Abstract
Intrahepatic bile ducts transport bile between bile canaliculi and the extrahepatic bile duct. The luminal surface of this tract is lined by a layer of biliary epithelial cells, or cholangiocytes, which secrete mucins consisting of scaffold proteins and O-glycosidically linked carbohydrate side chains. Although mucin core proteins have been extensively investigated, the structure and function of carbohydrate side chains have not. Here, we demonstrate that distinct sulfated glycans positive for MECA-79, R-10G, and 297-11A, but not 5D4, monoclonal antibodies are expressed in the cytoplasm of cells of large-sized ducts and in the apical membrane of cells in ductules, and that R-10G immunolabeling is partially eliminated by endo-β-galactosidase digestion, supporting the presence of N-acetylglucosamine-6-O-sulfated N-acetyllactosamine structures. We observed comparable apical membrane-predominant staining in ductular reactions seen during regeneration that occurs in various liver diseases and in cholangiolocarcinoma, a subtype of small duct-type intrahepatic cholangiocarcinoma (iCCA). Apical membrane expression of distinct sulfated glycans in large duct-type iCCA was negligible. Intriguingly, under pathological conditions, endo-β-galactosidase digestion almost completely eliminated R-10G immunoreactivity. These findings suggest that apical membrane expression of distinct sulfated glycans is a characteristic feature of ductules and their reactive and neoplastic counterparts
Keywords
Introduction
The biliary tract is an excretory system that drains bile produced by hepatocytes all the way to the duodenum. Anatomically, it is divided into extrahepatic and intrahepatic bile ducts, with the latter further divided into large- and small-sized intrahepatic bile ducts. Large-sized intrahepatic ducts, such as segmental and area ducts, are recognizable by visual inspection and accompanied by peribiliary glands; by contrast, small-sized intrahepatic ducts, such as septal and interlobular ducts and ductules, are identifiable only by light microscopy and are essentially not associated with peribiliary glands. 1 Among them, ductules are the most peripherally distributed intrahepatic bile ducts that connect to the canal of Hering, which is composed of both ductular cells and hepatocytes. Throughout the biliary tract, the luminal surface is lined by a layer of columnar to cuboidal biliary epithelial cells, or cholangiocytes, which secrete mucins and other molecules functioning in maintenance and regulation of the biliary system. 2 Although cholangiocytes are heterogeneous in size, shape, and function along the biliary tract, mucin secretion is a major function of all of them.
Mucins are epithelial glycoproteins, the primary function of which is to protect and lubricate the mucosal surface. 2 Structurally, they consist of a core protein decorated with numerous clustered oligosaccharides O-glycosidically linked to tandem repeat regions of the core protein rich in serine (Ser) and threonine (Thr). 2 Thus far, 20 mucin core proteins (MUCs) have been cloned in humans. Among them, MUC1, also known as epithelial membrane antigen (EMA), is expressed in the cytoplasm of cells in large-sized intrahepatic ducts and in the apical membrane of cells in small-sized intrahepatic bile ducts (except the canal of Hering). 3 Exploiting this differential expression pattern, apical membrane expression of MUC1 has been used in the histological diagnosis of cholangiolocarcinoma, 4 a subtype of small duct-type intrahepatic cholangiocarcinoma (iCCA) based on the latest World Health Organization (WHO) classification. 5 Besides MUC1, MUC6 is reportedly expressed in a scattered pattern in normal small-sized intrahepatic bile ducts, including the canal of Hering, and MUC6 expression significantly increases in ductular reactions, particularly those associated with chronic viral hepatitis. 6 However, the structure and function of carbohydrate side chains attached to MUCs are not fully understood.
We previously demonstrated that 6-sulfo (N-acetylglucosamine [GlcNAc]-6-O-sulfated) N-acetyllactosamine attached to extended core 1 O-glycans, which is recognized by MECA-79 monoclonal antibody, is expressed throughout intrahepatic bile ducts. 3 GlcNAc-6-O-sulfotransferase 2 (GlcNAc6ST-2), also known as high endothelial cell GlcNAc6ST (HEC-GlcNAc6ST) or L-selectin ligand sulfotransferase (LSST),7,8 is responsible for the expression of MECA-79-positive sulfated glycans in cholangiocytes. 3 Interestingly, similar to MUC1 core proteins, the expression of MECA-79-positive sulfated glycans in small-sized intrahepatic ducts is essentially localized to the apical membrane; on the contrary, in large-sized ducts, the expression of MECA-79-positive sulfated glycans is primarily cytoplasmic, most likely as intracytoplasmic mucins. We also have reported that the expression of MECA-79 glycoepitopes in the apical membrane of ductular structures characterizes cholangiolocarcinoma, 3 a subtype of small duct-type iCCA formerly called cholangiolocellular carcinoma. 4 These findings prompted us to ask whether apical membrane expression of sulfated glycans is a marker of bile ductules, either physiological or pathological.
Here, to gain insight into the structural basis of sulfated glycans expressed in intrahepatic bile ducts, we carried out immunohistochemical analysis of normal liver tissues using glycan sequence-specific monoclonal antibodies combined with various glycosidase pretreatments. That analysis allowed us to infer the most probable glycan structures. We conducted similar immunohistochemical analysis using neoplastic and reactive counterparts of bile ductules, namely, cholangiolocarcinoma and ductular reactions associated with chronic viral hepatitis, to assess similarities and differences with normal ductules.
Materials and Methods
Human Liver Tissues
Formalin-fixed, paraffin-embedded tissue blocks of normal healthy liver samples representative of tissues used for living donor liver transplantation (LDLT) (n=5) and tissues representing chronic viral hepatitis with prominent ductular reactions (n=5) and cholangiolocarcinoma (n=5), as well as tissues representing small (n=5) and large (n=5) duct-type iCCA, were retrieved from the pathology archives of both Shinshu University School of Medicine and the Faculty of Medical Sciences, University of Fukui. Detailed information relevant to sample sources is summarized in Table 1. Analysis of human liver tissues was approved by the Ethics Committee of Shinshu University School of Medicine (reference number 2766, approved July 7, 2014; reference number 5072, approved March 15, 2021) and that of the Faculty of Medical Sciences, University of Fukui (reference number 20160038, approved October 30, 2016).
Clinicopathological Information of Liver Tissues Examined in This Study.
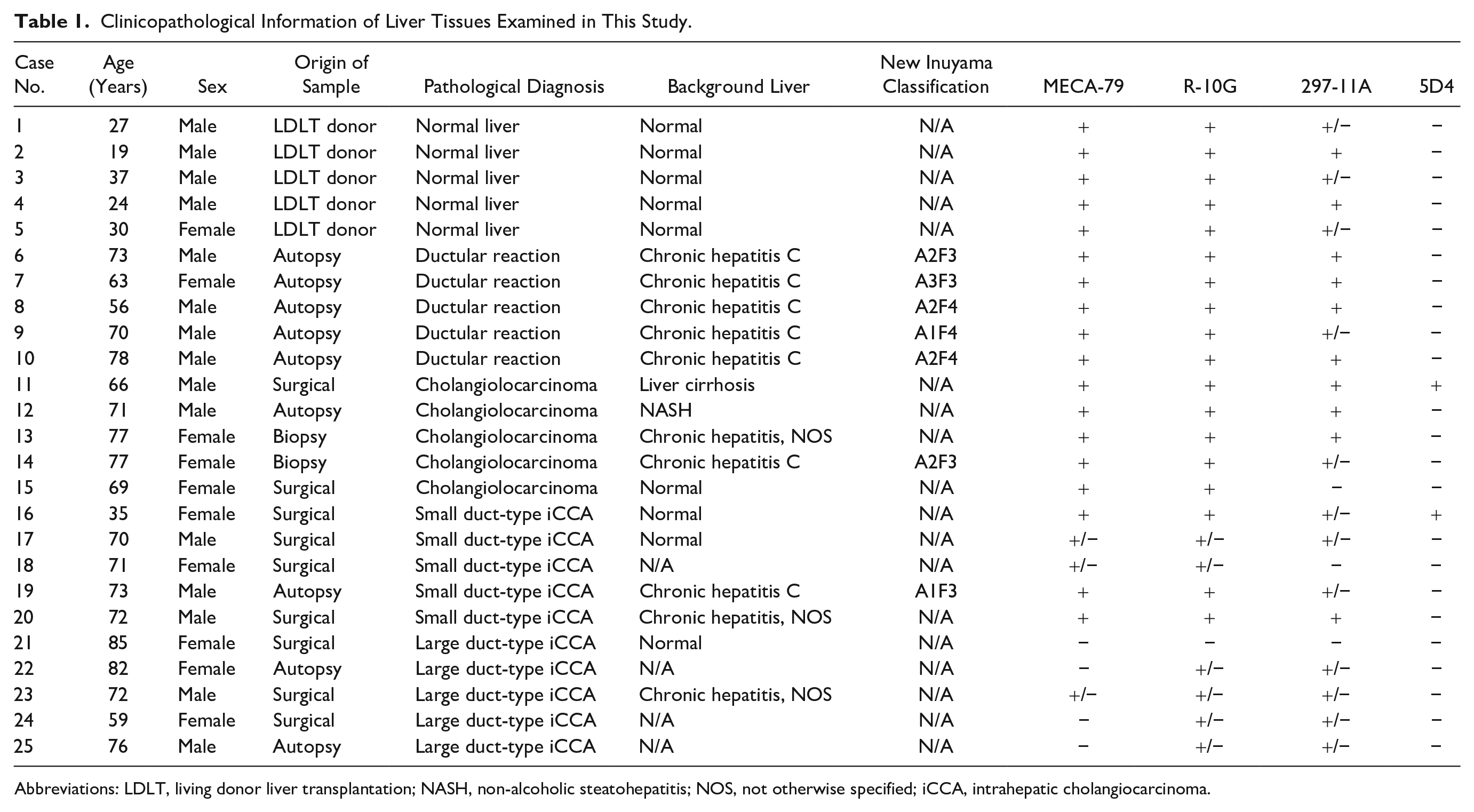
Abbreviations: LDLT, living donor liver transplantation; NASH, non-alcoholic steatohepatitis; NOS, not otherwise specified; iCCA, intrahepatic cholangiocarcinoma.
