Abstract
Recently, we demonstrated that treatment with all-
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease caused by persistent hyperactive inflammation resulting in deleterious effects in the kidney, skin, lung, heart, joints, and brain.1–5 Women of childbearing age are most commonly affected, and there is currently no known cure for lupus.5,6 SLE is induced following the generation of high-affinity autoantibodies against nuclear components, phospholipids, and self-antigens forming deposits of immune complexes (ICs) in the peripheral tissues.7,8 In addition, the complement system amplifies inflammation by mediating the recruitment of inflammatory leukocytes.9,10 Treatment for SLE includes anti-inflammatory, antimalarial, glucocorticoids, and an antibody therapy that was developed to interfere with autoimmunity, which are used to curtain the body’s ability to attack its own tissue.11–13 However, toxic side effects and increased susceptibility to infections warrant further investigations into the mechanism(s) regulating SLE and identification of novel drug targets for therapeutic intervention.14,15
Recently, treatment with all-
Materials and Methods
Ethics Statement
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The protocol was approved by the Institutional Animal Care and Use Committee (IACUC) of Virginia Tech College of Veterinary Medicine (Animal Welfare Assurance Number: A3208-01). All animal experiments were conducted under IACUC protocol #12-062. For anesthesia and euthanasia, isoflurane and CO2 were used, respectively, according to the IACUC protocol.
Mice and Vitamin A Administration
MRL/Mp (MRL) and MRL/lpr mice were purchased from the Jackson Laboratory (Bar Harbor, ME), and bred and maintained in a specific pathogen-free facility following the requirements of IACUC at Virginia Polytechnic Institute and State University. tRA and all-
Histological Preparation and Immunohistochemistry
After immersion-fixation in 10% neutral buffered formalin, fixed tissues were paraffin-embedded, sectioned, and stained for hematoxylin and eosin (H&E) at the Histopathology Laboratory at Virginia–Maryland Regional College of Veterinary Medicine. Coronal brains sections were collected at levels of the following structures: olfactory bulb, head of caudate nucleus, rostral level of hippocampus, caudal level of hippocampus, mid-level of cerebellum with underlying medulla oblongata, and caudal level of cerebellum with underlying medulla oblongata. Sections were analyzed with a Nikon ECLIPSE Ci-L microscope for histology, and pictures were taken by using NIS-Elements D 3.2 64-bit software under 20× objective lens (Nikon Plan 20×/0.40, OFN22 WD1.2) at room temperature. For immunohistochemistry, citrate buffer (10 mM sodium citrate, 0.05% Tween 20, pH 6.0) was used for antigen retrieval. Slides were dewaxed and stained for astrocytes using anti-glial-fibrillary acidic protein (GFAP; eBioscience, San Diego, CA), monocyte/macrophage using anti-Iba-1 (Novus Biologicals, Littleton, CO), plasma cells using anti-CD138 (BioLegend, San Diego, CA), adhesion molecules using anti-E-selectin (Santa Cruz, Heidelberg, Germany) and anti-ICAM-1 (eBioscience), brain depositions using anti-complement C3 (Cedarlane; Burlington, NC) and anti-mouse IgG (Sigma), and neurodegeneration using Fluoro-Jade C (EMD Millipore, Billerica, MA). All primary and secondary antibodies were used at a dilution factor of 1:20. Slides were mounted with Prolong Gold containing 4′,6-diamidino-2-phenylindole (DAPI; Life Technologies, Carlsbad, CA). Representative images pertaining to neuroinflammation and degeneration were shown from the rostral level of hippocampus. Immunohistochemistry slides were read and pictured with BX51 upright Olympus microscope (Olympus America; Center Valley, PA), a 20× objective and Stereo Investigator software (MBF Bioscience; Williston, VT). All stained sections were submitted for evaluation by a board certified neuropathologist blinded to the experimental groups. These findings were confirmed by a second blinded observer. For Fluoro-Jade C (FJC) staining, slides were stained in Fluoro-Jade solution (working concentration of 0.0001% in 0.1% acetic acid) for 10 min. Each stained slide was subjected to non-biased stereology to quantify the number of FJC-positive cells in the contoured cortex of three serial stained tissue sections. Using the optical fractionator probe from Stereo Investigator (MBF Bioscience) 10.30.1 software package and an upright Olympus BX51TRF motorized microscope (Olympus America), a blinded investigator quantified the total number of FJC-positive cells. A contour was placed over the entire right hemisphere cortex at 4× magnification, and a grid of 200 × 200 µm was placed over this area with a counting frame of 75 × 75 µm. The number of FJC-positive cells was randomly counted using the optical fractionator at 63× magnification. Positive and negative controls for immunohistochemistry were used as appropriate. Detailed information of antibodies is listed in Table 1.
Detailed Information of Antibodies.
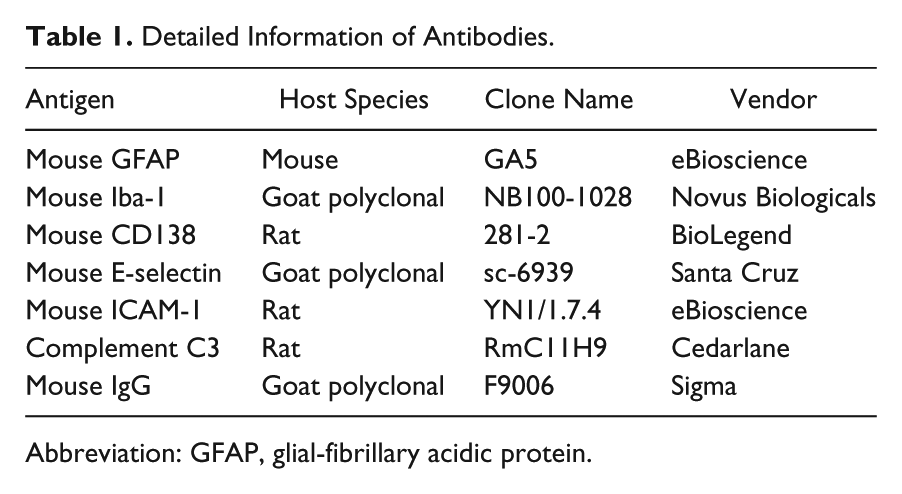
Abbreviation: GFAP, glial-fibrillary acidic protein.
