Abstract
The aim of this study was to evaluate the effect of sintered dicalcium pyrophosphate (SDCP) on fracture healing in an osteoporotic rat model. Female Sprague-Dawley rats (8 weeks old) were randomly allocated into five groups: sham-operated group, and bilateral ovariectomized group treated with SDCP, alendronate, calcitonin, or no treatment. Rats were sacrificed at 6 or 16 weeks after fracture. Fracture sites were examined by microcomputed tomography (microCT), histology, and mechanical testing. The results showed that SDCP mildly suppressed callus remodeling at 6 weeks, but not at 16 weeks. The lamellar bone in the callus area and new cortical shell formation in SDCP-treated group were similar to that of the sham group at 16 weeks after fracture, indicating there was no delayed callus remodeling into lamellar bone. At both 6 and 16 weeks after fracture, ultimate stress and elastic modulus were similar between the SDCP and sham groups, and the mechanical strength in these groups was better than that in other groups. Finally, analysis of the serum bone markers CTX-1 and P1NP suggested that SDCP decreased the bone turnover rate and promoted proper fracture healing. The effect of SDCP is superior to that of alendronate and calcitonin in the healing of osteoporotic fractures.
Introduction
Osteoporosis is a metabolic bone disorder that affects both men and women. It causes low bone mass and therefore increases bone susceptibility to fracture, even if it’s only a minor trauma. In developed countries, osteoporosis-associated fractures are a significant cause of morbidity and mortality (Johnell and Kanis 2006). Because most cases of osteoporosis are diagnosed when a fracture occurs and anti-osteoporosis medications are prescribed at that time, information on the influence of anti-osteoporosis drugs on fracture healing is essential. Bisphosphonates and calcitonin are two frequently prescribed medications used to treat osteoporosis (Cranney et al. 2002; Drake et al. 2008).
Alendronate was first approved by the US Food and Drug Administration in 1995, and subsequently three other bisphosphonates—risedronate, ibandronate, and zoledronic acid—were approved. Bisphosphonates suppress bone resorption by inhibiting osteoclast activity (Murakami et al. 1995), and long-term use further inhibits osteoclast differentiation (Li et al. 1999). With the concomitant reduction in the associated cost and need for health care, bisphosphonates have become widely recognized as a mainstay of treatment for osteoporosis (Yoon et al. 2011). However, a recent report has indicated a link between long-term bisphosphonate use and atypical non-spinal fractures (Yoon et al. 2011). Indeed, high-dose alendronate treatment, a bisphosphonate commonly used in clinical practice, induces the formation of a large, strong callus, but delays the structural and material restoration of the bone at the repair site (Manabe et al. 2009). Bisphosphonate use is associated with significant side effects, such as low bone turnover, hypocalcaemia, and osteonecrosis of the jaw (Drake et al. 2008). It has been suggested that severe and prolonged suppression of bone turnover may impair the ability of the bone to remodel, eventually leading to the accumulation of micro-damage and a diminution of bone strength (Watts and Diab 2010). In addition to micro-damage, the chronic suppression of bone turnover by alendronate may allow this secondary mineralization to continue to produce hyper-mineralized bone of a brittle nature (Odvina et al. 2005).
Calcitonin, on the other hand, inhibits osteoclastic bone resorption and increases bone density, and has been used to treat osteoporosis in postmenopausal women for more than 30 years (Cranney et al. 2002). A previous study has shown that calcitonin mildly suppresses callus remodeling, but does not appear to affect the natural fracture healing process and intrinsic material properties of the new bone (Manabe et al. 2009). However, the effects of calcitonin on bone cells and bone remodeling are not clear.
Although bisphosphonates and calcitonin have both been shown to be very useful in the treatment of osteoporosis, their limitations require the development of new treatments. Sintered dicalcium pyrophosphate (SDCP), composed of sintered beta-Ca2P2O7 with the addition of Na4P2O7.10H2O, is a pyrophosphate analog that has been shown to be biocompatible with bone in both in vivo and in vitro models (Lin et al. 1995; Sun et al. 1997; 2002; 2003a), demonstrating comparable effects to those of alendronate (Sun et al. 2002). Unlike bisphosphonates, SDCP is bioabsorbable and can be metabolized by endogenous phosphatases (Lin et al. 1995). Orally administered SDCP has been shown to increase bone mass in ovariectomized rats (Sun et al. 2002). Unlike bisphosphonates, SDCP has a high affinity for osteoblasts and induces osteoclast apoptosis by degradation of the Golgi apparatus in these cells (Sun et al. 2003a). Thus, although SDCP is a promising treatment for osteoporosis, the potential interaction between SDCP treatment and osteoporotic fracture healing must be examined.
The mode of action of medications used to treat osteoporosis is usually anti-catabolic/anti-resorptive or anabolic. As such, it is of interest to know whether these drugs affect not only normal bone remodeling but also fracture healing. Because osteoporotic patients are prone to fractures, physicians must face the possibility of fractures occurring in these patients while they are undergoing treatment for osteoporosis. The rat femoral osteotomy fracture model using intramedullary wiring has provided valuable information about fracture healing and the effects of pharmacological agents (Li et al. 1999; 2000; Sun et al. 2002). Thus, we used this model to explore the effect of SDCP and various anti-osteoporotic medications on fracture healing in osteoporotic rats.
Materials & Methods
Study Protocol
The experimental protocol was approved by the Institutional Animal Care and Use Committee of National Yang Ming University (Affidavit of Approval of Animal Use Protocol National Yang Ming University, IACUC 1000205). Sixty female Sprague-Dawley rats (8 weeks old) were purchased from the Laboratory Animal Center, National Yang Ming University (Taipei, Taiwan) and acclimated under standard laboratory conditions at 22 ± 2C and 50 ± 10% humidity. Standard rat chow and water were available ad libitum during the acclimation period.
Experimental Design
An osteoporotic fracture rat model was established to evaluate changes of bone microstructure and mineralized tissue during osteoporotic fracture healing as previously described, with minor modification (Hao et al. 2007). Briefly, the animals were randomly allocated into two groups: the sham-operated group (n=12) bilateral ovariectomy group (OVx, n=48). At 4 weeks after surgery (rather than 12 weeks), a femoral osteotomy and fixation was performed on all rats (n=60), as previous described (Li et al. 1999). A transverse osteotomy was made at the mid-shaft of the left femur, and the fracture fragments were reduced, stabilized and then fixed using an intramedullary stainless steel wire (diameter, 1.6 mm; Synthes, Switzerland). The wire was cut on the surface of the intercondylar groove to avoid motion restriction of the knee joint. Rats were permitted unrestricted activity after recovery from anesthesia.
