Abstract
Histochemistry of tumor sections is a widely employed technique utilized to examine cell death in preclinical xenograft animal models of cancer. However, this is under the assumption that tumors are homogeneous, leading to practices such as automatic cell counting across the entire section. We have noted that in our experiments the core of the tumor is largely or partially necrotic, and lacks evidence of vascularization (in contrast to the outer areas of the tumor). We note that this can bias and confound immunohistochemical analyses that do not take care to sample areas of interest in a way to take this into account. Design-based stereology with image analysis techniques is an alternative process that could be used to measure the volume of the necrotic region compared to the volume of the whole tumor.
Introduction
Xenograft tumor models are commonly employed in preclinical investigations of potential anticancer drugs.1,2 An advantage of subcutaneous implantation is that the tumor is readily palpable, and so tumor volume can be measured repeatedly over time using digital calipers. In addition to generating time course data, tumors measurable in situ allow researchers to begin drug treatment at a predetermined tumor, thus reducing between mouse variability and increasing statistical power. Hence, tumor volume and the effects of drug treatment can be assessed as the animal is still alive.3,4 By contrast, orthotopic models require tagged cells for in vivo imaging if this is to occur (with less precise measurements with current technology). 5 But, like orthotopic tumors, tumors from xenografts must be dissected before assays aimed at investigating mechanism of action can done.
Commonly Employed Analysis of Tumor Cell Death and Angiogenesis
Typically, tumors are cut into thin slices, between 5 and 10 µm, and examined with immunohistochemistry using various markers. Usually this involves examining apoptosis, angiogenesis, and specific proteins which drugs are thought to act on and/or regulate. However, when conducting tumor histology we found that xenograft non–small cell lung cancer tumors from male Nu/J mice after 14 days of drug administration (H3122 cells, see Bland et al. 6 for details) consisted of two distinct regions of cells (Fig. 1A). Highly dense cell regions with small nuclei were located mainly in the inner region of the tumors, and less dense cell populations with larger nuclei made up the surrounding tissue (Fig. 1A). The dense cell regions were heavily TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) stained in both the nuclei and the surrounding cytoplasmic regions (Fig. 1B). 