Flow Cytometry
Blood was collected and peripheral blood mononuclear cells were obtained with the addition of RBC lysis buffer (eBioscience). For surface marker staining, cells were blocked with anti-mouse CD16/32 (eBioscience), stained with fluorochrome-conjugated antibodies, and analyzed with BD FACSAria II flow cytometer (BD Biosciences, San Jose, CA). For identification of plasma cells, based on our experience with the cells from lupus-prone mice, both CD138 and the combination of CD44 and Ly6c are good markers for plasma cells in lupus (unpublished data). CD19 and B220, on the contrary, are not expressed on plasma cells. Results obtained with anti-CD44 (BioLegend) and anti-Ly6c (BD Biosciences) are shown. A minimum of 100,000 events were acquired and counted on the flow cytometer. Flow cytometry data were analyzed with FlowJo (FlowJo, LLC, Ashland, OR).
ELISA
Sera were collected from 14-week-old mice and subjected to ELISA. For detection of anti-double-stranded DNA (dsDNA) IgG, the plate was coated with 0.1 mg/ml of calf DNA (Sigma) in 1× saline-sodium citrate (SSC) buffer at 4C overnight, followed by washing with PBS containing 0.05% Tween-20 (PBS-T). Wells were then blocked with PBS containing 1% BSA for 1 hr at room temperature and washed. Samples were added and incubated for 1 hr at room temperature. After additional washing, horseradish peroxidase (HRP)–conjugated goat anti-mouse IgG-Fc secondary antibody (Bethyl Laboratories, Montgomery, TX) was added and incubated for 1 hr at room temperature, following by more washes with PBS-T. Then, 3,3′,5,5′-tetramethylbenzidine (TMB) substrate (BioLegend) was added. After the reaction was stopped, the plate was read at 450 nm with SpectraMax plate reader (Molecular Devices, Sunnyvale, CA). Total IgG concentration was determined with mouse IgG ELISA kit according to the manufacturer’s instructions (Bethyl Laboratories).
Statistical Analysis
For the comparison of two groups, unpaired student’s
Results
Effects of tRA and VARA Treatment on Brain Weight
To evaluate the effect of tRA on lupus pathogenesis in the brain, we treated 6-week-old, female MRL/lpr mice, orally and daily, with vehicle, tRA, or VARA for 8 weeks, then euthanized and evaluated brain tissue at 14 weeks of age. At this time, whole brain tissue was isolated and weighed in grams (g). VARA is a combination of tRA and the vitamin A supplement retinyl palmitate, which is more clinically relevant than tRA alone. Compared with MRL control mice, MRL/lpr mice had significantly lower brain weight (Fig. 1A) but significantly increased brain-to-body weight ratio (Fig. 1B), which are consistent with a previous report. 40 Eight weeks of tRA treatment on MRL/lpr mice resulted in a significant increase in brain weight (Fig. 1A), whereas VARA treatment significantly increased the weight ratio (Fig. 1B). tRA- and VARA-treated MRL control mice were unaffected (data not shown). The significant increase in brain weight with tRA treatment of MRL/lpr mice compared with vehicle treatment was correlated with a significant increase in neuroinflammation following histological examination (Fig. 1C). The inflammatory infiltrates were located primarily in the third ventricle and lateral ventricle (Fig. 1D), suggesting peripheral inflammation. Our previous findings showed no difference in total BW among MRL/lpr groups, indicating that the administered doses of tRA and VARA were not toxic to the mice.19,36 These data suggest that the increase in brain weight following tRA treatment may be the result of increased neuroinflammation.
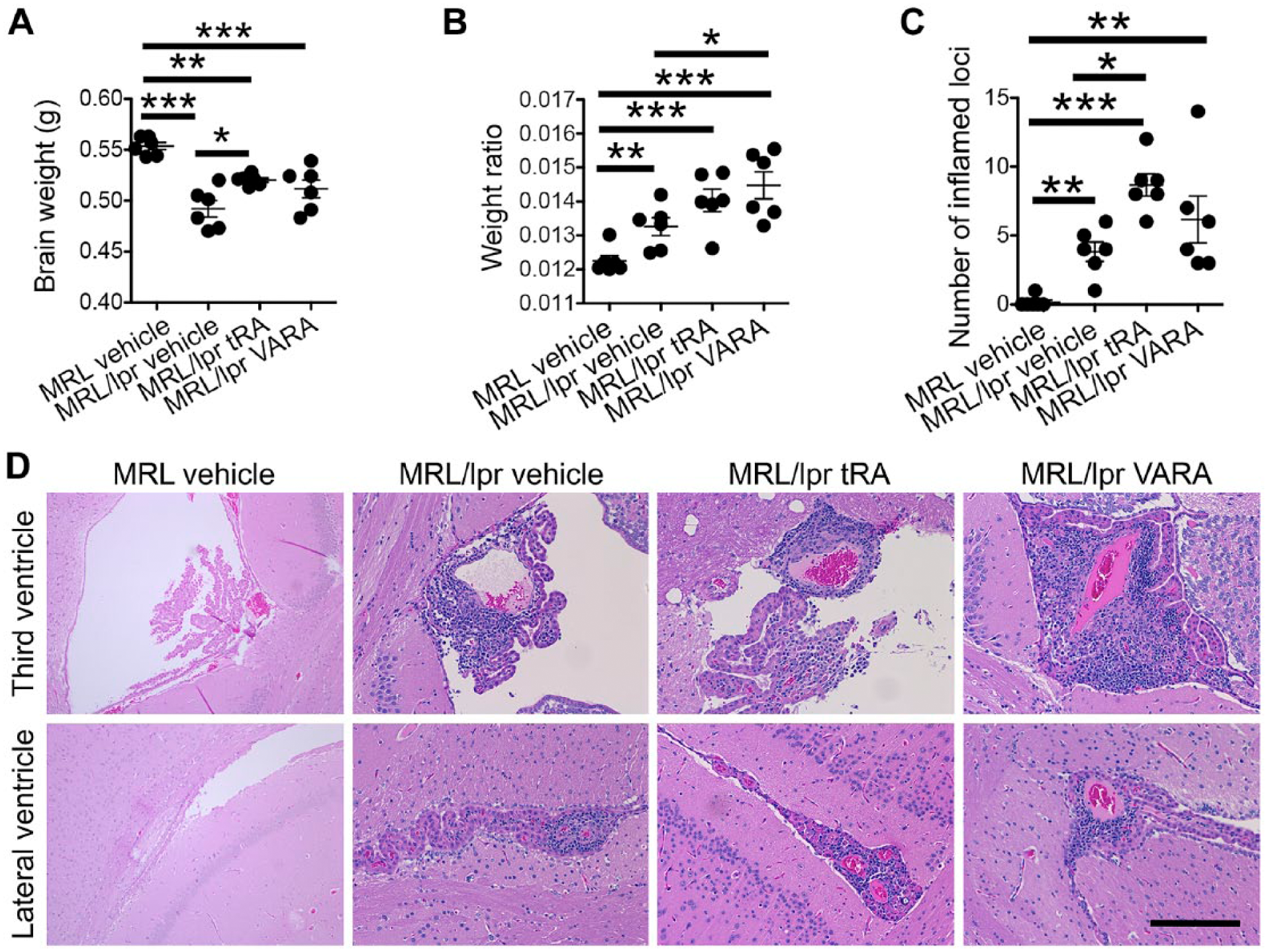
