Abstract
The correlation between long-QT and connexin 43 (Cx43) status and localization in elderly rats was determined to demonstrate a correlation between insulin resistance (I-R), ischemia-reperfusion, aging, and heart dysfunction. Male Wistar rats are grouped as 24-month-old rats (Aged-group), those with metabolic syndrome (8 months old; MetS-group), or controls (8 months old; Con-group). Both experimental groups have long-QT and low heart rate. Immunohistochemical imaging and quantification showed marked decreases in Cx43 staining of intercalated disc with less localizations in the Aged-group and MetS-group. The lateralization of Cx43 on longitudinal cell membrane was significantly high in the MetS-group than in the Con-group with no significant change in the Aged-group. Its significant cytoplasmic internalization was higher in the Aged-group than in the MetS-group. There were marked decreases in phospho-Cx43 (pCx43) staining of intercalated disc with less localizations in both groups than in the Con-group. Furthermore, lateralization of pCx43 was significantly low in the Aged-group and MetS-group, whereas there were no significant changes in the cytoplasmic internalization of both groups compared with the Con-group. Furthermore, the ratio of pCx43 to Cx43 was significantly small in both groups. We determined increases in RhoA and endothelin-1 in both groups, further supporting decreases in pCx43. Our data indicate the important role of I-R on long-QT in aging heart through alterations in both Cx43 protein level and localizations, leading to an abnormal spreading of ventricular repolarization in I-R heart:
Keywords
Introduction
The heart, as a mechanical pump, functions in a combination of various processes, such as involvement of active, passive, and/or endocrine factors. The regular heart rhythm and normal contractile activity of the heart can be maintained by a coordinated electrical activation in the heart. An electrical non-uniformity appears by the clustering of gap junction channels, which contributes to remodeling processes in heart disease.1,2 Any change in gap junctions, therefore, can have an important impact on the modulation of arrhythmogenic mechanisms in the heart, via disruption of synchronous activation. 3 Gap junctions are composed of several proteins, including the connexin family. Among them, connexin 43 (Cx43) is the major protein in the ventricular myocardium, and its relative amount, phosphorylation level, and distribution, as well as localization, can influence the conduction properties of cardiomyocytes, 4 as well as arrhythmogenesis in heart failure.4–6
Similar to metabolic syndrome (MetS), the aging population is increasing among humans worldwide, and hyperglycemia and insulin resistance are important factors to underline the tissue and cellular metabolic disorders, including the heart.7–13 One of the important common observation in either elderly individuals or those with MetS was apparent parallelism of similar cardiac dysfunction and marked insulin resistance with either high or normal blood glucose levels.14,15 Documents also point out the association between insulin resistance and advancing aging via metabolic disorders. Furthermore, organ-dependent insulin resistance such as myocardial insulin resistance in elderly individuals has been also mentioned.16–18 In these studies, the role of insulin resistance in abnormal electrocardiograms (ECGs) in patients with high blood pressure has been noted. A number of these studies also call attention to an association between insulin resistance and the development of cardiovascular disease through the involvement of insulin resistance in the longitudinal changes of cardiac repolarization heterogeneity.17,19 Moreover, some experimental studies suggest that hyperglycemia leads to the deterioration of myocardial intercellular communication mediated by Cx43 channels. 20 In these studies, their data strongly emphasize that there is a marked remodeling of Cx43 in the diabetic heart through high-glucose-depressed Cx43 gap junction and hemichannel function. 21 An association between the upregulation of phospho-Cx43 (pCx43) and the inhibition of RhoA/ROCK signaling in glomerular mesangial cells under high glucose exposure has also been shown, implying the effect of activated RhoA/ROCK signaling on Cx43 degradation in high-glucose-treated cells via endothelin-1 (ET-1)-induced RhoA activation.22–25 Besides these correlations, some studies also emphasized the role of gap junction remodeling with Cx43 in those with diabetes (DM) and in MetS mammalians, as well as in aged mammalians.21,26
The correlation between long-QT with increasing age and the increases in the risk of ventricular arrhythmias and cardiac mortality in elderly individuals is not exactly understood yet. 27 Previously, we have shown that a significant percentage of the aged rats (about 60–70%) have insulin resistance and long-QT-intervals, in a similar manner to those of high-carbohydrate diet–induced MetS rats.12,13 In addition, the single-cell action potential duration measured in left ventricular cardiomyocytes from both experimental animals was found to be significantly prolonged, at most, through significantly depressed voltage-dependent K+-channel currents.
As mentioned above, there is an association between anisotropic propagation of electrical signals and changes in the arrangement and number of gap junctions at the cellular level (i.e., concerning conduction velocities at the macroscopic level in the heart). It is not demonstrated how the complex spatial and temporal conduction properties of cardiac dynamics can be associated with connexin statuses such as their levels of phosphorylation, expression, or localization or all of them under insulin resistance. In addition, the literature data have shown a correlation between reduced insulin sensitivity and heart dysfunction, at most, via an increase in the QT-interval (long-QT) not only in diabetic patients28,29 but also in aged mammalians.30,31 Therefore, we aimed to examine a possible correlation between aging and associated heart dysfunction, through contribution of long-QT, at least, via alterations in connexin statuses, including not only their protein expression levels but also their cellular localizations. So, here, we examined the contribution of changes in the expression levels of pCx43 and Cx43 as well as their cellular localization and distribution patterns in the left ventricular part of the heart from insulin-resistant elderly rats with an apparent long-QT in their surface ECGs (Aged-group; 24 months old). For validation, the data of the elderly group were compared with either MetS rats (8-month-old rats fed with high-sucrose diet) or normal rats (Con-group; 8 months old). We also aimed to examine whether there are contributions of RhoA activation in the inhibition of Cx43 hemichannels, through ET-1 activation in the elderly left ventricle of the heart.
Materials and Methods
Experimental Animals
We used male Wistar rats in the three groups. The first experimental group are 24-month-old rats (Aged-group, n=12), and the second experimental group are rats with MetS (MetS-group) induced by feeding them daily with drinking water containing 32% sucrose for 24 weeks, starting from an age of 8 weeks (MetS-group; overall 8 months old, n=11). The third group are 8-month-old rats (Con-group, n=8). All animals were kept in a normal circadian rhythm and provided free access to tap water. 13 They were fed standard chow ad libitum daily. All animals were housed in standard rat cages with three rats per cage. The experimental protocol and handling of animals during the duration of the experiments were approved by the Ankara University ethics committee (No: 2015-12-137 and No: 2016-18-165).
Validation of Insulin Resistance and MetS in Rats
Before the experimental procedures, all rats had fasted overnight and blood samples were collected for insulin-level measurement using an enzyme immunoassay kit (E-EL-R2466; Elabscience, China). Blood glucose levels were assessed using standard glucose test strips (GlucoCheck Analyzer). To determine the oral glucose tolerance test (OGTT) values, the rats received an orogastric gavage of 1 g/kg glucose in double-distilled water, as described previously.12,13 Blood samples were collected from a small cut at the tip of the tail immediately before and at 0, 15, 30, 60, and 120 min following glucose administration. The total area under the curve was calculated using GraphPad Prism. Insulin resistance was evaluated by the homeostatic model assessment (HOMA) index, as described previously.12,13
In Situ ECG Recording
We used two local commercial electrodes (Commat Ltd; Ankara, Turkey) to record ECGs in situ from rats. The electrical activity of the heart in vivo was assessed by surface ECG measurements. Rats underwent inhalation anesthetics during the experiments. 32 Bipolar limb leads (leads I, II, III) were carried by carefully injecting 20-gauge needles into the forearms and hind limb. ECG data were acquired using an analog-to-digital converter BIOPAC MP35 (BIPOC; Goleta, CA) and processed with a high-cut (low-pass) filter at 50–500 Hz and analyzed using BIOPAC Student Lab Pro. All ECGs were recorded for 10 min in each rat. The durations of the heart rate, QRS-complex (combination of the Q-wave, R-wave, and S-wave), PR-segment (interval is the period that extends from the beginning of the P-wave until the beginning of the QRS-complex), and QT-interval (the time from the start of the Q-wave to the end of the T-wave) were analyzed from ECGs. The RR interval (the duration between successive heartbeats in ECG) was calculated from the heart rate. The corrected QT for each ECG trace was calculated according to the following formula: QTc = QT/(RR/100)1/2.