After fracture surgery, OVx rats were then further divided into four groups (n=12 in each group): vehicle (no treatment) group, SDCP group, alendronate (ALN) group, and calcitonin (CN) group. SDCP (Purzer Pharmaceutical Co., Taipei, Taiwan) was administered orally at a dosage of 1.0 mg/kg/day, 5 days per week, which is the optimal dosage based on prior study (Sun et al. 2002). Alendronate (Fosamax; Merck Sharp & Dome, Darmstadt, Germany) was administered orally at a dosage of 1.0 mg/kg/day, 5 days per week (Guy et al. 1993). Salmon calcitonin (Purzer Pharmaceutical Co., Taipei, Taiwan) was administered by subcutaneous injection at a dosage of 5 IU/kg/day, 5 days per week (Karsdal et al. 2008). The sham and OVx vehicle groups received the vehicle (isotonic sodium chloride solution) via a feeding tube through the mouth in the early morning after 12 hr of starvation, and this was continued until sacrifice.
Rats were sacrificed at 6 or 16 weeks after surgery (n=6 at each timepoint from each group). Left femurs were excised, all soft tissues were dissected and intramedullary wires were extracted. Specimens were collected for quantitative microcomputed tomography (microCT), mechanical testing, and histomorphometric study. Whole blood samples were obtained from all rats with plastic syringes via intra-cardiac puncture immediately following sacrifice. The blood samples were stored on ice, the serum was separated by centrifugation and divided into 500-µl aliquots in Eppendorf tubes, which were then stored at -80C until further analysis. A summary of the animal grouping and treatments is presented in Figure 1.
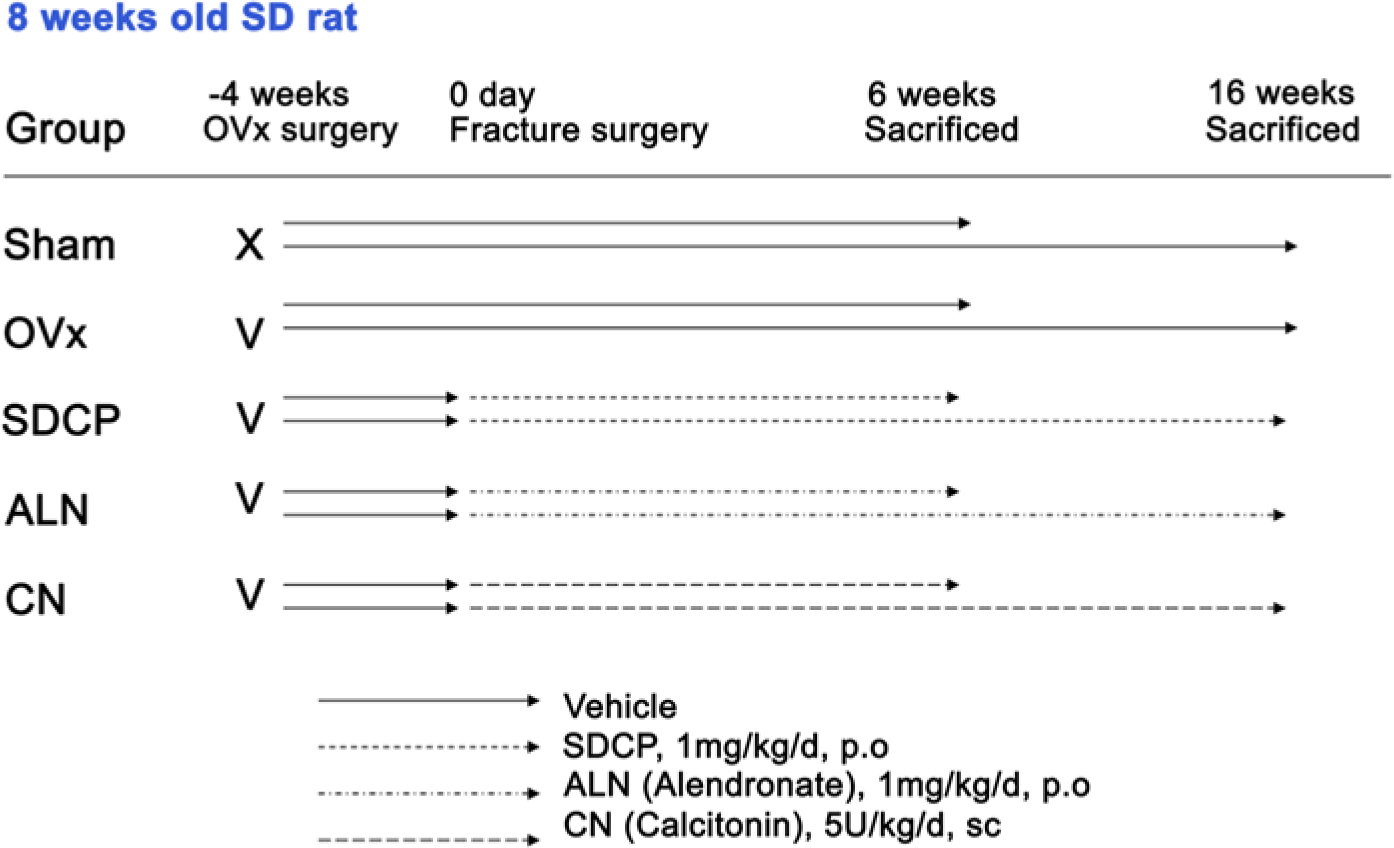
Summary of animal grouping and treatments. Animals were randomly allocated into two groups: sham group received a sham operation (n=12) whereas all remaining rats received bilateral ovariectomy (OVx, n=48). At 4 weeks after surgery, all rats were subjected to femoral osteotomy and fixation. The OVx rats were then further divided into four groups: vehicle group, sintered dicalcium pyrophosphate (SDCP) group, alendronate (ALN) group, and calcitonin (CN) group (n=12 in each group). The sham and OVx vehicle groups received the vehicle (isotonic sodium chloride solution) via a feeding tube. Rats were sacrificed at 6 or 16 weeks after surgery (n=6 in each group at each time point).
Radiological Examination and Microcomputed Tomography
Anteroposterior and lateral radiographs of the left femur were taken using a Triumph II X-ray device (General Electrical Medical System, Milwaukee, WI, USA) with the parameters of 50 kV, 2 mA, and 10 min.
The microarchitecture of the left femur was investigated using microCT (Triumph II; General Electrical Medical Systems). The voltage was 50 kV, the pixel size was 8 µm, and 512 projections were acquired over an angular range of 360° (angular step of 0.70°). The image slices were reconstructed, and the registered data sets were segmented into binary images. The trabecular bone was extracted by drawing ellipsoid contours with the analyzer software. Trabecular bone volume (trBV), trabecular total volume (trTV), bone volume percentage (BV/TV; %), trabecular thickness (Tb.Th), trabecular number (Tb.N), and trabecular separation (Tb.Sp) were calculated by peripheral quantitative computed tomography (pQCT).
Mechanical Testing
Immediately after microCT, a 3-point bending test was performed on the fracture site of the femur using a load mechanical testing machine (Instron, Series 4467 table top loading frame, Norwood, MA, USA). The femur was placed with its anterior surface upward on two lower supporting bars (21.9 mm apart), and the loading bar was positioned at the fracture site (anteroposterior position). A load was then applied at a rate of 10 mm/min until failure. The maximum force (N; maximum force of the load-deformation curve prior to failure) and stiffness (N/mm; the slope of the linear portion of the load-deformation curve) were calculated from the load-deformation curve. Because the structural properties depend on the size or geometry of the specimens, all specimen results were normalized using the cross-sectional moment of inertia (CSMI), as previously described (Li et al. 2001); this was then used to obtain intrinsic material properties such as ultimate stress (N/mm3) and elastic modulus (MPa), which are independent of cross-sectional size and shape.