Pretreatment of Tissue Sections
To remove N-glycans, tissue sections were incubated with 5000 U/ml (dissolved in 50 mM sodium acetate, pH 6.0) of peptide-N-glycosidase F (PNGase F; cat. no. P0704S; New England BioLabs, Ipswich, MA) at 37C for 90 min in a humidified chamber. 9 Similarly, to degrade keratan sulfate glycosaminoglycans, sections were incubated with 50 µg/ml (dissolved in 30 mM sodium acetate, pH 5.2) of recombinant keratanase II (from Bacillus circulans) prepared as described. 10 To cleave internal β1,4-galactosidic linkages in non-sulfated or GlcNAc-6-O-sulfated oligo- or poly-N-acetyllactosamine, sections were incubated with 50 µg/ml (dissolved in 30 mM sodium acetate, pH 5.2) of endo-β-galactosidase (from Citrobacter freundii) purified as described.11,12 As positive controls for enzymatic digestion, we performed immunohistochemistry with 5D4 antibody using porcine corneal tissues rich in N-glycosidically linked keratan sulfate glycosaminoglycans (Supplemental Figs. 1 and 2).
Monoclonal Antibodies
The following monoclonal antibodies served as primary antibodies: MECA-79 (rat IgM; BD Biosciences, Franklin Lakes, NJ) recognizing 6-sulfo (GlcNAc-6-O-sulfated) N-acetyllactosamine attached to extended core 1 O-glycans, Galβ1→4(SO4→6)GlcNAcβ1→3Galβ1→3GalNAcα1→Ser/Thr (in which GalNAc is N-acetylgalactosamine)13,14; R-10G (mouse IgG; Tokyo Chemical Industry, Tokyo, Japan) recognizing repeated N-acetyllactosamine tetrasaccharide, in which GlcNAc but not Gal residues are 6-O-sulfated, Galβ1→4(SO4→6)GlcNAcβ1→3Galβ1→4(SO4→6)GlcNAc1→R15 –17; 297-11A (mouse IgM) recognizing terminally positioned 6′-sulfo (Gal-6-O-sulfated) N-acetyllactosamine disaccharide, SO4→6Galβ1→4GlcNAcβ1→R, regardless of the GlcNAc-6-O-sufation 9 ; 5D4 (mouse IgG; Seikagaku Corporation, Tokyo, Japan) recognizing highly sulfated (both GlcNAc- and Gal-6-O-sulfated) oligo- or poly-N-acetyllactosamine in keratan sulfate glycosaminoglycans 18 ; EPR1023 (rabbit IgG; Abcam, Cambridge, UK) recognizing human MUC1; and MUC6/1553R (rabbit IgG; Abnova, Taipei, Taiwan) recognizing human MUC6.
Immunohistochemistry
Immunohistochemical staining for MECA-79 was carried out using an indirect method as described, 19 and that for R-10G, 297-11A, and 5D4 was undertaken using the Histofine system (Nichirei Biosciences; Tokyo, Japan) as per the manufacturer’s instructions. Briefly, after deparaffinization and rehydration, endogenous peroxidase activity was blocked by soaking sections in absolute methanol containing 0.3% H2O2 for 30 min. Antigens were then retrieved by autoclaving sections in 10 mM Tris/HCl buffer (pH 8.0) containing 1 mM ethylenediaminetetraacetic acid (EDTA) at 105C for 15 min. After blocking 30 min with 1% bovine serum albumin (BSA) in Tris-buffered saline (TBS), sections were incubated with primary antibodies at 4C overnight, followed by incubation with Histofine Simple Stain MAX PO (Nichirei Biosciences) for 30 min. The color reaction was developed using a Betazoid DAB Chromogen Kit (Biocare Medical; Pacheco, CA). Sections were briefly counterstained with hematoxylin. Double-immunofluorescence staining for MECA-79 plus either MUC1 or MUC6 was conducted essentially as described. 20
Quantification of Immunolabeling Intensity
ImageJ software version 2.1.0/1.53c (National Institutes of Health; Bethesda, MD) was used for the analysis. Grayscale processing and noise smoothing were performed on each target image with constant resolution and size. After that, a threshold value was set for the image, and the image was binarized. Of bits within the threshold, those considered background were eliminated. We then counted bits that formed clusters of a certain size.
Statistical Analysis
Data are expressed as means + SD of four images. Differences between groups were statistically analyzed by ordinary one-way ANOVA with Dunn’s multiple comparison test using GraphPad Prism 7 software (GraphPad Software; San Diego, CA). Values of p less than 0.05 were considered significant.
Results
Distinct Sulfated Glycans Are Expressed in Intrahepatic Bile Ducts
We first carried out immunohistochemical staining of human normal liver tissues with a battery of anti-sulfated glycan monoclonal antibodies, namely, MECA-79, R-10G, 297-11A, and 5D4. As shown in Fig. 1, cholangiocytes that constitute intrahepatic bile ducts, including the canal of Hering, were positive for MECA-79, R-10G, and 297-11A but not for the anti-keratan sulfate antibody 5D4. Interestingly, immunolocalization of glycoepitopes recognized by MECA-79, R-10G, and 297-11A differed depending on the caliber of intrahepatic bile ducts. For example, in large-sized ducts, glycoepitopes recognized by these antibodies were cytoplasmic, suggesting that the antibodies recognize an intracytoplasmic mucin (Fig. 1, left column). Comparable cytoplasmic immunolocalization was also observed in peribiliary glands (Fig. 1, left column, arrows). By contrast, in interlobular ducts and ductules, MECA-79 and R-10G glycoepitopes were expressed both on the plasma membrane and in the cytoplasm but in an apical membrane-predominant fashion (Fig 1, right column, second and third rows), whereas 297-11A glycoepitopes were expressed almost exclusively in the apical membrane of cholangiocytes (Fig. 1, right column, fourth row). We note that bile canaliculi of some hepatocytes adjacent to the canal of Hering also expressed sulfated glycans recognized by MECA-79, R-10G, and 297-11A antibodies (Fig. 1, right column insets, arrowheads). Hepatocytes were basically negative for MECA-79 and 297-11A antibodies, and only weak cytoplasmic signals for R-10G were observed (Fig. 1, right column, asterisks).
