Abstract
Histochemical analyses in plants are commonly performed on hand-made sections of fresh materials. The disadvantages of embedding in historesin, paraffin or paraplast® are the alterations to cellular contents, the high costs and few evident results, depending on the test. Polyethylene-glycol (PEG), as a low cost, hydrophilic medium that maintains most of the cellular features similar to fresh conditions, may be useful for obtaining good histochemical results in thinner and homogeneous sections. The current study aimed to compare the efficiency of PEG as an embedding medium for histochemical analyses of primary and secondary metabolites accumulation. Using hand-made sections of fresh samples (T1) as a comparison, we tested the influence of the use of Karnovsky’s solution as a fixative (T2) versus embedding in PEG (T3). The samples herein analyzed comprise leaves, stems, seeds and insect galls of different plant species. Neither the Karnovsky’s fixative nor the embedding in PEG altered the histochemical results for starch, lipids, terpenoids, proteins and reducing sugars in T1, T2, and T3. However, PEG binds to phenols, such as tannins, flavonoids and lignins, thereby presenting false negatives in T3.
Introduction
Obstacles in embedding and sectioning of plant materials are common in routine analyses of plant anatomy, especially for highly lignified materials, such as wood, and scleromorphic stems and leaves. On the other hand, some materials are thin and delicate, thus requiring embedding in paraffin, paraplast®, or even in more expensive medium, such as historesin (Leica Microsystems GmbH, Wetzlar, Germany). Historesin preserves the cytological features of the sample, but does not allow good images in histochemical analyses when compared with those obtained from hand-made sections using fresh material. Indeed, nowadays, several works have used sectioned fresh samples for histochemical reactions for lipids and terpenoids (Santos et al. 2008; Lopes et al. 2009; Coutinho et al. 2010; Oliveira and Isaias 2010; Oliveira et al. 2010; 2011; Marinho et al. 2011; Carneiro et al. 2014), even though the histological analyses were made in embedded material. Ventrella et al. (2013) suggested the use of fresh samples, although they mentioned the possibility of using historesin- or paraffin/paraplast®-embedded samples for some histochemical analyses. Nevertheless, these authors highlighted the artifacts that the histochemical tests on embedded sections may produce, which include more tenuous staining, reactions between the reagents and the embedding media, shrinkage of the sections and/or removal of compounds in the embedding process.
The polymers of paraffin and paraplast® are long hydrocarbons; i.e., long, hydrophobic molecules (Smith 1953), and the embedding process requires gradual solution changes; for instance, from alcohol to mineral oil to paraffin/paraplast®-replacement (Kraus and Arduin 1997). Thus, the dehydration associated with the process of embedding with paraffin and paraplast® removes the oils, terpenoids and other lipophilic reserves stored in the cytoplasm, thus hindering the histochemical detection of such compounds.
Beyond the aforementioned media, polyethylene-glycol (PEG) has been used in animal histology, histochemistry and immunocytochemistry (Smithson et al. 1983; Bard and Ross 1986; Gao and Godkin 1991). Plant anatomists have employed this medium for the common analysis of wood anatomy (Rupp 1964; Theiler 1973; Araújo and Costa 2007; Barbosa et al. 2010; Tamaio 2011).
Polyethylene-glycol is a polymer with a simple structure: HO-[CH2-CH2-O]n-H (Henning 2002). The compound is synthesized in industry through the polymerization of ethylene oxide (EO) with either water, mono ethylene glycol or diethylene glycol as a starting material, under alkaline catalysis (Henning 2002), resulting in a low cost of production. PEG is highly hydrophilic (Michel and Kaufmann 1973; Henning 2002; Barbosa et al. 2010; Sigma-Aldrich® 2014), and its aqueous solution has a very negative osmotic potential because of the several hydroxyl groups linked to the polymer (Michel and Kaufmann 1973; Henning 2002). The molecular mass of commercial PEG ranges from 200 to 10,000, according to the structure of the polymer. For plant material embedding, we have been using PEG 6,000. The melting temperature of PEG 6,000 (PEG with the molecular mass = 6,000) is 60C, and its solubility is high even in the presence of electrolytes because it is a nonionic substance (Henning 2002; Sigma-Aldrich® 2014). Because of these properties, PEG is used either as a dehydrating or embedding medium, and maintains the integrity of cellular contents even after all processes previous to the embedding (Rupp 1964; Barbosa et al. 2010). Henning (2002) has already highlighted the better results obtained with PEG when compared with paraffin, paraplast® and historesin. But, as far as we are concerned, PEG embedding for histochemical analyses has yet to be systematically tested. Then, taking PEG properties for granted, the current work analyzes the advantages and disadvantages, the efficiency and applicability of embedding plant material in PEG for basic histochemical tests.