Histological Preparation and Contact Microradiography
After mechanical testing, the specimens were repositioned and fixed in 10% formalin for microscopic evaluation. Formalin-fixed bones were decalcified with 7.5% nitric acid, embedded in paraffin, and cross-sectioned. Histological sections (7 µm thick) were stained with hematoxylin and eosin (H&E) prior to optical microscope examination.
Histomorphometric Analysis
Histomorphometric analysis was performed with an Aperio ScanScope CS (Aperio Technologies, Vista, CA, USA), which permitted scanning of the entire slide containing the regions of interest. The total area (TA), original cortical area, and the medullary area were measured, and the callus area (Ca.Ar) was calculated as described previously (Li et al. 1999). The lamellar bone area (La.Ar) and callus surface (Ca.S) were also measured. Osteoclast measurements were performed at 200× magnification in four standardized quarters: anterior, posterior, medial, and lateral aspects. Osteoclasts were defined as cells with more than three nuclei. Osteoclast number (N.Oc) was counted, and from this, the N.Oc/Ca.S ratio was calculated.
Biochemical Analysis
To specifically assess the degradation of type I collagen in bone, immunoassays for the level of type 1 carboxy-terminal collagen fragments (CTX-1) and the amino-terminal propeptide of type 1 procollagen (P1NP) in serum were performed. These markers have been demonstrated to provide a specific and sensitive index of bone resorption (CTX-1) and bone formation (P1NP) in several metabolic bone diseases, including osteoporosis (Garnero and Delmas 2003; Tsujimoto et al. 2011). Serum levels of CTX-1 fragments were measured using the RatLaps enzyme immunoassay (EIA; Immunodiagnostic Systems, Limited, Boldon, UK), and levels of P1NP were measured using the Rat P1NP EIA (Immunodiagnostics Systems Limited), according to the manufacturer’s instructions. The detection limit and range were 2.0 ng/ml and 0–188 ng/ml, respectively, for CTX-1 (ng/ml) and 0.7 ng/ml and 0–75 ng/ml, respectively for P1NP (ng/ml).
Statistical Analysis
The mean and standard deviation (SD) were calculated for each group. For comparisons among the five trial groups, one-way ANOVA was used to examine continuous variables. When a significant difference between groups was apparent, multiple comparisons were performed using the Bonferroni procedure with type-I error adjustment. Data were analyzed with SPSS 18.0 (SPSS, Inc., Chicago, IL), and a P value < 0.05 was considered statistically significant.
Results
Radiological Findings
Radiographs (Fig. 2A) and microCT images (Fig. 2B) of the femurs demonstrated that the fracture line was still present in all groups at 6 weeks post-fracture. All treatment groups had a lager callus than the sham group at 6 weeks post fracture. At 16 weeks post-fracture, the callus size of the SDCP group did not differ from that of sham group, whereas the ALN and CN groups had a larger callus than the other groups. The ALN and CN groups exhibited a large and woven-like callus at both 6 and 16 weeks post fracture, whereas the SDCP treatment group exhibited a shrunken and solid callus at 16 weeks as compared with that observed at 6 weeks post-fracture (Fig. 2).
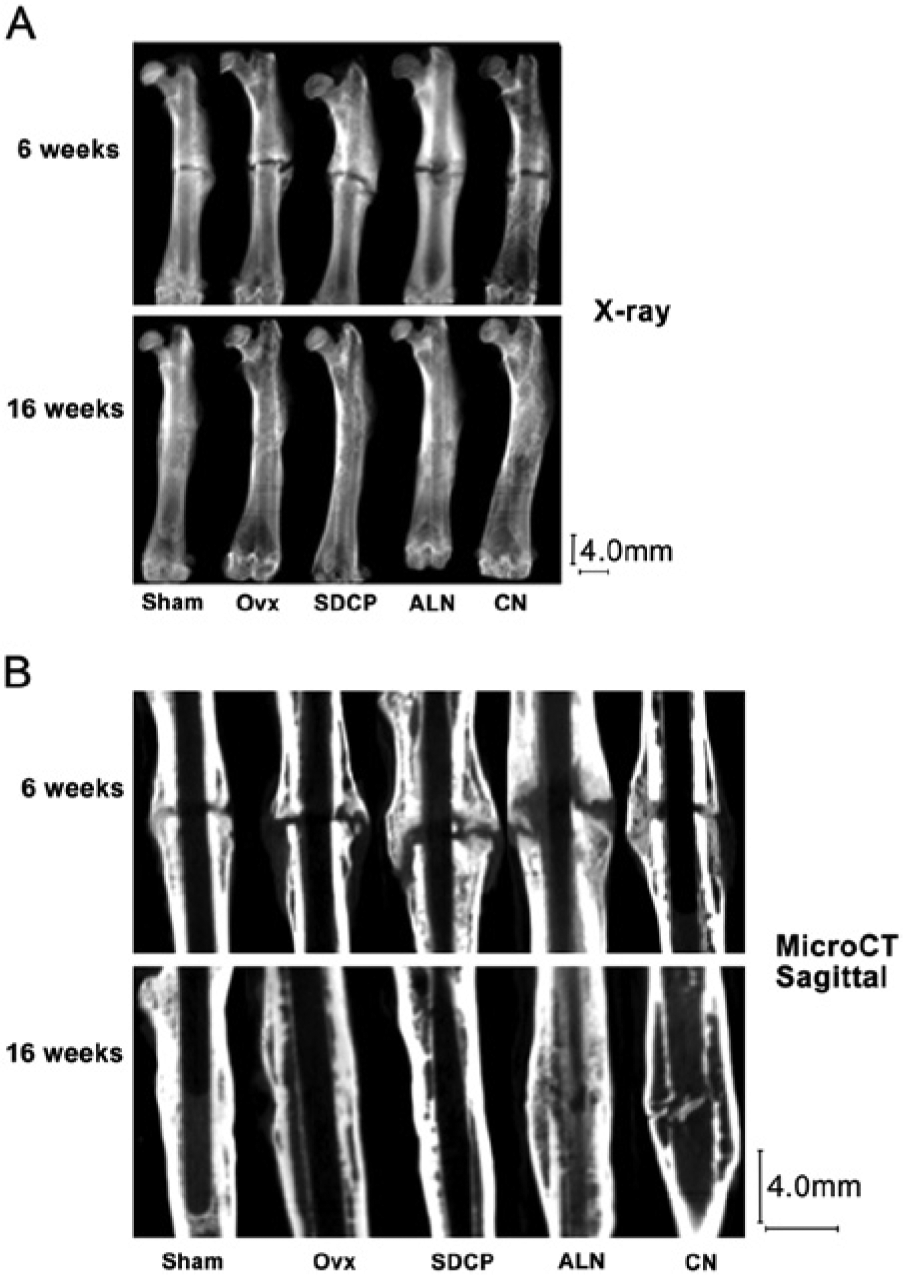
Radiographs (A) and microCT images (B) of the femurs of the study groups at 6 and 16 weeks after fracture. The ALN and CN groups exhibited a large and woven-like callus at both 6 and 16 weeks after fracture, whereas rats treated with SDCP exhibited a shrunken and solid callus at 16 weeks as compared with that observed 6 weeks after fracture. At 16 weeks post-fracture, the callus size of the SDCP group did not differ from that of the sham group. OVx, ovariectomy; SDCP, sintered dicalcium pyrophosphate; ALN, alendronate, CN, calcitonin. The vertical and horizontal bars are 4.0 mm.