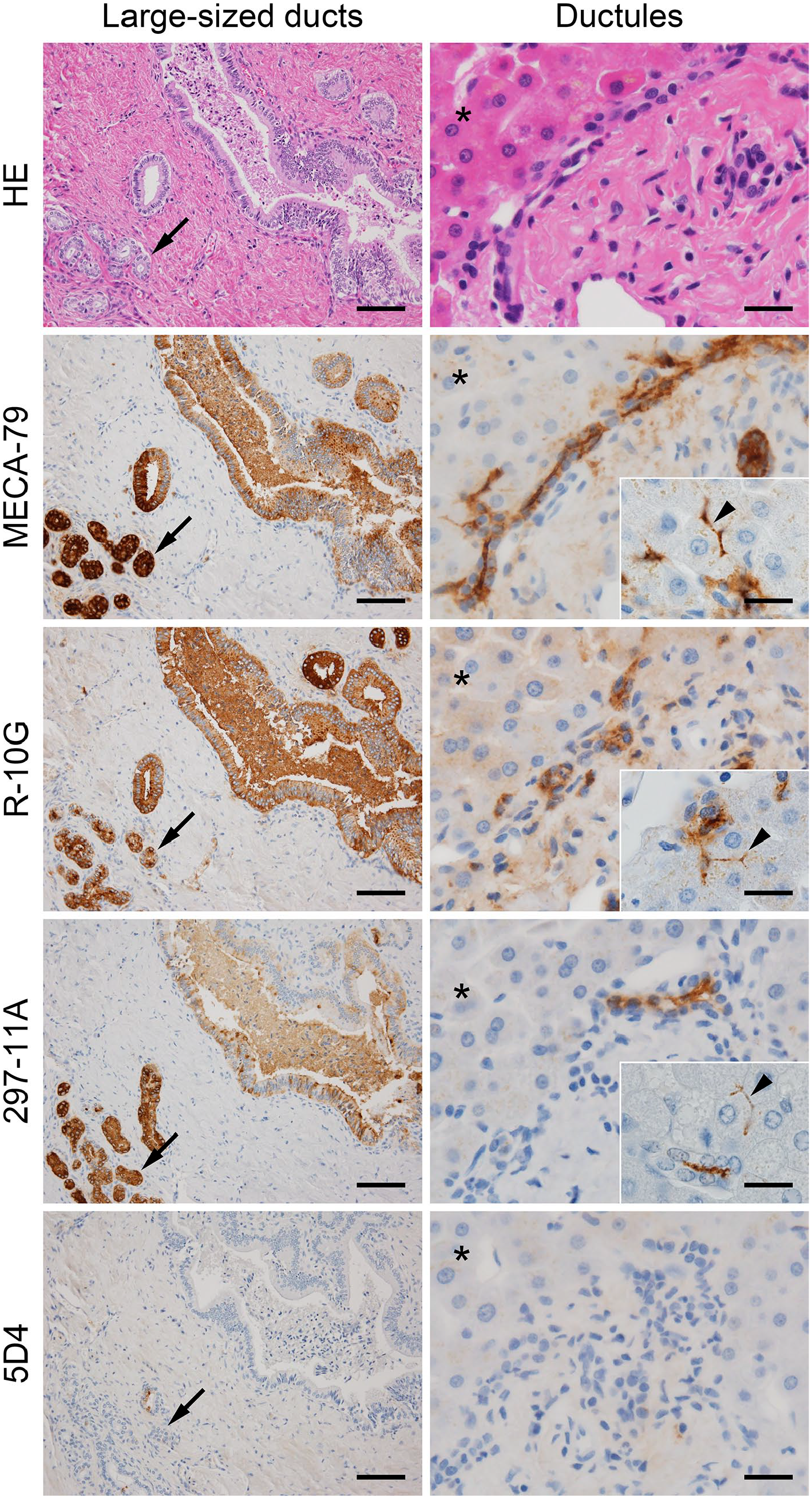
Expression of distinct sulfated glycans in large-sized ducts (left column) and ductules (right column). Serial tissue sections were stained with hematoxylin and eosin (H&E) and immunostained for MECA-79, R-10G, 297-11A, and 5D4. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Arrows in left column indicate peribiliary glands, arrowheads in right column insets indicate hepatocyte bile canaliculi adjacent to the canal of Hering, and asterisks in right column indicate hepatocytes. Bar = 100 µm for left column, 25 µm for right column, and 20 µm for right column insets. Similar immunohistochemical findings were obtained from five cases of normal liver. Thus representative photomicrographs are shown.
Sulfated Glycans Expressed in Intrahepatic Bile Ducts Are Present Primarily on O-glycans
As reported above, sulfated glycans expressed in intrahepatic bile ducts were completely negative for 5D4 (see Fig. 1, bottom row), indicating that those glycans are not keratan sulfate glycosaminoglycans. To corroborate this interpretation, before immunostaining for MECA-79, R-10G, and 297-11A, we treated tissue sections with keratanase II, an endo-β-N-acetylglucosaminidase (or, more precisely, an endo-β-
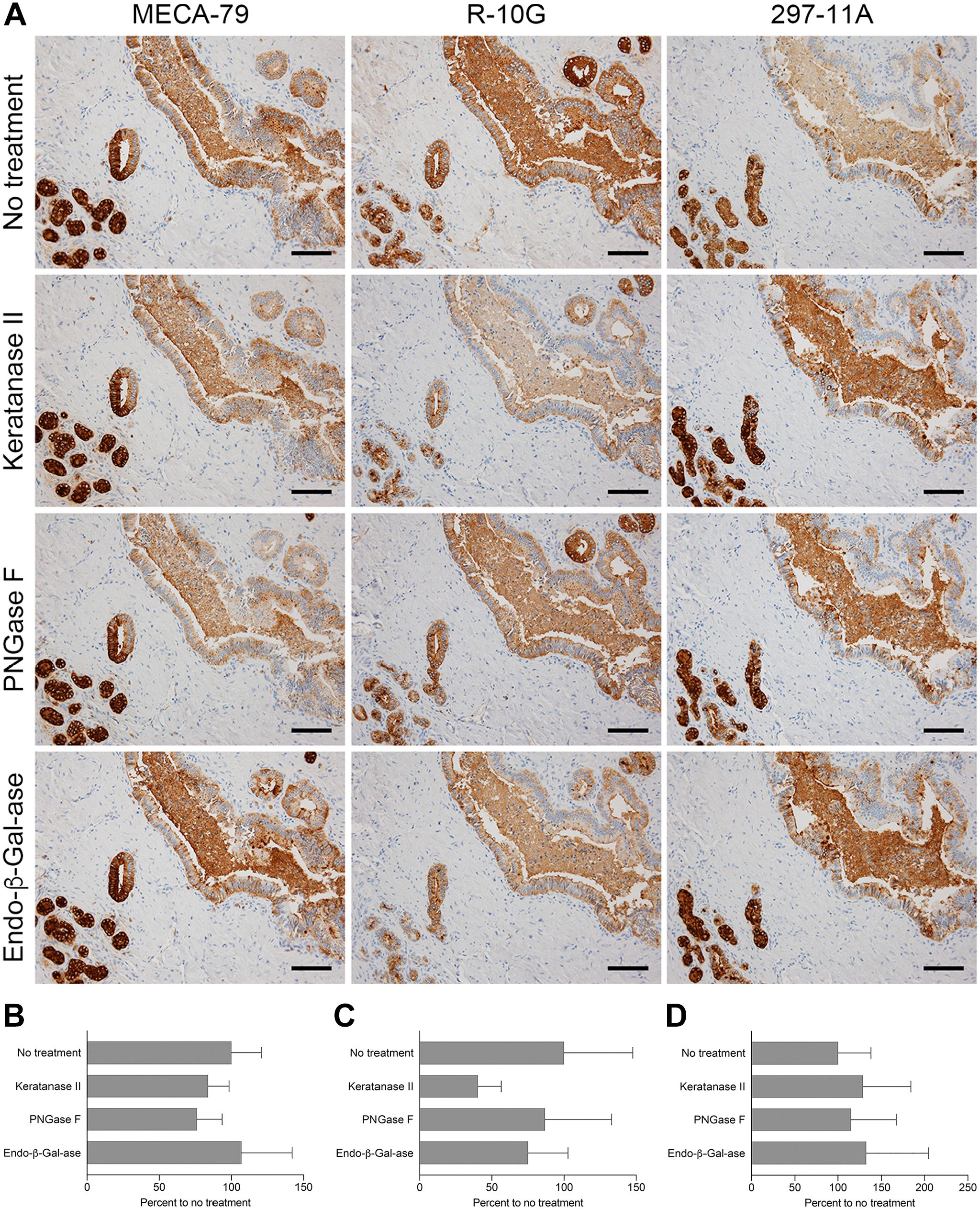
Immunohistochemical profile of sulfated glycans expressed in large-sized ducts with or without a series of glycosidase pretreatments. (A) Serial tissue sections were treated with keratanase II, peptide-N-glycosidase F (PNGase F), or endo-β-galactosidase (endo-β-Gal-ase) before immunostaining for MECA-79, R-10G, and 297-11A. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 100 µm. Photomicrographs in the top row are the same as their counterparts shown in Fig. 1. (B–D) Quantification of signal intensity of MECA-79 (B), R-10G (C), and 297-11A (D), with or without the indicated glycosidase pretreatments shown in panel (A). Data are presented as means + SD of four segments of each image. The X-axis represents the percentage relative to no treatment.