Immunohistochemical Examination
The left ventricular parts of the hearts were fixed in phosphate-buffered saline (PBS) with 10% formalin for 96 hr, and then were washed in running tap water, followed by dehydration and immersion in 75%, 96%, and 100% ethanol. After dehydration, these tissues were rinsed in xylene and then were embedded in paraffin wax. After the routine follow-up stages, 5-μm-thick sections were cut from paraffin blocks with a microtome (Leica RM 2125RT) and transferred onto microscope slides coated with poly-
The histological quantification of immunohistochemistry data is performed as described previously.33–36 First, we used a semiquantitative method for scoring “immunopositivity” for Cx43 and pCx43 as described in Table 1. Second, we quantified the positive immunostaining of Cx43 and pCx43 as pixels by using image analysis and presented them as percentage changes in comparison with those of controls. The results for Cx43 or pCx43 are expressed as arbitrary units, and then, their mean values are presented as bar graphs for each evaluated parameter.
The Histological Scoring Method, Used in the Evaluation of Immunohistochemistry Associated With the Status of Phospho-Cx43 (pCx43) and Cx43.
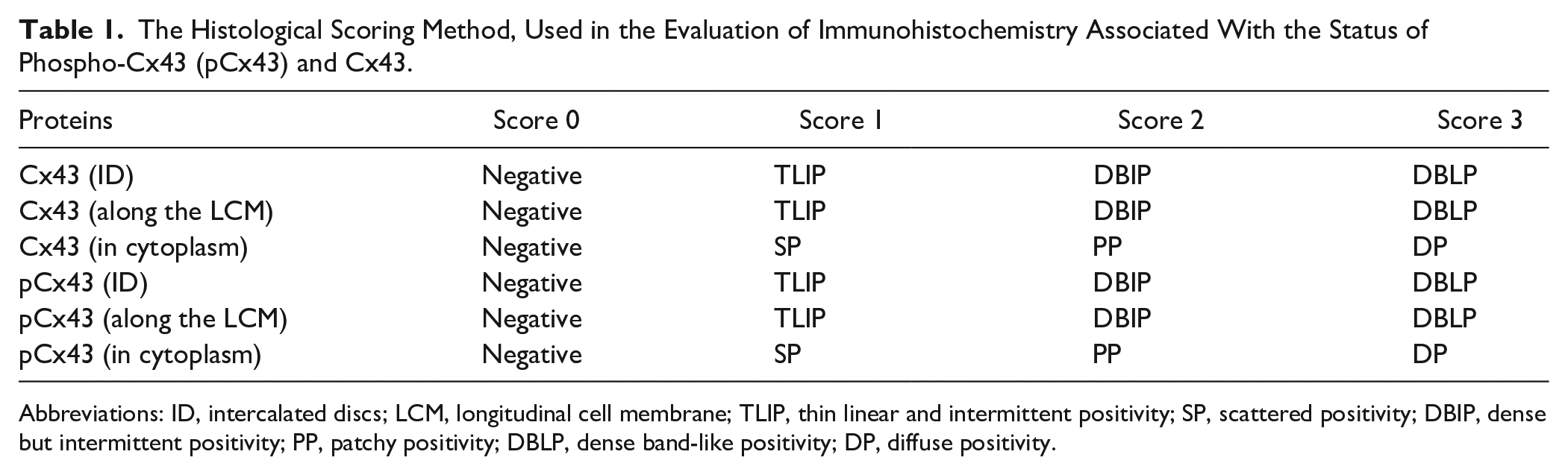
Abbreviations: ID, intercalated discs; LCM, longitudinal cell membrane; TLIP, thin linear and intermittent positivity; SP, scattered positivity; DBIP, dense but intermittent positivity; PP, patchy positivity; DBLP, dense band-like positivity; DP, diffuse positivity.
Western Blot Analysis
A Western blot analysis was performed to determine the relative protein and phosphorylation levels of Cx43, and the protein levels of RhoA and ET-1 compared with a reference protein GAPDH in the heart tissues. To prepare the tissue homogenates, frozen heart samples from the left ventricle were first crushed at liquid N2 temperature and then homogenized in ice-cold lysis buffer, as described previously. 37 Briefly, an equal amount of protein preparations (10–20 µg) were run on SDS-PAGE and blotted with specific antibodies for Cx43 (sc-9059, 1:500; Santa Cruz), pCx43 (ab194928,1:500; Abcam), RhoA (sc-418, 1:500; Santa Cruz), ET-1 (sc-21625, 1:500; Santa Cruz), and reference protein GAPDH (D16H11, 1:5000; Cell Signaling) to detect their protein levels.
Chemicals, Data Analysis, and Statistics
Reagents were obtained from Sigma-Aldrich (St. Louis, MO) unless otherwise noted. All antibodies were obtained from Santa Cruz Biotechnology. Second antibodies conjugated with horseradish peroxidase were purchased from Biolegend Biotechnology. Molecular weight markers, polyvinylidene difluoride (PVDF) membranes, and enhanced chemiluminescence (ECL) were also purchased from Biolegend Biotechnology.
The results are expressed as mean ± SEM in bar graphs. Statistical significance was evaluated by one-way ANOVA followed by Tukey’s post-test. The probability level of p<0.05 was considered statistically significant.
The statistical analysis of the scoring of immunohistochemical data is performed using Kruskal–Wallis one-way ANOVA and is expressed as means and standard deviations (SDs) as well as medians.
Results
General Findings of Experimental Animals
We used two experimental groups and one control group, as described in the “Methods and Materials” section. The first experimental group were 24-month-old male rats fed with a standard diet (Aged-group), whereas the second group were MetS-rats fed for 24 weeks with high sucrose (MetS-group; 8 months old). The control rats (Con-group) were 8-month-old male rats. The approximate comparative of our test groups, that is, the Aged-group, the MetS-group, and the Con-group, with human ages is 60, 22, and 22 years, respectively. 38 We did examine the aged rats without insulin resistance; there was no significant prolonged QT-interval (data not given). Therefore, here, we used only insulin-resistant aged rats.
The bodyweight of the Aged-group was not significantly different from that of the Con-group, whereas the bodyweight of the MetS-group was significantly higher than that of the Con-group (Fig. 1A). The heart weight of the Aged-group and the MetS-group was significantly higher than that in the Con-group (Fig. 1B). We also determined the ratio of the heart weight to body weight for every group. As can be seen in Fig. 1C, this ratio is significantly high in the Aged-group than in the MetS-group, while the ratios of both experimental groups are significantly higher than those in the Con-group.
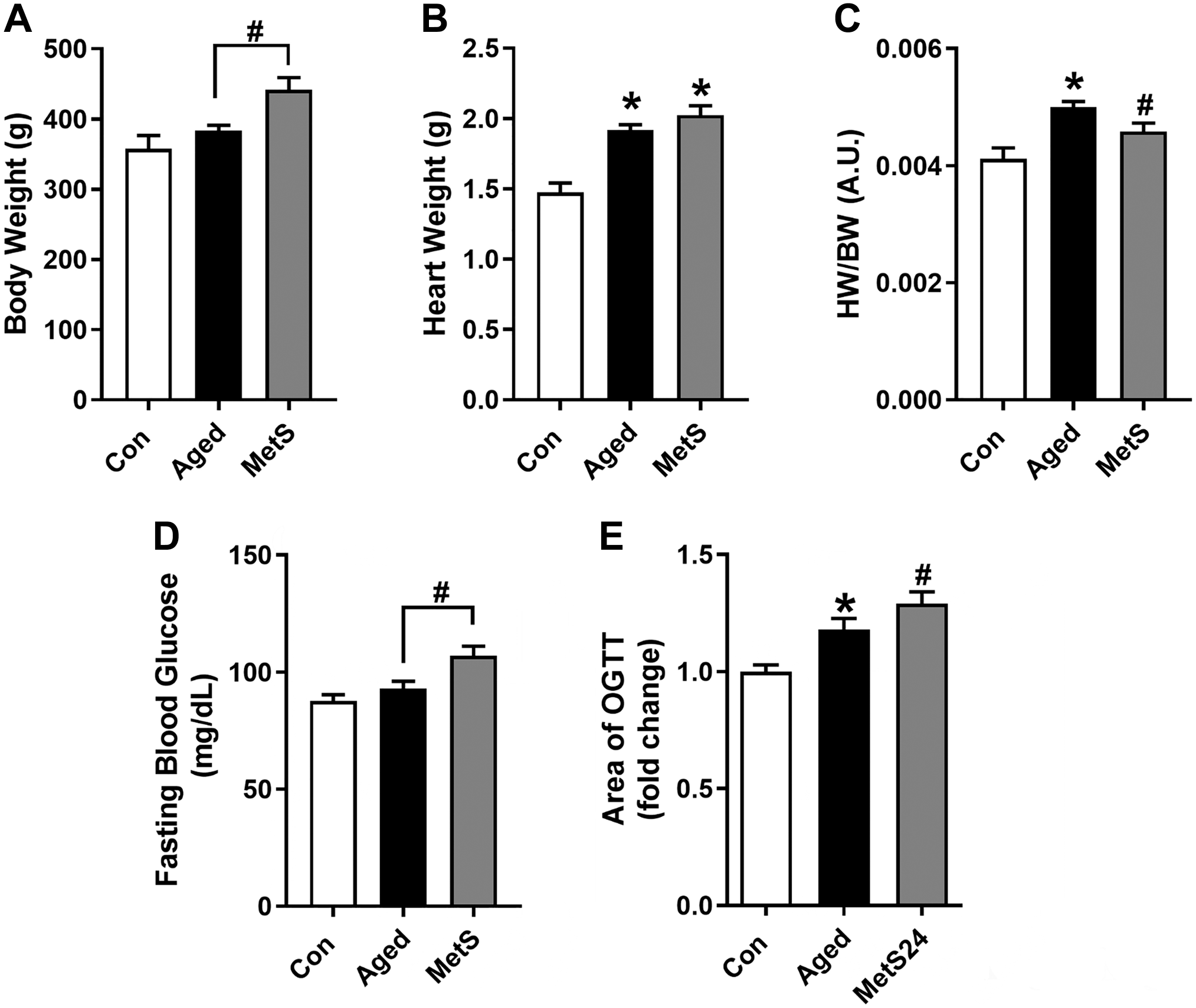
Systemic parameters of experimental animals. BW (A), HW (B), the ratio of HW to BW (C), fasting blood glucose levels (D), and changes in OGTT values calculated as the area under the curve determined with the peak blood glucose levels measured at 15, 30, 60, and 120 min following glucose loading during OGTT monitoring (E) of all groups. Experimental groups: the Aged-group, 24-month-old male rats fed with standard rat diet; the Con-group, 8-month-old male rats fed with standard rat diet; the Met-group, rats fed with high sucrose besides standard rat diet for 24 weeks (8 months old). All data were presented as mean ± SEM. The total number of rats/groups; n=8–12. Significance level, *p<0.05 vs Con-group. Abbreviations: HW, heart weight; BW, body weight; OGTT, oral glucose tolerance test.
