Abstract
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition typically targeting the axillary and anogenital regions of the body. The massive inflammatory cell infiltrate produced in this cryptogenic condition has led investigators in the attempt to link particular inflammatory cell fractions and cytokines to disease development, and ultimately to disease treatment. This study qualitatively and quantitatively analyzes the white blood cell fractions of macrophages, B-lymphocytes, T-lymphocytes, plasma cells, and granulocytes in 104 HS lesions on formalin-fixed paraffin-embedded tissues using immunohistochemistry (IHC). Four dermis-associated epithelial categories were investigated from persons with HS: 15 unaffected HS skin (US), 19 distended but unruptured follicle epithelium (UF), 62 migrating stratified squamous epithelium (MSSE) from ruptured follicles, and 35 degraded migrating epithelial sheets (DMES). In addition, 27 control skin (CS) from persons without HS were evaluated. Analysis of cell counts indicated that non-migratory dermal epithelium (CS, US, and UF) stimulated very little inflammatory response. However, contrary to previous studies which indicated macrophages to be the chief inflammatory cell in HS, this study showed that plasma cells were the primary cell type present in early-stage HS lesions (MSSE), whereas granulocytes were the major cell population seen in late-stage HS lesions (DMES):
Introduction
Chronic inflammatory diseases are understood to be the result of a dysregulated immune response. Studies on chronic skin diseases such as psoriasis and atopic dermatitis have led to the discovery of individual signal pathways that are disrupted, thereby identifying specific targets and resulting in effective treatment options. 1 Hidradenitis suppurativa (HS) is a chronic inflammatory condition of apocrine-bearing skin characterized by follicular hyperplasia and occlusion, recurring cysts, and sinus tract formation. Many in-depth molecular studies have been performed to understand which inflammatory signals predominate in HS.1–6 However, these signals are produced by multiple types of cells and stimulate several other leukocytes. The extreme and complex inflammatory reaction seen in HS has thus far eluded identification of a primary pathogenic mechanism, and consequently, there is no universal effective treatment. 1 To understand these molecular signaling pathways more clearly, the leukocyte populations must be identified. Although some studies have described the leukocytes present in HS, most have not comparatively quantified them. Moreover, many studies investigating the inflammatory response have small sample sizes.7–10 This study evaluated more than 100 samples to assess more accurately the infiltrates seen in this condition that affects 1%–4% of the population. 11 Beyond immunological dysfunction, several studies have investigated the role keratinocytes play in the pathogenesis of HS. Keratinocytes singly, in small groups, and in sheets are identified within the perifollicular infiltrate long after cystic rupture. Keratinocytes are known to invoke an inflammatory response when they penetrate beyond the epidermis.2,4,12 Hotz et al. 12 demonstrated that keratinocytes in HS skin produce an increased amount of proinflammatory cytokines, contributing to the already exaggerated inflammatory response. It has also been suggested that keratinocytes in HS have an intrinsic defect, which may contribute to follicular occlusion. 12 In addition, clinically unaffected HS perilesional skin has epidermal and inflammatory alterations in excess of those found in persons without HS.2,4,13 These subclinical defects point to an underlying predisposition to HS and a possible link between architectural and immunological alterations. For a greater understanding of the immunological response in HS, this study qualitatively and quantitatively investigated the leukocyte populations found in unaffected HS skin (US) compared with dermal infiltrates associated with unruptured follicle epithelium (UF), early-stage ruptured follicles, and late-stage HS lesions.
Materials and Methods
A total of 104 pathologist-confirmed HS lesions were evaluated using hematoxylin and eosin (H&E) stain for the presence of occluded and distended but UF and for ruptured follicles (Fig. 1). Early- to mid-stage lesions contained intradermal sheets of migrating stratified squamous epithelium (MSSE) with intact basement membranes which have branched off from ruptured follicle epithelium. Late-stage HS lesions were identified by the presence of intradermal groups of keratinocytes without intact basement membrane and were termed degraded migrating epithelial sheets (DMES) by the investigators. Frequently, the DMES areas were contiguous with MSSE. US distant to active lesions in persons with HS were evaluated, as well as were normal control skin (CS) from persons without HS. The CS served as control tissues and were selected from routine breast reductions and from panniculus specimens removed during cosmetic abdominoplasty in patients without HS. The five tissue categories, CS, US, UF, MSSE, and DMES, were designated by the investigators to indicate control tissues and to indicate the degree of HS lesional progression based on morphological characteristics on H&E. All tissues were obtained from the University of Tennessee Health Science Center shared tissue resource, and all experiments were performed with internal review board approval (19-07040-NHSR).
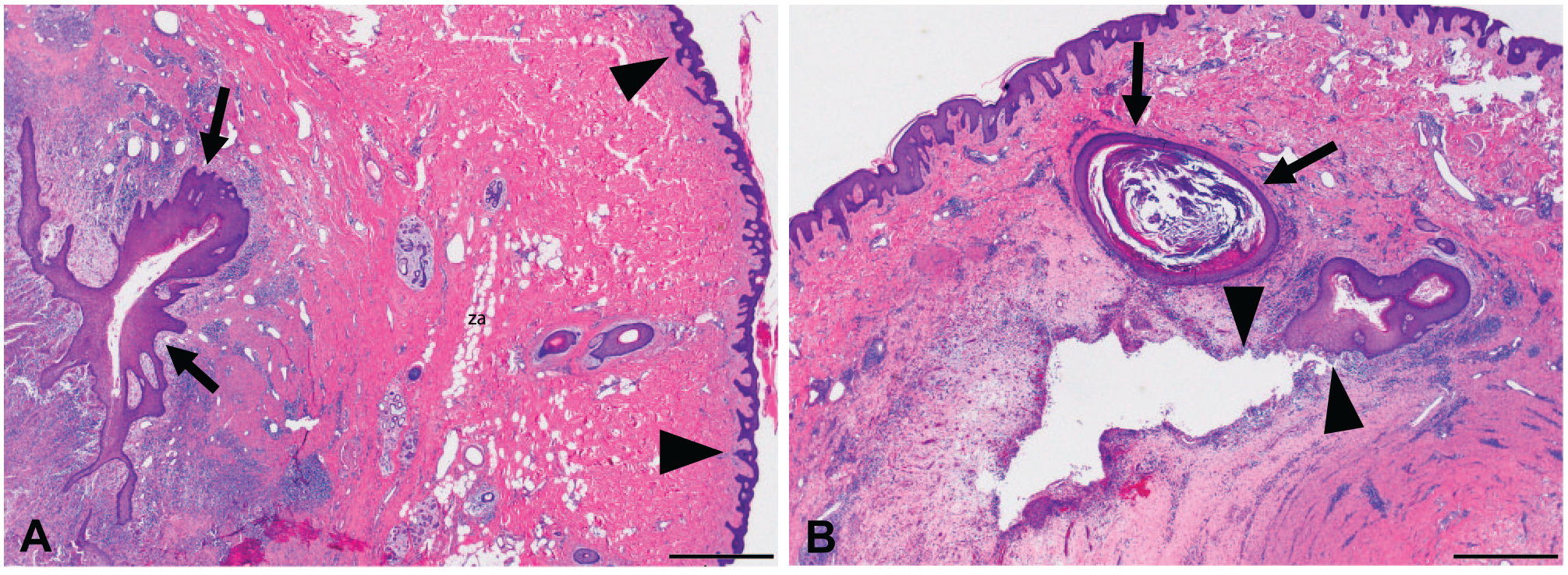
Hidradenitis suppurativa (HS). Low-power views of HS lesions demonstrating four epithelial HS categories investigated in this study stained with H&E. In (A), US is seen to the right (arrowheads), whereas MSSE with intact basement membranes (arrows) is seen surrounded by a sea of chronic inflammation at the left. In (B), UF (arrows) is seen with distended, keratin-filled lumen and intact, compressed epithelium. A DMES is shown at the arrowheads where an MSSE has begun to break down. While not appreciable at this low-power magnification, the edges of the cavity exhibit occasional small clusters of degenerated keratinocytes. Abbreviation: US, unaffected HS skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets; H&E, hematoxylin and eosin. Scale bar = 1 mm.
