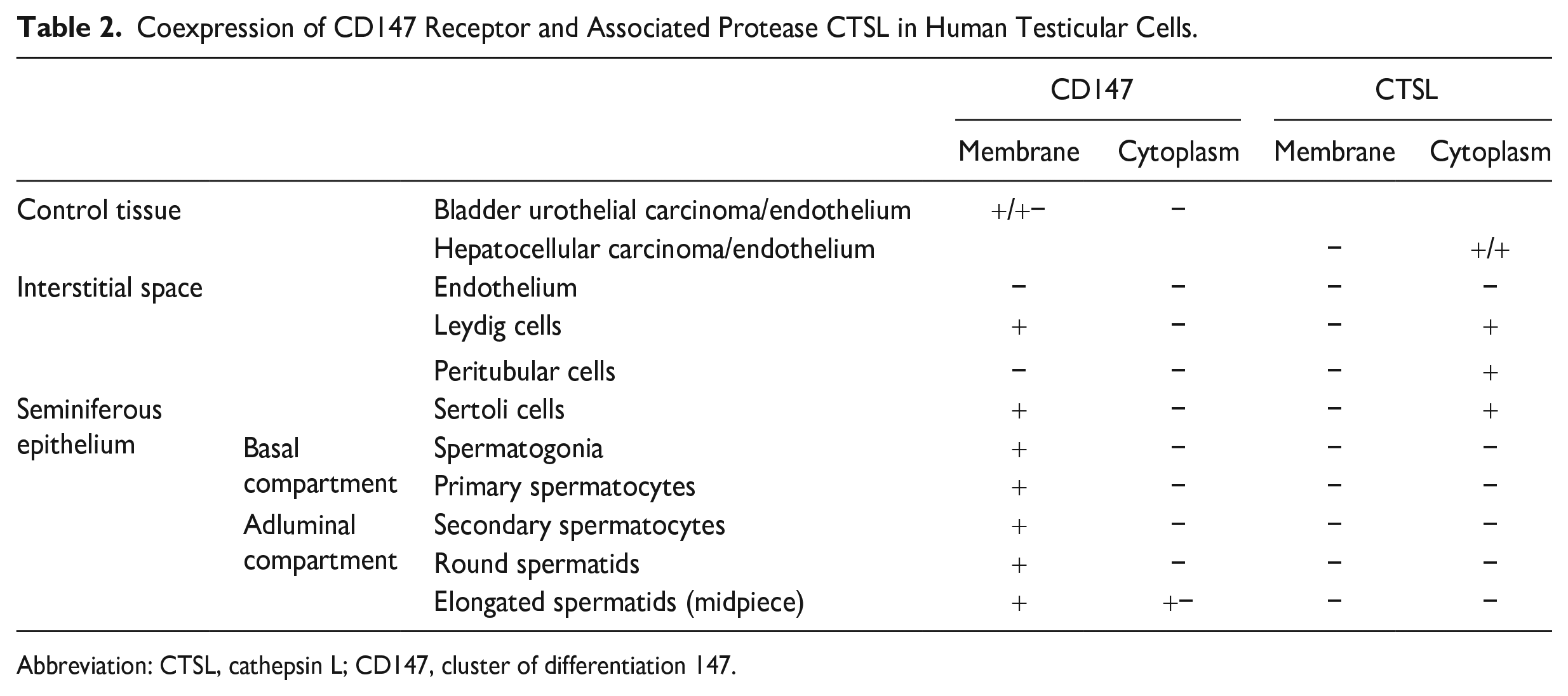
Abbreviation: CTSL, cathepsin L; CD147, cluster of differentiation 147.
CTSL Protein Expression in Human Testicular Cells
Cathepsins, a particular class of protease, recycle cellular and matrix proteins within lysosomes. 30 The testis’ epithelium has been characterized as harboring CTSL,52,57 a cysteine protease that is primarily responsible for degrading protein antigens derived from pathogens’ endocytosis.61,63
CD147-associated protease, CTSL IHC was performed in 15 cases (5 of SCOS, 5 of MA, and 5 of HP). Hepatocellular carcinoma tissue was included for comparison. The CTSL protein was detected in the cytoplasm of tumor cells and endothelium (Fig. S4).
In testicular tissue, evident staining was observed in the membrane of small cytoplasmic vesicles in PTCs, LCs, and SCs (basal region). No staining was observed in the endothelium or germ cells (Figs. 8–15; Table 2).