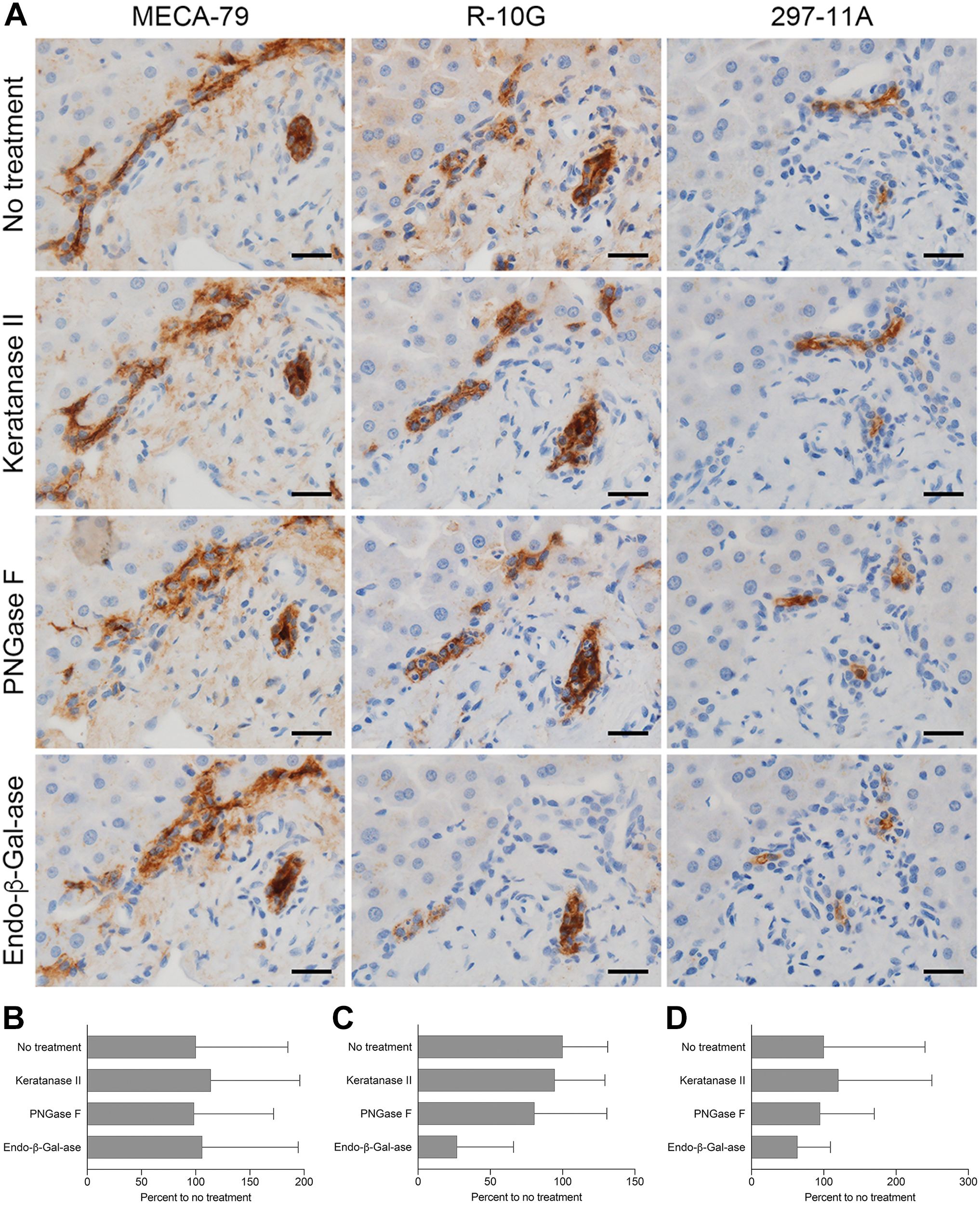
Immunohistochemical profile of sulfated glycans expressed in ductules with or without a series of glycosidase pretreatments. (A) Serial tissue sections were treated with keratanase II, peptide-N-glycosidase F (PNGase F), or endo-β-galactosidase (endo-β-Gal-ase) before immunostaining for MECA-79, R-10G, and 297-11A. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 25 µm. Photomicrographs in the top row are the same as their counterparts shown in Fig. 1. (B–D) Quantification of signal intensity of MECA-79 (B), R-10G (C), and 297-11A (D), with or without the indicated glycosidase pretreatments shown in panel (A). Data are presented as means + SD of four segments of each image. The X-axis represents the percentage relative to no treatment.
We then asked whether these sulfated glycans are displayed on N-glycans [GlcNAc attached to asparagine (Asn)] or O-glycans (GalNAc attached to Ser or Thr). To do so, we treated tissue sections with PNGase F, which cleaves the innermost GlcNAc and Asn residues, thus removing a substantial proportion of N-linked oligosaccharides from glycoproteins (see Supplemental Figs. 1 and 2). As shown in Figs. 2A and 3A (third row of both), signals for all three antibodies persisted even after PNGase F digestion, indicating that sulfated glycans are not borne on N-glycans but on O-glycans.
We also treated tissue sections with endo-β-galactosidase, which cleaves β1,4-galactosidic linkages to GlcNAc when the Gal residue is not 6-O-sulfated, regardless of the sulfation on adjacent GlcNAc. 15 After endo-β-galactosidase digestion, R-10G signal intensity in ductules decreased to ~25% of that seen in untreated tissues (Fig. 3C), while signals for MECA-79, R-10G, and 297-11A antibodies were comparable (Figs. 2A and 3A, bottom row of both). This finding indicates that a substantial proportion of R-10G-reactive glycan side chains contains endo-β-galactosidase-resistant carbohydrate sequences.
Distinct Sulfated Glycans Are Expressed in Ductular Structures Formed in Pathological Conditions
We previously demonstrated that MECA-79-positive sulfated glycans are expressed on the luminal surface of ductular structures formed in cholangiolocarcinoma. 3 In that study, however, we did not assess R-10G and 297-11A antibody immunoreactivity against these sulfated glycans. Moreover, we have not yet determined whether these sulfated glycans are also expressed in ductular reactions, defined as reactive processes at the interface of portal and parenchymal compartments in human liver that arise in various diseases and injuries.21,22 Thus, we carried out the same immunohistochemical analysis as reported above in normal liver, using liver tissue sections from specimens representing cholangiolocarcinoma and chronic viral hepatitis with prominent ductular reactions. As shown in Fig. 4 (left column), MECA-79, R-10G, and 297-11A stained both the luminal surface and cytoplasm of ductular structures formed in cholangiolocarcinoma but in an apical membrane-predominant fashion, as seen in normal ductules (see Fig. 1, right column); however, the immunohistochemical profile after a series of glycosidase treatments differed in ways from that seen in normal ductules. First, as shown in Fig. 5A (left column), MECA-79 signals increased after digestion with keratanase II (to ~360% of untreated tissues; Fig. 5B) and with endo-β-galactosidase (to ~280% of untreated tissues; Fig. 5B). Second, endo-β-galactosidase digestion almost completely eliminated R-10G immunolabeling (Fig. 5A, middle column, bottom panel; compare with that seen in Fig. 3A). Third, as shown in Fig. 5A (right column), 297-11A signals markedly decreased after PNGase F digestion (to ~20% of untreated tissues; Fig. 5D) and after endo-β-galactosidase digestion (to ~30% of untreated tissues; Fig. 5D). Moreover, we also observed an immunohistochemical profile comparable with that seen in cholangiolocarcinoma in ductular reactions associated with chronic viral hepatitis (Figs. 4 and 6). These findings collectively suggest that apical membrane expression of distinct sulfated glycans is not only a characteristic of normal ductules but also a feature of their reactive and neoplastic counterparts, and that glycosidase susceptibility is altered in these pathological conditions.
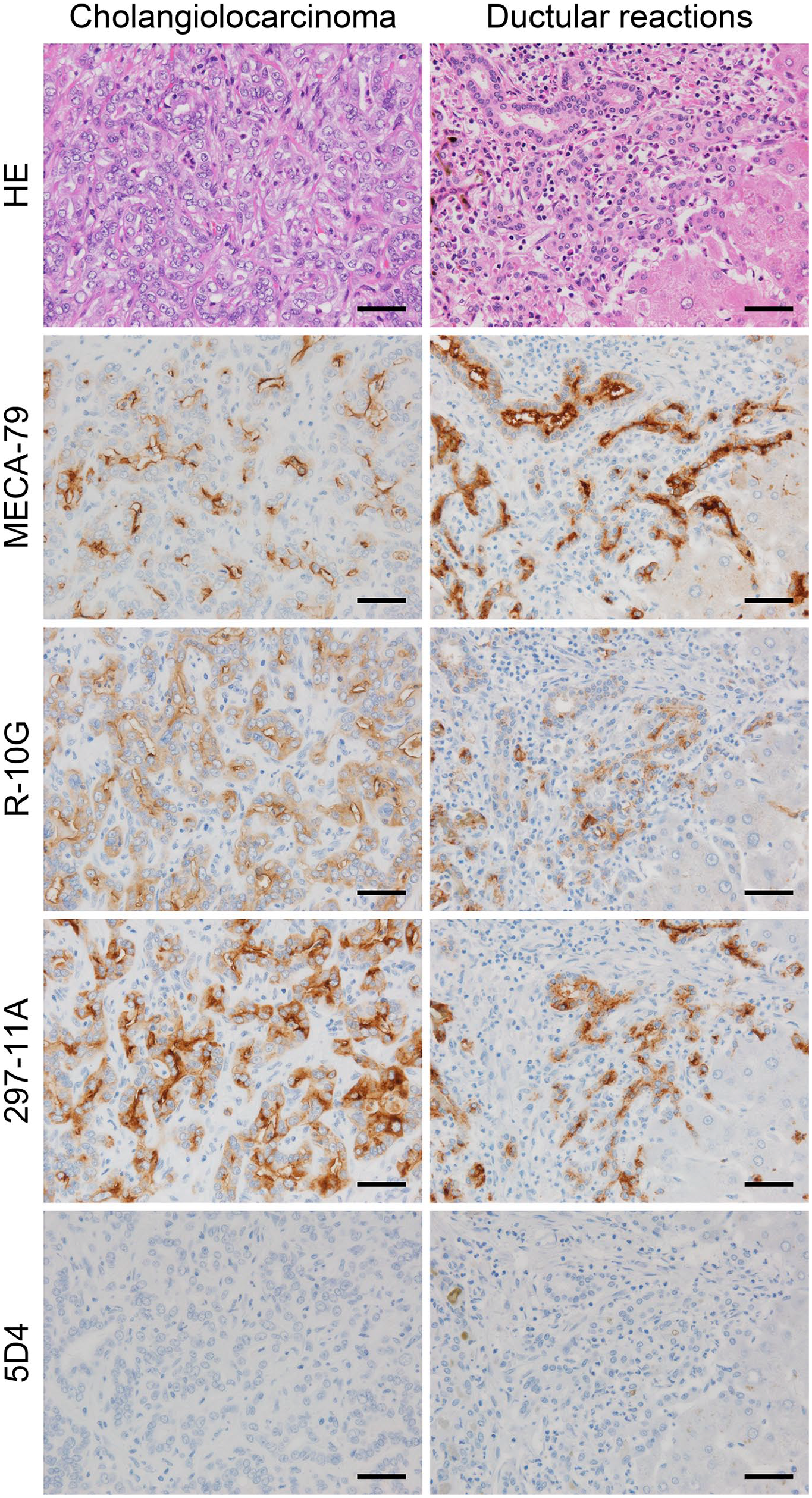
Expression of distinct sulfated glycans in cholangiolocarcinoma (left column) and ductular reactions associated with chronic viral hepatitis (right column). Serial tissue sections were stained with hematoxylin and eosin (H&E) and immunostained for MECA-79, R-10G, 297-11A, and 5D4. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 50 µm. Similar immunohistochemical findings were obtained from five cases each of cholangiolocarcinoma and chronic viral hepatitis specimens with prominent ductular reactions. Thus, representative photomicrographs for each disease are shown.