The fasting blood glucose level of the MetS-group was significantly higher than that of the Con-group, whereas the Aged-group had a similar blood glucose level (Fig. 1D). The OGTT values of the groups were calculated as the area of the curves obtained at the peak blood glucose levels measured at 15, 30, 60, and 120 min following high glucose loading. The Aged-group, similar to the MetS-group, has a significantly higher OGTT than that of the Con-group (Fig. 1E).
For the Aged-group (24 months old), the insulin level and HOMA-IR (Homeostatic Model Assessment for Insulin Resistance) were 0.57 ± 0.07 pmol/l and 19.7±4.6, respectively, compared with their controls (0.22 ± 0.03 pmol/l and 10.9 ± 3.1), whereas for the MetS-group, those values were 0.67 ± 0.04 pmol/l and 28.9±5.5, respectively, compared with their controls. When we compared these parameters for the Aged-group and the MetS-group, the differences between these two experimental groups are not significantly different.
ECG Parameters Calculated for Experimental Animals
We calculated the parameters of heart function from the ECGs of these two experimental groups in comparison with that of the Con-group. As shown in Fig. 2B, the heart rates of the Aged-group and the MetS-group are significantly slower compared with the Con-group. We also calculated PR-, RR-, QRS-, and QT-intervals (Fig. 2C–F). These measured intervals were significantly prolonged in the Aged-group and the MetS-group compared with the Con-group. The calculated levels of QTc are given in Fig. 2F (right). The QTc values for these two experimental groups were significantly longer than the Con-group, whereas both the QT (Fig. 2F, left) and QTc of the Aged-group were longer than those of the MetS-group. The representative ECGs for the groups are given in Fig. 2A.
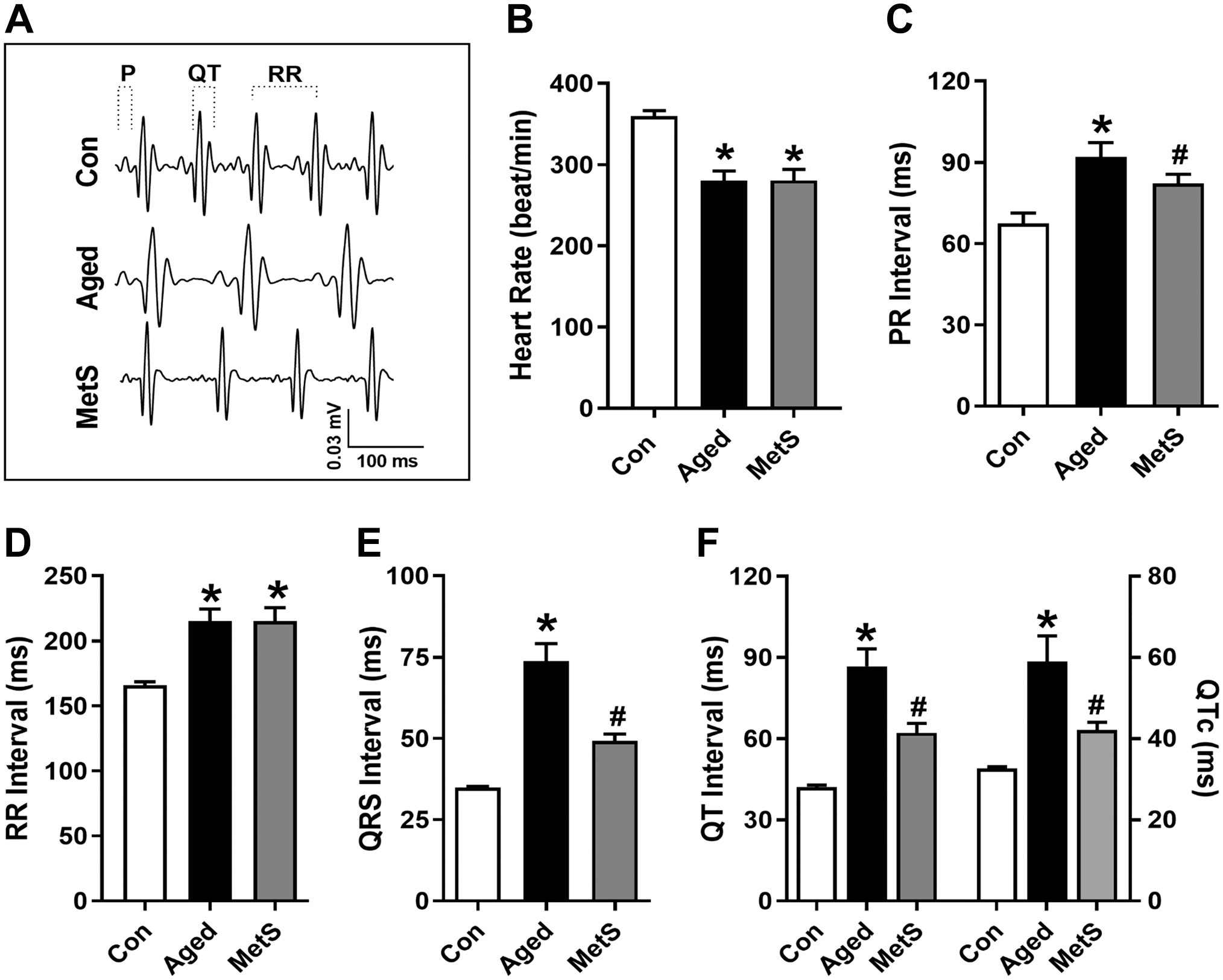
The parameters of electrocardiograms (ECGs) in experimental rats. The representative ECGs for the groups (A), heart rates (B), and the calculated parameters of ECGs such as PR-interval (C), RR-interval (D), QRS-complex (E), and QT-interval (F, left). RR-intervals were calculated from the heart rate, and the calculated QTc values (F, right) for groups are given in Fig. 1. Data are presented as mean (±SEM) values. The total number of rats/groups; n=4–5. Significance levels at *p<0.05 v. Con-group, #p<0.05 vs Aged-group.