Brain weight in 14-week-old MRL/lpr mice treated with tRA or VARA. MRL mice were given, orally and daily, tRA (6 mg/kg BW) or VARA (11.2 mg retinyl palmitate/kg BW, equivalent to 6 mg retinol/kg BW, mixed with 0.6 mg tRA/kg BW that was 10% of the amount of retinol) from 6 to 14 weeks of age when brain and body weights were collected. (A) Brain weight in grams (g) graphically represented for MRL and MRL/lpr vehicle (canola oil)-treated and MRL/lpr tRA- or VARA-treated mice. (B) Brain weight as a ratio of total BW in the four groups. (C) Number of inflamed loci with leukocyte infiltration on each H&E slide that contained coronal brains sections of the following structures: olfactory bulb, head of caudate nucleus, rostral level of hippocampus, caudal level of hippocampus, mid-level of cerebellum with underlying medulla oblongata, and caudal level of cerebellum with underlying medulla oblongata. *
Prominent Astrogliosis, Ventriculitis, and Meningitis Following tRA and VARA Administration
Reactive astrogliosis is a response of astrocytes in the brain that involves morphological and functional changes, including hypertrophy, cellular division, and upregulation of intermediate filaments, such as GFAP, as well as release of inflammatory mediators, which occurs following the destruction of neurons in response to central nervous system trauma, infection, ischemia, autoimmune response, and neurodegeneration. 41 To begin addressing the effects of tRA and VARA on astrogliosis, we subjected coronal brain tissue sections, at the rostral level of the hippocampus and dorsal third ventricle (DV3), to antibodies against GFAP. The extent of GFAP labeling was dramatically increased in tRA- and VARA-treated MRL/lpr mice around the DV3 and meninges between the dorsal aspect of the cerebral hemispheres and dorsal to the corpus callosum (Fig. 2D–D1 and 2F–F1, respectively) compared with vehicle treatment (Fig. 2B–B1). Focal astrogliosis was evident in these regions following treatment. Astrogliosis was not observed in the cortex or any other gray matter or white matter structures. Prominent cellular accumulation is evident in tRA-treated mice in the meninges between the dorsal aspects of the cerebral hemispheres and dorsal to the corpus callosum at the level of the habenular nuclei (Fig. 2C). The choroid plexus of the fourth ventricle contains many plasma cells around interstitial capillaries and extending into the epithelial layer and adjacent ventricular lumen (data not shown). Additional immunofluorescence labeling of these cells for markers against plasma cells (CD138), monocyte/macrophages (Iba-1), and cell adhesion molecules (ICAM and E-selectin) was performed. The meningeal infiltrate contains many plasma cells, including those containing Russell bodies, smaller numbers of other lymphocytes, and occasional macrophages. Other large meningeal inflammatory foci are seen in the medulla oblongata, and contain fewer plasma cells and more lymphocytes and macrophages by H&E. A region of adjacent gray matter surrounding the DV3 and meninges is finely vacuolated and contains increased reactive astrocytes, as demonstrated in Fig. 2. The cellular infiltrates localized in the DV3 and meninges (Fig. 3A) contained CD138-positive (Fig. 3B) and Iba-1-positive cells (Fig. 3C), indicating plasma cell and peripheral-derived monocyte/macrophage identity, respectively. High expression of ICAM-1 was also observed, indicating their inflammatory nature (Fig. 3D and F). Expression of E-selectin (Fig. 3E and F) was minimal. These data suggest that tRA and VARA administration results in chronic ventriculitis, meningitis, and focal astrogliosis in the MRL/lpr mouse model.