The results of the histological examination of the femurs at 6 and 16 weeks post fracture are shown in Figure 3. In the sham group, the callus decreased with time, and the formation of a new cortical shell reached almost 100% at 16 weeks post-fracture in these rats. At 6 weeks post-fracture, the callus sizes for rats in the SDCP, ALN, and CN groups were much larger than those in the sham group. At 16 weeks, the callus sizes in the sham and SDCP groups were smaller, with the appearance of the new cortical shell. Comparatively, the extent of callus reduction was less in the ALN and CN groups than in the other groups at this later time point.
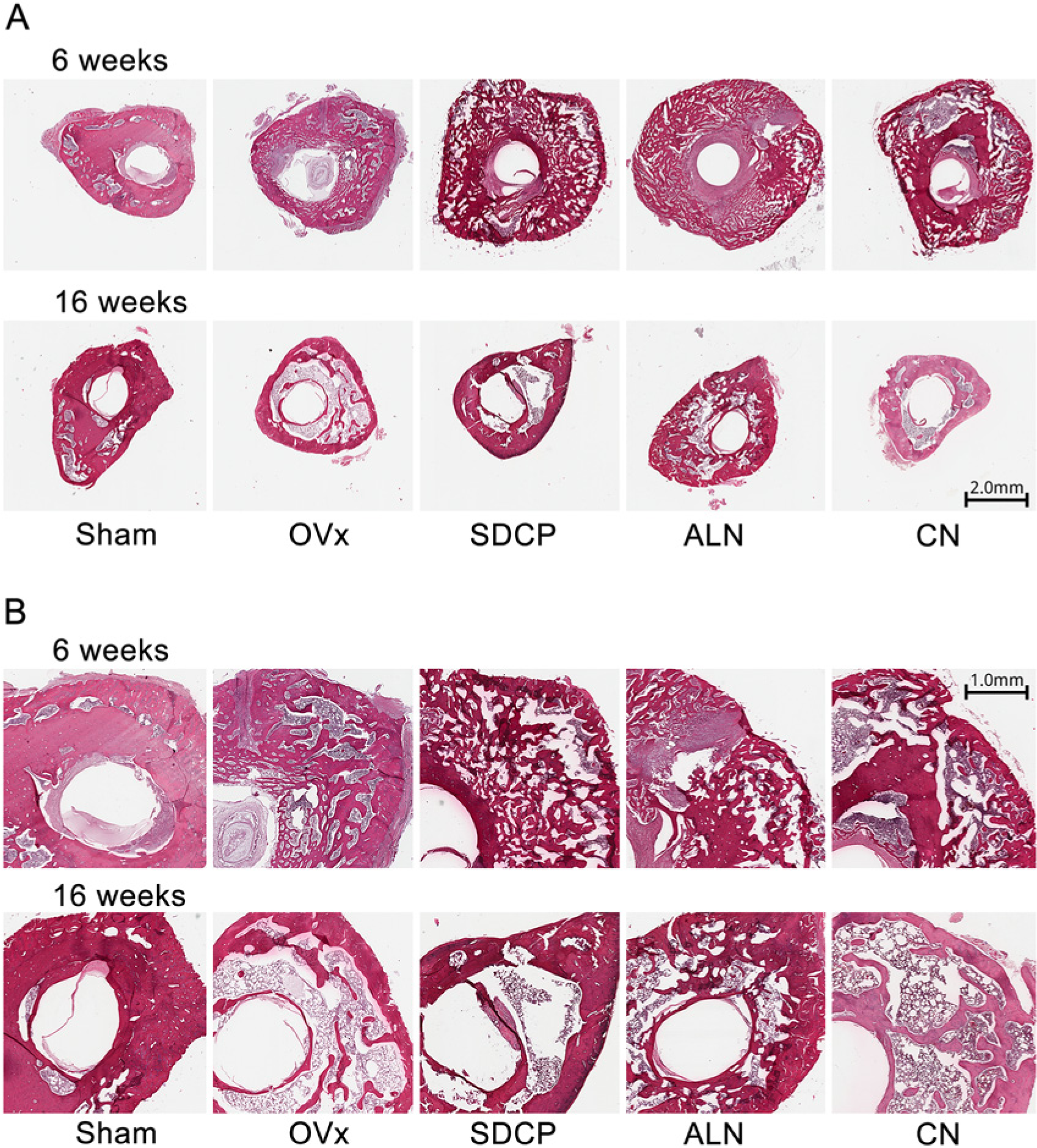
Histological images of femoral longitudinal sections taken at 6 and 16 weeks post-fracture. The SDCP, ALN, and CN treatment groups exhibited a much larger callus than the sham group at 6 weeks after fracture. At 16 weeks, the callus size in the sham and SDCP groups had decreased, with the appearance of a new cortical shell. OVx, ovariectomy; SDCP, sintered dicalcium pyrophosphate; ALN, alendronate, CN, calcitonin. (hematoxylin and eosin; 3A, scale bar = 2.0 mm; 3B, scale bar = 1.0 mm).
pQCT Analysis
The trabecular microarchitectural parameters are presented in Table 1. Our analysis showed that Tb.Th of the contralateral (unfractured) femur was all smaller in the OVx rats than in sham-treated rats (non-ovariectomy group). This result confirms that the ovariectomy procedure was successful. There was no significant difference in trBV between the SDCP group and other groups (OVx, ALN, and CN) both at 6 and 16 weeks. The trTV of the ALN-treated group was significantly higher than sham (at 6 and 16 weeks) and OVx (at 16 weeks) groups. The BV/TV for rats in the SDCP group was significantly lower than that in the OVx group at 16 weeks (p<0.001) but not at 6 weeks. There was no significant difference in Tb.Th between the SDCP group and the other groups (OVx, ALN, and CN) both at 6 and 16 weeks. There was no significant difference in the contralateral Tb.Th between the SDCP group and the other groups (OVx, ALN, and CN) at both time points. Tb.N in the SDCP group was significantly lower than that in the OVx group at 16 weeks (p<0.001) but not at 6 weeks. Tb.N in the SDCP group was significantly lower than that in the ALN group at 6 weeks (p=0.001), but not at 16 weeks. The Tb.Sp for rats in the SDCP group was significantly greater than that in the OVx group at 16 weeks (p=0.001), but not at 6 weeks.
Trabecular Microarchitectural Data of the Five Groups at 6 and 16 Weeks Post-Fracture.