Immunohistochemical Analysis of Cx43 in the Heart Tissue
Positive immunostaining for Cx43 was detected in the sections obtained from the left ventricular parts of the hearts. We first evaluated the staining level of Cx43 in the intercalated discs between cardiomyocytes in the samples. As can be seen in Fig. 3 (left), the detected stainings of Cx43 in the intercalated discs, cytoplasm, and along the cell membrane are in normal appearance and had no myocardial abnormalities with neat Cx43 stainings in the Con-group. We detected marked decreases in Cx43 immunostaining in the intercalated discs of cardiomyocytes from both the Aged-group and the MetS-group (Fig. 3, middle and right, respectively). In the same images, we also detected marked increases in the staining of Cx43 internalization in the cytoplasm, observed as small and randomly distributed dot stainings, in the samples from both the Aged-group and the MetS-group. We also detected a marked Cx43 staining along the sarcolemma (indicating Cx43 lateralization) in the MetS-group but not in the Aged-group. The negative controls of imagings for Cx43 were performed using a non-immune IgG and are presented in Fig. 3B
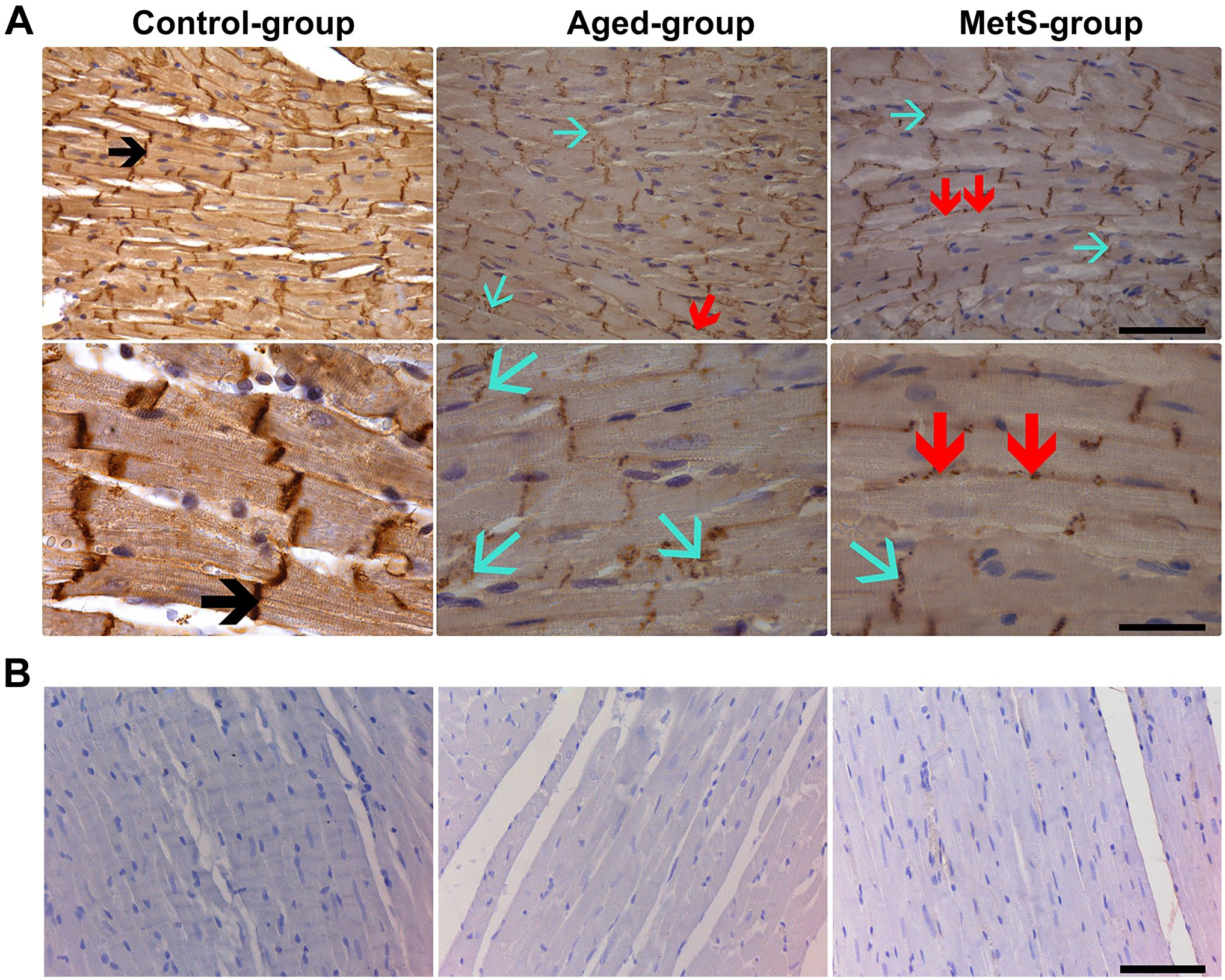
Immunohistochemical analysis of heart tissues for connexin 43 (Cx43). (A) Cx43 immunostaining in left ventricular heart tissue from the Con-group, the Aged-group, and the MetS-group. Cx43 immunostaining level with high density was located in the intercalated discs of cardiomyocytes (black arrow) in the Con-group. In both the Aged- and the MetS-groups, the Cx43 immunostaining levels were markedly decreased in the intercalated discs of cardiomyocytes (blue arrow) as small dot shape stainings distributed randomly in the cytosol together with the observed Cx43 lateralization (red arrow) along the cell membrane. Magnifications in the upper row (×40): scale bar = 100 µm and in the lower row (×100): scale bar = 40 µm. (B) Immunohistochemical staining of negative controls for Cx43. Magnification, ×40 and scale bar = 100 µm.
We also detected pCx43 immunostaining levels in the heart tissue samples. As seen in Fig. 4 (left), the pCx43 immunostaining density was detected with a normal distribution and localization in the intercalated discs, cytoplasm, and along the membrane of cardiomyocytes in the Con-group. In both the Aged-group and the MetS-group, the detected pCx43 immunostaining levels were markedly decreased in the intercalated discs of cardiomyocytes, whereas these decreases were markedly higher in the Aged-group than in the MetS-group (Fig. 4, middle and right, respectively). Furthermore, we detected slightly less lateralization of pCx43 in the samples from both experimental groups compared with the Con-group. The internalizations of pCx43 detected in both experimental groups were not different from the Con-group. The negative controls of imagings for pCx43 were performed using a non-immune IgG and are presented in Fig. 4B
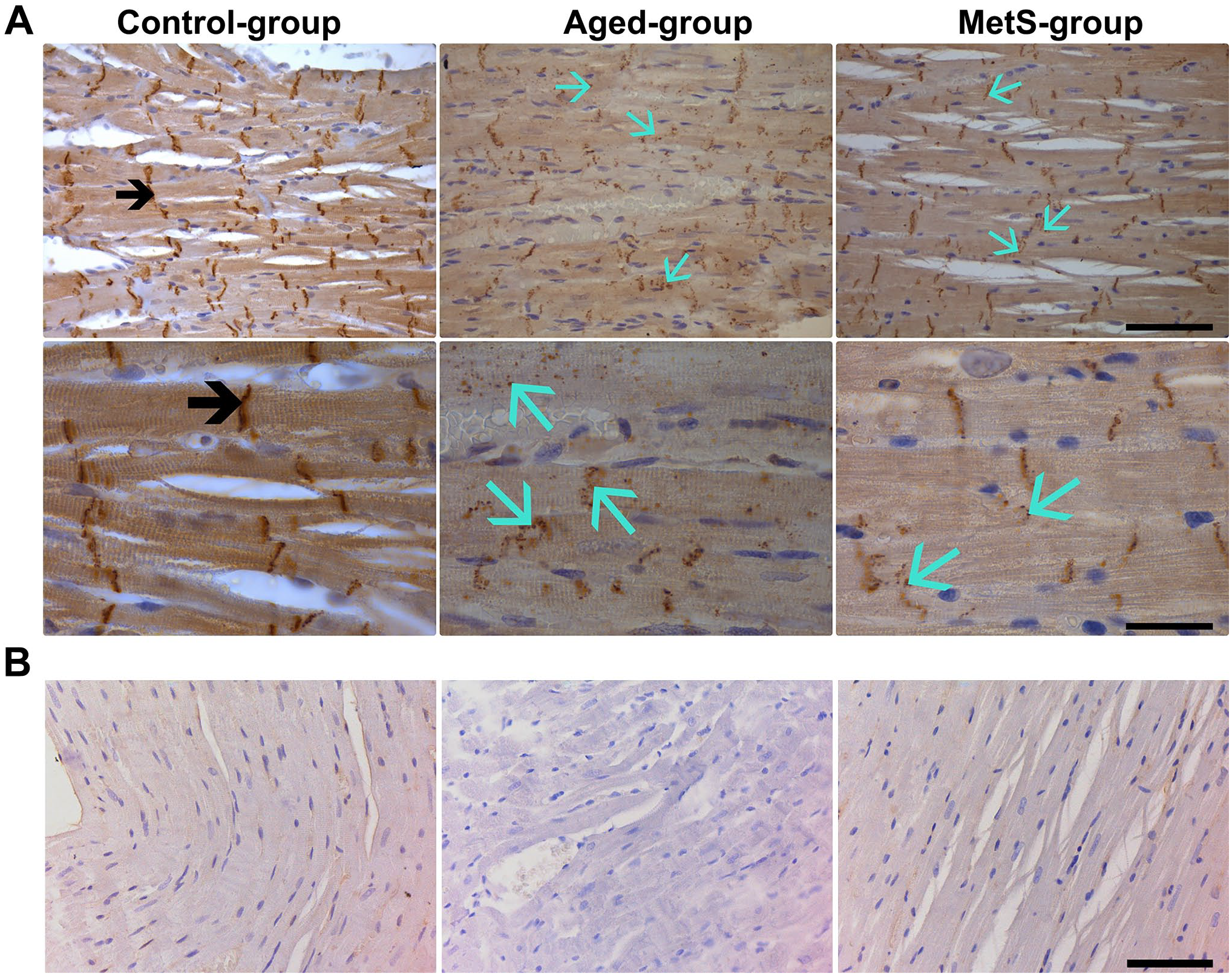
Immunohistochemical analysis of heart tissues for phospho-connexin 43 (pCx43). (A) The pCx43 protein levels in the heart tissues from the Con-group, Aged-group, and MetS-group. pCx43 immunostaining level with high density and normal distribution was located in the intercalated discs of cardiomyocytes (black arrow) in the Con-group. In both the Aged-group and the MetS-group, pCx43 immunostaining levels were markedly decreased in the intercalated discs of cardiomyocytes and there was the internalization of these proteins slightly observed as small dot shape stainings (blue arrow). Magnifications in the upper row (×40): scale bar = 100 µm and in the lower row (×100): scale bar = 40 µm. (B) Immunohistochemical staining of negative controls for pCx43. Magnification, ×40 and scale bar = 100 µm.