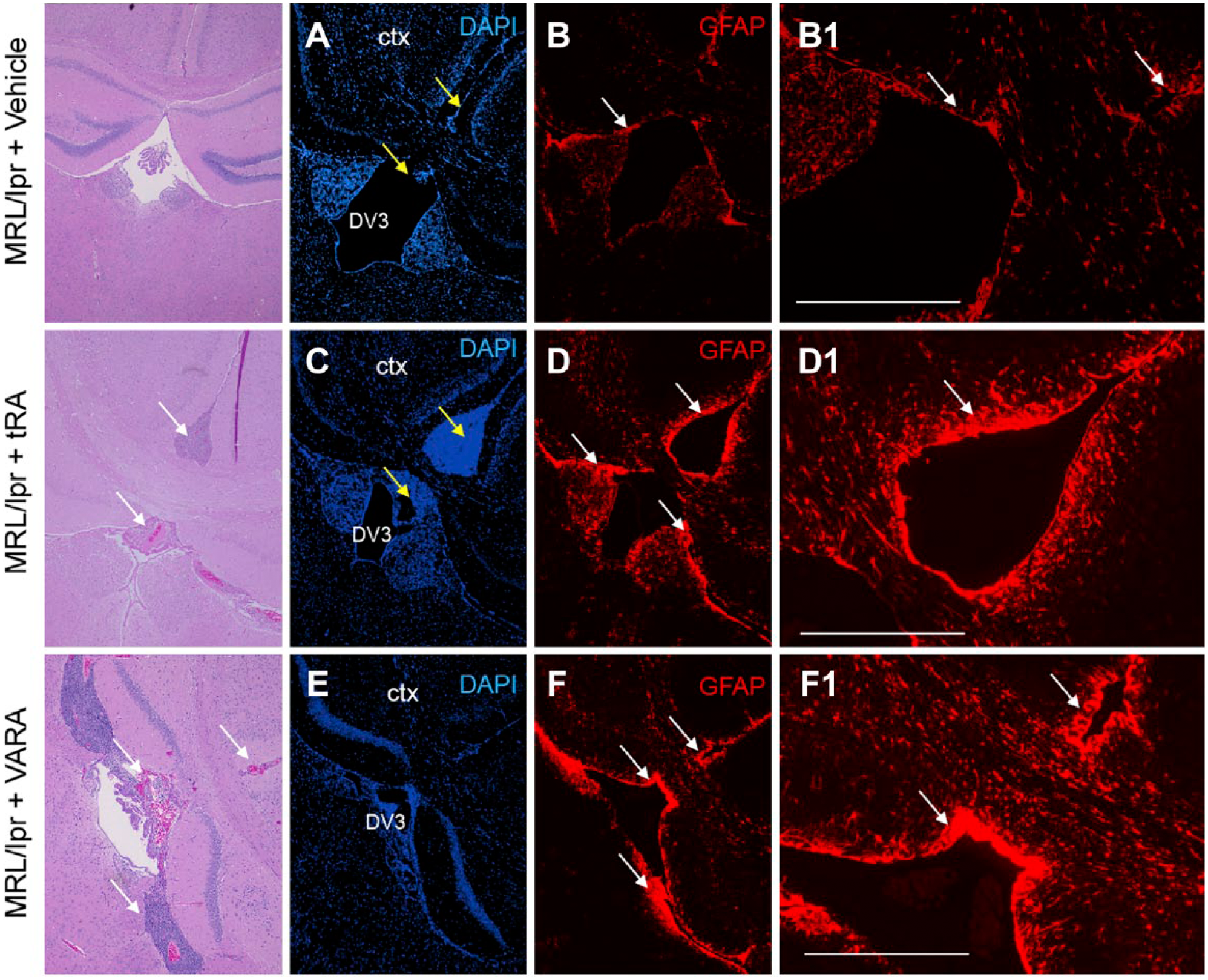
Astrogliosis in the brain of MRL/lpr mice treated with tRA or VARA. Immunohistochemistry of brain sections from MRL/lpr mice treated with vehicle (A and B), tRA (C and D), or VARA (E and F), using antibodies against GFAP. GFAP staining was substantially increased around the DV3 and meningeal lining between the two cortical hemispheres (white arrows) in tRA- and VARA-treated compared with vehicle-treated MRL/lpr mice. Scale bar = 400 µm. Abbreviations: tRA, all-
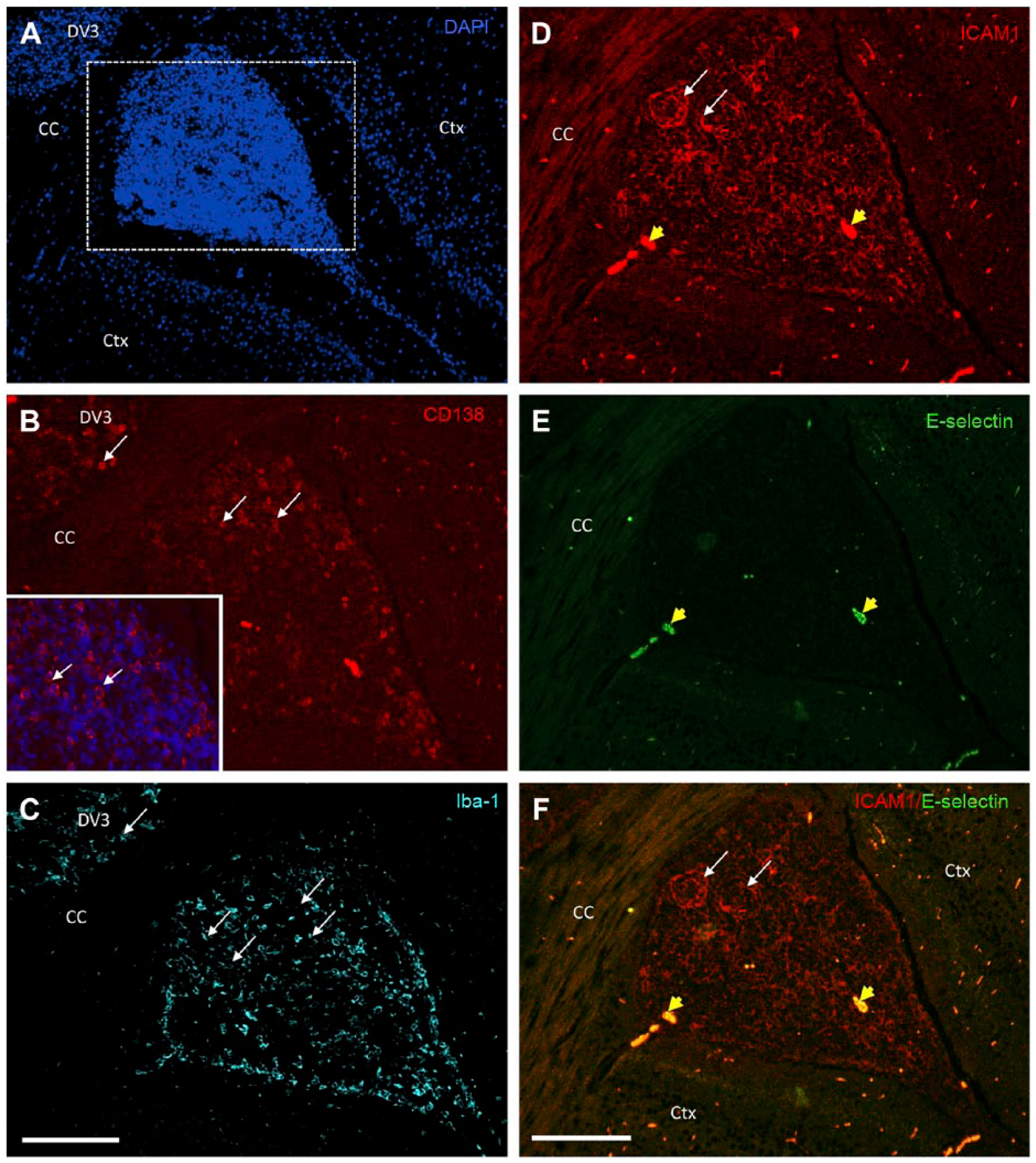
Identification of cellular infiltrates in the meninges of MRL/lpr mice treated with tRA. Brain sections of MRL/lpr tRA-treated mice were used to identify the cellular infiltrate in the meninges between the dorsal aspect of the cerebral hemispheres and dorsal to the CC at the level of the habenular nuclei (A). Antibodies against plasma cell-specific protein CD138 (red) showed accumulation of plasma cells in the DV3 and meninges (white arrows) (B) as well as monocyte/macrophages as seen by anti-Iba-1 staining (C). Sections were further characterized for inflammatory adhesion molecules ICAM-1 and E-selectin (D–F). High expression of ICAM (white arrows; D) was seen indicating their inflammatory nature whereas E-selectin was absent (E) in the infiltrates. Non-specific staining was also observed in several areas indicated by yellow arrow heads. Scale bar = 200 µm. Abbreviations: tRA, all-
Complement C3 and IgG Deposition in the Brain Following tRA and VARA Administration
Complement C3 and IgG deposition is the hallmark of glomerular injury in SLE. 42 These active immune components are also minimally observed in the brains of several SLE mouse models. Here, using double immunofluorescence labeling, we show a significant increase in the accumulation of C3 and IgG expression in the DV3 and meninges of tRA (Fig. 4E and F, respectively) and VARA (Fig. 4H and I, respectively) administered MRL/lpr mice compared with vehicle controls (Fig. 4A–C). tRA- or VARA-treated MRL control non-diseased mice fail to display C3 or IgG deposition (data not shown), indicating that drug dosage alone is not causing neurotoxic effects and subsequent immune activation. These results do suggest that the actions of tRA and VARA severely compromise the immuno-privileged nature of the brain under lupus-prone or autoimmune conditions. It also suggests that tRA or VARA may divert C3 and IgG deposition from the kidney to the brain, through as yet undetermined mechanisms.
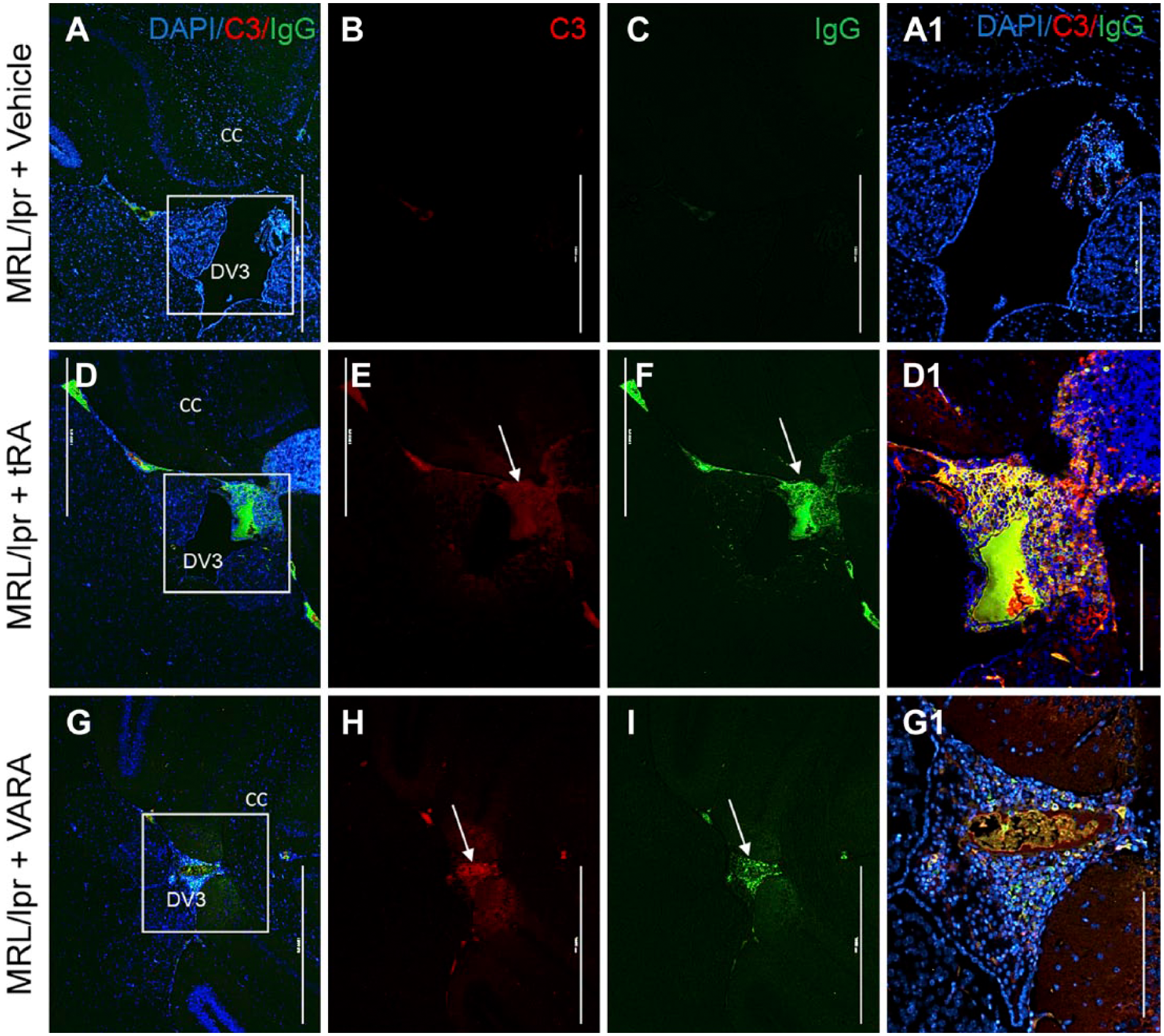