Materials & Methods
A set of available plant material with distinct tissues and patterns of metabolite accumulation was handled for the analyses. Leaves, stems, and stem galls induced by a Lepidoptera on Marcetia taxifolia (Melastomataceae); leaves, stems, and leaf galls induced by Clinodiplosis sp. (Cecidomyiidae) on Croton floribundus (Euphorbiaceae); and imbibed bean seeds (Phaseolus vulgaris, Fabaceae) were submitted to three processes (T1, T2 and T3), as described below. The galls were herein included for they have been proved to be good models for histochemical studies, due to a magnification of reserves of primary and secondary metabolites in several systems (Moura et al. 2008; Oliveira and Isaias 2010; Oliveira et al. 2010; 2011; Vecchi et al. 2013; Ferreira and Isaias 2013; In Press; Carneiro et al. 2014). They can also be useful in testing the possible influence of the stressed cells on the methods.
T1 (fresh material): Fresh collected samples hand-sectioned with razor blades were used as the control tests.
T2 (fixed material): Samples were fixed with 2.5% glutaraldehyde and 4.5% formaldehyde in 0.1 M phosphate buffer, pH 7.2 (Karnovsky 1965), and stored in 70% ethanol. The fixed material was re-hydrated, and hand-sectioned with razor blades.
T3 (samples fixed and embedded in PEG): Fragments were fixed (Karnovsky 1965), washed in 0.1 M phosphate buffer (30 min), washed in distilled water (30 min), and then embedded with PEG 6,000. The process of embedding began with a solution of 25% PEG 6,000 in distilled water (25% w/v) in a 60C stove. The total volume was demarcated and when half of the volume had evaporated, the solution was completed with pure PEG 6,000 and maintained at 60C for at least 48 hr. The mounting solution was a 90% PEG solution (w/v) maintained at 60C with 2% Arabic gum, added at the moment of mounting (Kraus and Arduin 1997). The melted mounting solution was placed in cubic paper boxes, and the samples were placed and positioned immediately thereafter in the melted block. The solidified blocks were stored in the freezer (-20C). For sectioning in rotary and sliding microtomes (20-40 µm), the blocks of PEG were adhered to wood blocks with melted 90% PEG solution, and then stored in freezer. To prevent microtomized sections from crumbling, the blocks were moisturized using a wet paint-brush prior to sectioning. The sections were placed in 40-50C waterbath, and washed in distilled water.
Histochemical Analyses
Immediately after sectioning, the sections were immersed in water, and submitted to the histochemical reagents (Table 1), and to the control tests suggested by each author. Thereafter, the sections were placed on the slides with a drop of water, covered with coverslips, immediately analyzed and photographed using a light microscope (Leica ICC50 HD, Leica Microsystems GmbH, Wetzlar, Germany).
Histochemical Analyses Used in Current Work.