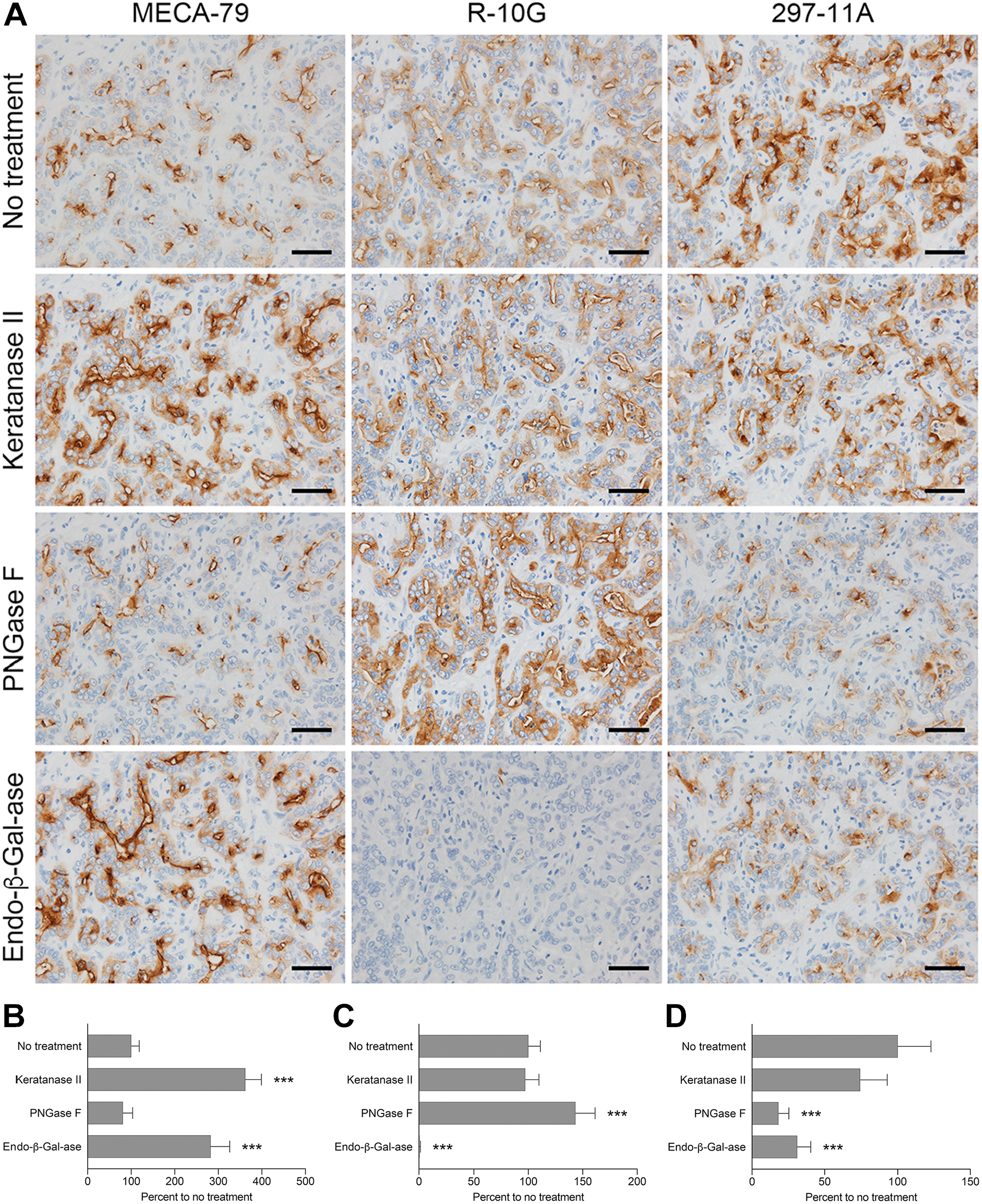
Immunohistochemical profile of sulfated glycans expressed in cholangiolocarcinoma with or without a series of glycosidase pretreatments. (A) Serial tissue sections were treated with keratanase II, peptide-N-glycosidase F (PNGase F), or endo-β-galactosidase (endo-β-Gal-ase) before immunostaining for MECA-79, R-10G, and 297-11A. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 50 µm. Photomicrographs in the top row are the same as their counterparts shown in Fig. 4. (B–D) Quantification of signal intensity of MECA-79 (B), R-10G (C), and 297-11A (D), with or without the indicated glycosidase pretreatments shown in panel (A). Data are presented as means + SD of four segments of each image. The X-axis represents the percentage relative to no treatment. ***p<0.001.
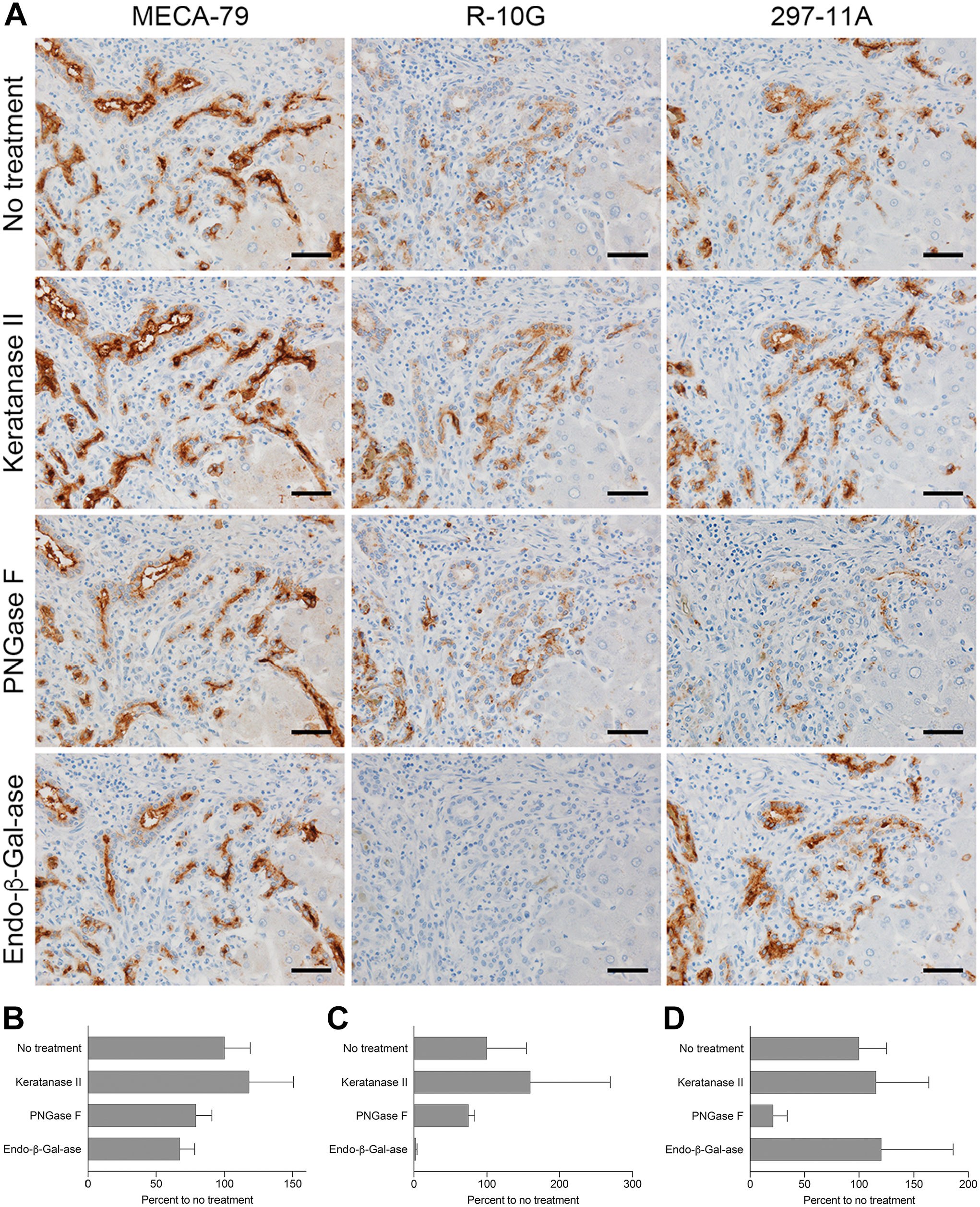
Immunohistochemical profile of sulfated glycans expressed in ductular reactions associated with chronic viral hepatitis with or without a series of glycosidase pretreatments. (A) Serial tissue sections were treated with keratanase II, peptide-N-glycosidase F (PNGase F), or endo-β-galactosidase (endo-β-Gal-ase) before immunostaining for MECA-79, R-10G, and 297-11A. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 50 µm. Photomicrographs in the top row are the same as their counterparts shown in Fig. 4. (B–D) Quantification of signal intensity of MECA-79 (B), R-10G (C), and 297-11A (D), with or without the indicated glycosidase pretreatments shown in panel (A). Data are presented as means + SD of four segments of each image. The X-axis represents the percentage relative to no treatment.
