Abstract
Giant cell tumors of bone (GCTBs) are locally aggressive tumors with the histological features of giant cells and stromal cells. Denosumab is a human monoclonal antibody that binds to the cytokine receptor activator of nuclear factor–kappa B ligand (RANKL). RANKL inhibition blocks tumor-induced osteoclastogenesis, and survival, and is used to treat unresectable GCTBs. Denosumab treatment induces osteogenic differentiation of GCTB cells. In this study, the expression of RANKL, special AT-rich sequence-binding protein 2 (SATB2, a marker of osteoblast differentiation), and sclerostin/SOST (a marker of mature osteocytes) was analyzed before and after treatment with denosumab in six cases of GCTB. Denosumab therapy was administered a mean of five times over a mean 93.5-day period. Before denosumab treatment, RANKL expression was observed in one of six cases. After denosumab therapy, spindle-like cells devoid of giant cell aggregation were RANKL-positive in four of six cases. Bone matrix–embedded osteocyte markers were observed, although RANKL was not expressed. Osteocyte-like cells were confirmed to have mutations, as identified using mutation-specific antibodies. Our study results suggest that treatment of GCTBs with denosumab results in osteoblast–osteocyte differentiation. Denosumab played a role in the suppression of tumor activity via inhibition of the RANK–RANKL pathway, which triggers osteoclast precursors to differentiate into osteoclasts.
Introduction
Giant cell tumors of bone (GCTBs) are locally aggressive and typically occur in young adults. The most common site of origination around the knee joint is the metaphysis to the epiphysis. Histologically, GCTB is composed of neoplastic stromal cells and reactive multinucleated osteoclast-like giant cells. 1 The rate of recurrence of GCTB ranges from 30% to 50% after curettage. 2 The rate of recurrence after curettage with adjuvant therapies—including after the use of liquid nitrogen, phenol, ethanol, and polymethylmethacrylate bone cement—ranges from 7% to 10%.2–4 Approximately 85–95% of the GCTB cases harbor a histone H3F3A (H3.3) gene mutation, resulting in a substitution of glycine 34-to-tryptophan (G34W). A minor subset of H3F3A mutations harbor other substitutions at the rate of 1–2% that include G34L/M/R/V.5–7 Immunohistochemical analysis to detect the H3F3A mutation has been of diagnostic value in the G34W substitution. 8
The receptor activator of nuclear factor–kappa B (RANK) ligand (RANKL), a transmembrane protein from the tumor necrosis factor superfamily, plays an important role in osteoclastogenesis.9,10 Denosumab is a human monoclonal antibody that targets RANKL, a key bone resorption mediator. Denosumab strongly suppresses bone resorption by downregulating the differentiation and activation of osteoclasts and is widely used for the treatment of osteoporosis and bone metastasis.11,12 The expression of RANKL by neoplastic stromal cells promotes the recruitment of reactive multinucleated osteoclast-like giant cells, leading to the osteolytic nature of GCTB. Indications for the treatment of GCTBs are not limited to unresectable cases, such as recurrent lesions of the spine and pelvis, but also cases with the metastatic lung lesions. 13
Histological changes after treatment with denosumab are depicted by the characteristic disappearance of multinucleated osteoclast-like giant cells and the prominent proliferation of spindle cells. Bone formation is also recognized within the lesion, with the osteoid matrix resembling osteosarcoma-like tissue. These findings suggest that treatment of GCTBs with denosumab results in osteoblastic differentiation and bone formation. 8 In this study, RANKL expression is analyzed for biological activity as well as osteoblast differentiation to confirm the hypothesis that tumor cells differentiate into osteoblasts–osteocytes following treatment with denosumab.
Materials and Methods
Materials
Among the GCTB cases treated surgically in our hospital, those treated preoperatively with denosumab for 60 days or more were examined. Before surgery, the diagnosis of GCTB was confirmed histologically via the use of a core needle or open biopsy. The diagnosis was confirmed using resected specimens. Preoperatively, denosumab was injected subcutaneously. The dosing schedule was 120 mg every 4 weeks on days 1, 8, 15, 29, and 57. The injection regimen was not strictly regulated, and the injection dates were variable. Despite this, the interval between injections did not exceed 1 month. 14 This study was approved by the institutional review board of Kyoto University (R2499).
Immunohistochemical Analysis
Immunohistochemistry was performed on formalin-fixed, paraffin-embedded specimens using an automated immunostainer (BenchMark Ultra, Ventana Medical Systems, Tucson, AZ). The primary antibodies used were those against H3.3G34W (clone RM263, dilution 1:100, RevMAb Biosciences, South San Francisco, CA), RANKL (Polyclonal, dilution 1:100, RayBiotech, Peachtree Corners, GA), special AT-rich sequence-binding protein 2 (SATB2; EP281, dilution 1:100, Cell Marque, Rocklin, CA), and sclerostin/SOST (Polyclonal, dilution 1:100, Proteintech, Rosemont, IL).