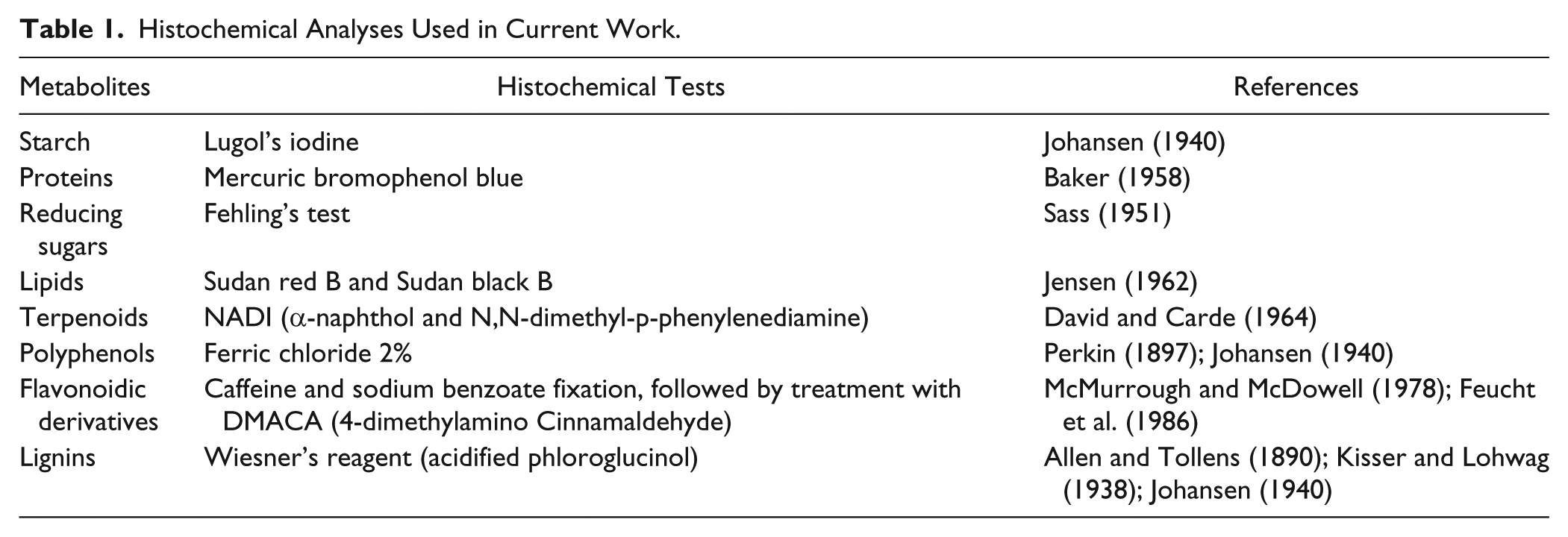
Results
The process of fixation in Karnovsky’s solution (T2) did not alter the detection of any handled positive results for all samples and tested metabolites (Table 2). The detection of primary metabolites was similar in all treatments (T1, T2 and T3). The results of the Lugol’s test were more evident in thin sections of material embedded in PEG (T3) (Fig. 1A–1D). On the other hand, the detection of proteins was more evident in fixed material (T2) (Fig. 1E–1I). The detection of reducing sugars and lipids was not different among T1, T2 and T3 (Fig. 1J–1O).
Detection of Metabolites in Fresh Samples (T1), Fixed (T2), and Embedded in PEG (T3) in Marcetia taxifolia (Melastomataceae), Croton floribundus (Euphorbiaceae) and Phaseolus vulgaris (Fabaceae).
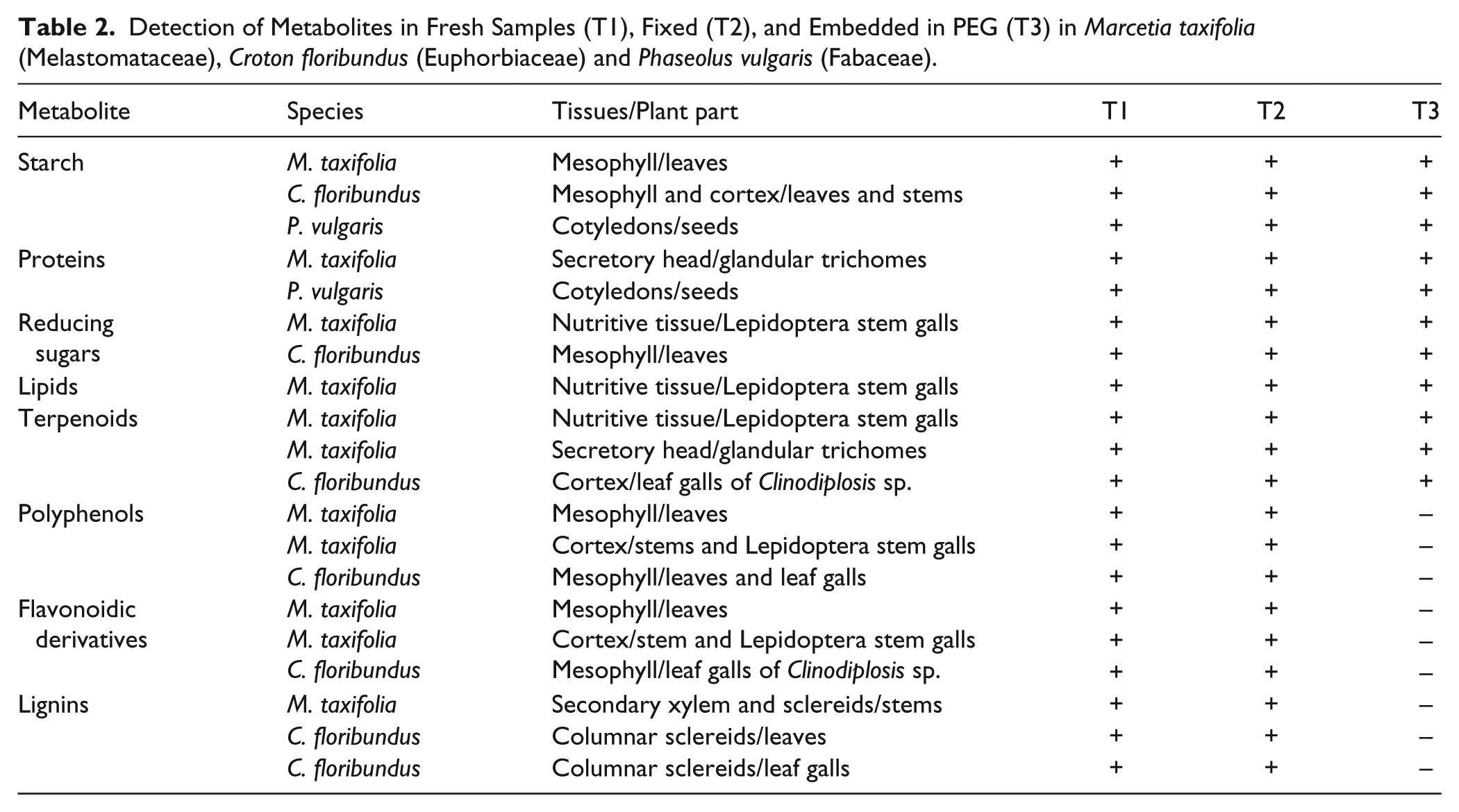
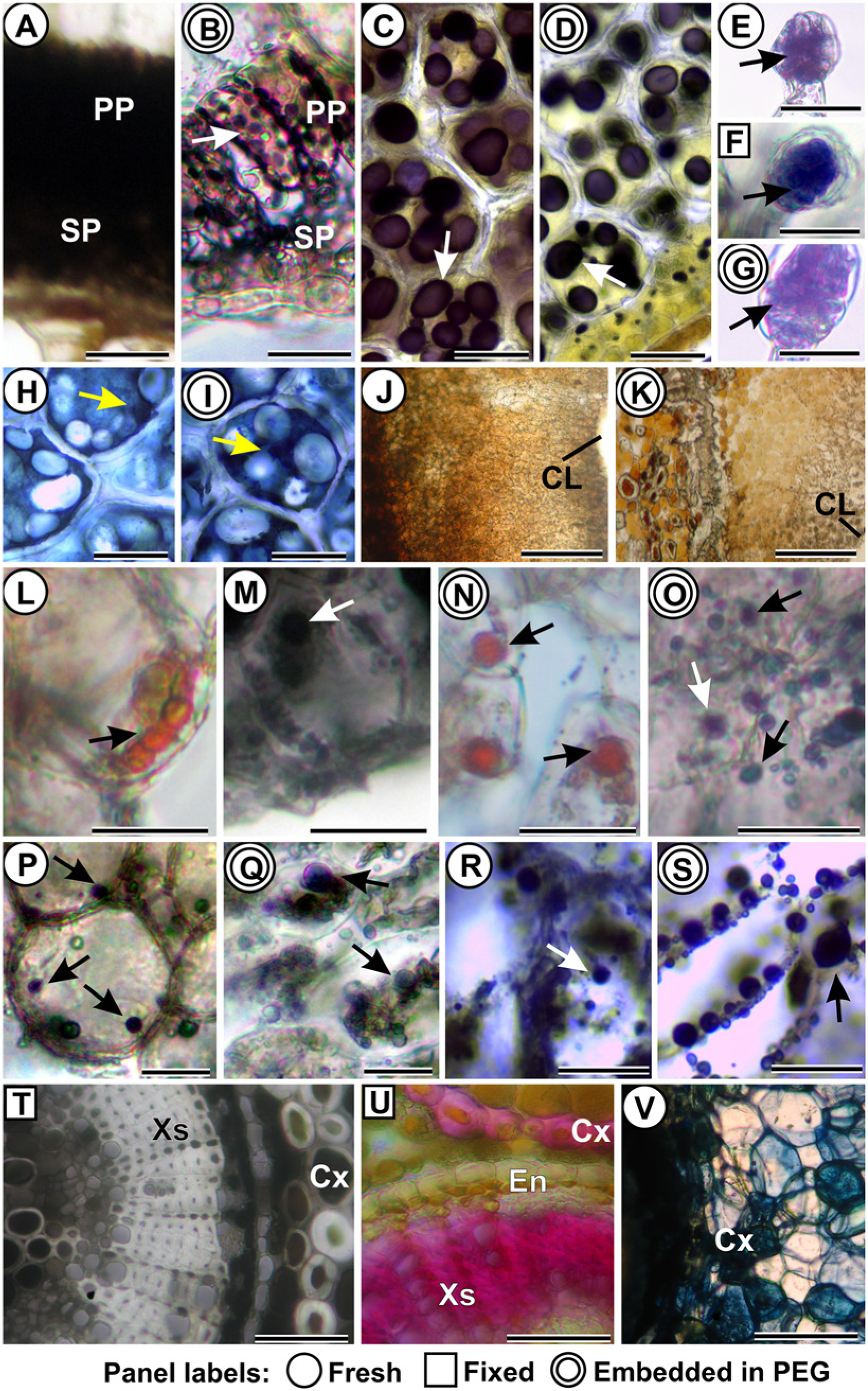
Histochemical analyses of fresh samples (T1; one ring around panel labels), fixed samples (T2; square around panel labels), and samples fixed and embedded in PEG (T3; 2 rings around panel labels). (A–D) Lugol’s reaction for starch. (A–B) Leaf of Marcetia taxifolia. Arrow: starch grains. (C–D) Cotyledon of the seed of Phaseolus vulgaris. Arrows: starch grains. (E–I) Mercuric bromophenol blue