Tissue sections were cut at a thickness of 4 µm and were placed on plus-charged slides for staining. After drying in a 60C oven for 2–24 hr, sections were deparaffinized through three changes of xylene (3 min each), two changes of absolute ethanol (15 sec each), and one rinse of 95% ethanol for 15 sec before hydration in deionized water. All sections were stained with H&E to identify epithelial structures. The presence of these structures was verified with ready-to-use cytokeratin AE1/AE3 (PA0094; Leica Biosystems, Buffalo Grove, IL), antigen retrieval (AR) solution pH 6.0 paired with Bond Polymer Refine Detection Kit (DS9800; Leica). The detection kit was used by the investigators to perform manual immunohistochemistry (IHC) using staining trays coupled with the protocol described below. Antibodies used to identify specific subgroups of white blood cells included T-cell marker CD3 (103R-96; Cell Marque, Rocklin, CA) 1:400, AR pH 6.0; granulocyte marker CD15 (115M-15; Cell Marque) 1:50, AR pH 9.0; macrophage markers CD68 (PA5-32330; Invitrogen, Rockford, IL) 1:100 mixed with CD163 (163M-16; Cell Marque) 1:200, AR pH 9.0; plasma cell marker MUM1 (CRM352B; BioCare Medical, Pacheco, CA) 1:200, AR pH 6.0; and B-cell marker PAX-5 (CM2070; BioCare) 1:50, AR pH 6.0. After AR, sections were first treated with 1× tris-buffered saline (TBS) Automation Wash Buffer (TWB945M; BioCare), pH 7.7, before endogenous peroxidases were inactivated using dilute hydrogen peroxide for 5 min. After rinsing with TBS, the cell-specific primary antibody was applied to the sections for 20 min. Thereafter, the detection kit reagents were applied in order and included post primary (8 min), polymer (8 min), diaminobenzidine chromagen (2 min), and hematoxylin (5 min), with intervening TBS rinses. The slides were allowed to air dry and were briefly immersed in xylene before coverslipping. Antibody-appropriate positive and negative tissue and reagent controls were assayed at the same time and all stained appropriately.
Using the cytokeratin AE1/AE3 IHC stain, MSSE and DMES were identified, and an area of analysis was selected on 4× for each lesion. Selection of regions of interest (ROIs) using low-power magnification and cytokeratin assay on light microscopy was performed to minimize bias for inclusion/exclusion of one cell type over another. Selected ROIs on serial sections from each case were photographed on 40× magnification using CellSens imaging software, resulting in an area of analysis of 0.1 mm2. The cells staining positively in each image were enumerated independently by the investigators and the values averaged. Discrepant counts were resolved jointly by the investigators before statistical analysis.
Statistical analysis was performed using Microsoft Excel data analysis tools, one-way ANOVA, and t-Test: Two-Sample Assuming Equal Variances. Because this study consisted of five experimental groups, a Bonferroni correction was implemented and the standard p value of 0.05 was divided by 5 to give a statistically significant p value of 0.01 used for analysis.
Results
Sections containing epithelium and dermis associated with CS, US, UF, MSSE, and DMES were identified on H&E and were verified using a cytokeratin IHC stain (Figs. 1 and 2). One hundred four tissue blocks containing pathologist-verified HS lesions were analyzed for the presence of five experimental groups. In cases where two or more experimental groups were present in a single tissue section and were clearly independent of one another (>2 mm apart), more than one category was evaluated, bringing the total number of evaluated ROIs to 131. Fifteen lesions were categorized as US, 19 as UF, 62 as MSSE, and 35 as DMES. As seen in Fig. 1A, there are US and MSSE more than 2 mm apart. In cases such as these, the white blood cell populations associated with the epithelium in these categories could be independently interpreted with confidence. In contrast, Fig. 1B shows an example of UF and DMES with overlying skin. Because of the close proximity of these structures to each other, the skin and UF could not be considered entirely independent. However, a portion of the DMES was >2 mm distant from the other structures and was used for analysis in that lesion. When two or more structures within the same category (e.g., 2 MSSE) were seen in a single lesion, only one (typically the larger one or the one furthest from a resection margin) was selected to evaluate. In addition, 27 CS samples were analyzed from normal breast and abdominal skin in persons without HS.