The nuclear expression of histone H3.3 G34W, sclerostin/SOST, and SATB2 antibodies was evaluated, and the cytoplasmic expression of RANKL was assessed before and after denosumab therapy. Nuclear staining in more than 50% of tumor cells was interpreted as a positive result for RANKL expression. The faint expression of RANKL was judged to be negative. After denosumab administration, osteocyte-like cells in bone tissue and spindle cells were assessed separately.
Tartrate-resistant Acid Phosphatase Activity
Deparaffinized sections were incubated at 37C in 0.1 M acetate buffer (pH 5) containing 220 μM naphthol AS-MX phosphate/dimethyl formaldehyde solution, 2 mM fast red violet LB salt, 50 mM l-(+)-sodium tartrate, and 1 M MgCl2 for 30 min (all Sigma agents). Sections were subsequently counterstained with hematoxylin.
Results
Clinical Features
GCTBs showed a 2:1 incidence of male to female ratio. The mean age of patients was 41.2 years (range, 26–61 years). GCTB was diagnosed in each case at the humerus (proximal), ring finger, femur (distal), tibia (proximal), lumbar spine (L1), or sacrum. According to Campanacci’s classification, the severity of cases was grade 1 (latent, 0 cases), grade 2 (active, 4 cases), and grade 3 (aggressive, 2 cases) (Table 1). The mean number of doses of denosumab received was 5 (range, 4–6). The mean duration between doses was 93.5 days (range, 66–101 days) (Table 2).
Summary of Clinical Data for Six Giant Cell Tumor of Bone Cases.
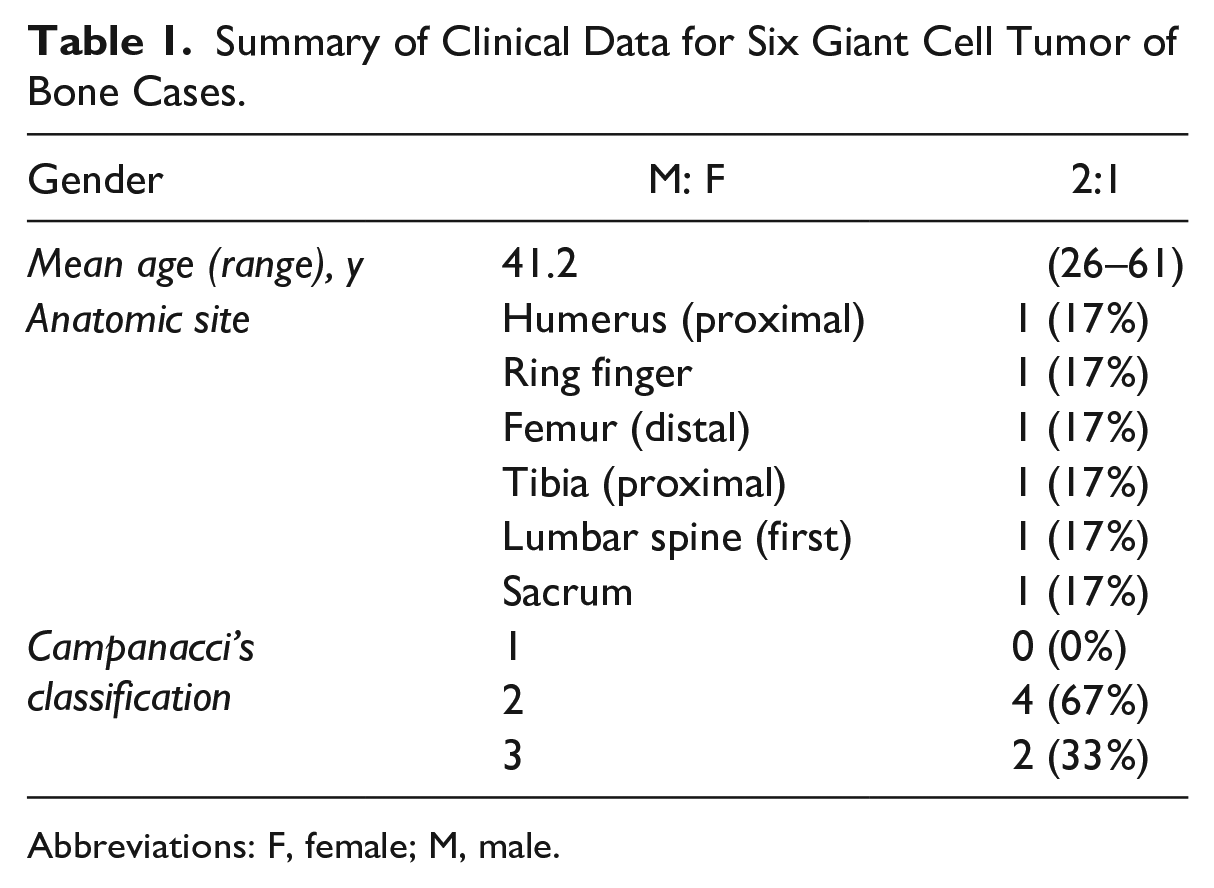
Abbreviations: F, female; M, male.