detection of proteins. (E–G) Secretory heads of trichomes on M. taxifolia (arrows). (H–I) Cotyledon of the seed of P. vulgaris. Arrows: proteins. (J–K) Fehling’s reaction for reducing sugars. Decreasing gradient towards the larval chamber (CL) in Lepidoptera stem gall on M. taxifolia. (L–O) Lipids (arrows) in the nutritive tissue of Lepidoptera stem galls on M. taxifolia. (L, N) Sudan red B. (M, O) Sudan black B. (P–S) NADI reaction for terpenoids (arrows). (P, Q) Nutritive tissue of Lepidoptera stem galls on M. taxifolia. (R, S) Cortex of leaf galls of Clinodiplosis sp. on Croton floribundus. (T) 2% ferric chloride detection of polyphenols (black content) in fixed stems of M. taxifolia. (U) Acidified phloroglucinol detection of lignins (pink cell walls) in secondary xylem (Xs) and some cortex cells (Cx) in fixed stems of M. taxifolia. (V) DMACA (4-dimethylamino Cinnamaldehyde) detection of flavonoidic derivatives (dark blue) in cortex cells (Cx) of stems of M. taxifolia, in sections of fresh material. Abbreviations: CL: larval chamber. Cx: cortex. En: endoderm. PP: palisade parenchyma. SP: spongy parenchyma. Xs: secondary xylem. Scale bars: (A, B, L, M): 10 µm; (C, D, H–K; T–V): 50 µm; (E–G; N, O; R, S): 20 µm; (P, Q): 15 µm.
The detection of secondary metabolites was not affected by the fixation (T2), and the detection of terpenoids was not altered by embedding in PEG (T3) (Fig. 1P–1S). However, embedding with PEG resulted in false-negatives in the detection of total polyphenols, including flavonoidic derivatives and lignins (Table 2), which were assumed based on the comparative positive results with the other treatments (T1 and T2) (Fig. 1T–1V).
Discussion
Our results demonstrate that the process of fixation (T2) does not alter the results obtained by the tests with the fresh material (T1), even with the material affected by the biotic stress of galling insects. The Karnovsky’s solution has been demonstrated to be advantageous for it does not destroy or alter the contents of the cells and tissues, because of the rapid stabilization of the semipermeable membranes (Karnovsky 1965; Paljärvi et al. 1979). The glutaraldehyde is a much more efficient fixative for cell membranes than formaldehyde; however, the former has a slower rate of tissue penetration than the latter. Thus, Karnovsky’s fixative provides an ideal mixture of these two compounds for an efficient fixation because formaldehyde can “pre-stabilize” the tissue while waiting for the glutaraldehyde to diffuse through the sample (Paljärvi et al. 1979).