Quantification of Immunohistochemical Analysis of Cx43 and pCx43
Similar to previously published studies by others,33,39,40 the immunohistochemistry of Cx43 and pCx43 in the myocardium as markers of aging-associated heart dysfunction in samples was detected by scoring the staining degrees. As can be seen in Table 2, we detected less Cx43 staining of its distribution and localization in the intercalated discs of the samples from both experimental groups compared with the Con-group (score number: 2.75 ± 0.46), whereas these detections were more marked in the Aged-group (score number: 1.25 ± 0.45) than in the MetS-group (score number: 1.64 ± 0.50). We also detected marked increases in the staining of Cx43 internalization appearing as small and randomly distributed dot stainings in the cytoplasm of the MetS-group (score number: 0.55 ± 0.32) and the Aged-group (score number: 0.92 ± 0.28) compared with the Con-group (score number: 0.38 ± 0.21), with more prominence in the Aged-group. However, we determined marked increase in Cx43 lateralization along the sarcolemma in the MetS-group (score number: 1.27 ± 0.64) than in the Con-group (score number: 0.63 ± 0.31) with no marked change in the Aged-group (score number: 0.52 ± 0.21).
Quantification of Immunohistochemical Data for Cx43 Using the Scoring Method.
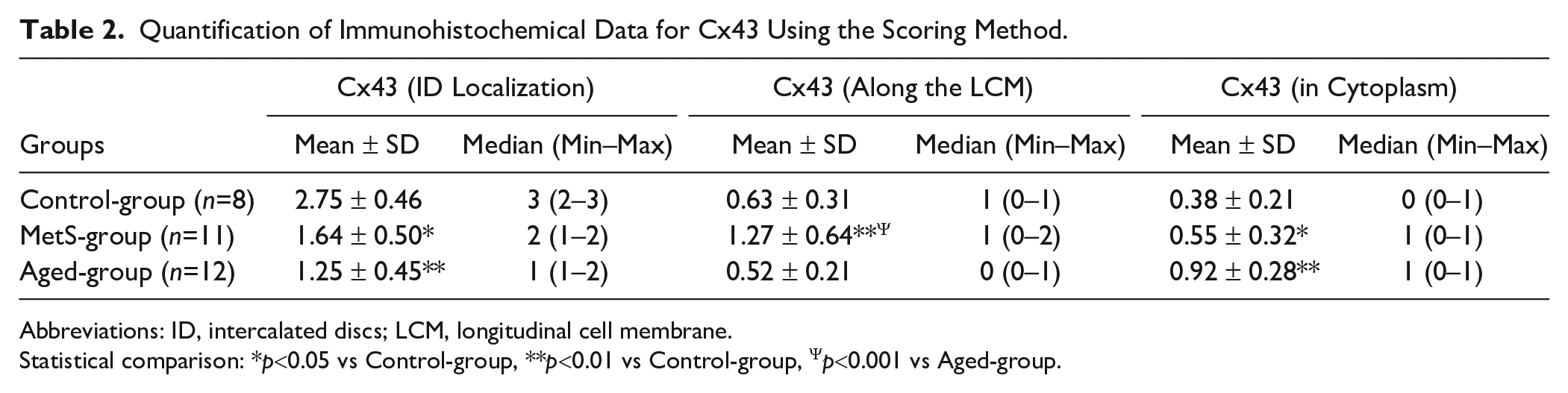
Abbreviations: ID, intercalated discs; LCM, longitudinal cell membrane.
Statistical comparison: *p<0.05 vs Control-group, **p<0.01 vs Control-group, Ψp<0.001 vs Aged-group.
We also detected pCx43 immunostaining in the heart tissues. The pCx43 immunostaining levels were significantly decreased in the intercalated discs of cardiomyocytes in both the Aged-group (1.08 ± 0.28) and the MetS-group (1.36 ± 0.50), compared with the Con-group (2.25 ± 0.46). Furthermore, this decrease in the Aged-group was markedly higher than that in the MetS-group. Moreover, we detected slight lateralization of pCx43 in the samples from both experimental groups using scored evaluation (0.42 ± 0.41 vs 0.27 ± 0.26 in the Aged-group vs MetS-group) compared with the Con-group (0.75 ± 0.70), whereas there was similar degree of laterilization in both experimental groups (Table 3).
Quantification of Immunohistochemical Data for pCx43 Using the Scoring Method.
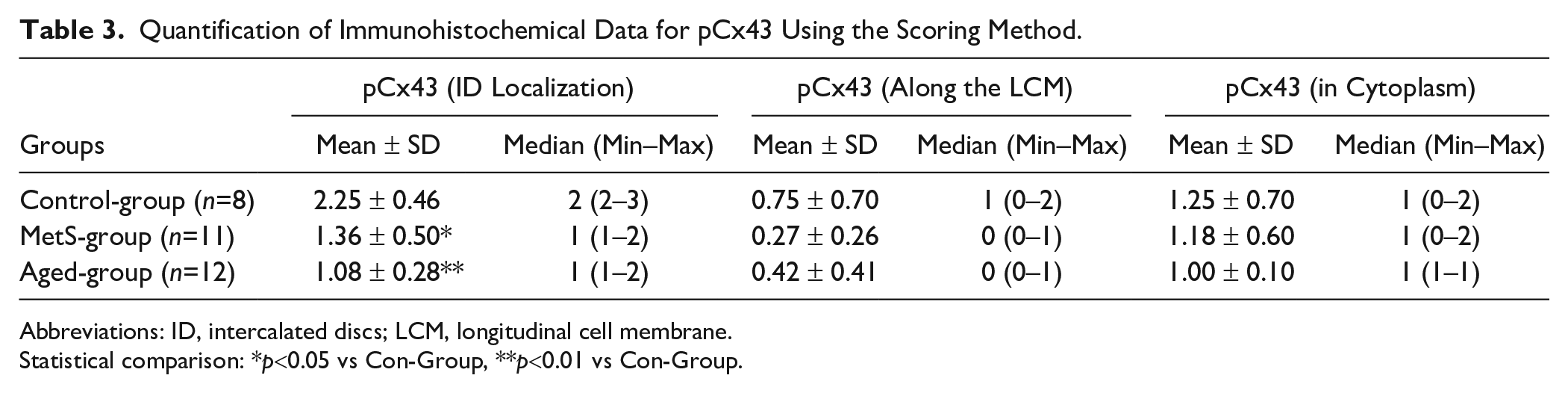
Abbreviations: ID, intercalated discs; LCM, longitudinal cell membrane.
Statistical comparison: *p<0.05 vs Con-Group, **p<0.01 vs Con-Group.
We also determined pCx43 internalization in the samples. As can be seen in Table 3, the statuses of pCx43 immunostainings (observed as small dot shape stainings in the cytoplasm) using scored evaluation were found to be widely distributed in all samples, whereas there were no significant differences between the experimental groups and the Con-group.