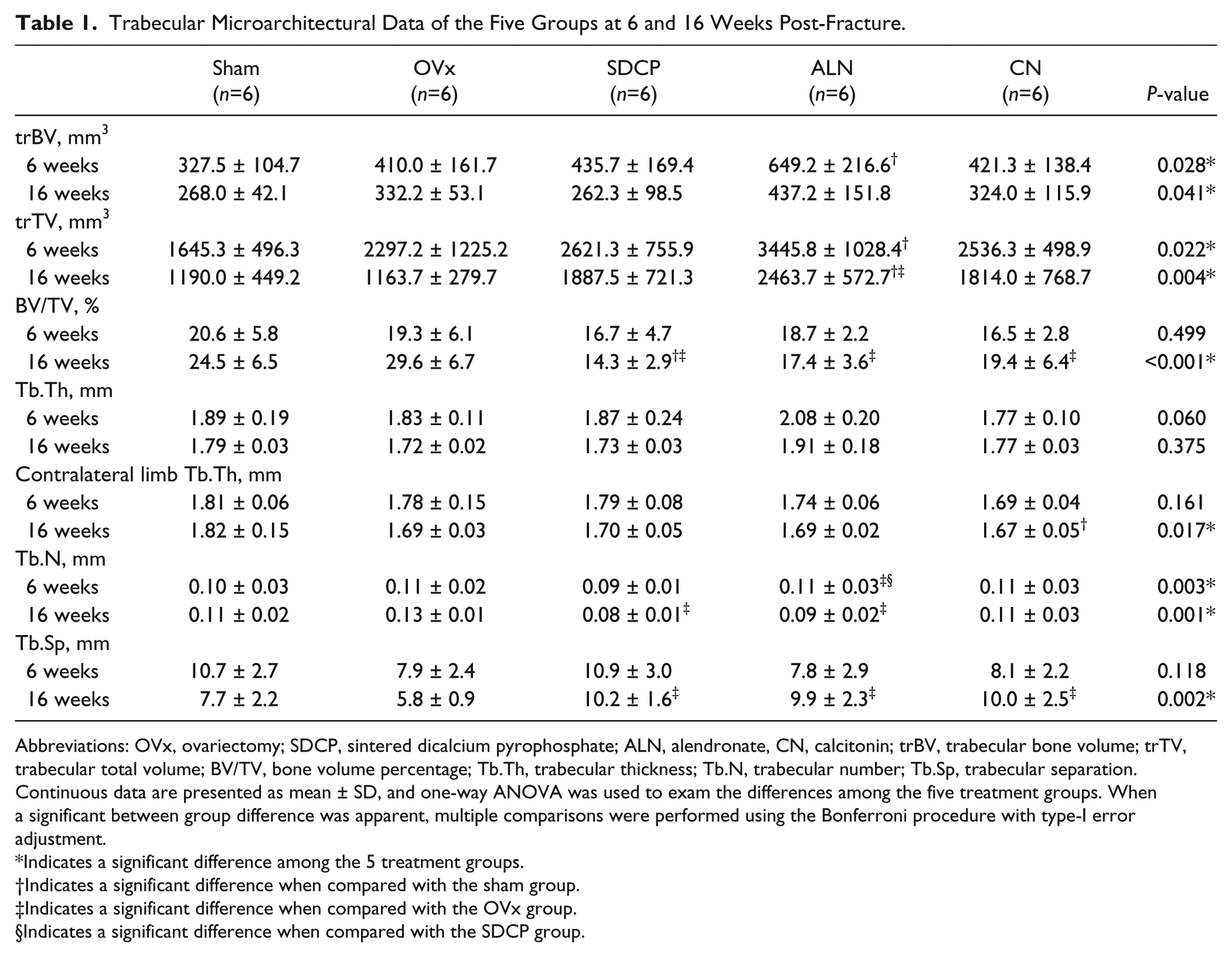
Abbreviations: OVx, ovariectomy; SDCP, sintered dicalcium pyrophosphate; ALN, alendronate, CN, calcitonin; trBV, trabecular bone volume; trTV, trabecular total volume; BV/TV, bone volume percentage; Tb.Th, trabecular thickness; Tb.N, trabecular number; Tb.Sp, trabecular separation.
Continuous data are presented as mean ± SD, and one-way ANOVA was used to exam the differences among the five treatment groups. When a significant between group difference was apparent, multiple comparisons were performed using the Bonferroni procedure with type-I error adjustment.
Indicates a significant difference among the 5 treatment groups.
Indicates a significant difference when compared with the sham group.
Indicates a significant difference when compared with the OVx group.
Indicates a significant difference when compared with the SDCP group.
Mechanical Testing
As compared with measurements at 6 weeks post-fracture, the maximum force, stiffness, ultimate stress, and elastic modulus were all greater at 16 weeks in all experimental groups (Fig. 4).
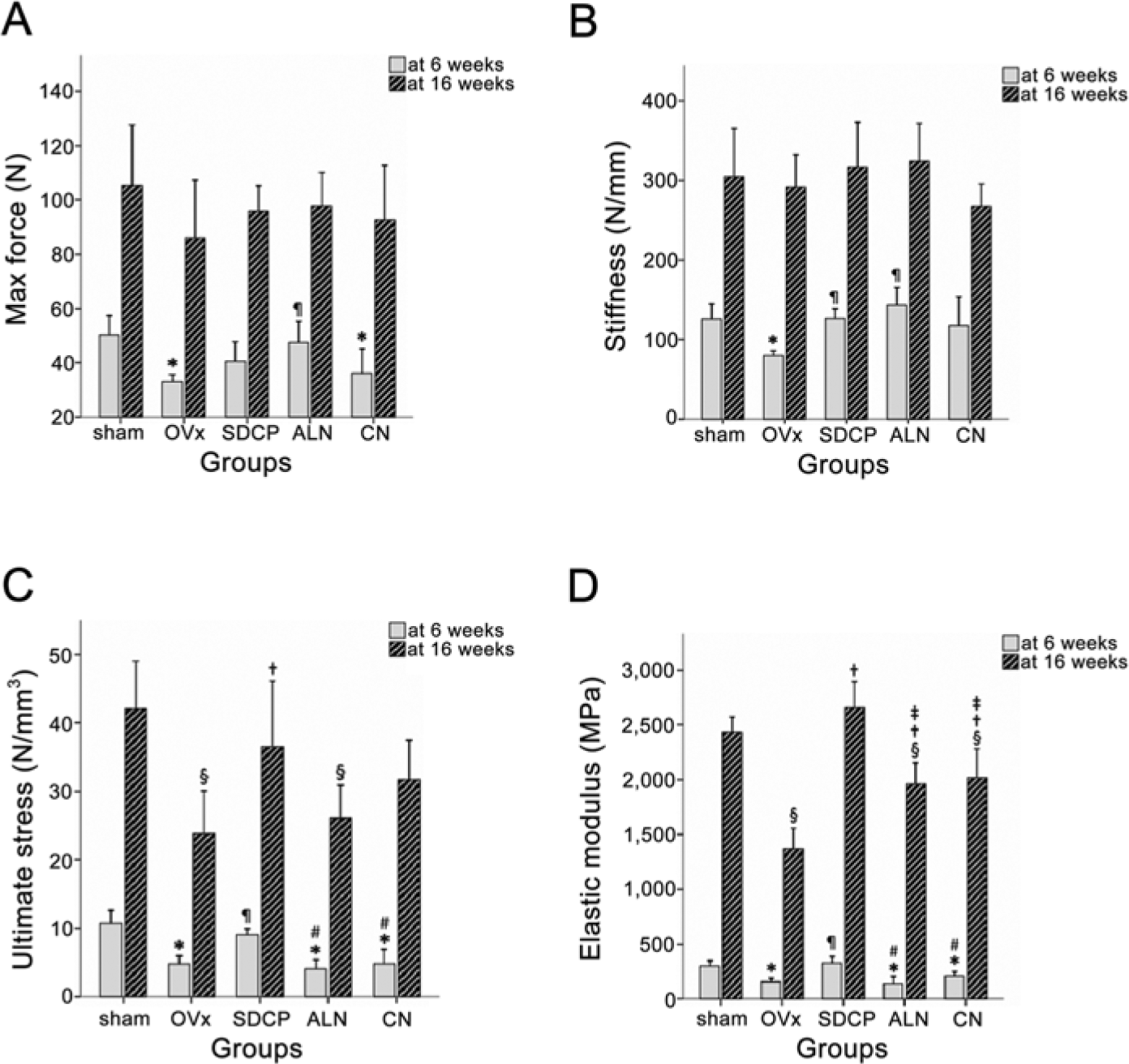
The mechanical testing of the fractured femur. A) Maximum force (N). B) Stiffness (N/mm). C) Ultimate stress (N/mm3). D) Elastic modulus (MPa). The ultimate stress and elastic modulus were significant higher in the sham and SDCP groups than other groups at both 6 weeks and 16 weeks after fracture. * Significant difference as compared with the sham group. ¶ Significant difference as compared with the OVx group. # Significant difference as compared with the SDCP group. § Significant difference as compared with the sham group. † Significant difference as compared with the OVx group. ‡ Significant difference as compared with the SDCP group.