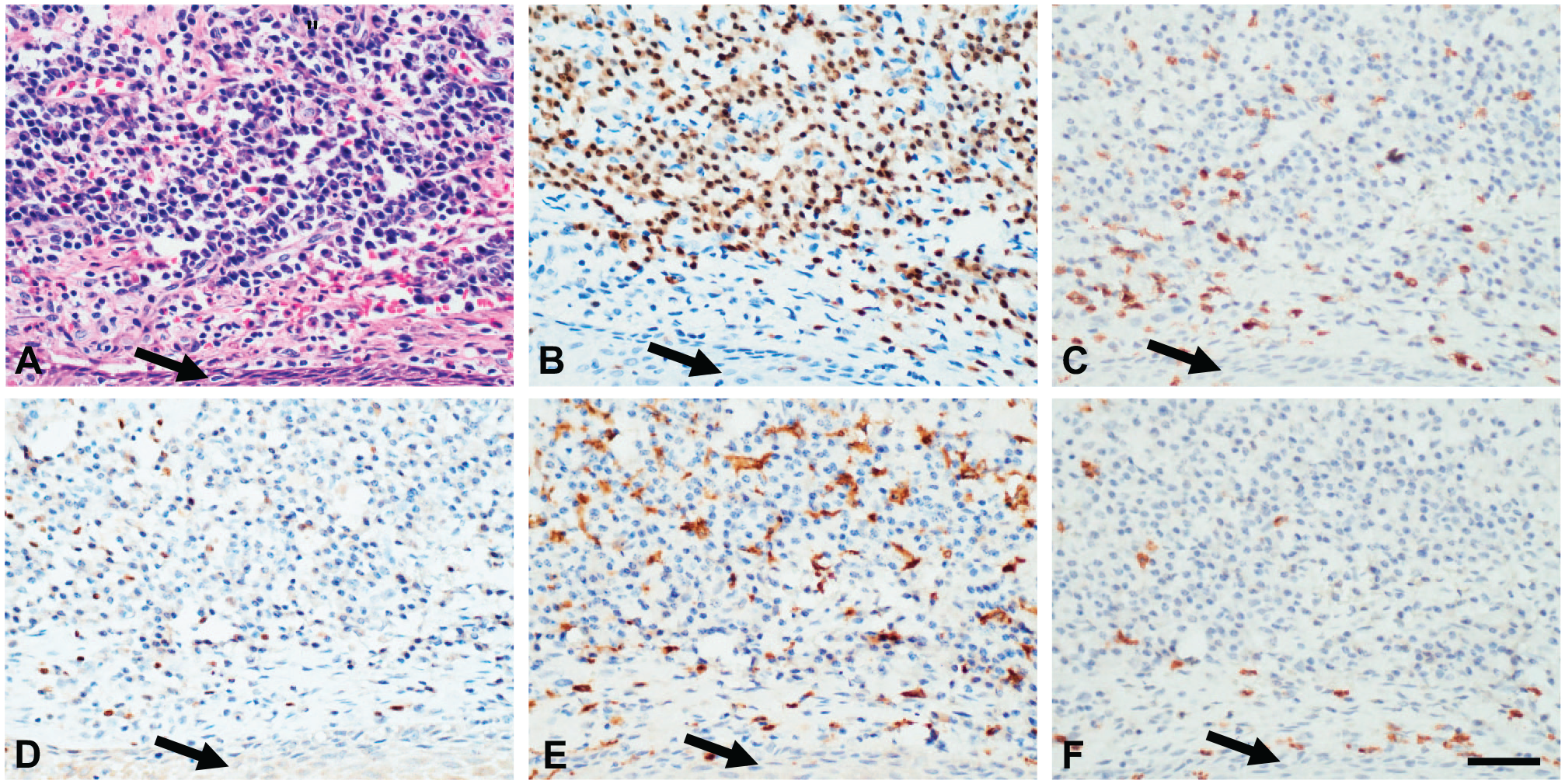
Hidradenitis suppurativa (HS). High-power images of IHC assays on serial sections in a lesion of HS associated with stratified squamous epithelium (arrows) from a ruptured follicle and the associated inflammatory response. (A) H&E stain, (B) MUM1 IHC assay for plasma cells, (C) CD3 IHC assay for T-lymphocytes, (D) PAX-5 IHC assay for B-cells, (E) combined CD68/CD163 IHC assay for macrophages, and (F) CD15 IHC assay for granulocytes. Abbreviation: H&E, hematoxylin and eosin. Scale bar = 50 µm.
On the H&E stain, DMES were defined by the absence of basement membrane in dermal/hypodermal stratified squamous epithelium. On cytokeratin stain, DMES were also identified by individual and clustered keratinocytes in a background of inflammatory cells and blush of background IHC positivity representing free keratin in the extracellular matrix (Fig. 2). Cell counts were performed on images acquired on 40×, representing a 0.1-mm2 area (Table 1; Figs. 3 and 4).
Average Number of White Blood Cell Fractions Found in CS, US, UF, MSSE, and DMES per 0.1 mm2.
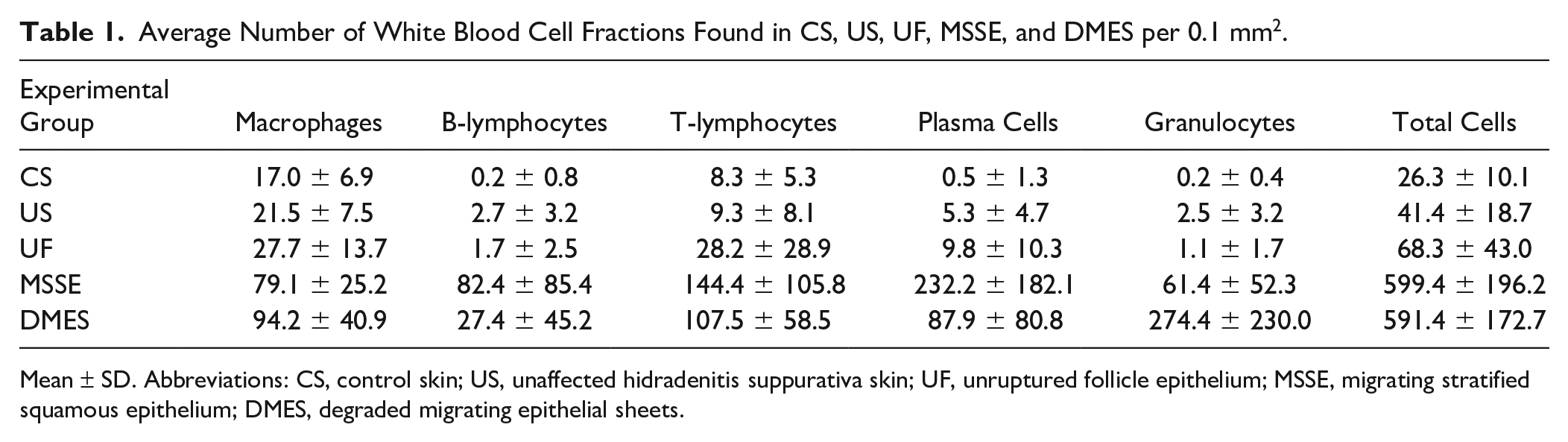
Mean ± SD. Abbreviations: CS, control skin; US, unaffected hidradenitis suppurativa skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets.