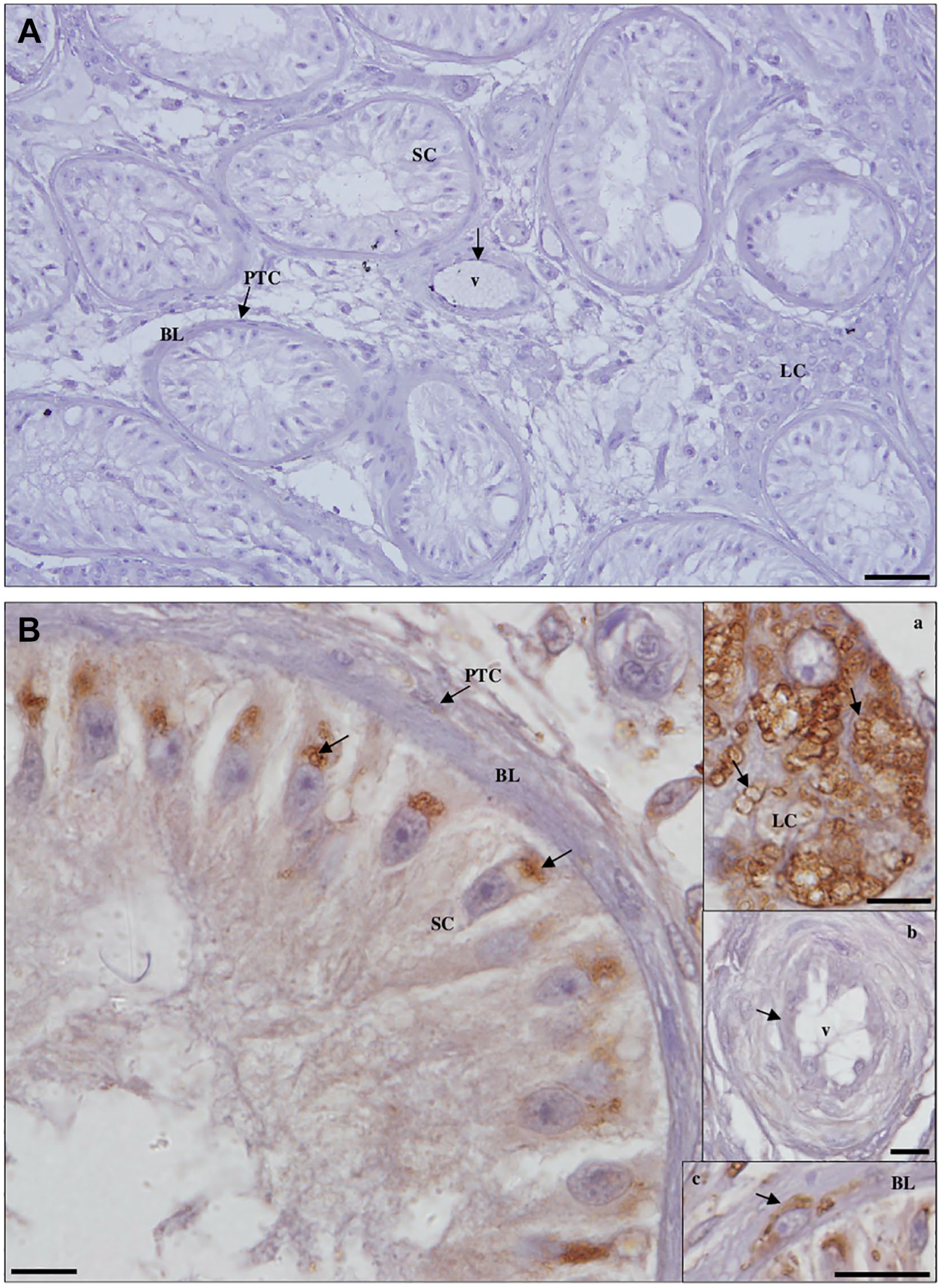
Detection of cathepsin L protein in Sertoli cell-only syndrome testicular tissue by immunohistochemistry. (A) Negative control. Scale bar, 50 µm. (B) Staining is observed in the membrane of cytoplasmic vesicles in Leydig cells (LCs; a, inset) and peritubular cells (PTCs; c, inset; scale bar, 20 µm) of the basal lamina (BL), being in Sertoli cells (SCs) concentrated at the cytoplasmic base. The endothelium (arrow) of blood vessels (v; b, inset) did not stain. Scale bars, 10 µm.
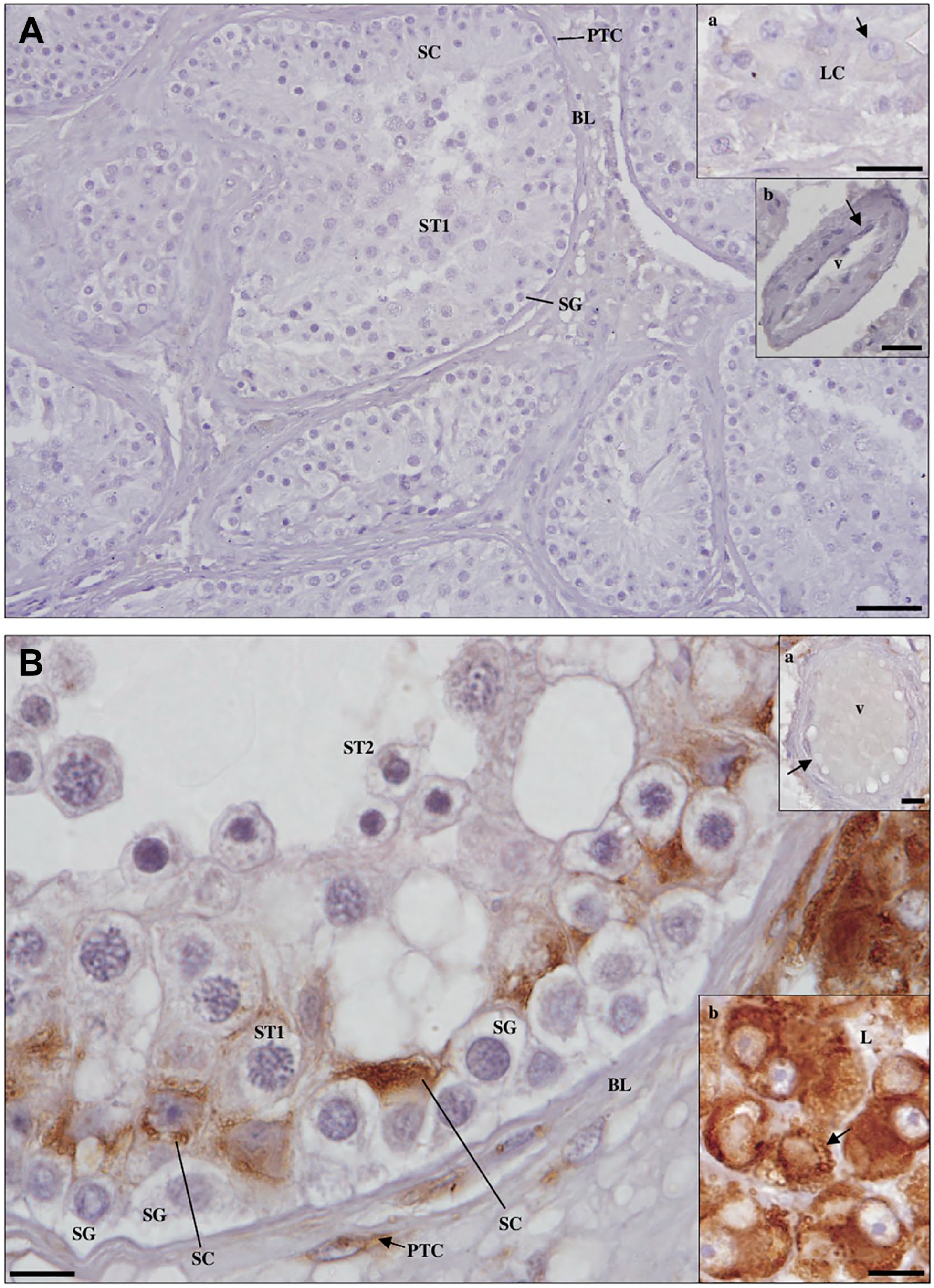
Detection of cathepsin L protein in maturation arrest testicular tissue by immunohistochemistry. (A) Negative control. Scale bar, 50 µm. (a, inset): Leydig cells (LCs); (b, inset): blood vessel (v). Scale bars, 20 µm. (B) Staining is observed in the membrane of cytoplasmic vesicles in LCs (b, inset) and peritubular cells (PTCs) of the basal lamina (BL), being in Sertoli cells limited to the basal cytoplasm. The endothelium (arrow) of v (a, inset), spermatogonia (SG), primary spermatocytes (ST1), and secondary spermatocytes (ST2) did not stain. Scale bars, 10 µm.
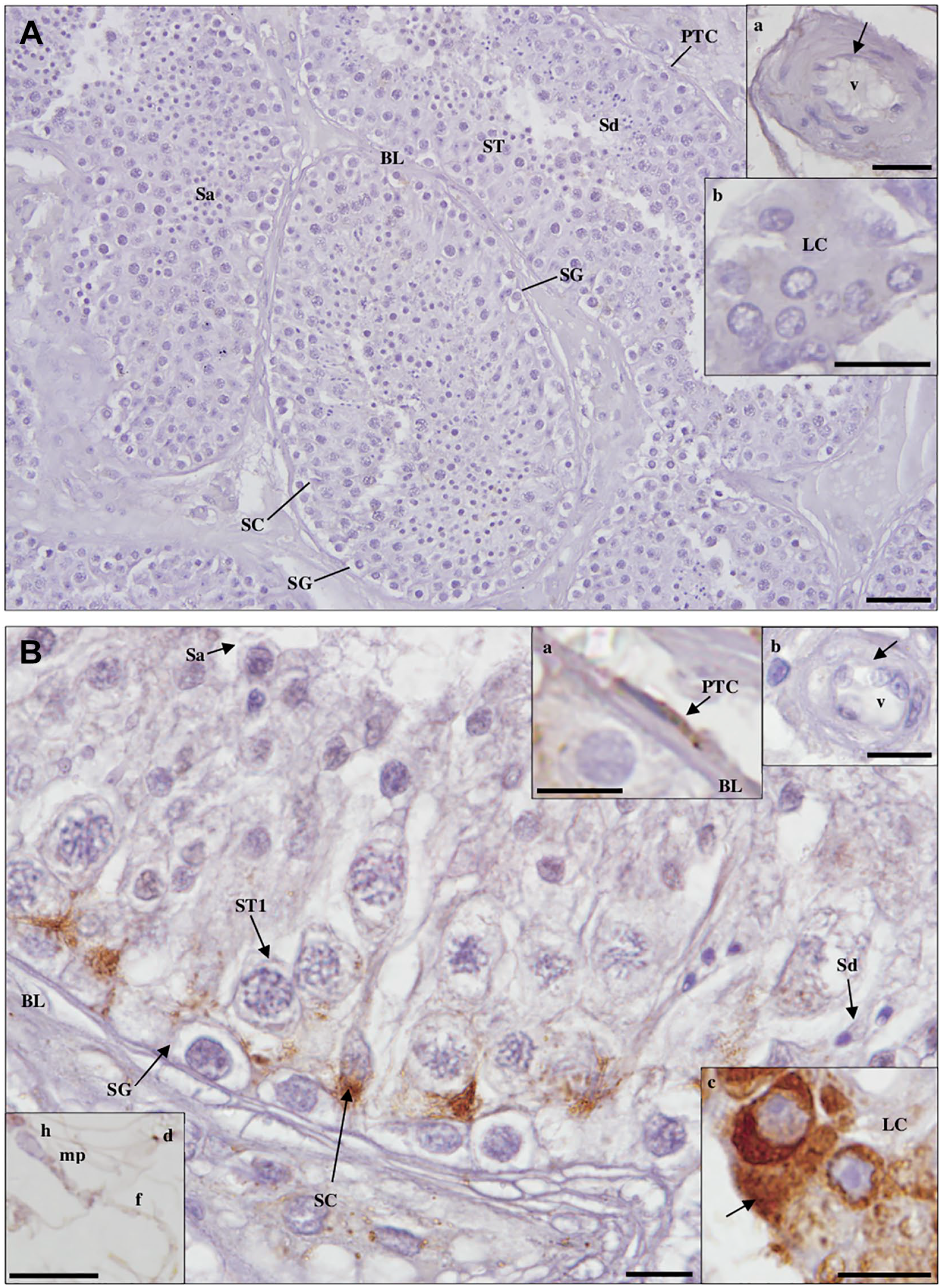
Detection of cathepsin L protein in hypospermatogenesis testicular tissue by immunohistochemistry. (A) Negative control. Scale bar, 50 µm. (a, inset): blood vessel (v); (b, inset): Leydig cell (LC). Scale bars, 20 µm. (B) Staining is observed in the membrane of cytoplasmic vesicles in LCs (c, inset) and peritubular cells (PTCs; a, inset, scale bar, 20 µm.) of the basal lamina (BL), being in Sertoli cells (SCs) concentrated at the cytoplasmic base. Spermatogonia (SG), primary spermatocytes (ST1), round spermatids (Sa), elongated spermatids (Sd), and the endothelium (arrow) of v (b, inset) did not stain. (d, inset): spermatid head (h), midpiece (mp), and flagellum (f). Scale bars, 10 µm.