Maximum Force
There was no significant difference in the maximum force between the SDCP group and the sham group. Maximum force in the OVx and CN groups was lower than that in the sham group, whereas rats in the ALN group showed higher maximum force than those in the OVx group at 6 weeks (Fig. 4A).
Stiffness
Stiffness in the SDCP and ALN groups was significantly higher than that in the OVx group (Fig. 4B; p=0.001) at 6 weeks post fracture, but not at 16 weeks.
Ultimate Stress
At 6 weeks, the ultimate stress in the SDCP group was significantly higher than that in the OVx, ALN, and CN groups (Fig. 4C; all p<0.001). At 16 weeks, the ultimate stress in the SDCP group was significantly higher than that in the OVx group (Fig. 4C; p=0.004).
Elastic Modulus
At both 6 and 16 weeks, the elastic modulus in the SDCP group was significantly higher than that in the OVx, ALN, and CN groups. The elastic modulus in the OVx group was significantly lower than that in the ALN and CN groups (Fig. 4C and 4D; all p<0.001).
Histomorphometry
The histomorphometry data are presented in Table 2. The total area (TA) of the fracture for rats in the SDCP group was significantly greater than that measured in the OVx and CN groups at 6 weeks (both p<0.001), and significantly lower than that in the ALN group at 16 weeks (p<0.001). The callus area (Ca.Ar) for rats in the SDCP group was significantly greater than that for rats in the OVx and CN groups at 6 weeks (both p<0.001), and significantly lower than that for rats in the ALN group at 6 and 16 weeks (both p<0.001). The ratio of callus area to total area (Ca.Ar/TA ratio) in the SDCP group was significant lower than that in the ALN group at 16 weeks (p<0.001), but not at 6 weeks. The ratio of lamella bone area to callus area (La.Ar/Ca.Ar ratio) for tissue from rats in the SDCP group was significantly greater than that in the ALN group at 6 and 16 weeks (both p<0.001). The ratio of osteoclasts to callus surface (N.Oc/Ca.S ratio) in the SDCP group was significantly lower than that of the OVx group at 6 and 16 weeks (both p<0.001).
Histomorphometry of the Five Groups at 6 and 16 Weeks Post-Fracture.
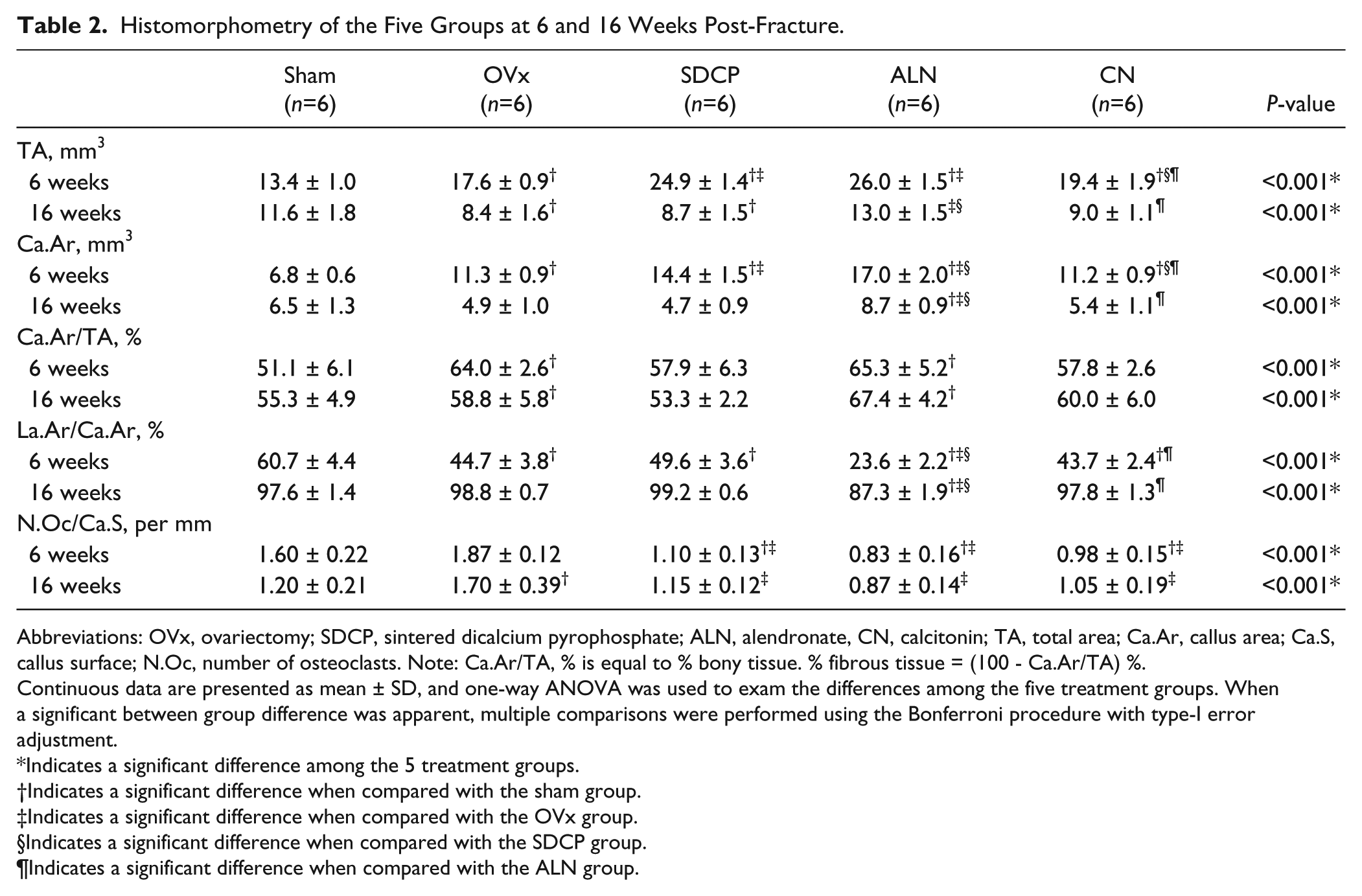
Abbreviations: OVx, ovariectomy; SDCP, sintered dicalcium pyrophosphate; ALN, alendronate, CN, calcitonin; TA, total area; Ca.Ar, callus area; Ca.S, callus surface; N.Oc, number of osteoclasts. Note: Ca.Ar/TA, % is equal to % bony tissue. % fibrous tissue = (100 - Ca.Ar/TA) %.