RANKL Expression in Giant Cell Tumor of Bone Pre- and Post-denosumab Treatment.
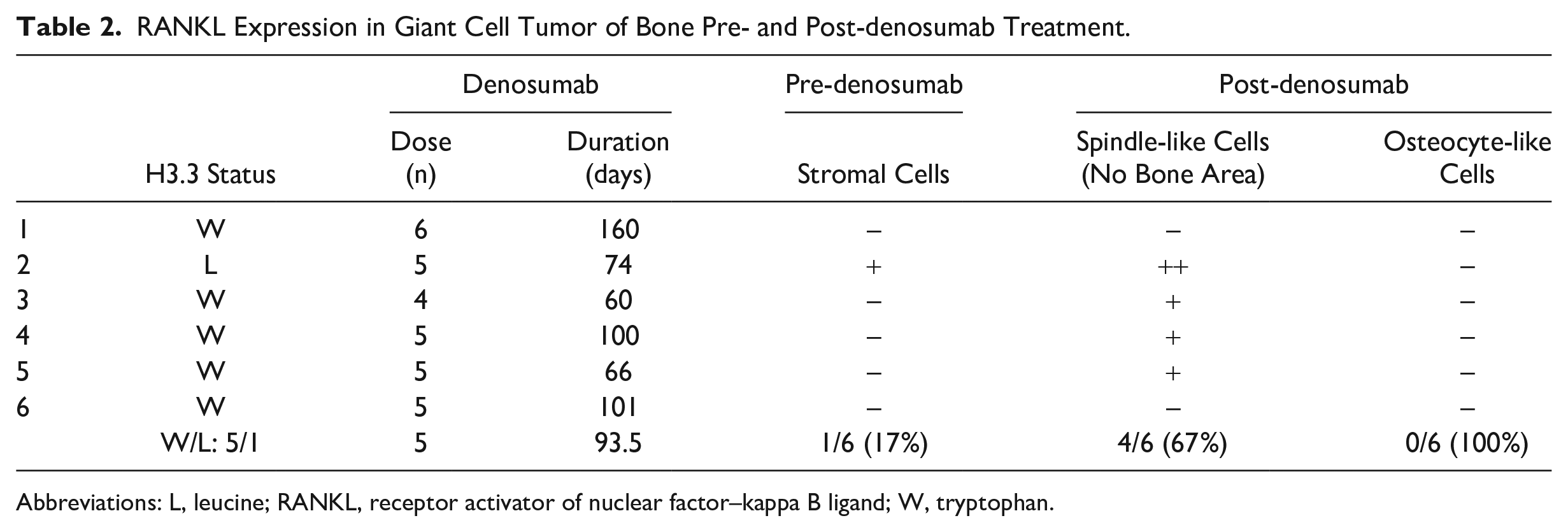
Abbreviations: L, leucine; RANKL, receptor activator of nuclear factor–kappa B ligand; W, tryptophan.
Histological Features
All six biopsied GCTB specimens had typical neoplastic stromal cells and multinucleated osteoclast-like giant cells (Fig. 1). After denosumab treatment, histologically evaluated GCTBs exhibited the following typical morphological changes that included the depletion of multinucleated giant cells and an area of spindle cell proliferation with the appearance of osteoid and bone tissue. Spindle cell proliferation was arranged in a storiform pattern similar to fibrohistiocytic tumors. Formed bone comprised immature and mature bone matrix and was rimmed by osteoblast-like cells such that the embedded cells looked like osteocytes (osteocyte-like cells).