Both MUC1 and MUC6 Function as Scaffold Proteins for Distinct Sulfated Glycans
We previously demonstrated that MUC1 is a scaffold protein for MECA-79 sulfated glycans. 3 To determine whether MUC6 performs a similar function, we conducted double-immunofluorescence staining for MECA-79 and MUC6 using tissue sections of cholangiolocarcinoma and ductular reactions. In the cholangiolocarcinoma samples, as we reported previously, 3 MECA-79 and MUC1 signals colocalized (Fig. 7, top row). In addition, MUC6 was not expressed in cholangiolocarcinoma in any specimens tested (Fig. 7, second row). However, in ductular reactions, MUC1 was minimally expressed (Fig. 7, third row) and MUC6 was expressed chiefly in the cytoplasm (Fig. 7, bottom row). Moreover, a substantial proportion of MECA-79 signals colocalized with signals for MUC6. Taken together, these findings suggest that MUC1 and MUC6 are distinct MUCs preferentially expressed in cholangiolocarcinoma and ductular reactions, respectively, and both may function as a scaffold protein for unique cholangiocyte-derived sulfated glycans.
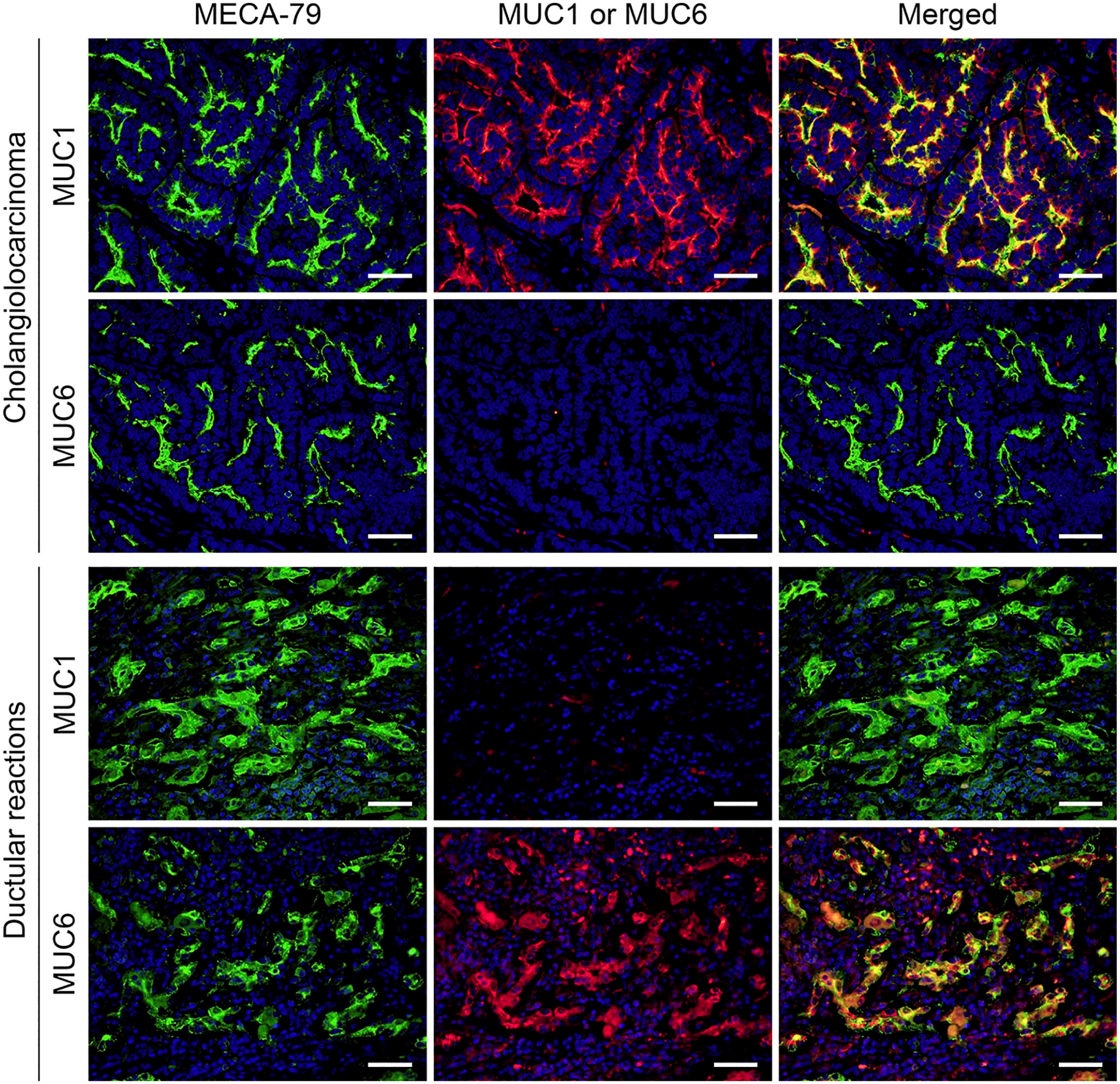
Double-immunofluorescence staining of cholangiolocarcinoma (upper two rows) and ductular reactions (lower two rows) for MECA-79 sulfated glycans (green) and either MUC1 (top and third rows) or MUC6 (second and bottom rows) core proteins (red). Yellow signals in merged panels indicate antigen colocalization. Sections were stained with 4′,6-diamidino-2-phenylindole (DAPI) to mark nuclei. Note that the expression of MECA-79 sulfated glycans is observed in both the apical membrane and cytoplasm but in apical membrane-predominant fashion, particularly in cholangiolocarcinoma. Bar = 50 µm.
Distinct Sulfated Glycans Are Expressed in Small Duct-type Relative to Large Duct-type iCCA
We then assessed immunohistochemical profiles of sulfated glycans expressed in other iCCA subtypes. In small duct-type iCCA (Fig. 8, left column), we observed an apical membrane-predominant expression pattern similar to that of sulfated glycans seen in cholangiolocarcinoma. This expression pattern was consistent in four of five cases of small duct-type iCCA (see Table 1). Also like results seen in cholangiolocarcinoma, endo-β-galactosidase digestion completely eliminated R-10G immunolabeling (Fig 9A, middle column, bottom panel, and Fig. 9C) and significantly decreased 297-11A signals (to ~50% of untreated tissues; Fig 9A, right column, bottom panel, and Fig. 9D). By contrast, in large duct-type iCCA, expression of MECA-79-positive sulfated glycans was negative in four of five specimens (see Table 1). Expression of R-10G- and 297-11A-positive sulfated glycans varied among specimens, but apical membrane expression of both was negligible (Fig. 8, right column, third and fourth rows).