Continuous data are presented as mean ± SD, and one-way ANOVA was used to exam the differences among the five treatment groups. When a significant between group difference was apparent, multiple comparisons were performed using the Bonferroni procedure with type-I error adjustment.
Indicates a significant difference among the 5 treatment groups.
Indicates a significant difference when compared with the sham group.
Indicates a significant difference when compared with the OVx group.
Indicates a significant difference when compared with the SDCP group.
Indicates a significant difference when compared with the ALN group.
Biochemical Analyses
CTX-1
CTX-1 in the SDCP group was significantly lower than in the CN group at 6 weeks (Fig. 5A; p<0.001). At 16 weeks, CTX-1 in the SDCP, ALN and CN groups was significantly lower than that in the OVx group (Fig. 5A, p<0.001).
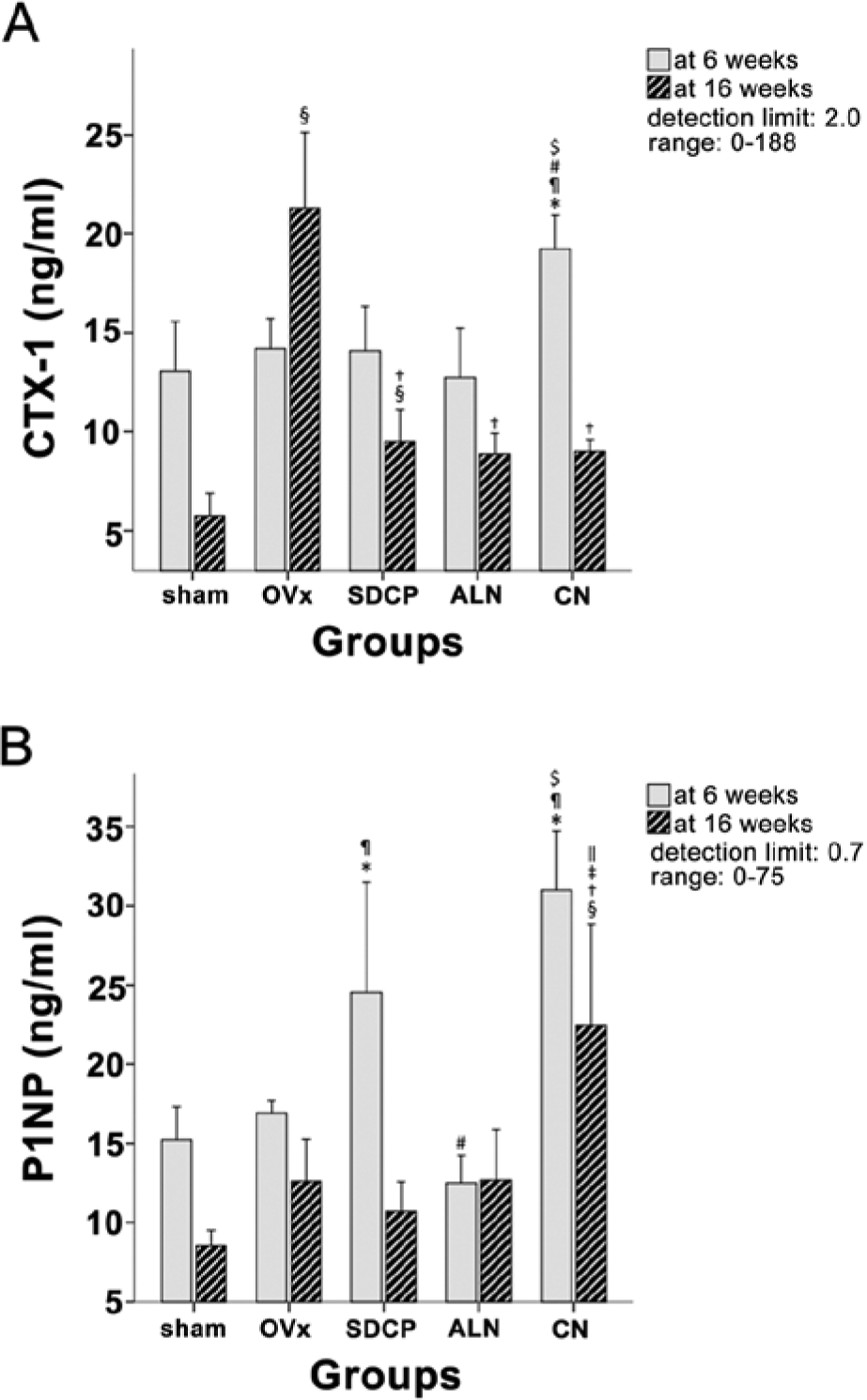
(A) CTX-1 (ng/ml) and (B) P1NP (ng/ml), as determined by EIA. CTX-1 was significantly greater in the CN group than in the other groups at 6 weeks, and in the OVx group at 16 weeks after fracture. Increases in P1NP were observed in the SDCP and CN groups at 6 weeks after fracture. * Significant difference as compared with the sham group. CTX-1, type 1 carboxy-terminal collagen fragments; P1NP, amino-terminal propeptide of type 1 procollagen. ¶ Significant difference as compared with the OVx group. # Significant difference as compared with the SDCP group. $ Significant difference as compared with the ALN group. § Significant difference as compared with the sham group. † Significant difference as compared with the OVx group. ‡ Significant difference as compared with the SDCP group. || Significant difference as compared with the ALN group.
P1NP
At 6 weeks, P1NP was significantly greater in the SDCP group than in the OVx and ALN groups (Fig. 5B; both p<0.002). At 16 weeks, P1NP in the SDCP group was significantly lower than that in the CN group (Fig. 5B; p<0.001).