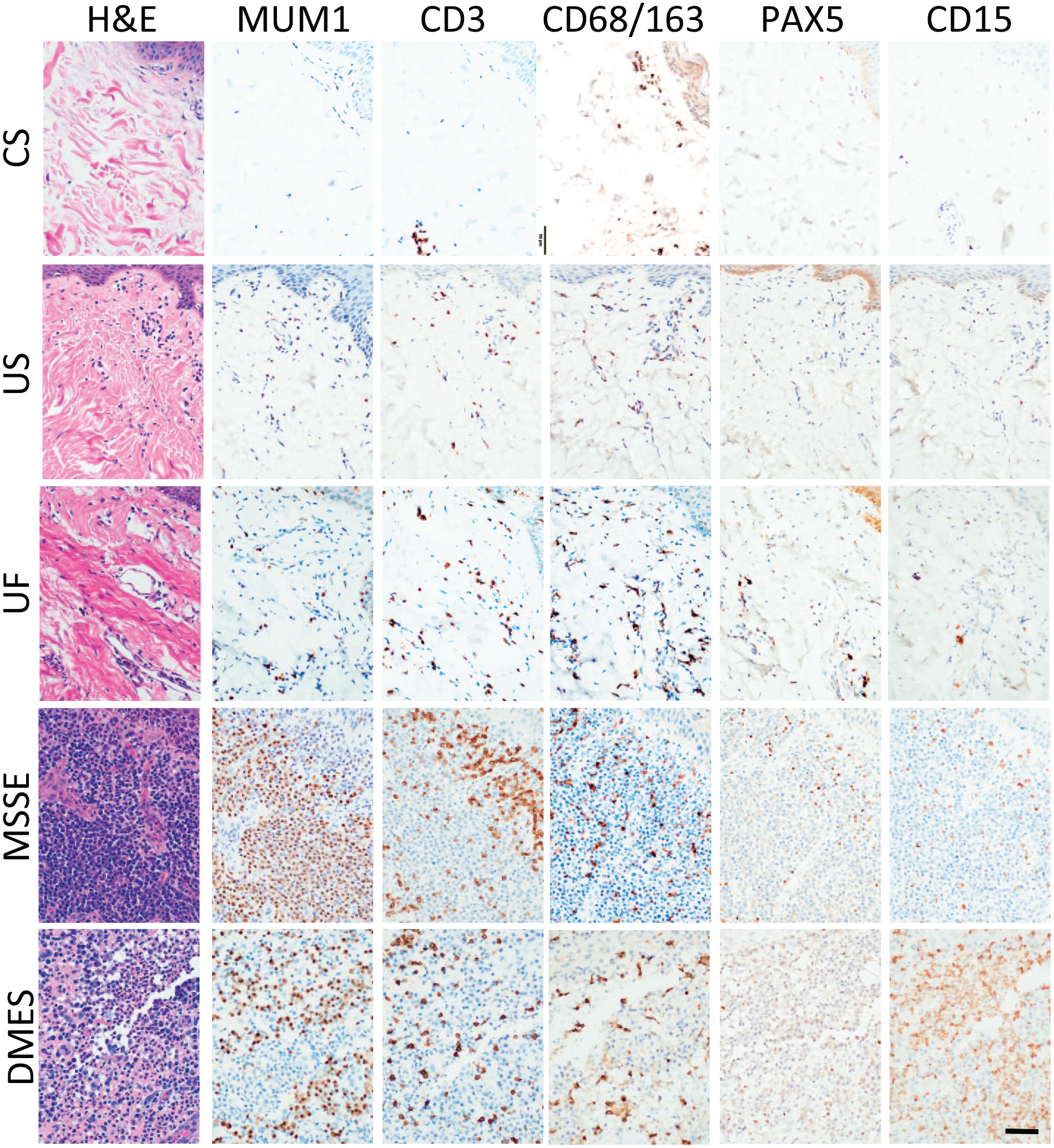
Hidradenitis suppurativa (HS). Serial sections of experimental dermal groups CS, US, UF, MSSE, and DMES stained with H&E and IHC assays MUM1, CD3, CD68/163, PAX-5, and CD15 represent the average case for each category. Intact epithelium is seen in the upper right-hand corner of each image except for DMES where a fissure instead represents where epithelium once was but only fragments of keratinocytes remain among the inflammatory cells. Abbreviation: CS, control skin; US, unaffected HS skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets; H&E, hematoxylin and eosin. Scale bar = 50 µm.
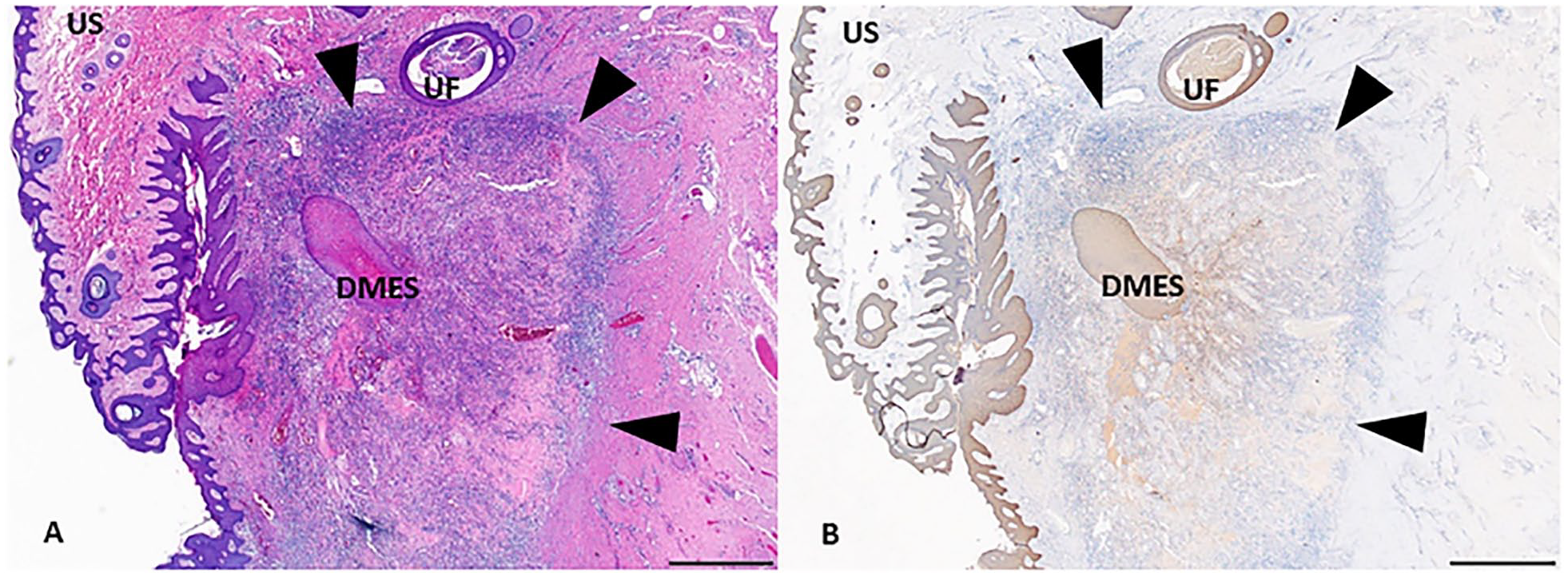
Hidradenitis suppurativa (HS). Serial sections from an HS lesion stained with H&E (A) and with cytokeratin (B). An example of DMES is demonstrated in the center of each image surrounded by inflammation with an abrupt transition to unaffected dermis (arrowheads). A blush of beige cytokeratin stain is seen radiating from the DMES toward the arrowheads and represents keratin debris spilled into the dermis from a ruptured follicle. Abbreviation: H&E, hematoxylin and eosin; DMES, degraded migrating epithelial sheets. Scale bar = 1 mm.