In the last group of examinations, we evaluated the immunohistochemistry data by image analysis and presented the results as percentage changes in the experimental groups with respect to the control group. As can be seen in Fig. 5, the quantifications of immunohistochemical imaging showed that there were significantly less localizations of Cx43 in intercalated discs in Aged-group samples (60 ± 27% vs Con-group) with slightly less in the MetS-group (41 ± 26% vs Con-group). The lateralization of Cx43 on longitudinal cell membrane was significantly high in the MetS-group than in the Con-group (202 ± 45%) with no significant change in the Aged-group (33 ± 25% vs Con-group). Furthermore, its significant internalization in cytoplasm was high in the Aged-group (242 ± 48% vs Con-group) than in the MetS-group (145 ± 36% vs Con-group). We also detected significant decreases in pCx43 (pCx43) of intercalated discs in both experimental groups (55 ± 13% in the Aged-group and 40 ± 19% in the MetS-group) than in the Con-group. Furthermore, the laterization of pCx43 was significantly low in both the Aged-group (56 ± 21%) and the MetS-group (64 ± 18%) than in the Con-group, whereas there were no significant changes in internalization in the cytoplasm of both experimental groups compared with the Con-group.
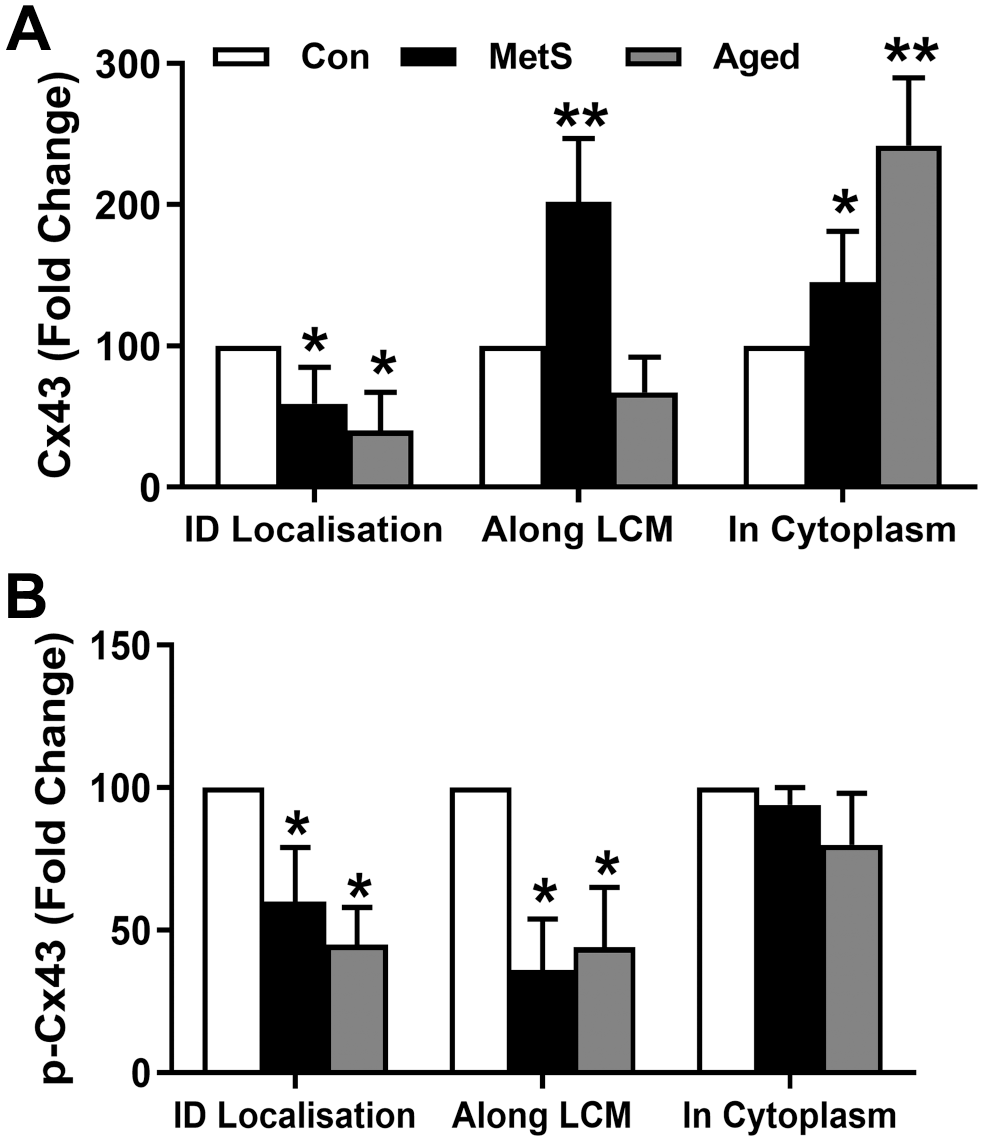
The quantification of immunohistochemistry imagings. The histological quantification of immunohistochemistry data is performed following semiquantitative investigation of scoring by quantification of positive immunostainings of Cx43 (A) and pCx43 (B) as pixels by using image analysis. The quantification of experimental groups for each investigated parameter is presented as percentage changes compared with control. The results for Cx43 or pCx43 are expressed as arbitrary units, and the bars represent mean values (±SEM). The samples for each investigation are 4–5/group, and the statistical significance was obtained with Mann–Whitney test for independent samples: *p<0.05 vs Con-group, **p<0.01 vs Con-group.
Biochemical Analysis of the Phosphorylation and Protein Levels of Cx43 in Left Ventricular Heart Tissues
We analyzed the phosphorylation and protein levels of Cx43 in left ventricular tissues of rat hearts from experimental groups (Aged-group and MetS-group) in comparison with the Con-group using the Western blot analysis. The representative Western blot bands for pCx43 and the total protein expression levels of Cx43 are given in Fig. 6A and B. The pCx43 and Cx43 levels were significantly decreased both in the Aged-group and in the MetS-group (Fig. 6C and D). We also calculated the ratio of pCx43 to Cx43 for the groups. As can be seen in Fig. 6E, the ratio of pCx43 to Cx43 was significantly low in both the Aged-group and the MetS-group than in the Con-group, with more prominence in the MetS-group than in the Aged-group.
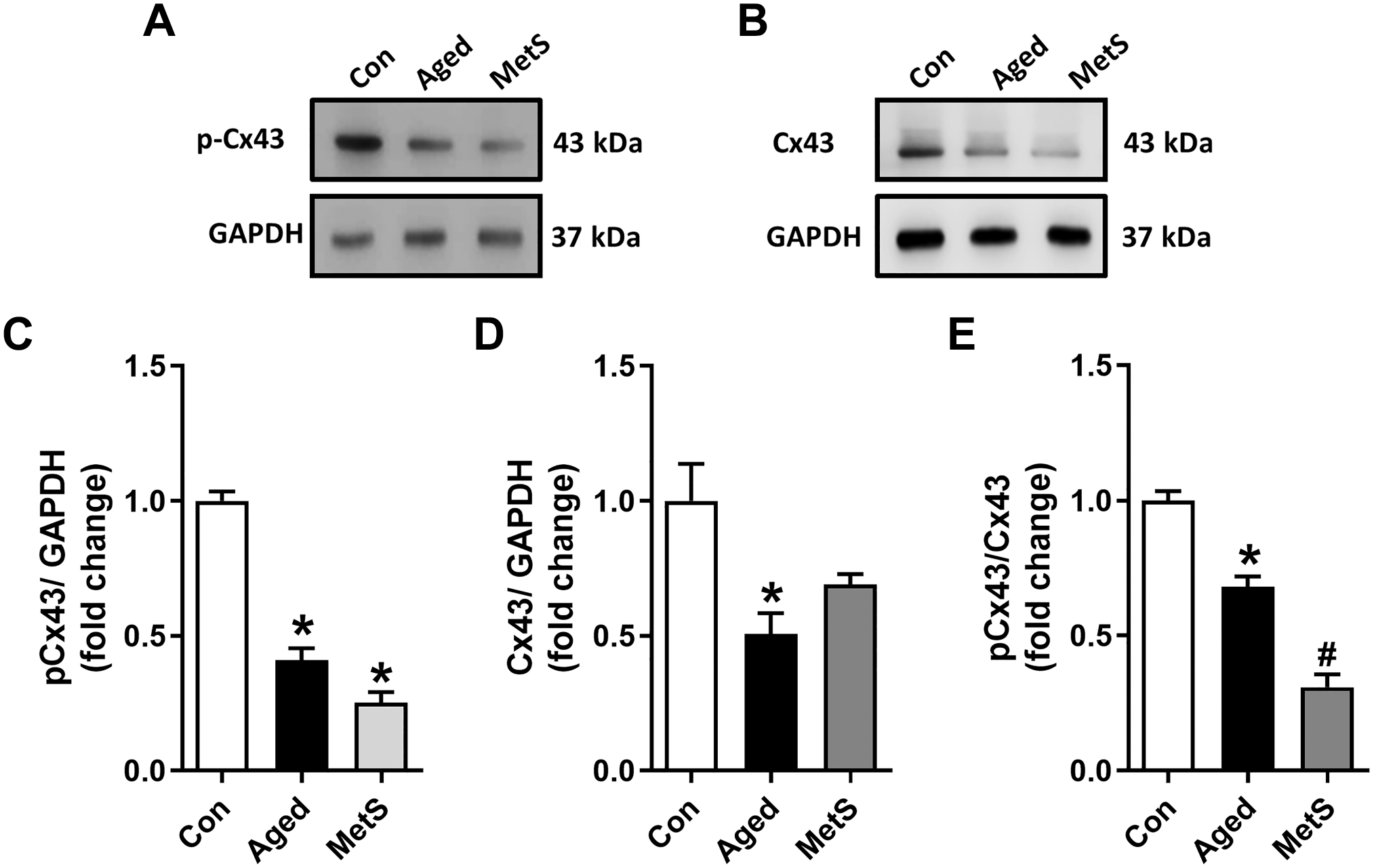
The phosphorylation and protein expression levels of connexin 43 (Cx43) in left ventricular heart tissue from experimental animals. Representative Western blot bands for the phosphorylation (pCx43) and protein expression (Cx43) level of connexin 43, respectively, in (A) and (B). The average Western blot analysis of pCx43 and Cx43 for a reference protein GAPDH in heart samples from the experimental groups is given in (C) and (D). The ratio of pCx43 to Cx43 in (E). Bars represent changes as mean (±SEM). The hearts used from rats are 4–5/group. *p<0.05 vs Con-group, #p<0.05 vs Aged-group.