Coexpression of CD147 Receptor and CTSL Protease in Human Testicular Cells
CD147-mediated entrance of SARS-CoV-2 into the host cell involves the intracellular processing of SARS-CoV-2 S protein, which involves the lysosomal cysteine proteases cathepsins, namely, the CTSL.63,64
The coexpression of CD147 and CTSL in the human testis was limited to somatic SCs, which play a role in testis formation and provide an essential environment for spermatogenesis, 65 and LCs, which produce androgen and play a key role in spermatogenesis regulation. 66
Coexpression of ACE2 Receptor and CTSL Protease in Human Testicular Cells
ACE2 and CTSL coexpression was also found to be restricted to the interstitial space, specifically in SCs, LCs, and PTCs, the latter of which not only provide structural integrity to the seminiferous tubule and are involved in the transport of spermatozoa and testicular fluid but also play a role in the regulation of spermatogenesis and testicular function by secreting a variety of substances including extracellular matrix components and growth factors. 67
Discussion
Data indicating that SARS-CoV-2 infection causes testicular complications is insufficient and contradictory. 68 The initial steps of SARS-CoV-2 infection involve receptor binding and fusion of the viral lipid envelope with cellular membranes, specifically through S glycoprotein. 69 According to reports, ACE2 is the primary host receptor responsible for binding SARS-CoV-2.31,70 Other host proteins, such as CTSL, which has been shown to facilitate S cleavage and promote infection, 31 or CD147, whose role in binding S is debatable,43,58 may also influence virus entry and infectivity. Tissue tropism is likely influenced by the cell-type-specific expression of proteins and other molecules involved in SARS-CoV-2 binding and fusion. 71
SARS-CoV-2 primarily infects cells in the respiratory tract but can also spread to other organs. The most sensitive methods for detecting the presence of SARS-CoV-2 in tissues are RT-qPCR-based methods that detect viral mRNA, and viral mRNA has been detected outside of the respiratory tract, including in the testis. The presence of viral mRNA does not always imply the presence of an infectious virus, but the presence of transcripts and proteins in testicular cells was anticipated to be standardized by large-scale single-cell research, which has resulted in the creation of huge databases through consortiums on transcriptomics and proteomics. However, different outcomes showed that this was not the case. In addition, numerous authors reported their findings for the testis without mentioning the precise cellular level of expression or spatial location within the cell. Moreover, although both the receptor and the protease are required for viral entry into the host cell, coexpression for either ACE2/TMPRSS2 or CD147/CTSL was not usually observed (Tables 3–5). Furthermore, despite the fact that some testicular cells express significant levels of receptor mRNA or protein, no such protease expression was detected, contributing to the contradictory results.
mRNA Expression of Receptor ACE2 and Associated Protease TMPRSS2 in Testicular Cells Based on Large-Scale Databases.