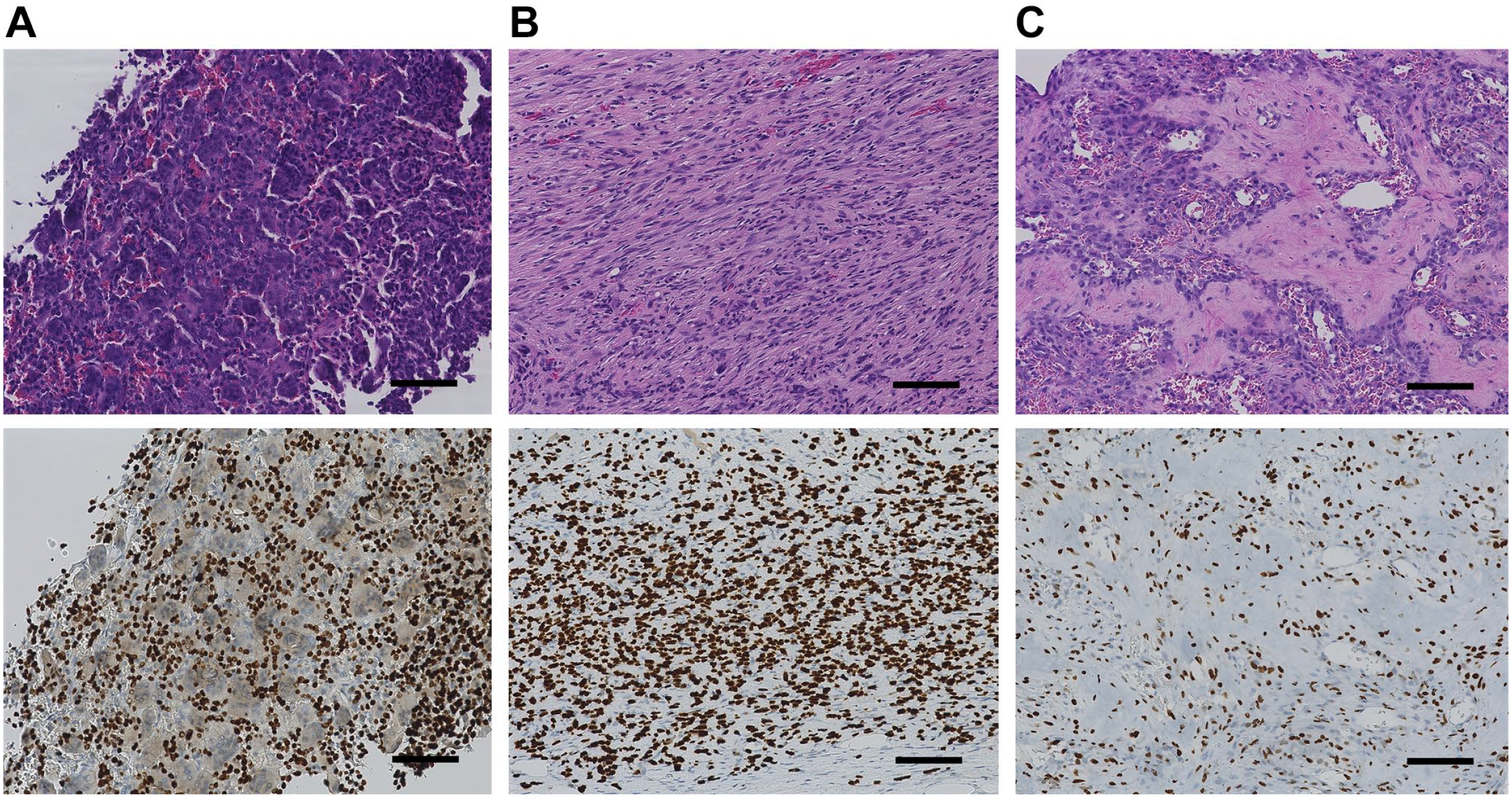
Giant cell tumor of bone. Microscopic findings of giant cell tumors are composed of multinucleated giant cells and mononuclear stromal cells (A-top). After denosumab treatment, the giant cells disappear, and spindle cell proliferation becomes prominent (B-top) and osteoid formation is observed (C-top). Immunohistochemistry for the histone H3.3 G34W antibody was positive in mononuclear stromal cells but negative in multinucleated giant cells (A-bottom). After denosumab treatment, histone H3.3 G34W antibody positivity was diffused in spindle cells (B-bottom) and osteocyte-like cells embedded in bone tissue (C-bottom) (scale bar = 100 µm).
H3.3 G34W Antibody Expression and H3.3 Mutation Status
H3.3 G34W positivity was only observed in stromal cells, but not in the multinucleated giant cells of five of the six cases analyzed. The negative result in response to the H3.3 G34W antibody was because the amino acid glycine (G) mutated to leucine (L) by direct sequence. After denosumab administration, H3.3 G34W antibody positivity persisted in all the same cases that were positive before denosumab administration. Immunoreactivity of H3.3 G34W was diffusely positive in fibroblast-like cells. Osteocyte-like cells were embedded in trabeculae (Fig. 1). The same five cases were also positive for inter-trabecular spindle cells. The positive H3.3 G34W antibody findings suggest that the osteocyte-like cells were tumor cells of GCTB harboring the H3.3 G34W mutation.