Complement C3 and IgG deposition in the DV3 of MRL/lpr mice treated with tRA and VARA. Double immunofluorescence labeling of complement protein C3 (red) and IgG deposition (green) in the brain of vehicle (A–C), tRA (D–F), and VARA-treated (G–I) MRL/lpr mice at 14 weeks. Scale bar = 1000 µm. Expression of C3 protein and IgG accumulation was substantially increased following tRA and VARA administration compared with vehicle control (D1, G1, and A1 insets, respectively). Scale bar = 400 µm. Abbreviations: tRA, all-
Increased Circulating Autoantibodies Following tRA and VARA Administration
We next investigated the humoral immune response that is the cause of type III hypersensitivity in SLE disease. 43 Peripheral blood mononuclear cells (PBMCs) isolated from the blood of vehicle, tRA-, and VARA-treated MRL/lpr mice were subjected to immune-labeling using antibodies against CD44 and Ly6c, then quantified by flow cytometry at 14 weeks of age. The number of CD44+Ly6c+ cells was increased significantly in tRA- and VARA-treated MRL/lpr mice compared with vehicle-treated MRL/lpr mice (Fig. 5A). Coincident with these changes, we found both anti-dsDNA and total IgG levels were enhanced significantly in the tRA- and VARA-treated groups compared with the vehicle-treated MRL/lpr mice (Fig. 5B and C, respectively). These results indicate that tRA and VARA increased circulating autoantibodies in MRL/lpr mice, presumably by inducing plasma cell differentiation or expansion.31,32 This may also explain the source of plasma cells and IgG deposition found in the brain.
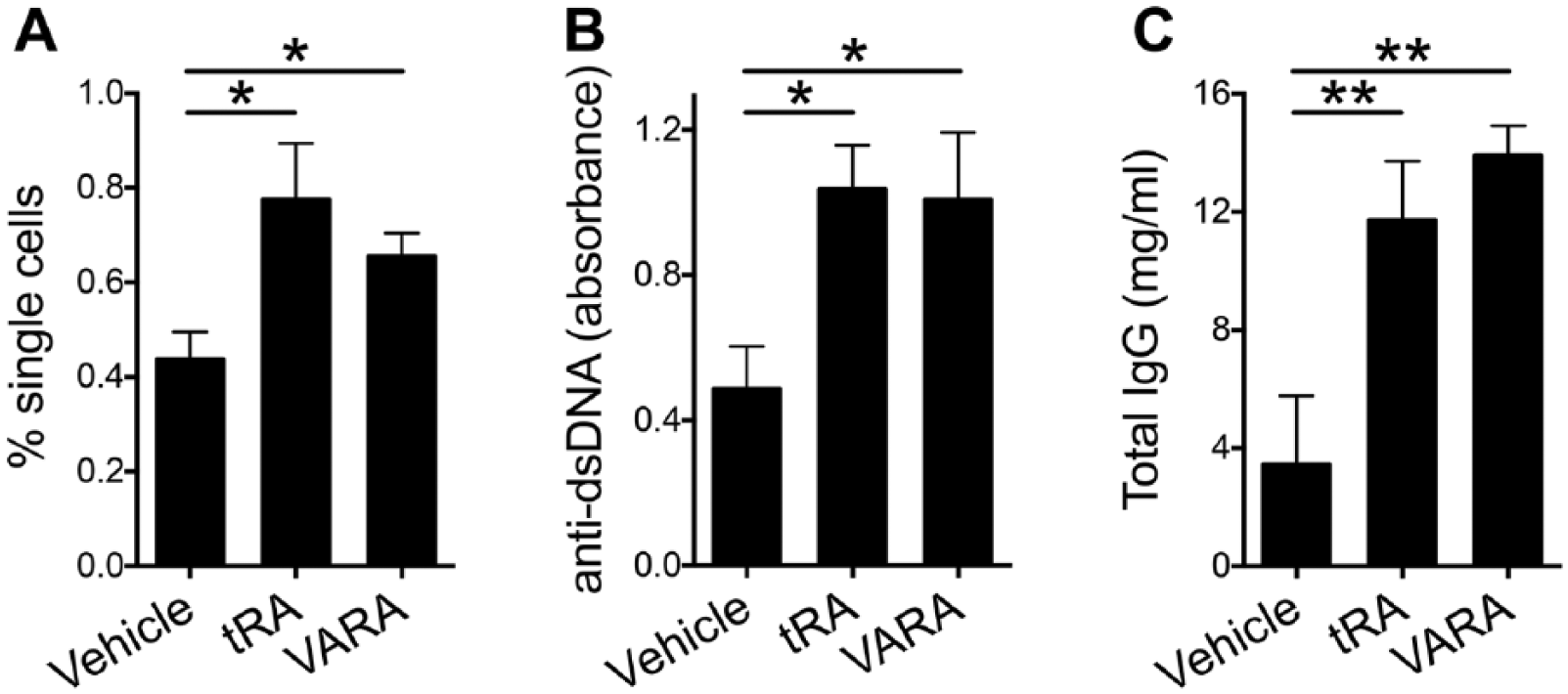
Frequency of CD44+Ly6c+ cells and levels of antibodies in the blood of MRL/lpr mice treated with tRA and VARA. (A) Percentage of CD44+Ly6c+ cells in the blood of tRA- and VARA-treated MRL/lpr mice was quantified using flow cytometry. A significant increase was observed in tRA and VARA compared with vehicle-treated MRL/lpr mice. (B) Anti-dsDNA IgG in the plasma of 14-week-old MRL/lpr mice as detected by ELISA. (C) Total IgG concentrations (mg/ml) in the plasma were also detected by ELISA. Data show significant increases in both anti-dsDNA and total IgG levels in tRA- and VARA-treated mice. *
Increased Fluoro-Jade C and GFAP Staining in the Brain Following tRA and VARA Administration