6 TUNEL stain cannot discriminate between apoptosis, necrosis, and autolytic cell death,7–9 and so we hypothesize that in the center of the tumors necrosis has occurred due to hypoxia and/or glucose deprivation.10,11 Consistent with this hypothesis, within these dense regions there are TUNEL-positive nuclei with both karyorrhexis and karyolysis (lysis and fragmentation of the nucleus and nuclear content) within the cytoplasm. This was also evident by the size of the nuclei, where the TUNEL-positive nuclei are smaller compared with the TUNEL-negative nuclei, indicating nuclear shrinkage (Fig. 1B).12,13 We further hypothesized that this was due to rapid tumor growth resulting in insufficient vascularization of the tumor core to maintain cell viability, a phenomenon well characterized in rapidly growing human solid tumors.14–16 Corroborating this, CD105 staining, which indicates the presence of endothelial cells, was only present in the outer regions of the tumors (Fig. 1C). 6
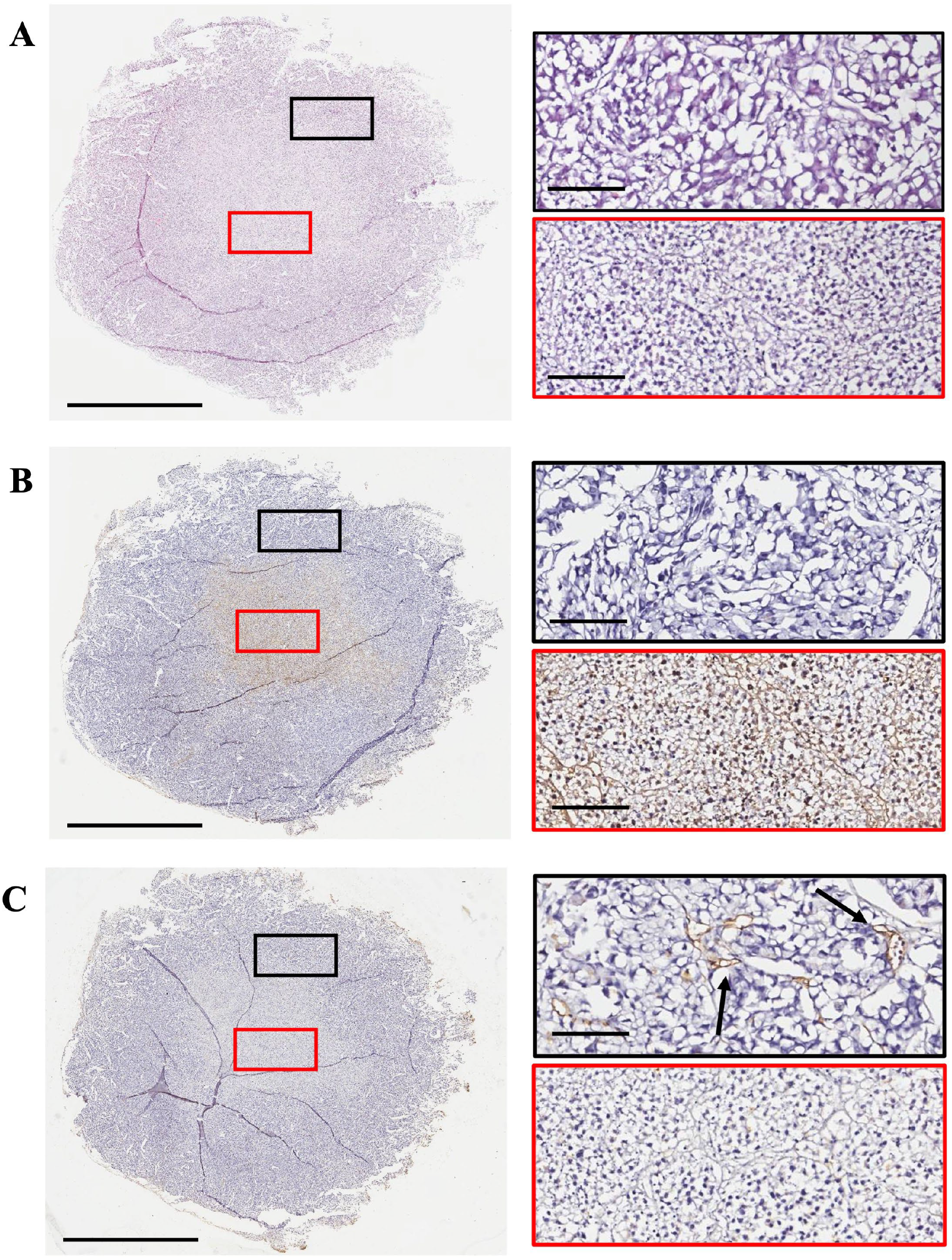
Tumor histology, apoptosis, and microvessel density staining from a Nu/J mice bearing a H3122 xenograft. The tumor slices were stained for (A) histology (using haematoxylin and eosin staining), (B) apoptosis (using TUNEL staining), and (C) microvessels (using CD105 staining). Haematoxylin was used as the counterstain for all sections. Images were taken using an Aperio Image ScanScope System (Leica Biosystems). The scale bar represents 3 mm for the images on the left and 200 µm for the images on the right. Red/black boxes on the right correspond to the respective region within the left image. Arrows point to microvessels.
Implications of Inadequate Tumor Analysis
The implication of this is that, as the inner region of the tumors is presenting as necrotic cores, caution must be taken when examining apoptosis and angiogenesis as measures of drug action. The same caution should extend to all other markers, such as cell migration and growth signaling proteins, as the live/dead regions within the tumors will alter the levels of these factors. Moreover, although in our experiments some tumors had only one region of necrosis (located within the core), others had two or more such regions, making it difficult to subtract these regions from analysis. When using tissue taken from mouse cancer xenografts, sections should be first investigated for the presence of a necrotic core and subsequent analyses be designed in such a way as to take this into account.