Tartrate-resistant Acid Phosphatase Staining
Tartrate-resistant acid phosphatase (TRAP)-positive multinucleated giant cells and scattered mononuclear cells were positive in areas with less multinucleated giant cells. After denosumab treatment, TRAP-positive cells were absent in areas with spindle cells as well as in areas of bone formation (Fig. 2).
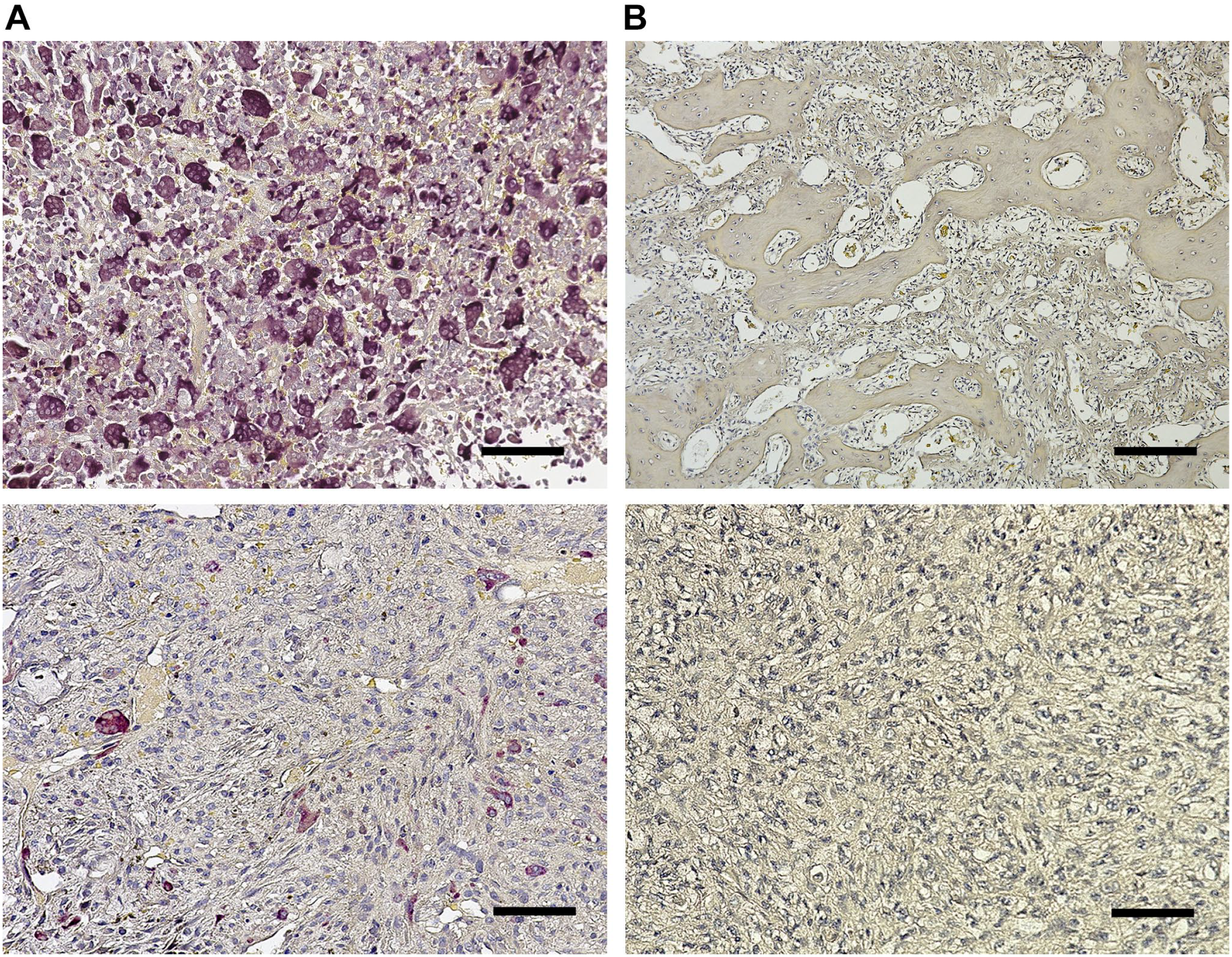
Giant cell tumor of bone. Before denosumab treatment, TRAP-positive multinucleated giant cells are scattered (A-top). Before denosumab treatment, TRAP-positive mononuclear cells resembling stromal cells in less multinucleated cells are recognized (A-bottom). After denosumab treatment, there are no TRAP-positive cells in the bone formation area (B-top) and the spindle cell area (B-bottom) (scale bar = 100 µm). Abbreviation: TRAP, tartrate-resistant acid phosphatase.