To address whether prominent astrogliosis, ventriculitis, and meningitis negatively affected neuronal health, we performed Fluoro-Jade staining to identify degenerating neurons in the cortex at 14 weeks following the regimen of vehicle, tRA, and VARA treatment in MRL/lpr mice. FJC is a high-affinity anionic fluorescein derivative that stains neurons undergoing degeneration.44,45 MRL/lpr mice treated with vehicle showed limited signs of neuron degeneration in the cortex at 14 weeks of age (7.0 ± 2.1 FJC-positive cells; Fig. 6A), whereas tRA (26.3 ± 1.9 FJC-positive cells) and VARA (28.8 ± 3.0 FJC-positive cells; Fig. 6B and C, respectively) administration significantly increased the presence of FJC-positive cells (Fig. 6J). These findings correlate with increased astrogliosis, inflammatory cell infiltration, and immune complex deposition in the brain. Although the exact mechanism(s) mediating neuronal cell death following tRA and VARA is unclear, it is well established that chronic inflammation associated with reactive astrogliosis and meningitis, as well as heightened systemic inflammation and leukocyte infiltration, leads to release of cytotoxic mediators such as reactive oxygen intermediates, proteases, cytokines, and excitatory amino acids that may participate in neuronal injury.46–48 The data overall imply that controlling vitamin A metabolism may play an important protective role in the brain during the onset of autoimmunity in MRL/lpr mice. Increased exposure to vitamin A metabolites may attenuate renal injury in SLE, however, paradoxically inducing neuroinflammation and neurodegeneration. At this time, it is unclear whether these effects are directly linked to the actions of tRA and VARA on brain physiology or are secondary to changes in the peripheral auto-inflammatory milieu.
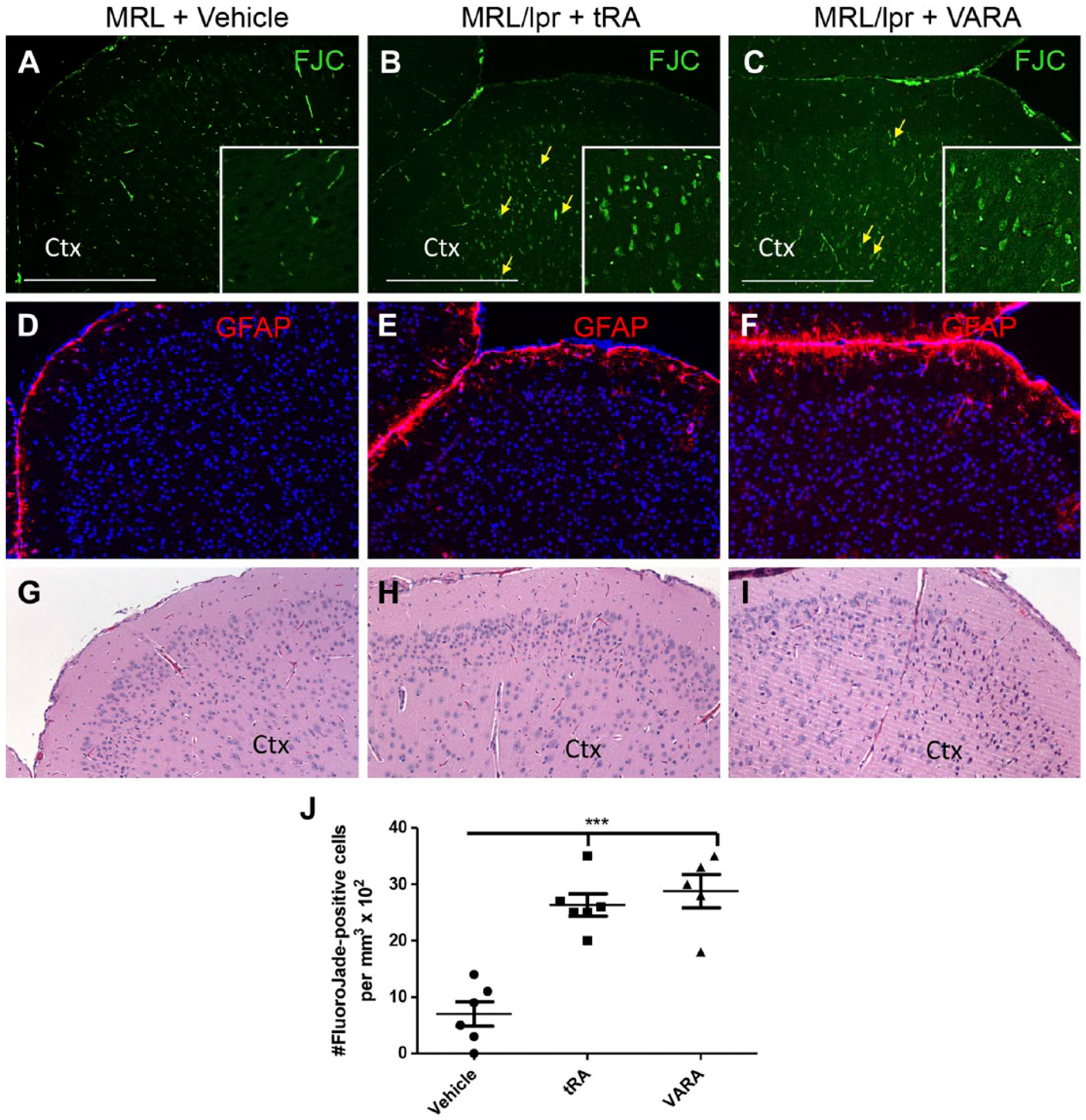
Neurodegeneration and astrogliosis in the cortex of tRA- and VARA-treated MRL/lpr mice. FJC staining (green) was used to visualize neurodegeneration in the cortex of (A) vehicle, (B) tRA-, and (C) VARA-treated MRL/lpr mice at 14 weeks. Compared with vehicle-treated mice, tRA and VARA display a significant increase in Fluoro-Jade positive neurons (yellow arrows), indicating ongoing neurodegeneration in the cortex following administration of these retinoic acid derivatives. Scale bar = 500 µm. (D–F) Representative images of anti-GFAP staining (red) in vehicle (D), tRA- (E), and VARA-treated MRL/lpr mice (F). (G–I) Representative H&E of the same brain region of the three groups of mice, respectively. (J) Quantified data of the estimated number of FJC-positive cells in the cortex. ***
Discussion