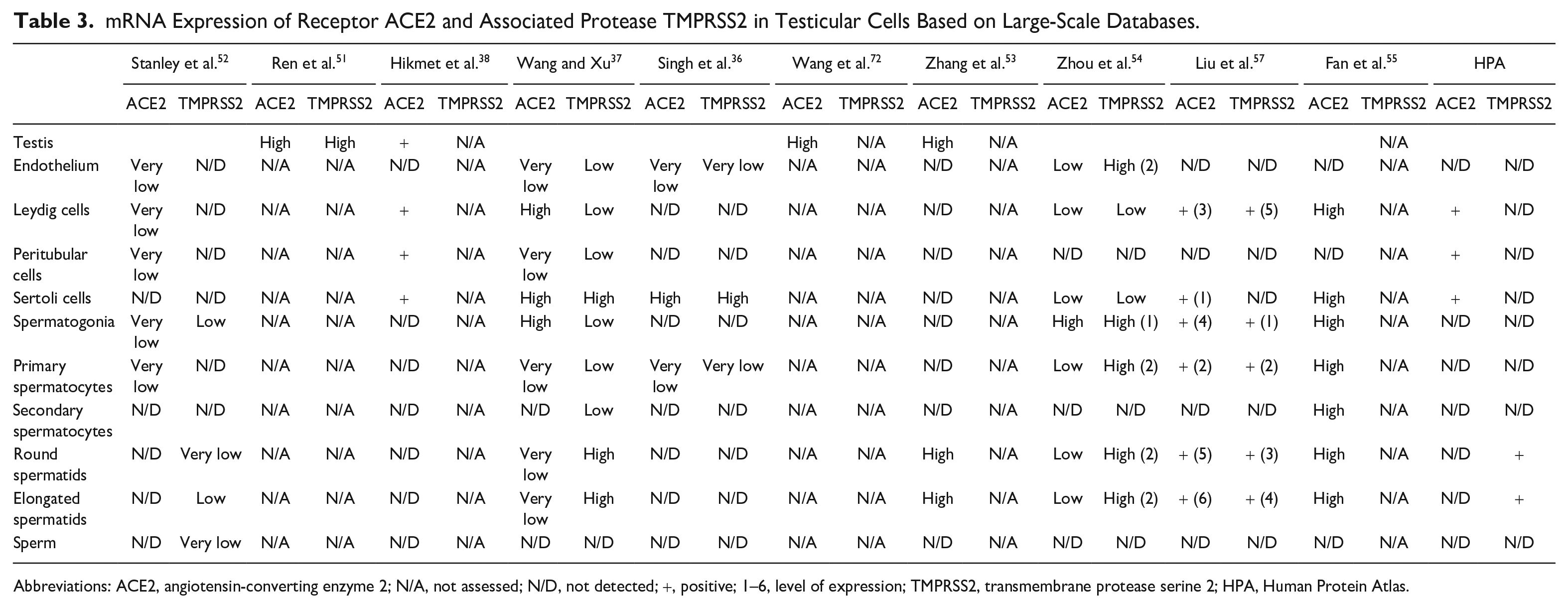
Abbreviations: ACE2, angiotensin-converting enzyme 2; N/A, not assessed; N/D, not detected; +, positive; 1–6, level of expression; TMPRSS2, transmembrane protease serine 2; HPA, Human Protein Atlas.
Large-Scale Database Results Regarding the Protein Expression of Receptor ACE2 and Associated Protease TMPRSS2 in Testicular Cells.
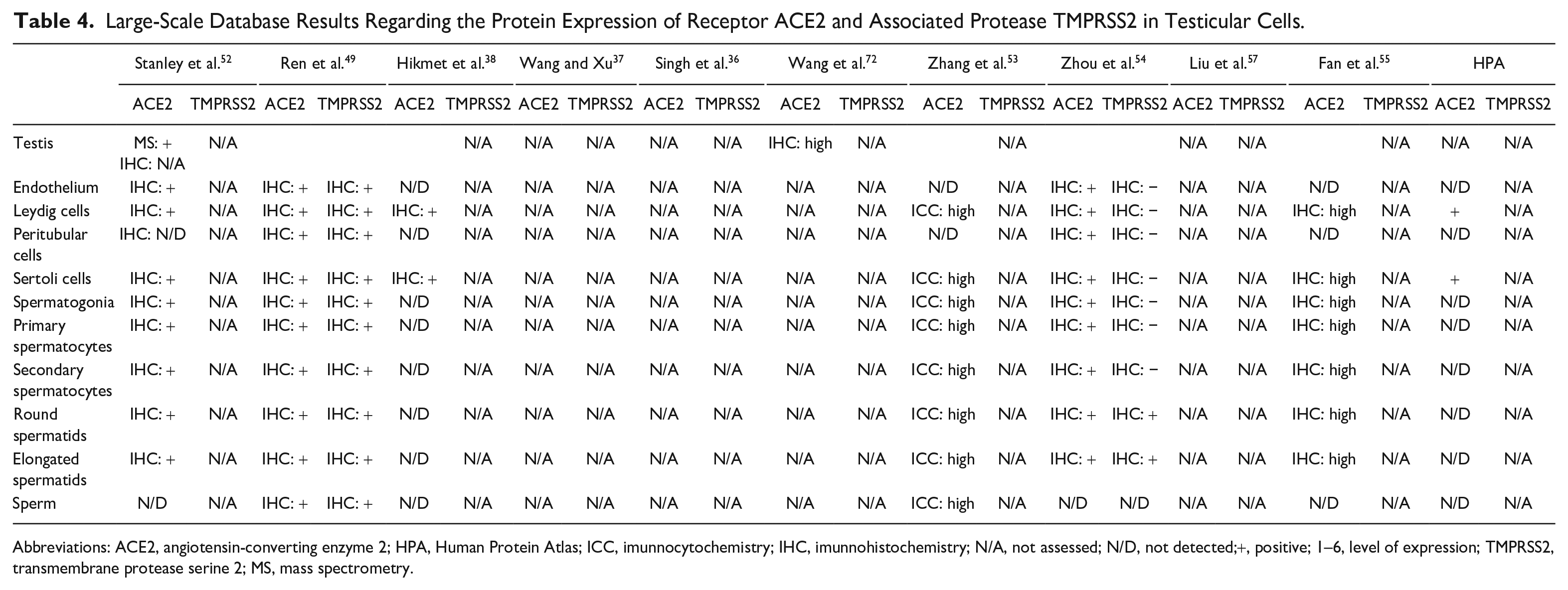
Abbreviations: ACE2, angiotensin-converting enzyme 2; HPA, Human Protein