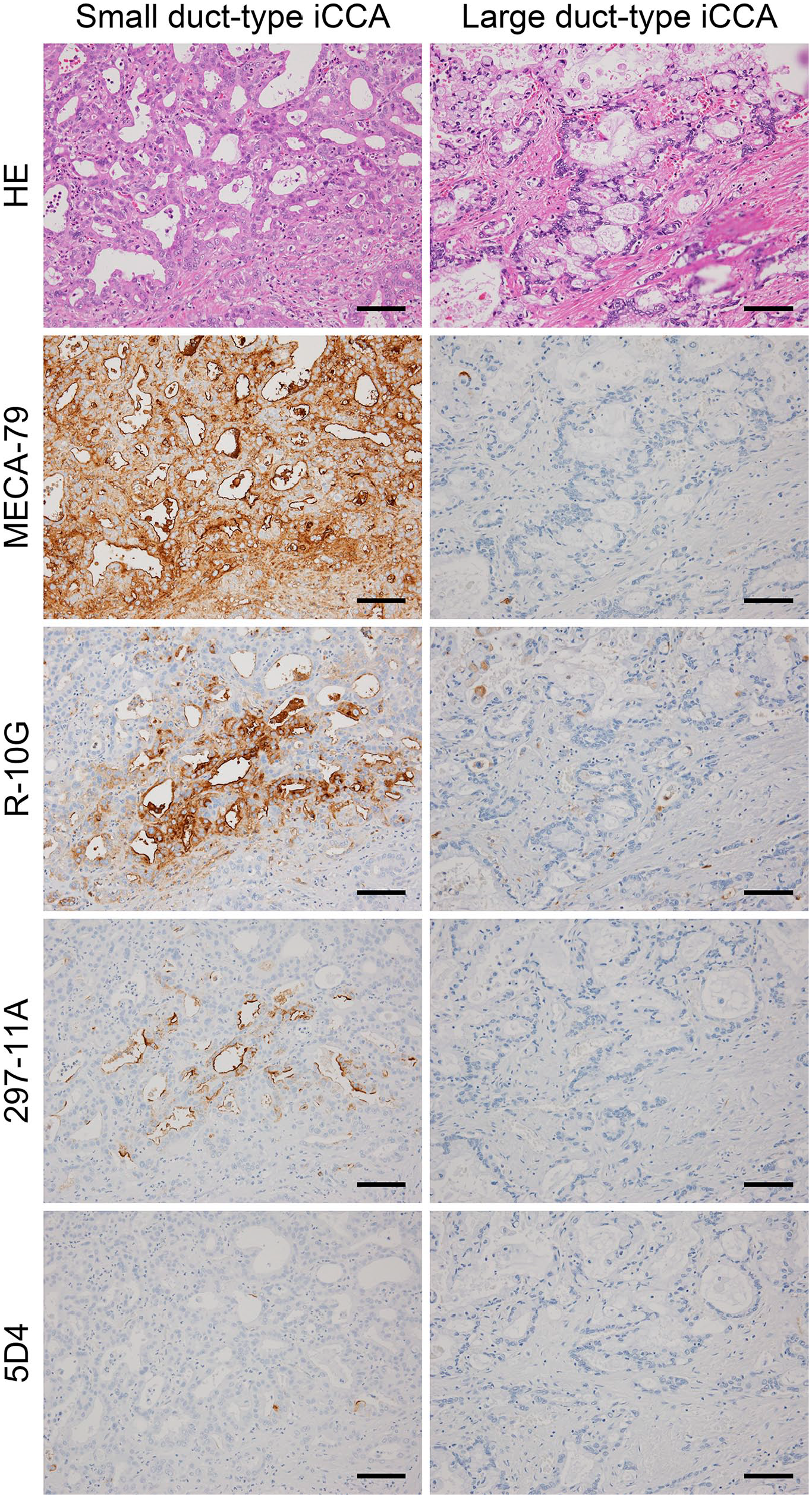
Expression of distinct sulfated glycans in small duct-type (left column) and large duct-type (right column) iCCA. Serial tissue sections were stained with hematoxylin and eosin (H&E) and immunostained for MECA-79, R-10G, 297-11A, and 5D4. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 100 µm. Similar immunohistochemical findings were obtained in four of five cases of small duct-type iCCA, but staining seen in large duct-type iCCA varied from sample to sample (see Table 1). Thus representative photomicrographs for each disease are shown. Abbreviation: iCCA, intrahepatic cholangiocarcinoma.
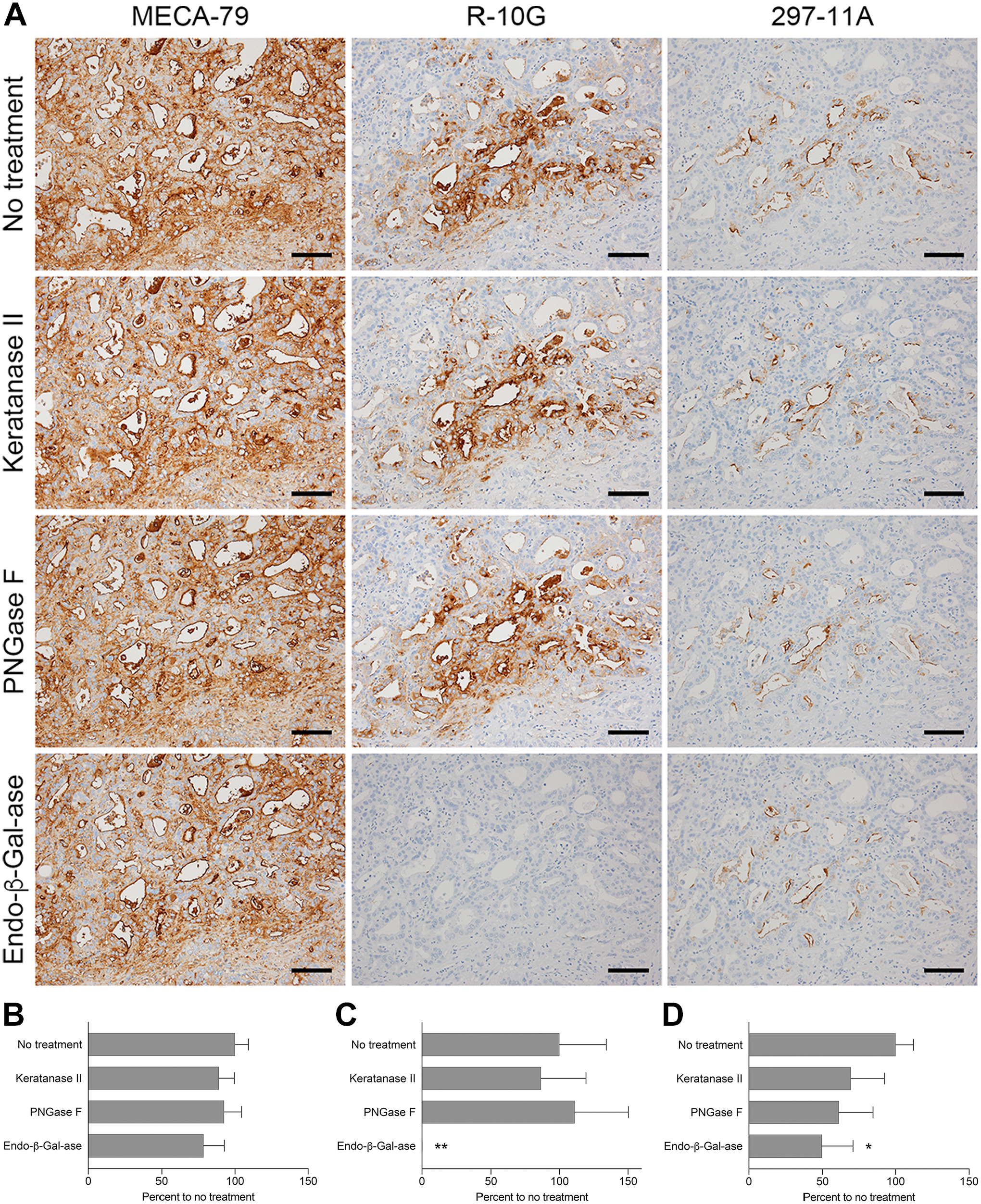
Immunohistochemical profile of sulfated glycans expressed in small duct-type iCCA with or without glycosidase pretreatments. (A) Serial tissue sections were treated with keratanase II, peptide-N-glycosidase F (PNGase F), or endo-β-galactosidase (endo-β-Gal-ase) before immunostaining for MECA-79, R-10G, and 297-11A. Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 100 µm. Photomicrographs in the top row are the same as their counterparts shown in Fig. 8. (B–D) Quantification of signal intensity of MECA-79 (B), R-10G (C), and 297-11A (D) with or without the glycosidase pretreatments shown in panel (A). Data are presented as means + SD of four segments of each image. The X-axis represents the percentage relative to no treatment. Abbreviation: iCCA, intrahepatic cholangiocarcinoma. *p<0.05, **p<0.01.
Discussion
In this study, we demonstrate that distinct sulfated glycans positive for MECA-79, R-10G, and 297-11A, but not for 5D4 antibodies, are expressed in normal human ductules in an apical membrane-predominant fashion. We also observed apical membrane immunolabeling with all three antibodies in ductular reactions seen in the reactive process of liver injury and also in cholangiolocarcinoma, which is now defined as a specialized histological variant of small duct-type iCCA in the latest WHO classification. 5 These findings suggest that apical membrane expression of distinct sulfated glycans is a characteristic feature of ductules and their reactive and neoplastic counterparts.