Cell Counts
Total Cells
Overall, the three categories associated with intact epithelium (CS, US, and UF) demonstrated expectedly fewer total infiltrating white blood cells than early-stage MSSE and late-stage DMES. There were no areas of heavy inflammation in US and UF when not in close proximity to MSSE or DMES. There was no statistical difference between MSSE and DMES for total inflammatory cells present. Similarly, there was no difference between US and UF for total cell counts. However, all other comparisons among the five sample types were significantly different (p<0.01).
Macrophages
Statistically significant differences in macrophage counts were not seen between DMES and MSSE. However, these two groups showed large increases and statistical significance in macrophage populations when compared with CS, US, and UF. Although CS and US were not significantly different, and US and UF were not significantly different, the UF macrophage count was significantly increased when compared with CS.
B-lymphocytes
For B-lymphocyte analysis, US, UF, and DMES were found to be statistically similar, However, B-lymphocytes found in CS were significantly fewer than in the other four epithelial types. Similarly, B-lymphocytes found in MSSE were significantly greater in number than those in the other four epithelial types.
T-lymphocytes
The T-lymphocyte counts mirrored the macrophage counts, if not in overall numbers, then in relative ratios. MSSE and DMES counts were similar to each other, but were each significantly more than CS, US, and UF. CS and UF were both similar to US, but were significantly different from each other.
Plasma cells
Plasma cells exhibited the most marked differences among sample groups. The only two groups in this analysis which were similar were US and UF. All other groups were significantly different in number when compared with each other. MSSE contained the greatest number of plasma cells, whereas CS contained the fewest.
Granulocytes
DMES lesions contained by far the greatest number of granulocytic cells, whereas MSSE lesions contained the second largest counts. DMES lesions differed significantly from MSSE, and both of these lesions differed significantly from CS, US, and UF. Among CS, US, and UF, average cell counts ranged from 0.2 cells/0.1 mm2 to 2.5 cells/0.1 mm2, but were significantly different between CS and US only, with US having the higher counts.
To compare the distribution of cell types with each other for each category, a set of scatter plots were graphed (Fig. 7). Macrophages were the only leukocyte population that was present in all samples and in all categories, and was consequently used as the x-axis value, to which all other cell types were compared. For all categories, as the populations of macrophages increased, so did the T-cells, albeit not to the same degree. The categories of CS, US, and UF exhibited similar scatter plots. B-cell and granulocyte counts were flat to minimally increased as macrophage counts increased. Plasma cells and T-cells showed a proportional relationship to macrophage counts. The MSSE and DMES categories displayed different relationships compared with the non-migratory epithelial categories. In MSSE, macrophages demonstrated a proportional relationship with both T-cells and granulocytes, but were inversely proportional to plasma cells and B-cells. In DMES, T-cells were directly proportional to those of macrophages, but granulocytes, plasma cells, and B-cells demonstrated an inversely proportional relationship.
Discussion
HS is a poorly understood dermatological condition that causes pilosebaceous units to become occluded with keratin. The resultant distention and rupture of epithelium at the base of the hair follicle stimulate an inflammatory response,10,14,15 which may lead to painful and disfiguring lesions within the dermis and epidermis. Microscopic evaluation of H&E and cytokeratin-stained HS specimens resulted in four classifications of pilosebaceous epithelium: US, UF, MSSE, and DMES. CS taken from persons without HS made up a fifth experimental category. As expected, US pilosebaceous units maintained normal architecture, and the epithelium was unaffected. In contrast, the keratin-occluded hair shafts observed in UF were surrounded by flattened epithelial layers. UF also exhibited compression of dermal collagen, fibroblasts, and accessory structures into concentric layers around the distended follicle (Fig. 2). Like UF, MSSE frequently exhibited broad keratin-filled spaces with stratified squamous epithelium. However, in this early lesional stage, the individual keratinocytes were not compressed. Instead, distinct spaces were found between cellular junctions, indicating the cells were spread widely with the epithelial sheets expanding and projecting into the dermis. Similar to findings from previous studies, the migrating epithelial basement membrane typically acquired a feathered edge appearance.16–18
Cell analysis yielded marked differences in average numbers of cells found in the five experimental groups (Table 1; Figs. 5 and 6). Several cell fractions had large standard deviations, particularly in MSSE and DMES, reflecting the dynamic nature of these lesions and the inflammatory response to them. B-cells, for example, were rarely seen “in moderation.” Typically, they were present in very low or very high numbers, suggesting that areas analyzed may have occasionally involved tertiary lymphoid follicle formation as described by van der Zee et al. 7 Indeed, the investigators noted two tissues that exhibited follicular or pseudofollicular arrangements of lymphocytes, but neither follicle was in close proximity to each epithelial ROI which was directly evaluated in this study. However, the authors cannot, with confidence, deny enumeration of B-cells originating from a follicle in which the periphery only was sampled. Plasma cells were noted to cluster, and while often seen in large aggregates in MSSE, they were found in smaller discrete groups in DMES. A limitation of this study is that a limited area of inflammation (0.1 mm2) adjacent to the epithelium was sampled in each lesion. Data analysis demonstrated that despite large standard deviations seen within some cell fractions, the differences in cell populations were often statistically significant (Table 2).