Administration of tRA has been demonstrated to reduce lupus nephritis in several models.17,36,38,39 Our previous findings using lupus-prone MRL/lpr mice confirmed a beneficial role of tRA in reducing glomerulonephritis and proteinuria during onset of lupus. 19 Interestingly, the current findings reveal that although tRA mediates renal protection in MRL/lpr mice, there is an inverse effect on immune activation and complex deposition in the brain. Although tRA seems to ameliorate glomerular inflammation, 39 it exacerbates neuroinflammation as evidenced by astrogliosis, and leukocyte accumulation in the DV3 and meninges. Furthermore, we showed that tRA significantly increased the level of circulating autoantibodies, which strongly correlated with exacerbated pathology of the brain. IgG and complement C3 deposition are highly visible in these regions as well, which correlates with positive staining for neurodegeneration marker, FJC indicating the heightened tRA-induced inflammatory milieu may cause neuronal damage secondary to the overactive autoimmune response.
Retinoid signaling not only exerts key immune-modulatory effects in the body 22 but plays an important role in the developing and mature brain.49,50 The liver is the major site of dietary retinoid storage and processing in the body51,52; however, the RA-synthesizing retinaldehyde dehydrogenases (RALDHs) are expressed in the postnatal basal ganglia, olfactory bulb, and hippocampus,53–56 indicating that RA is made locally in the brain during discrete phases of development. However, RALDHs are also expressed in the pia mater, meninges, and blood vessels of the brain.56,57 Moreover, the distribution of retinoid receptor proteins in the adult nervous system is unique from that seen during development 50 and suggests that RA signaling could also play a physiological role in multiple brain regions, including the cortex, amygdala, hypothalamus, hippocampus, and striatum. 58 Interestingly, a number of neuronal-specific genes contain recognition sequences for the retinoid receptor proteins that indicate neuronal function could be directly regulated by retinoids. 59 Several studies have provided evidence that RA signaling plays a role in normal brain functions such as learning and memory. 49 These and other findings suggest that RA may regulate a number of key physiological functions in the brain; however, it is unclear whether there is a threshold for the level of RA that may alter homeostasis. MRL control mice showed no indication of neurodegeneration, reactive gliosis, immune cell infiltration following tRA and VARA treatments, but the same treatments led to substantial exacerbation of these events under a lupus-prone environment. It is currently unclear whether this effect is related to changes in RA-synthesis or receptor expression in defined brain regions under these circumstances. Because peripheral-derived immune cells primarily accumulated in the meninges and third ventricle, neurodegeneration is likely secondary to astrogliosis and/or toxic factors released from lupus-induced immune cells. It also suggests that meningeal RA-synthesis and/or RA receptor expression may be compromised in these mice.