RANKL Expression
RANKL expression was observed in the cytoplasm of stromal cells in one of six GCTB cases (17%) before denosumab therapy. After denosumab treatment, RANKL expression was prominent in four of six GCTB cases (67%). In the bone formation area of all cases, RANKL was undetected in osteocyte cells embedded in immature and mature bone matrices (0%); although RANKL was expressed in spindle cells including those located in areas of bone formation (Table 2, Fig. 3).
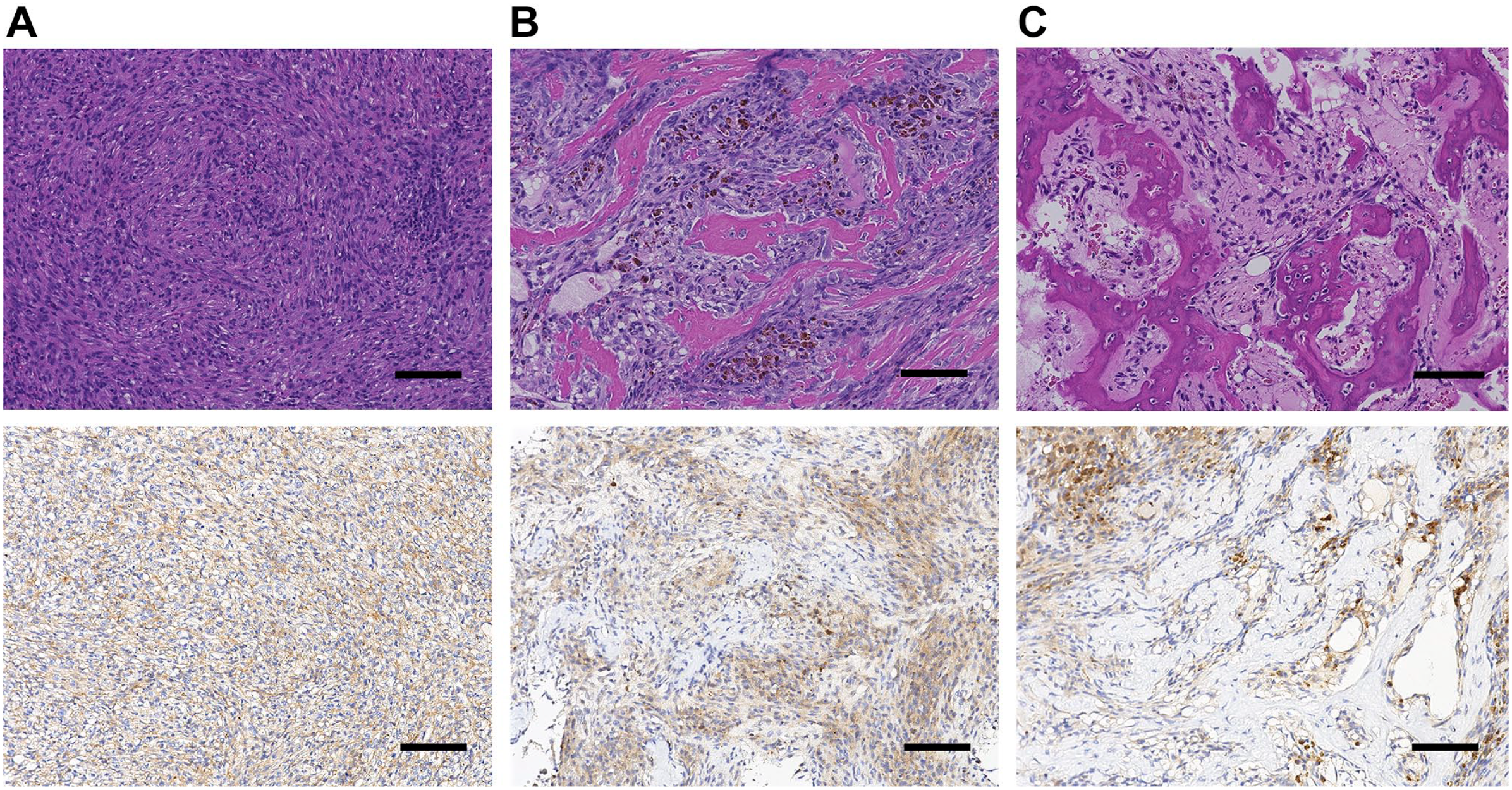
Giant cell tumor of bone. After denosumab treatment, RANKL expression was observed in the spindle cell area in this case (A). In areas of bone formation after denosumab treatment, RANKL expression was absent in osteocyte-like cells embedded in immature bone matrix (B) and mature bone matrix (C). Note the positive expression in intertrabecular spindle cells (B and C) (scale bar = 100 µm). Abbreviation: RANKL, receptor activator of nuclear factor–kappa B ligand.