Atlas; ICC, imunnocytochemistry; IHC, imunnohistochemistry; N/A, not assessed; N/D, not detected;+, positive; 1–6, level of expression; TMPRSS2, transmembrane protease serine 2; MS, mass spectrometry.
Expression of Receptor CD147 and Associated Protease CTSL in Testicular Cells Based on Large-Scale Databases.
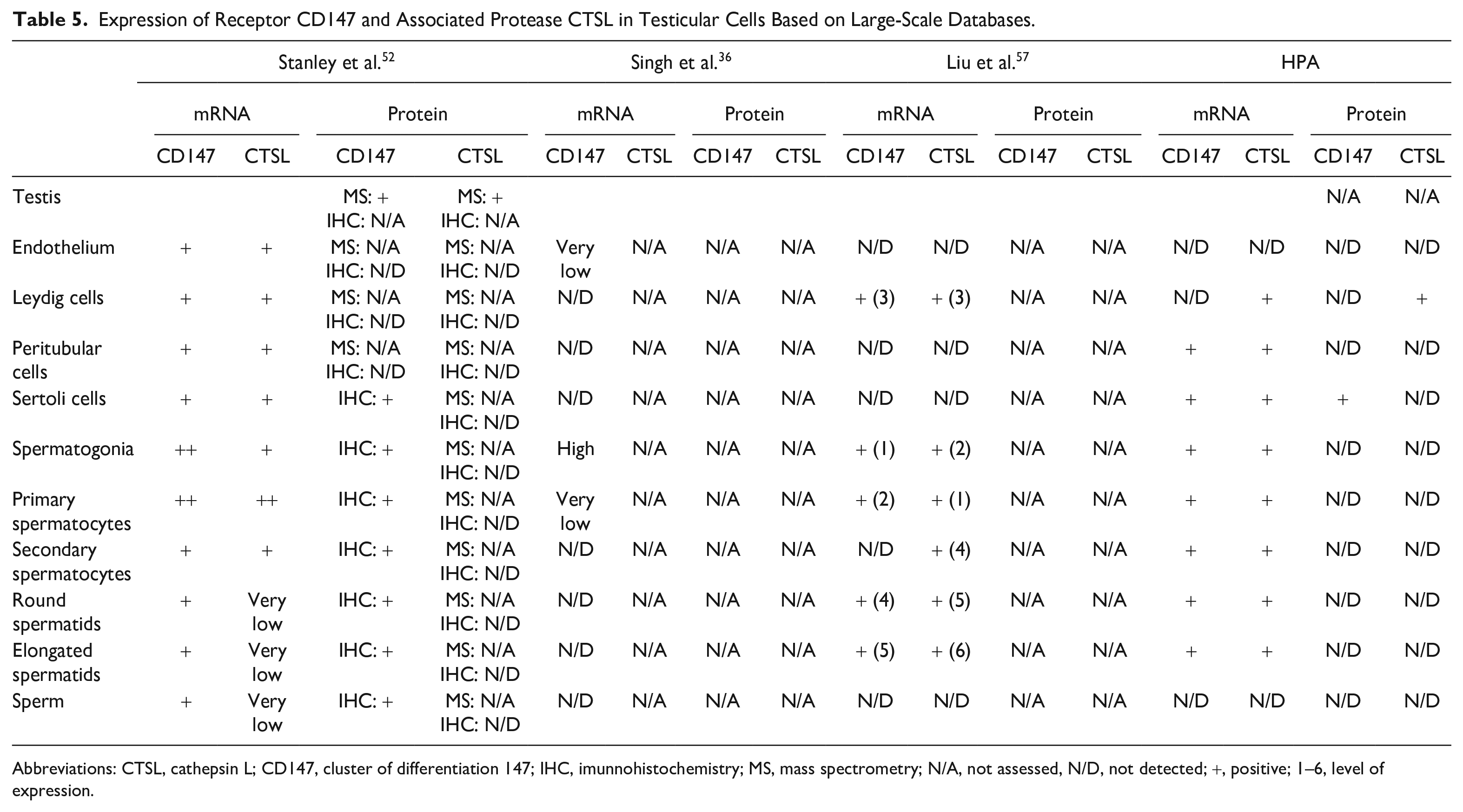
Abbreviations: CTSL, cathepsin L; CD147, cluster of differentiation 147; IHC, imunnohistochemistry; MS, mass spectrometry; N/A, not assessed, N/D, not detected; +, positive; 1–6, level of expression.