Increased Protein Levels of RhoA and ET-1 in Left Ventricular Heart Tissues
As studies indicate a role for the RhoA pathway in the expression and phosphorylation of Cx43, we analyzed the activation of RhoA and ET-1 by determining their protein levels in the left ventricular heart tissues of the rats in the experimental groups. The levels of these proteins in the Aged-group were significantly higher than those in the Con-group (Fig. 7A and B, respectively). Moreover, there were no significant differences in RhoA and ET-1 levels compared with the Aged-group and the MetS-group.
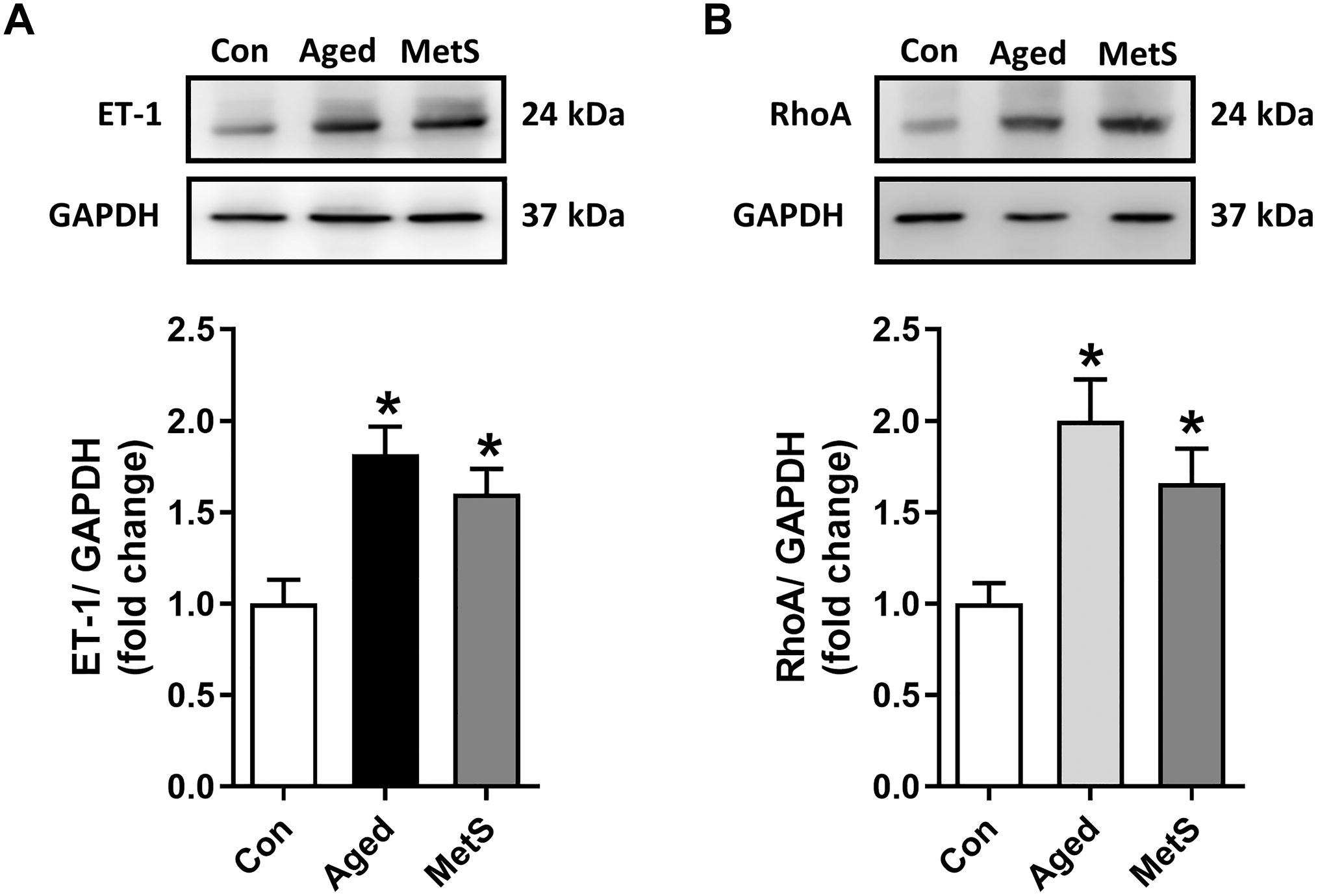
Protein expression levels of endothelin-1 (ET-1) and RhoA in left ventricular heart tissue from experimental animals. The protein expression level of ET-1 (A) and RhoA (B) was measured with Western blot analysis for a reference protein GAPDH in heart samples from the experimental groups given in Fig. 1. Representative Western blot bands of proteins are given in the upper part of the bar graphs. The bars represent changes as mean (±SEM). The hearts used from the rats are 4–5/group. *p<0.05 vs Con-group.
Discussion
This study presents, for the first time, the important role of insulin resistance on long-QT and low heart rate in the aging heart through not only reductions in phosphorylation and expression levels of Cx43 but also their apparent localization in the ventricular cardiomyocytes. Furthermore, we determined similar alterations in these parameters in MetS rat ventricular cardiomyocytes, characterized by insulin resistance. Moreover, we have previously shown that altered ECG parameters (importantly, prolongations in the parameters of ECGs such as PR-, RR-, and QT-intervals and QRS-complex with low heart rates) of MetS rats were very similar to those of 24-month-old rats and insulin resistance with normal bodyweight.12,13 Interestingly, when we compared two groups of MetS rats with either 16-week high-carbohydrate feeding or 24-week high-carbohydrate feeding, the shorter period–fed MetS rats presented short-QT with high heart rate,37,41 whereas longer feeding induced long-QT and low heart rate 12 although both groups have definite insulin resistance with different levels. These differences can be a consequence of the early and chronic MetS development in mammals. Correspondingly, one can propose that development of insulin resistance can be closely associated with alterations in the electrical activity of rats in both in vivo and in vitro conditions. Interestingly, in other studies with MetS and/or obese mice models induced by high-fat diet, authors have shown that high-fat diet for different application durations induced significantly increased body weight, plasma leptin, and aldosterone levels in systemic levels and increased heart rate, stroke volume, and cardiac output at organ level without an effect on blood pressure, as well as increased sympathetic activity.42,43 However, L’Abbate et al. 44 investigated the effect of high-fat diet in C57BL6 mice for 13 weeks and showed that there was an abnormal ventricular repolarization (long-QTc on ECG) together with impaired left ventricular filling rate and increased left ventricular mass. A more interesting finding of this group study is to demonstrate higher heart rate, which seems to oppose the long-QT in ECG in these animals. The differences observed between these two groups such as high-fat diet in C57BL6 mice for 13 weeks and high-carbohydrate diet in adult rats for 16 weeks are long-QT vs high heart rate and short-QT vs high heart rate. Animal differences, diet differences, or both can underline the different alterations in the heart. Supportingly, early studies also mention different cardiovascular responses in rats to either high-fat or high-carbohydrate diet. 45 In another study, authors studied the effects of a 1-week intracerebroventricular leptin infusion on various parameters of energy balance in male Wistar rats of different age groups (from 3 to 24 months): Through their data, a promotion of resistance to both hypermetabolic and anorexigenic actions by obesity has been demonstrated, especially in old rats but not young rats. 46
As mentioned in previous studies,34,47 these changes, such as statuses and localizations of Cx43, seem to lead to an abnormal spreading of ventricular repolarization in the insulin-resistant aging rat heart. Furthermore, we, previously, have also demonstrated that there was similar prolongation in action potential duration determined in left ventricular cardiomyocytes isolated from either MetS rat heart or aged rat heart, at most, in the repolarization phase of action potentials, through depressed voltage-dependent K+-channel currents.12,13 In addition, our previous organ- and cell-level findings in the same groups of animals strongly imply that similar alterations in the heart parameters can be related to insulin resistance and associated alterations in cardiometabolic disturbances.