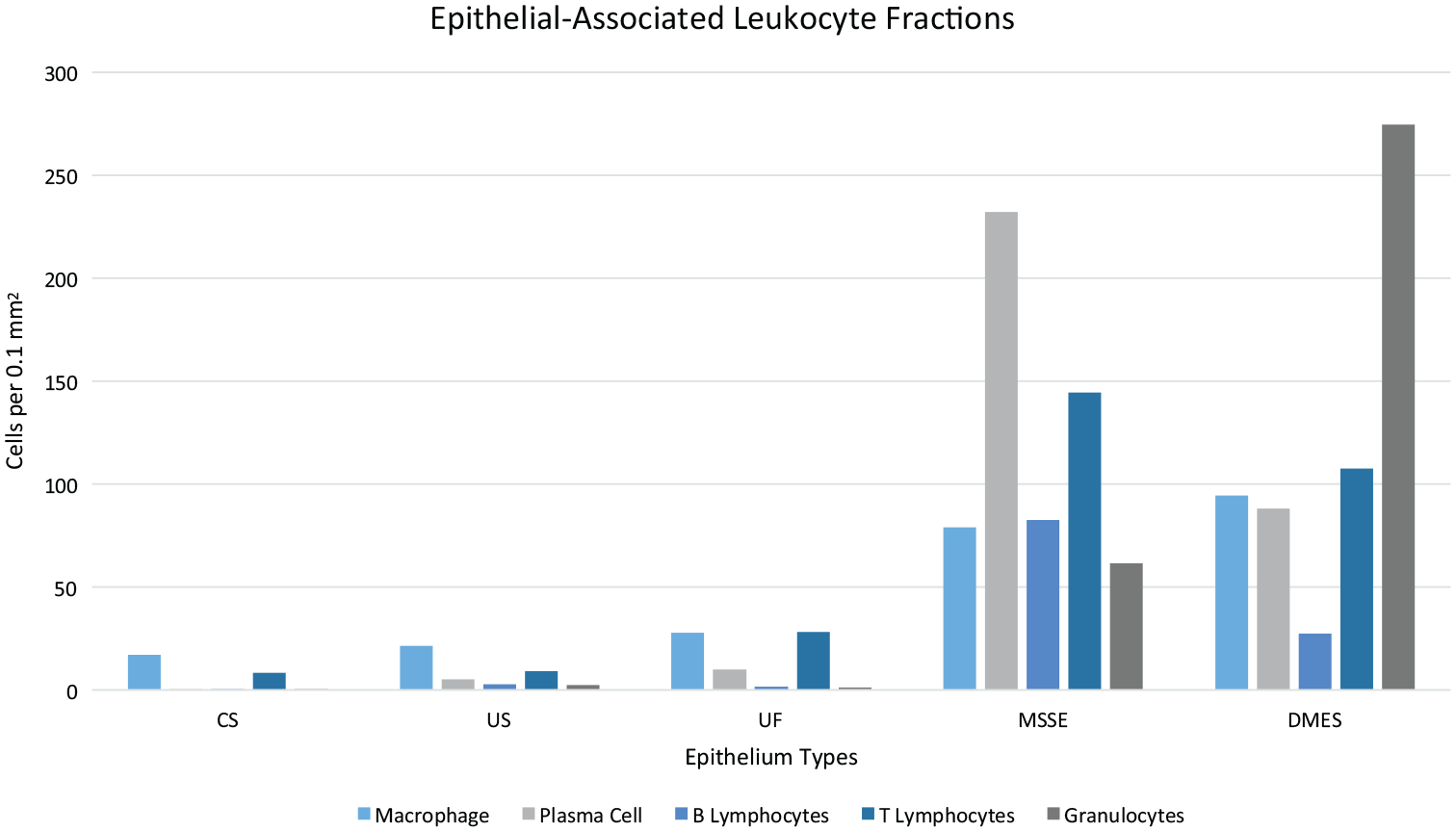
Hidradenitis suppurativa (HS). Cell counts seen in each epithelial-associate sample type. Overall, WBC counts were lower in CS, US, and UF compared with MSSE and DMES. The largest inflammatory cell fraction in MSSE was plasma cells, and the largest fraction in DMES was granulocytes. Abbreviation: CS, control skin; US, unaffected HS skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets.
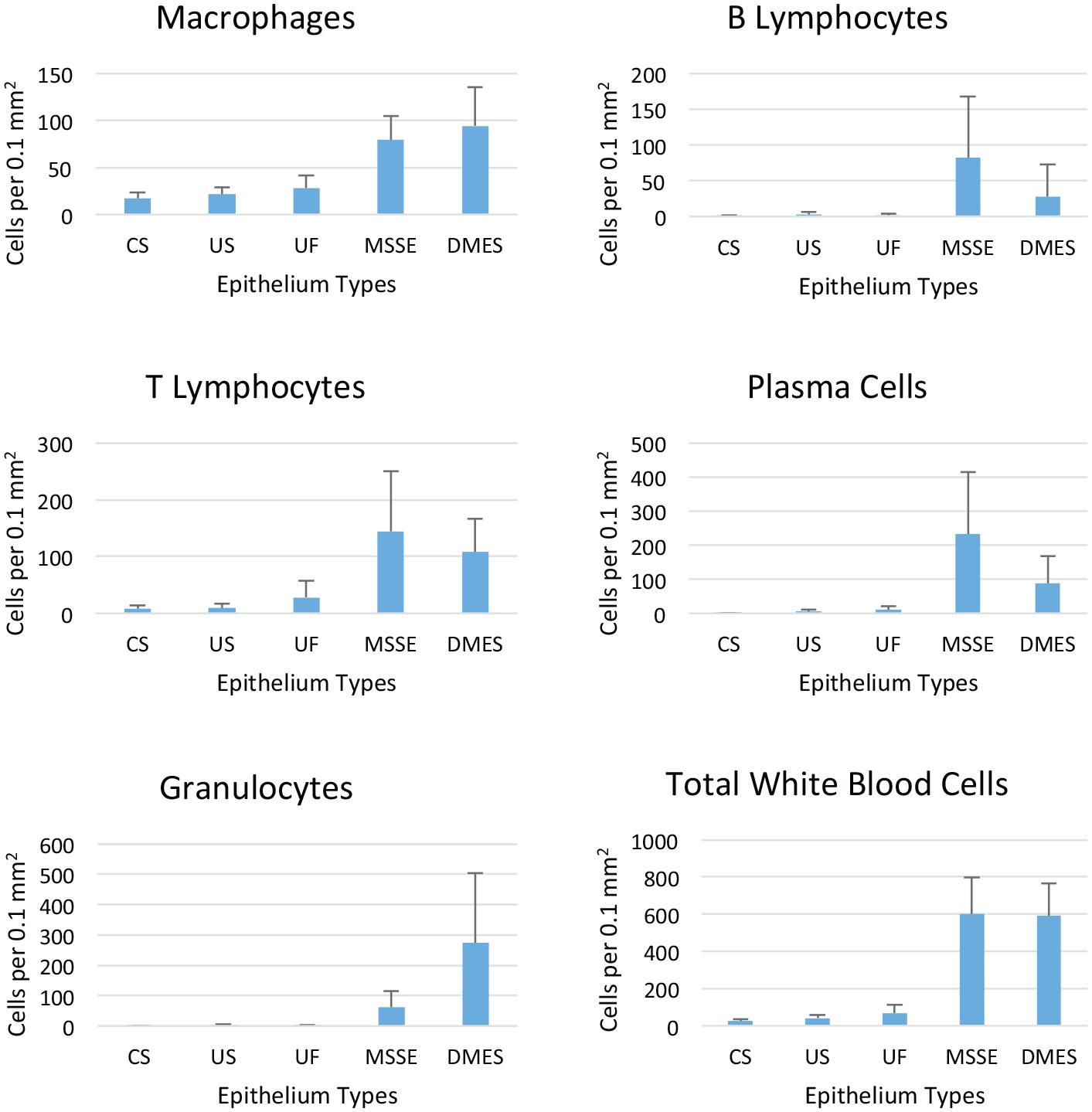
Hidradenitis suppurativa (HS). White blood cell fractions seen in dermis associated with epithelium in normal skin and HS lesions. Abbreviation: CS, control skin; US, unaffected HS skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets. Mean ± SD.