IHC and immunofluorescence (IF) study of cells and tissues can give a last piece of information to these extensive analyses despite lacking the capability of proteomics. Large-scale databases make use of different antibodies, and the few IHC and IF reports that do not use these large-scale databases also use a variety of antibodies. Given that a single antibody or a single combination of antibodies may not be representative, as the prior findings revealed by the various staining patterns acquired, this should not be viewed as a problem but rather as a significant addition of information.
Our most recent evaluation of IHC and IF publications that did not use extensive databases revealed a severe lack of thorough characterisation of the various testicular cells, mostly because there were no high-resolution images available. This inspired us to use high-resolution microscopy and a novel panel of antibodies to define the morphological spatial placement of the SARS-CoV-2 receptors (ACE2 and CD147) and related proteases (TMPRSS2 and CTSL) in order to provide a more accurate testicular cell characterisation. Four new antibodies that were not previously used in the investigation of the geographic distribution of SARS-CoV-2 receptors and associated proteases are added in this work.
Our results show that SARS-CoV-2 receptors and their S protein priming-host cell proteases are expressed specifically in the human testis at both seminiferous tubule epithelium and testicular interstitial cells.
The current findings confirm the presence of the ACE2 receptor and its associated protease, TMPRSS2, as well as demonstrate the existence of the alternative receptor CD147 and validate the presence of its related protease, CTSL, in LCs. The results further corroborate the presence of the primary SARS-CoV-2 receptor, ACE2, and the protease CTSL, in SCs, as well as demonstrate the presence of the alternative receptor CD147 and the primary protease, TMPRSS2, for the first time. This coexpression of SARS-CoV-2 receptors and respective proteases in LCs and SCs are in line with the risk of hypogonadism highlighted in COVID patients. The assessment of male gonadal dysfunction in patients with active COVID-19 revealed significantly increased serum luteinizing hormone (LH) levels and significantly decreased serum testosterone (T) levels to LH ratio, as well as serum levels of follicle-stimulating hormone (FSH) to LH ratio, causing a hypergonadotropic hypogonadism.73–77 Indeed, in the absence of T, FSH is not sufficient to maintain spermatogenesis. The altered hormonal status in patients infected by SARS-CoV-2 has been attributed to injuries at the level of LCs and SCs, but the altered hypothalamus-pituititary-testis axis may be attributed to disease anxiety and the use of corticosteroids in the disease management. Fortunately, reports have shown that hormone levels return to normal after recovery from the disease.7,13
The fact that the current findings were also obtained in SCOS, a pathology first described by Del Castillo in 1947 as having no germ cells, SCs with normal cytoplasm within seminiferous tubules, no histological degeneration in the testes, and a reduced testicular volume with normal secondary sexual characteristics, 78 provides confirmation that elevated serum LH, as well as FSH and lowered serum T, indicates LC and germ cell failure. 79 Nonetheless, the long-term risk of hypotestosteronemia and hypogonadism in recovered-COVID patients needs to be investigated.
The current findings also corroborate the presence of the receptor CD147 in SG, spermatocytes, and spermatids, as well as the presence of the main SARS-CoV-2 receptor, ACE2, and its main associated protease, TMPRSS2, for the first time. However, they do not support the presence of CTSL protease in SG, spermatocytes, or spermatids, ruling out coexpression of CD147 and CTSL in the seminiferous tubule epithelium.
Despite the presence of the ACE2 and CD147 receptors, as well as the protease TMPRSS2, in seminiferous tubule epithelium cells, the virus has not been found in the semen of infected patients, whether recovering or in the acute phase of the disease, nor has viral RNA been found in a testicular biopsy performed on a COVID-19 patient. 9 Our findings are consistent with the findings of Shen et al., who discovered that the expression of ACE2 in the testis is related to age, with a peak around 30 years and a low from 60 years onward. 80 Even so, as all receptors and proteases studied were found to be coexpressed in LCs and SCs, older men and young children are not immune to the risk of reproductive disorders, particularly those governed by endocrine regulation. Furthermore, 1 month after recovery from COVID-19, patients with moderate symptoms had lower sperm quality, with one presenting testicular discomfort, than those with mild symptoms who did not require hospitalization and showed no alterations in sperm quality. 10 However, other studies found no changes in semen parameters in patients who tested positive for SARS-CoV-2. 8 This report’s clarification of the existence of SARS-CoV-2 receptors and proteases on testicular cells confirms the SARS-CoV-2 tropism for the human testis in addition to the airways, but its presence do not necessarily lead to virus infection of the testis. Although SARS-CoV-2 appears to be unable to cross the human blood-testis-barrier (BTB) or have access to the seminiferous tubule epithelium, if we look at the general principles of viral uptake by the testis, the male reproductive system is indeed vulnerable to viral infections because the BTB does not completely prevent virus entry. 81 Whether the testicular effects of a virus are minor or part of a larger syndrome, it should be noted that viruses such as Zika, Ebola, human immunodeficiency virus, and human papilloma virus have been found in testis and semen.82,83 It remains unknown where SARS-CoV-2 falls on this spectrum.