Increasing evidence suggests that blood brain barrier (BBB) disruption occurs in MRL/lpr mice
60
and is often observed in SLE patients that present with neuropsychiatric conditions.
61
BBB disruption is thought to be a major contributor to neuropsychiatric SLE, which comprises 20% to 70% of all SLE patients. Microthrombi-mediated ischemia and immune-mediated attack of the endothelium leading to local cytokine production are also implicated in the BBB breakdown.
61
Indeed, disruption of the BBB observed in MRL/lpr mice has been shown to be regulated by tumor necrosis factor (TNF) signaling.
60
Components of the complement system have been shown to alter the integrity of the BBB via regulation of tight junction-associated protein expression.
62
Interestingly, glutamate receptor antibodies such as anti-N-methyl-
Overall, the current study suggests that tRA and VARA treatment may exacerbate the systemic-derived influences on cellular infiltration and neurodegeneration in MRL/lpr mice. However, recent evidence points to brain-intrinsic factors in regulating degeneration in MRL/lpr mice. Despite transfer of healthy MRL bone marrow to MRL/lpr mice, neurodegeneration, cellular infiltration, and BBB disruption occurred even in the presence of attenuated systemic disease. 60 Given the exacerbation of effects in tRA- and VARA-treated MRL/lpr mice, future studies should focus on the endogenous contribution of retinoid signaling in the brain of lupus-prone mice using chimeric and pharmacotherapy studies.
Footnotes
Acknowledgements
We thank Melissa Makris for the use of flow cytometry core facility. We thank Dr. Bernard S. Jortner for performing histopathological analysis. X. Liao is a Stamps Fellow in the Biomedical and Veterinary Sciences graduate program.
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health (NIH) or any other funding agency.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: MHT and XML designed the experiments; MHT and XML wrote the paper; JBS, XL, and JR executed the experiments; and all authors have read and approved the manuscript as submitted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by R01 NS096281 (M.H.T.), R15 NS081623 (M.H.T.), and Virginia Tech’s (VT) Open Access Subvention Fund (OASF). We recognize the Virginia–Maryland College of Veterinary Medicine for student and financial support.