Statistical Significance (p<0.01) Between Epithelial Categories for Each Leukocyte Fraction.
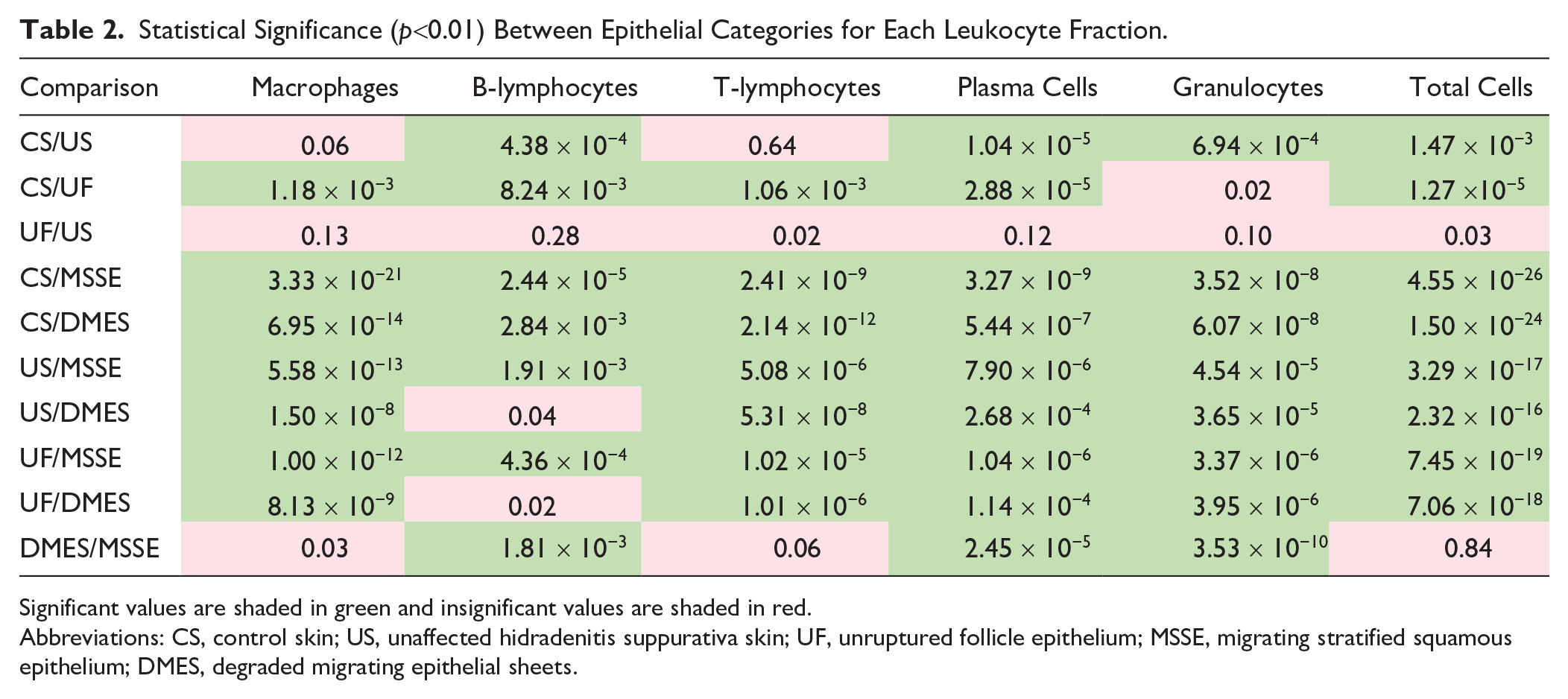
Significant values are shaded in green and insignificant values are shaded in red.
Abbreviations: CS, control skin; US, unaffected hidradenitis suppurativa skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets.
In this study, normal CS demonstrated the lowest number of inflammatory cells and exhibited a macrophage and T-cell signature, key components of the innate and adaptive immune systems in the skin. Macrophages are present to detect pathogenic invasion and subsequently activate T-lymphocytes that serve as a bridge between innate and adaptive immune responses. 19 This relationship can be seen in the consistent correlation between macrophages and T-lymphocytes in Figs. 5 and 7. The other two non-migratory epithelial categories, US and UF, evidenced higher numbers of inflammatory cells overall when compared with CS, suggesting that the inflammatory mechanism in HS may be inherently dysregulated. US differs from normal CS in its leukocyte profile of plasma cells, B-lymphocytes, and granulocytes. This proinflammatory environment suggests an immune dysfunction is critical to HS development. Plasma cells were the most markedly increased fraction in US and UF compared with CS, suggesting that HS patients may have spillover antibody production from nearby active lesions. Our results mirror increased plasma cells found in previous studies.1,7,20 B-lymphocytes are a hallmark of chronic skin conditions, such as eczema, as well as autoimmune disorders, 21 and their increase is not unexpected in HS. Overall, leukocyte populations between US and UF did not demonstrate any statistically significant differences. The homogeneous but minimal inflammatory response between US and UF indicates that the precipitating inflammatory event in HS is follicular rupture.