Here, we found that the canal of Hering, in addition to other regions of intrahepatic bile ducts, was positively stained with MECA-79, R-10G, and 297-11A. The canal of Hering, which comprises a slit-like space lined by cholangiocytes on one side and hepatocytes on the other, 2 not only functionally links the hepatocyte canalicular system with intrahepatic bile ducts to facilitate bile drainage but is also recognized as a progenitor cell niche for hepatocytes and cholangiocytes and is a source for ductular reactions seen in virtually all hepatobiliary diseases. 23 Indeed, in this study, chronic viral hepatitis-associated ductular reactions were also positively immunostained by all three anti-sulfated glycan antibodies, and their cytoplasmic staining was more intense than in normal cells. Such immunolocalization patterns are comparable with those seen in staining for MUC6 core proteins, in which signals are detected in both the apical membrane and cytoplasm of epithelial cells that constitute ductular reactions, particularly those associated with chronic viral hepatitis. 6 Moreover, double-immunofluorescence for MECA-79 and MUC6 revealed colocalization of both antigens in cells comprising ductular reactions. Overall, these observations raise the possibility that, in ductular reactions, MUC6 may function as a scaffold protein for distinct sulfated glycans.
We also observed that bile canaliculi of some hepatocytes adjacent to the canal of Hering express sulfated glycans positive for MECA-79, R-10G, and/or 297-11A. It would be of interest to know what proteins serve as scaffold proteins for these sulfated glycans. It is reported that CD10 is preferentially expressed in hepatocyte bile canaliculi, 24 and similar expression patterns have been reported for CD13 and biliary glycoprotein 1 (BGP1), the latter known as carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1). 25 Although these molecules are not canonical MUCs, they may function as scaffold proteins for sulfated glycans analyzed here, as they harbor multiple Ser and Thr residues, which serve as potential O-glycosylation sites.
In normal intrahepatic bile ducts, we observed that some R-10G signals were resistant to endo-β-galactosidase digestion, regardless of the duct caliber. As the R-10G epitope, Galβ1→4(SO4→6)GlcNAcβ1→3Galβ1→4(SO4→6)GlcNAc, contains the minimum structural requirements for endo-β-galactosidase, namely, GlcNAcβ1→3Galβ1→4GlcNAc (indifferent to GlcNAc-6-O-sulfation), 12 endo-β-galactosidase should degrade the R-10G epitope. It is possible that, in normal intrahepatic bile ducts, additional modifications such as Gal-6-O-sulfation and/or fucosylation on the carbohydrate side chains capped with R-10G-positive GlcNAc-6-O-sulfated oligo-N-acetyllactosamine may sterically hinder access of endo-β-galactosidase to glycans or interfere with enzyme activity. 26 On the contrary, in pathological settings such as cholangiolocarcinoma and ductular reactions, these modifications may be negligible, resulting in almost complete elimination of R-10G immunolabeling after endo-β-galactosidase digestion.
We also observed that in pathological conditions, 297-11A reactivity against sulfated glycans substantially decreased after PNGase F digestion, indicating that most 297-11A glycoepitopes exist on N-glycans in pathological settings. It has been shown that epithelial cells involved in cholangiolocarcinoma and ductular reactions express neural cell adhesion molecule (NCAM), 27 a hepatobiliary progenitor cell marker modified exclusively by N-glycosidically linked carbohydrate side chains. 28 Thus, N-glycans on NCAM proteins, rather than O-glycans on MUCs such as MUC1 and MUC6, may be preferentially modified with the 297-11A glycoepitope in pathological settings.
Here, we observed what may be viewed as a paradoxical increase in MECA-79 signal intensity in cholangiolocarcinoma after digestion with keratanase II or endo-β-galactosidase and a similar increase in R-10G signal intensity after PNGase F digestion in this type of tumor. It is plausible that in untreated tissue sections, epitopes recognized by these antibodies are blocked sterically by glycan structures in the same molecule, 26 and that digestion with these three glycosidases removes those interfering structures, making epitopes more accessible to antibodies.
We also observed comparable immunohistochemical profiles between small duct-type iCCA and cholangiolocarcinoma. This finding is expected as cholangiolocarcinoma is a special subtype of small duct-type iCCA, and these two tumors reportedly originate from peripherally distributed small-sized bile ducts, including interlobular ducts, ductules, and the canal of Hering. 5 However, immunohistochemical profiles of large duct-type iCCA, which arise in large-sized intrahepatic bile ducts near the hepatic hilum (proximal to the right and left hepatic ducts), 5 differed from those seen in small duct-type iCCA. Despite the cytoplasmic expression of distinct sulfated glycans in non-neoplastic large-sized ducts and peribiliary glands, the expression of MECA-79-positive sulfated glycans was negative in four of five large duct-type iCCA cases. Furthermore, the expression of R-10G- and 297-11A-positive sulfated glycans varied from sample to sample, but apical membrane expression of these sulfated glycans was negligible. These facts prompted us to hypothesize that sulfotransferases involved in the biosynthesis of cholangiocyte-derived sulfated glycans, that is, GlcNAc6ST-23 and keratan sulfate Gal-6-O-sulfotransferase (KSGal6ST), 18 are likely downregulated in large duct-type iCCA, however, further studies are required to test this hypothesis.
In various cancers, aberrant O-glycosylation of MUCs is associated with malignancy.29,30 Chou et al. reported that in breast cancer cells, overexpression of core 1 β1,3-galactosyltransferase 1 (C1GalT1), which synthesizes the core 1 O-glycans that serve as precursors for many mucin-type O-glycans, increased shedding of the MUC1 extracellular domain and simultaneously increased association of the MUC1 cytoplasmic tail with β-catenin. 31 Moreover, those C1GalT1-overexpressing cancer cells exhibited increased cell growth and invasivity relative to mock-transduced cells. 31 As cyclin D1 is reportedly a target of Wnt/β-catenin pathway 32 and association of the MUC1 cytoplasmic tail with β-catenin regulates cyclin D1 expression, 33 results of Chou et al. suggest that aberrant O-glycosylation of MUC1 activates Wnt signaling pathway in the context of breast cancer. It is possible that distinct sulfated O-glycans expressed in cholangiolocarcinoma have a similar effect on MUC1, contributing to tumor malignancy.
The physiological role of sulfated glycans expressed on the luminal surface of ductules has not yet been elucidated. Bile acid molecules have a hydrophobic steroid backbone exhibiting one to three hydroxyl groups, 34 and before excretion from hepatocytes, they are conjugated with glycine or taurine, converting them into negatively charged bile acid molecules. 35 It is tempting to speculate that repulsive electrostatic interactions between negatively charged sulfate group moieties in cholangiocyte-derived sulfated glycans and negatively charged bile acid molecules may function to prevent clogging of ductules and the canal of Hering. Further studies are required to clarify the role of cholangiocyte-derived distinct sulfated glycans.
There are several limitations in this study. As the number of tissue samples evaluated in this study was small (n=5 for each disease), the conclusions drawn should be corroborated by a larger number of samples. In addition, the age of the patients, which was not adjusted between normal and pathological samples, may have influenced the results of this study. It was difficult to obtain a larger set of samples partly due to the fact that this study was a collaborative effort by a small number of research institutions. To analyze a larger number of samples in the future, a multi-institutional collaborative study is necessary.
Supplemental Material
sj-pdf-1-jhc-10.1369_00221554211035730 – Supplemental material for Apical Membrane Expression of Distinct Sulfated Glycans Is a Characteristic Feature of Ductules and Their Reactive and Neoplastic Counterparts
Supplemental material, sj-pdf-1-jhc-10.1369_00221554211035730 for Apical Membrane Expression of Distinct Sulfated Glycans Is a Characteristic Feature of Ductules and Their Reactive and Neoplastic Counterparts by Hitomi Hoshino, Tomoya O. Akama, Kenji Uchimura, Mana Fukushima, Akifumi Muramoto, Takeshi Uehara, Yasuni Nakanuma and Motohiro Kobayashi in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
The authors thank Mr. Hisataka Kato and Ms. Maiko Yamanaka for technical assistance, and Dr. Elise Lamar for critical reading of the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Participation was as follows: HH designed and performed the research (immunohistochemistry), analyzed the data, and wrote the manuscript; TOA designed and performed the research (purification of glycosidases) and analyzed the data; KU and YN analyzed the data and edited the manuscript; MF, AM, and TU performed the research (histopathological evaluation); and MK conceived of and designed the research, analyzed the data, and wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research (C) 17K08758 from the Japan Society for the Promotion of Science (to H.H.), a Grant for Life Cycle Medicine from the Faculty of Medical Sciences, University of Fukui (to H.H.), a Grant Award from the Hokuto Foundation for Bioscience (to H.H.), and in part by a Grant-in-Aid for Scientific Research (B) 21H02702 from the Japan Society for the Promotion of Science (to M.K.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