Even though fixed samples have been more rarely used in plant histochemistry, some authors have been successfully using FAA (formaldehyde, acetic acid and ethyl alcohol solution) (Farías et al. 2009) or Karnovsky’s solution in their tests (Cahill et al. 1989). The results of the current study reinforce the applicability of Karnovsky’s solution for histochemical analyses, and its usefulness when fresh material is not available. Fresh samples (T1) are commonly used in basic histochemical analyses (Johansen 1940; Jensen 1962; Kraus and Arduin 1997; Santos et al. 2008; Lopes et al. 2009; Coutinho et al. 2010; Oliveira and Isaias 2010; Oliveira et al. 2010; 2011; Marinho et al. 2011; Ventrella et al. 2013) and are therefore herein considered as controls for the purpose of comparison. Neither fixation nor embedding in PEG alters the histochemical results, with the exception of the false-negative results obtained when using T3 for the detection of polyphenols, lignins and flavonoidic derivatives. This was most likely attributed to the precipitation of phenols and tannins (Henning 2002) through strong hydrogen bonds between hydroxyl groups in phenols and PEG (Silanikove et al. 2001). Patel and Foss (1964) showed that PEG molecules bind with phenols, and Gehrig et al. (2000) described a protocol of RNA (ribonucleic acid) extraction that uses PEG to remove an excess of polyphenols. Thus, PEG binds to the phenolics—a group comprising lignins, tannins and flavonoidic derivatives (Boerjan et al. 2003; Castro and Demarco 2008; El Gharras 2009)—during the staining and thus causes the histochemical reagents to fail (Table 1). Phloroglucinol (1,3,5-trihydroxy-benzene) is another phenolic compound which may bind to PEG polymers (Ning et al. 2008; Wang et al. 2012) similar to the lignin polymers, and thus would block the histochemical reaction for Wiesner’s stain for the detection of lignins.
In theory, PEG-embedded material can be used for PAS (periodic acid-Schiff) and Feulgen’s techniques for the detection of polysaccharides and nucleic acids, respectively. The principle of both procedures is the formation of colored addition complexes with the aldehydes (Jensen 1962). In the keto-enol tautomerism of the DNA, the -aldehyde forms (Watson and Crick 1953) are more common than the -enol forms (with hydroxyls). As PEG cannot bind to the aldehydes through hydrogen bonds, the sites for aldehyde-basic fuchsin linkages are available either for PAS or Feulgen’s test, and PEG-embedded material can be used for both histochemical techniques.
Also theoretically, sections of plant material embedded in PEG may be suitable for immunocytochemical analyses—as is true for animal tissues—assuming that the embedding process does not alter the properties of plant tissues (Smithson et al. 1983; Bard and Ross 1986; Gao and Godkin 1991). Moreover, PEG does not exhibit significant autofluorescence under the emission spectra between 190–540 nm wavelengths (Chiu et al. 2012), which is advantageous for immunocytochemistry. In any case, pilot tests similar to our analyses are recommended to attest the efficiency of embedding in PEG for PAS, Feulgen’s and plant immunolocalization. Our results confidently attest to the usefulness of Karnovsky’s solution as a fixative for the histochemical detection of starch, proteins, soluble sugars, lipids, terpenoids, polyphenols, lignins, and flavonoidic derivatives. PEG prevents the removal of lipophilic substances stored in the cells and it is possible to perform histochemical analyses with a wide range of plant materials. However, PEG should not be used as an embedding medium for samples that will be tested for phenolic substances (polyphenols, lignins, and flavonoidic derivatives).
Footnotes
Acknowledgements
We thank Renê Gonçalves da Silva Carneiro (UFMG) and Cibele Bedetti de Souza (UFMG) for the critical reading of the manuscript, and Prof. Dr. Geraldo L. G. Soares (UFRGS) for the remarkable advice on chemical reactions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) and FAPEMIG (Fundação de Amparo à Pesquisa do Estado de Minas Gerais) for the financial support.