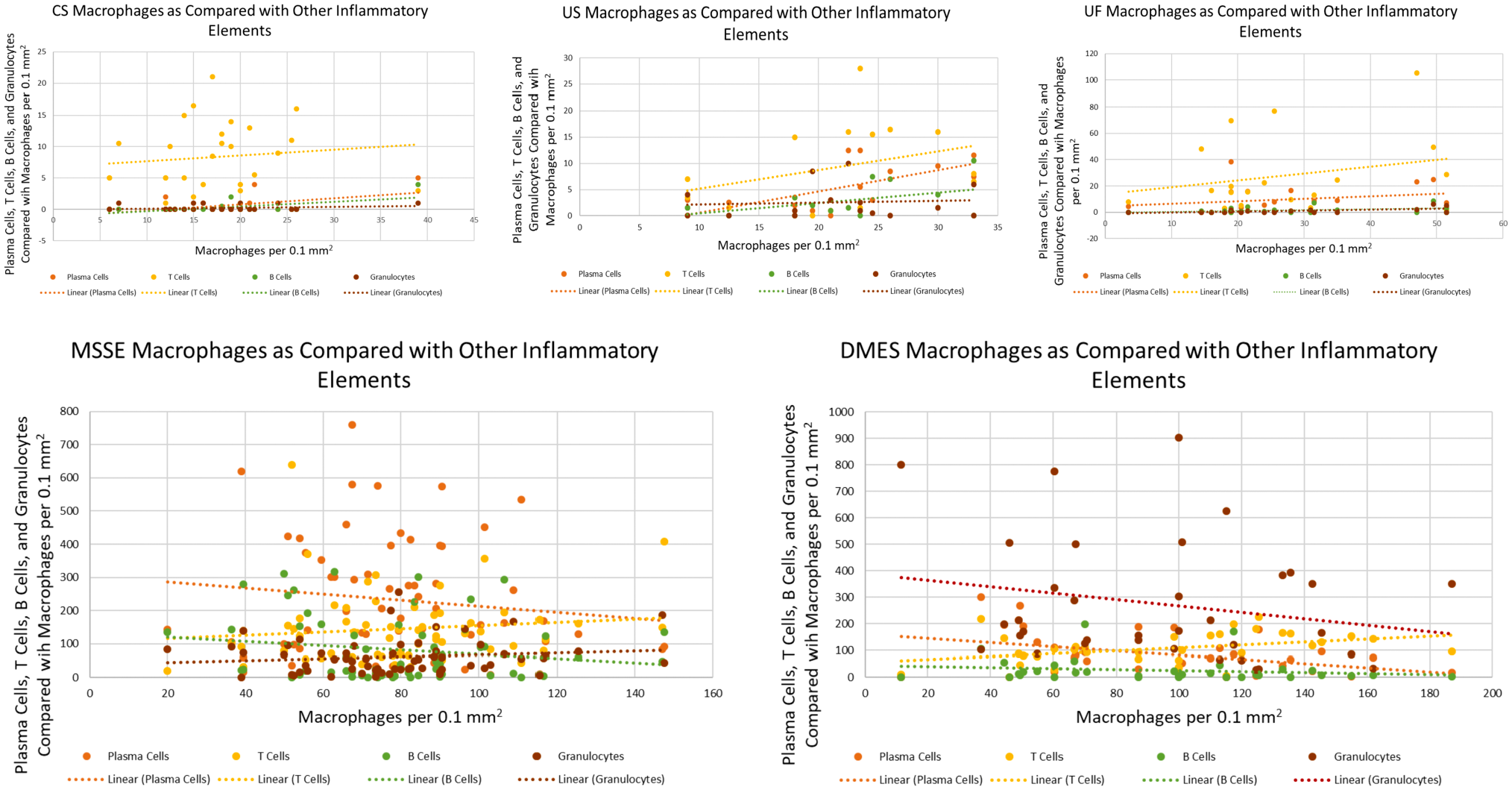
Hidradenitis suppurativa (HS). Scatter plots demonstrating the relationship of macrophages to other leukocyte fractions in each category analyzed. Abbreviation: CS, control skin; US, unaffected HS skin; UF, unruptured follicle epithelium; MSSE, migrating stratified squamous epithelium; DMES, degraded migrating epithelial sheets.
Among other cells and debris introduced to the inflamed dermis at rupture, keratinocytes likely induce the unique inflammatory alterations observed in early to advanced stages of HS.22–24 Upregulated interleukin (IL)-17 signaling inducing keratinocytes to produce a proinflammatory microenvironment via IL-1β has been observed in subclinical and lesional HS skin.3–5,12 HS keratinocytes have also been noted to produce increased cytokines IP10 and CCL5 which attract T-helper 1–type lymphocytes. Keratinocyte-produced antimicrobial peptides attract monocytes and further increase IL-1β,5,12 thus sustaining an inflammatory environment.
Plasma cells are the largest fraction of inflammatory cells seen in association with MSSE, the earliest stage of post-follicle rupture. The data suggest that the prevalence of B-cells and plasma cells in the dermis may be due to exposure of the dermal compartment to keratinocytes, which provokes an autoantibody response.20,22–24 The extreme immune response likely weakens the extracellular matrix surrounding the epithelium, via various matrix metalloproteinases, allowing for deeper epithelial migration.1,6,25,26 Hydration and expansion of corneal layers, 27 as well as the rapid inflammatory response that expands the dermis, increase the distance between MSSE epithelial edges, making reparative efforts unsuccessful. A lack of contact inhibition allows injured epithelium to proliferate 28 and is therefore likely responsible for the formation of sinus tracts from two unrelated sheets of colliding MSSE. The results of this study suggest that when a fully formed sinus cannot be achieved, the abundant inflammatory cells in the dermis attack and degrade the intact epithelium of the MSSE forming later-stage DMES.
Previous studies have implicated macrophages as major contributors to HS inflammation.5,8 In this study, macrophages were found to be the most abundant cell type in CS and US, and macrophages along with T-cells were the predominant white blood cell types in UF. However, in MSSE, macrophages comprised only 13% of the total inflammatory infiltrate, whereas plasma cells and T-lymphocytes made up 39% and 24% of the leukocyte population, respectively. In DMES, macrophages remained essentially unchanged at 16% of the total inflammatory cell population despite representing the highest number of macrophages among the experimental groups. Granulocytes predominated DMES, averaging 46% of the white cell count. Lima et al. 4 explain the prominence and role of granulocytes in later-stage HS as proinflammatory and promoting continued IL-17 signaling.
Together, these data suggest that macrophages control prelesional (US and UF) HS, whereas early and advanced HS lesions (MSSE and DMES) are associated with upregulated plasma cells and granulocytes, respectively. The current findings also suggest that this inflammatory response leads to unsuccessful reparative action in lesional epithelium, resulting in cyclic epithelial production, migration, and degradation until the hair follicle is completely destroyed or until a sinus tract is formed.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SLC designed the study. SRA and SLC performed microtomy, H&E staining, and IHC. SRA and SLC drafted and revised the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