Electron micrographs of autopsied patient tissues revealed SARS-CoV-2 viral-like particles inside endothelial cells. 84 However, it is unclear whether the presence of virus inside the endothelial cell reflects viruses that are replicating or viruses that have only entered tissues.
Our findings also describe the presence of the ACE2 and TMPRSS2 proteins in endothelial cells and PTCs for the first time. Testicular endothelial cells play an important role in the germline stem cell niche. This is especially true for male cancer survivors after chemotherapy who are infertile after chemotherapy because spermatogonial stem cells are highly susceptible to cytotoxic injury. 85 This is not significant not only because of the treatment of COVID-19 patients and their effects on testis but also because of the hypoxemia state of COVID-19 patients, which is a marker of disease severity. 86 It has been reported that hypoxia conditions may cause BTB injury and immune infertility due to decreased ZO-1 protein levels and increased BTB permeability. 87 Other underlying mechanisms of SARS-CoV-2 infection, such as hypoxia-induced testicular, may thus exacerbate the massive cytokine release also in the testis via LCs and SCs, the only cells found to coexpress all four proteins studied. As a result, it remains unclear to what extent SCs elicit immune privilege to germinal cells via the BTB and suppress inflammation or limit virus-associated testicular damage.
The presence of the SARS-CoV-2 in testicular cells does not explain the contradictory results of the literature regarding male reproductive potential, but it is a critical piece of the puzzle to rule out the possibility of testicular cells being directly infected by SARS-CoV-2 or the BTB be ruptured, allowing the virus entry, anticipating an increased risk of the virus spreading through sperm. However, this is merely speculative, implying that more research is required.
The role of these receptors and proteases in normal spermatogenesis is also worth investigating further. Normal spermatogenesis is a well-organized process in which diploid spermatogonial stem cells collaborate with somatic cells such as SCs, PTCs, and LCs to differentiate into spermatocytes and haploid spermatozoa. Despite the fact that we were able to determine the expression of the four proteins using three NOA histopathologies, we are aware that the roles and mechanisms of genes in regulating normal and abnormal spermatogenesis may differ, making this study limited. Even so these pathologies allowed for a unique identification of the SARS-CoV-2 receptors and proteases identification in testicular cells.
Supplemental Material
sj-tif-1-jhc-10.1369_00221554231168916 – Supplemental material for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells
Supplemental material, sj-tif-1-jhc-10.1369_00221554231168916 for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells by Maria Rita Ribeiro, Ana Margarida Calado, Ângela Alves, Rute Pereira, Mário Sousa and Rosália Sá in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-2-jhc-10.1369_00221554231168916 – Supplemental material for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells
Supplemental material, sj-tif-2-jhc-10.1369_00221554231168916 for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells by Maria Rita Ribeiro, Ana Margarida Calado, Ângela Alves, Rute Pereira, Mário Sousa and Rosália Sá in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-3-jhc-10.1369_00221554231168916 – Supplemental material for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells
Supplemental material, sj-tif-3-jhc-10.1369_00221554231168916 for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells by Maria Rita Ribeiro, Ana Margarida Calado, Ângela Alves, Rute Pereira, Mário Sousa and Rosália Sá in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-4-jhc-10.1369_00221554231168916 – Supplemental material for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells
Supplemental material, sj-tif-4-jhc-10.1369_00221554231168916 for Spatial Distribution of SARS-CoV-2 Receptors and Proteases in Testicular Cells by Maria Rita Ribeiro, Ana Margarida Calado, Ângela Alves, Rute Pereira, Mário Sousa and Rosália Sá in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
The authors would like to thank António Couceiro, MD, Department of Pathology, Centro Hospitalar de Vila Nova de Gaia/Espinho, for testicular tissue samples; Paula Monteiro, MD, PhD (Director), Department of Pathology, Instituto Português de Oncologia do Porto Francisco Gentil EPE (IPO Porto), Porto, Portugal, for cancer tissue samples; Paula Teixeira, Department of Microscopy Lab technician, for all the support in the laboratory; and Nuno Mendes, PhD, IPATIMUP-I3S, for kidney cell lines that were used previously to the present study.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: conceptualization (RS), methodology (MRR, RS, MS), formal analysis (MRR, MS, RS), investigation (MRR, RS), technical assistance (AA), visualization (MRR, MS, RS), supervision (MS, RS), writing-original draft (MRR), writing-review and editing (MS, RS), critical review of the final manuscript (MRR, AMC, RP, RS), resources (MS), funding acquisition (MS). All authors have read and agreed to the order of appearance and of the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ICBAS-UP and UMIB/ITR. The UMIB-Unit for Multidisciplinary Research in Biomedicine is funded by the Foundation for Science and Technology (FCT) Portugal (grant numbers UIDB/00215/2020 and UIDP/00215/2020) and ITR-Laboratory for Integrative and Translational Research in Population Health (LA/P/0064/2020).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