Osteoblast and Osteocyte Lineage Markers of SATB2 and Sclerostin/SOST
The SATB2 is a reliable marker of osteoblast differentiation. Sclerostin/SOST, antagonistic of Wnt signaling, is secreted by osteocytes. 15 Before denosumab therapy, the osteoblast and osteocyte lineage markers, SATB2 and sclerostin/SOST, respectively, were detected in mononuclear stromal cells in four of six (67%) cases and three of six (50%) cases, in that order. After denosumab therapy, SATB2 and sclerostin/SOST expression were observed in three of six (50%) and six of six (100%) cases, respectively. In all six cases, osteoblast-like cells rimming bone trabeculae were positive for SATB2, and osteocyte-like embedded cells were positive for sclerostin/SOST (100%) (Table 3, Fig. 4).
SATB2 and Sclerostin/SOST Expression.

Abbreviation: SATB2, special AT-rich sequence-binding protein 2.
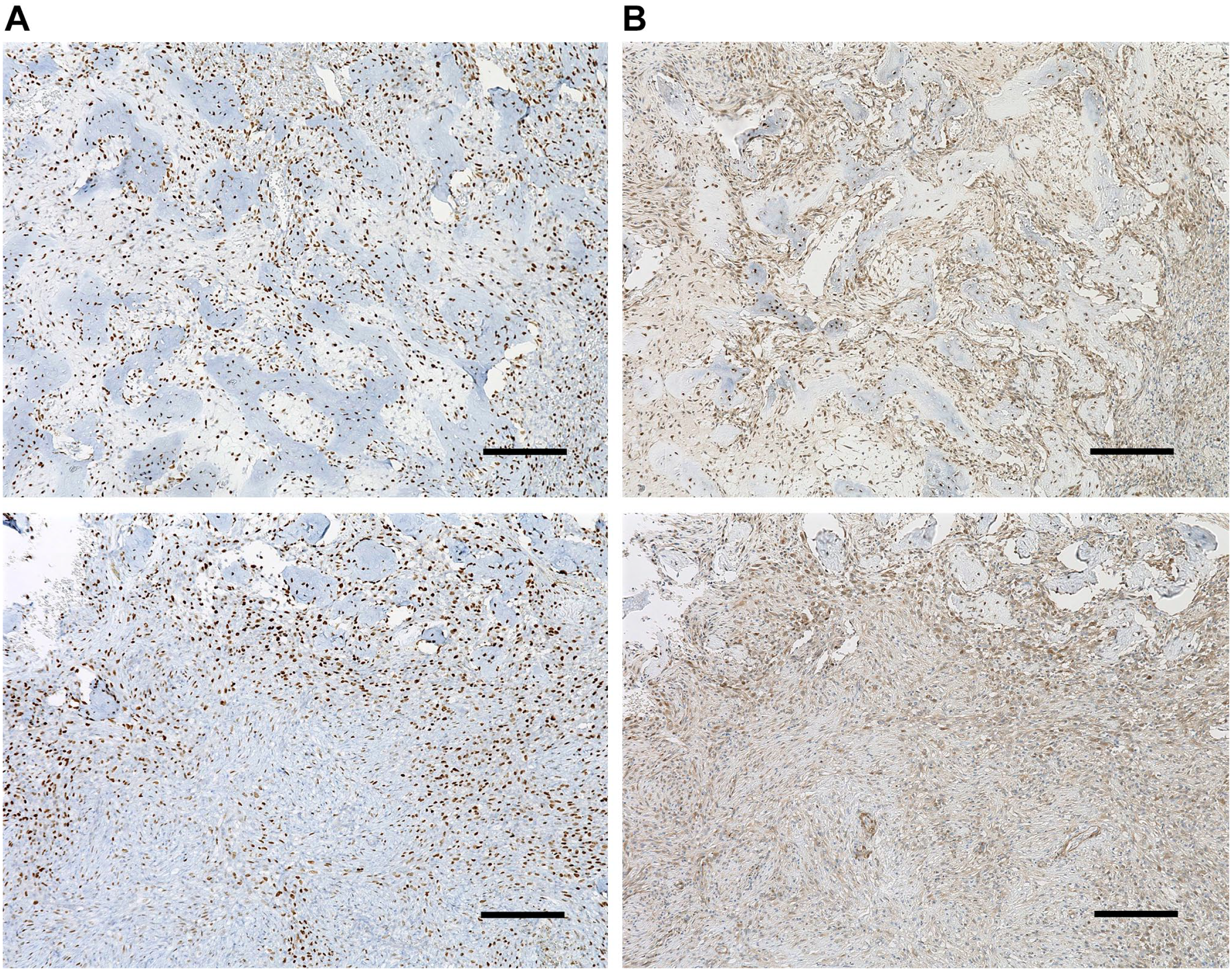
Giant cell tumor of bone. After denosumab treatment, positive expression of SATB2 (A) and SOST/sclerostin (B) was found not only in osteocyte-like cells embedded in bone but also in spindle cells between trabeculae. Spindle cells outside of the bone formation area showed weak positivity for SATB2 (A-bottom) (scale bar = 100 µm). Abbreviation: SATB2, special AT-rich sequence-binding protein 2.