We, here, demonstrated marked alterations in not only phosphorylation and expression levels of a gap-junctional protein Cx43 but also their localization in the cells. Under the light of literature documents,5,48 one can propose that these changes in Cx43 status may play an important role in the unsynchronized distribution of electrical conduction in the heart, particularly under pathological stimuli such as insulin resistance. Correspondingly, we determined almost similar changes in ECGs of both aged and MetS rats, which seemed to be parallel to the statuses and expression levels of Cx43. Also, the calculated ratio of pCx43 to Cx43 for the experimental groups was significantly smaller than that for the Con-group with more prominence in the MetS-group than in the Aged-group. This difference may be related to higher levels of blood glucose and OGTT results in the MetS-group than in the Aged-group. However, both the long-QT and long-QTc values were significantly prominent in the Aged-group than in the MetS-group. On the contrary, these significant differences in electrical parameters between the Aged-group and the MetS-group were also supported by the observations in our previous studies, as well.12,13 For instance, we observed marked arrhythmic action potentials in isolated left ventricular cardiomyocytes from the Aged-group but not from the MetS-group. These differences can explain, in part, the observed differences among the Aged-group and the MetS-group. Similar to findings in animals, in the aged human population (65 years and above), cardiac arrhythmia is a leading cause of mortality. 49 The mechanism responsible for cardiac arrhythmias, although multifactorial, can be broadly subdivided into disorders of automaticity and those of impulse conduction. These abnormalities seem to be related to the alteration in the expression, phosphorylation, and localization of Cx43, resulting in a change in anisotropy in the heart. 47 Accordingly, downregulation of Cx43 and abnormal topology of Cx43 in the aged guinea pigs’ atria have demonstrated the occurrence of inducible atrial fibrillation. 34 However, in that study, the authors note that atrial fibrillation is difficult to induce in young guinea pigs that have high levels of Cx43. In addition, they demonstrated reduced levels of not only total Cx43 expression but also its functional phosphorylated forms in aged guinea pigs’ atria. Their data are consistent with our present findings in the rats from the Aged-group.
Our present data provided more important information about the role of Cx43, including its expression, phosphorylation, and distribution in gap-junctional localizations (i.e., increases in lateralization, internalization, and degradation). Associated with those abnormalities, there should be other factors such as changes in the RhoA signaling pathway. For instance, some studies demonstrated that there is a close relationship between the activation of the RhoA pathway and insulin resistance. 50 Furthermore, inhibition of RhoA signaling, administration of a Cx43 mimetic peptide, or both could provide important cardioprotective therapeutic approaches to abnormalities in electrical activity and then the mechanical activity of the heart under metabolic disorders and the aging process. More importantly, the association between long-QT and hyperinsulinemia has also been detected in aged non-diabetics, which is supporting our present findings. 51 In this case, our present data are in line with the published data. Of note, it is widely accepted that cell-to-cell electrical coupling in the heart occurs mainly via gap junctions. 52 Similarly, in previous studies, a conduction of reduction in cell coupling in Cx43 knockdown rabbit myocytes such as slowing conduction and enhanced atrial arrhythmogenicity in the aged atrium has been demonstrated.53,54 These studies support the idea that not only a decrease in the expression of Cx43 but also its localization and phosphorylation processes can result in slowing of electrical conduction and increased susceptibility to cardiac arrhythmias, leading to marked dispersion of repolarization between epicardial and deeper myocardial layers. The above statements have corroborated our present findings. Correspondingly, some studies, besides ours, have shown that the role of some intracellular factors including changes in the level of intracellular Ca2+ can be associated with the phosphorylation and expression of Cx43 in the heart.4,12,55 Supporting this, it has been mentioned that the conduction velocity of ventricular muscle was reduced by the reduced Cx43 through the activation of the above proteases and phosphatases in the diabetic heart or hyperglycemic heart cells. 20 Similarly, recent studies address the fact that there is a close relationship between the activation of RhoA/ROCK and Cx43 activity, particularly in the functioning of the heart.23,56 Considering the important role of RhoA/ROCK in the regulation of Cx43-hemichannel activity under pathophysiological conditions 22 such as hyperglycemia, 57 the abnormal activation of the RhoA/ROCK pathway has an important action on abnormal cardiac electrical conduction through Cx43. Moreover, as one recent study shows the amelioration of cardiac remodeling and arrhythmogenesis with the administration of Cx43 mimetic peptide in rats with heart failure, 58 our present data (for MetS-group and Aged-group) also strongly support the contribution of RhoA activation to the abnormal expression and location of Cx43 and their relation with abnormal electrical conduction in failing hearts. With further data on parallel activation of ET-1 with RhoA,25,59 the findings of this study address the underlying mechanisms of long-QT in the insulin-resistant heart of aged mammals.
For clinical aspects, our data are supported by the study of Lucero et al. 58 that demonstrates the benefits of administration of Cx43 mimetic peptide Gap27 to rats suffering from heart failure via amelioration of cardiac remodeling and arrhythmogenesis. As discussed with several important original and review articles,60–63 maintenance of tissue homeostasis with connexins opens the door for not only scientific improvements but also commercial innovations of connexin-based therapeutics. It has been also well documented that the number of inherited human disorders stems from mutations in connexin genes, highlighting the indispensable role of gap junctions in a tissue dysfunction under many pathological stimuli. 64 As shown in our present and other data, studies of humans with heart diseases also reveal changes in connexin expression (particularly downregulation of Cx43 in the ventricular myocytes) and its subcellular localization in the damaged heart. 26 In these studies, it has been documented that there is a heterogeneous reduction in Cx43 expression and disordering in gap junction distribution in human ventricular disease and it correlates with electrophysiologically identified arrhythmic changes and contractile dysfunction in these samples. They also demonstrated the role of redistribution of gap junctions from the intercalated disc to the lateral borders of cardiomyocytes in the remodeling of the heart under failing conditions. 62 The observed lateralized Cx43 in these damaged hearts is concluded as the representation of altered anisotropic conduction, providing a substrate for arrhythmia, which further promotes cardiomyocyte death. 63 Overall insight to this subject: innovation is needed for the production of connexin-based therapeutics, although there is little information about the use of connexin-based therapeutics in the clinic for the treatment of either arrhythmia or any other heart disease with an enhancement of gap-junctional communication (i.e., improvement of lowered conduction) via treatment with a connexin-based therapeutic.65–67
Overall, we, here, demonstrated that not only reduced levels of pCx43 and Cx43 but also their apparent internalizations and lateralizations may be associated with abnormal ventricular repolarization, which in turn leads to long-QT in the Aged-group under insulin resistance, similar to the MetS-group. In this regard, although the underlying mechanisms of these two syndromes include alterations in electrophysiological properties of cardiomyocytes besides changes in the heart tissues, further studies are needed to clarify the exact mechanism responsible for the existence of these changes under insulin resistance. More importantly, we, for the first time, demonstrated that increased heterogeneity in ventricular repolarization of elderly heart, at least, through both reduced levels of pCx43 and Cx43 as well as their apparent internalizations and lateralizations can be an important risk factor for sudden cardiac death. Further examination of the relations emphasized here should bring new insights into the underlying mechanisms of this complex interaction and new preventive and/or therapeutic approaches.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
BT conceived the study and wrote the manuscript; YO and DB performed the majority of the experiments.
Data Availability
Data are available from the authors upon request.
Ethical Approval
All experimental procedures were approved by the local ethics committee of Ankara University (Decision No.: 2015-12-137 & No: 2016-18-165) and complied with the standards of the European Community Guidelines, the Directive 2010/63/EU, on the care and use of laboratory animals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants (No. SGAB-119S661) from The Scientific and Technological Research Council of Turkey.