Considerations for Xenograft Study Design
Although rapid tumor growth and insufficient vascularization are the most common factors resulting in necrotic tumor cores, there are a number of other considerations that researchers must take into account. This includes the tumor age (i.e. the length of time between cell implantation and tumor excision), the location of the tumor and operator/injector error. These factors will also influence the vascularization of the tumor and hence, the presence of a necrotic region. Adequately powered experiments, along with the use of proper controls, will reduce any bias in the amount of vascularization between mice. Furthermore, when preparing the samples for immunohistochemical analysis of drug action, caution must be taken with pre-analytic factors. 17 Bagchi et al. (2021) reported when preparing organs for immunohistochemistry, the delay to fixation and the time in the fixative reagent can have significant implications on many cancer biomarkers. With a longer delay to fixation (>2 hours), some biomarkers were reduced (c-MET, p53 and pAKT), whilst others were increased (EGFR and CD10), when compared to a shorter time (1 hour). CD10 and c-MET were also reduced when the sections were in the fixative reagent for 72 hours, compared to the sections in 12 hours. The authors suggest that the delay to fixation should be below 1 hour and the time in the fixative reagent should be 12-24 hours to prevent bias from these pre-analytical factors. 17 Therefore, when designing and conducting a tumor experiment, factors such as the tumor age, location of the tumor and pre-analytic factors should be considered.
Recommendations for Tumor Analysis
Simply excluding the region of necrosis is not sufficient to accurately measure drug action, as the area immediately surrounding the dead tissue may be undergoing cell death (due to the hypoxia gradient within the tumor, and therefore, creating a gradient of altered cellular phenotypes). Measuring the necrotic core is also essential when examining cancer drugs with known effects of vascularization. For example, VEGF antagonists prevent new blood vessels being formed around the tumor, 18 and therefore, are likely to increase the size of the necrotic core. Furthermore, the appearance of necrosis may not be seen immediately and cells may be undergoing early stages of cell death. In order to prevent the issues addressed in this piece, we discuss morphometric analysis recommendations that if performed accurately, along with considerations that should be used in the initial tumor study design, could allow appropriate screening of drug markers within xenograft tumors.
The minimum requirements should include information on the total area of the tumor, the region that has been analyzed and the area of the necrotic region (if applicable) along with location of the areas analyzed. This would exclude the bias of random, small fields that may not take into account the necrotic core of the tumor. However, we advise that to adequately measure tumor biomarkers, the researcher should not overlook the heterogeneity of the tumor by only examining one tumor section, a common practice employed when undertaking a xenograft study. If only one slice of the tumor is used, bias is created due to the variability across the whole tumor. For example, if the section is taken near the edge of the tumor, a necrotic region may not be present, and the researcher is selecting a population of cells closer to the blood vessels formed around the tumor, as opposed to cells within the center of the tumor which are exposed to hypoxia and ischemia. In order to correct this issue, sampling protocols that incorporate design-based stereology should be utilized.19-21 Design-based stereology incorporates a higher precision design, and reducing bias, by utilizing rigorous sampling protocols. For a tumor study, this would involve using more tumor sections and fields of view (i.e. the whole tumor section as opposed to small, random areas). This would allow the researcher to take into account the understanding that tumors are complex, heterogenic, 3D structures, as opposed to 2D planes. Therefore, although considered more time consuming, stereology combined with image analysis, can examine the whole tumor and the volume that is necrotic compared to live, increasing the validity of the results and reducing bias.20,21 This data can also be correlated to the volume measurements taken from the animal, using digital callipers, before the animal was euthanized and the tumor excised. Apoptosis can also be measured across the tumor by using a apoptosis-specific markers, such as cleaved caspase-3, bim, bax or bcl-2.22,23 As mentioned earlier, we do not recommend using the TUNEL stain as this cannot accurately discriminate between apoptosis, necrosis and autolytic cell death.7-9 This stereology and image analysis process could provide new information on drug.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ARB was involved in data curation, formal analysis, investigation, writing—original draft, and writing—review and editing. JCA was involved in conceptualization, formal analysis, methodology, project administration, resources, software, supervision, and writing—review and editing. Both authors have read and approved the manuscript prior to submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a University of Otago School of Biomedical Sciences Deans Bequest Grant.