Discussion
Administration of denosumab has reportedly increased the rate of recurrence of GCTBs after curettage. 14 In the current study, after denosumab treatment, GCTB mononuclear stromal cells became spindle-shaped. New bone formation was also observed. Because spindle cells are located between newly formed bones, they are trapped and difficult to curettage. Osteoblast-like cells which rimmed the bone, and osteocyte-like cells embedded in the bone, expressed the osteoblast and osteocyte differentiation markers, SATB2 and sclerostin/SOST, respectively, in all six cases following denosumab therapy. Moreover, spindle cells expressed these osteoblast–osteocyte differentiation markers, particularly after denosumab treatment. Also, immunohistochemistry with the H3.3G34W antibody was used to detect osteoblast-like cells and osteocyte-like cells with mutations, suggesting these osteoblast-like and osteocyte-like cells are tumor cells.
Homeostasis of calcium/phosphate is important during osteoblast–osteocyte differentiation. Fibroblast growth factor (FGF)-23 is a circulating phosphaturic hormone predominately produced by osteocytes to maintain phosphate homeostasis. FGF-23 is required for normal skeletal development and for the preservation of bone integrity. 16 Elevated levels of circulating FGF-23 with hypercalcemia were reported following the discontinuation of denosumab. 17 Denosumab may influence osteoblast–osteocyte differentiation in GCTBs in terms of calcium/phosphate homeostasis.
The histone variant H3.3 affects transcription and chromatin structure, and interacts with RNA processing mechanisms. The proliferative activity of stromal cells was elevated in the presence of a H3.3 G34W substitution, and a role for H3.3 in RNA processing and chromatin modulation—blocked by the G34W substitution—potentially drives the tumorigenic process in GCTBs, leading to hyper-proliferative activity. 18 Therefore, GCTB cells are capable of regrowth after the discontinuation of denosumab. In the current study, RANKL expression was prominent in the spindle-shaped cells and became more conspicuous after the administration of denosumab. Conversely, osteoid-embedded tumor cells failed to express RANKL, which may mean that the tumor cells embedded in bone were no longer active or able to recruit multinuclear giant cells.
TRAP activity revealed multinucleated giant cells and mononuclear TRAP-positive cells. After treatment with denosumab, TRAP-positive cells were undetectable, not only in giant cells but also in mononuclear cells in both the spindle cell and bone forming areas. Recently, osteoclast recycling was discovered during the process of bone resorption. RANKL-stimulated osteoclasts often fission into smaller daughter cells called osteomorphs that can accumulate and fuse together to form larger functional osteoclasts. 19 This phenomenon resembles the absence of giant cells when GCTBs are treated with denosumab. 20
In the current study, GCTB cells harboring the H3.3 mutation were embedded within the bone matrix during osteocyte differentiation and exhibited SATB2 and sclerostin/SOST expression, after denosumab treatment. In addition, these osteocyte-like cells no longer expressed RANKL whereas nearby interosseous cells did express RANKL. These results suggest that after denosumab treatment, osteocyte-differentiated GCTB cells lack biological stimuli via the RANK–RANKL signaling pathway to recruit and activate osteoclasts. Denosumab may play a role in the inactivation of tumor cells.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
TN and AS conceptualized the study design and drafted the manuscript. YM and KM planned the study and the experiment. MH and YY are pathologists and were responsible for the pathological samples and diagnosis. JT and SM critiqued the manuscript. All authors approved the final submitted version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Accessibility Statement
The data that support the findings of this study can be made available upon request from the corresponding author.