Discussion
Osteoporotic patients are prone to fractures; therefore, physicians must face the possibility of fractures occurring in these patients while they are receiving a treatment regime for this disorder. Satisfactory healing of an osteoporotic fracture is critically important to functional recovery and quality of life. Some therapies for osteoporosis may affect the processes associated with bone repair. Therefore, it is important to be aware of the effects of drugs used for the treatment of osteoporosis on fracture healing. Oral bisphosphonates are the most widely prescribed class of anti-osteoporotic drugs. Recent reports, however, suggest a link between prolonged bisphosphonate use and atypical low-energy, subtrochanteric fractures (Schindeler et al. 2008). Although some studies have shown that patients prescribed bisphosphonates could attain complete healing of femoral fractures (Gong et al. 2012; Kim et al. 2012), many others show delayed fracture healing after bisphosphonate therapy (Egol et al. 2013; Schneider et al. 2012).
The results of this study show that the healing and remodeling processes of the SDCP group were most similar to that of the sham group, both radiologically and histologically (Figs. 2, 3). The trBV of the SDCP-treated group at 6 weeks was higher than that in the sham and OVx groups, and the larger trabecular bone volume and cross-sectional area indicate that SDCP mildly enhanced callus formation at 6 weeks. The lamellar bone in the callus area and new cortical shell formation in the SDCP-treated group were similar to that of the sham group at 16 weeks after fracture, indicating there was no delayed callus remodeling into lamellar bone. There was no significant difference in BV/TV between the SDCP and sham groups (Table 1). At both 6 and 16 weeks after fracture, ultimate stress and elastic modulus were similar between the SDCP and sham groups, and the mechanical strength in these groups was superior to that in the OVx, ALN, and CN groups (Fig. 4).
Fracture healing is an extremely important biological process for survival, and should return bone strength to its full pre-fracture strength as soon as possible. To this end, a short phase of endochondral external callus formation occurs, which is then followed by a prolonged remodeling period. The structural properties depend on the size and geometry of the fractured bone. The results of this study showed that maximum force and stiffness were not different at 16 weeks among the groups, but stress and elastic modulus were. Maximum force and stiffness were calculated to obtain information about the intrinsic material properties, such as ultimate stress and elastic modulus, by normalizing the cross-sectional moment of inertia. The differences at 16 weeks among the groups suggest that the quality of bone in the callus was different among the groups. This implies that a larger volume and cross-sectional area of callus compensates for a poorer quality of bone in the callus to achieve mechanical recovery of the fractured bone (Li et al. 1999). The results also indicate that SDCP enhances early osteoporotic bone healing without interfering with bone remodeling.
Both bone density and quality are important determinants of bone strength. Bone quality is described by matrix characteristics, such as bone structural characteristics, and is important for the reinforcement of bone strength. It was found that ALN and alfacalcidol (ALF), a prodrug of calcitriol, increased bone strength with distinctive effects on bone mass and quality. ALN formed larger calluses and delayed woven bone remodeling into lamellar bone, whereas ALF induced lamellar bone formation to transform the callus to a native bone pattern via enzymatic cross-linking effects (Saito et al. 2010). The histomorphological data of our study showed similar results for the ALN and CN groups. We also demonstrated that the quality of callus formation in the SDCP treatment group was much improved as compared with these other treatment groups, as shown by a smaller total area (TA), callus area (Ca.Ar), and ratio of callus area to total area (Ca.Ar/TA ratio), but greater ratio of lamella bone area to callus area (La.Ar/Ca.Ar ratio) (Table 2) and comparable or even better mechanical strength (Fig. 4).
Early in the fracture healing process there is rapid production of cartilage and woven bone, which provide initial strength during the healing process. Woven bone is then remodeled to lamellar bone, and a new cortical shell is formed to restore the original structure and mechanical strength (Schindeler et al. 2008). During fracture healing, osteoclastic bone resorption plays an important role in endochondral ossification and remodeling of woven bone to lamellar bone (Marsell and Einhorn 2011; Schindeler et al. 2008). The biology and mechanics of the whole sequence of fracture repair—from inflammation to angiogenesis, cartilage formation, calcification, and remodeling—are of immense importance. Bisphosphonates inhibit osteoclast activity (Larsson and Fazzalari 2014; Murakami et al. 1995), and long-term use inhibits osteoclast differentiation (Larsson and Fazzalari 2014; Li et al. 1999). A previous study showed that a high dose of alendronate induces the formation of a large, strong callus, but delays the structural and material restoration of the callus (Manabe et al. 2009). Calcitonin, on the other hand, mildly suppresses callus remodeling, and has no effect on the natural fracture healing process and material properties of the new bone (Manabe et al. 2009). Our study demonstrated that the SDCP treatment group had a lower ratio of osteoclasts to callus surface (N.Oc/Ca.S ratio), and this ratio is indicative of osteoclast cellular activity (Table 2). This result indicates that SDCP does not impair the remodeling process in late bone fracture healing as does ALN and CN.
Correct fracture healing depends on the synergy between biomechanical, molecular and cellular factors. The efficacy of fracture healing is determined by three ideal conditions: adequate blood supply, good contact between bone fragments, and good stability. In osteoporosis, bone quality adversely affects the tissue structural competence, increasing the risk of complicated fracture healing. The qualitative and quantitative alterations established at the cellular level during osteoporosis explain the progressive deterioration of bone tissue healing ability (Tsujimoto et al. 2011). The current study showed that SDCP resulted in fracture healing of osteoporotic bone that was more consistent with the healing process of normal bone than ALN or CN. The dosage used in the experimental design was the optimal dosage based on prior study (Sun et al. 2002). Further, previous studies have demonstrated that osteoporosis can be established as early as 4 weeks after ovariectomy and that the bone mass of an ovariectomized rat can be increased after 4 weeks of SDCP treatment (Kuo et al. 2012; Sun et al. 2002). The exact mechanism by which SDCP influences callus remodeling could not be establish in the present study, though it seems to be quite different from that of bisphosphonates.
SDCP can be metabolized by endogenous phosphatases (Lin et al. 1995). SDCP both activates osteoblasts and inactivates osteoclasts, but might have less effect on the inhibition of osteoclasts such that it might not delay remodeling as compared to ALN and CN. The callus size in the SDCP group was smaller than that of the ALN and the CN groups at 16 weeks post-fracture. In addition, the SDCP treatment group exhibited a shrunken and solid callus at 16 weeks as compared with that at the 6-week post-fracture time point [Fig. 2, 3]. Osteoblasts originate from mesenchymal stem cells that reside in the bone marrow together with hematopoietic stem cells, and osteoblast numbers might relate to progenitor cell numbers. The two cell types cooperate through direct cell-to-cell interactions, and release of cytokines and growth factors (Benayahu et al. 1992). In our previous study (Sun et al. 1999), we showed that SDCP enhanced the promotion of osteoblast differentiation and the increased the synthesis of prostaglandin E2. Here, we show that the level of the bone formation maker, P1NP, in the SDCP group was significantly greater than that in the OVx and ALN groups at 6 weeks post-fracture (Fig. 5B). This suggests that SDCP led to the production of mature, bone-forming cells, and thus has a positive effect on bone formation during fracture healing. The finding that SDCP increased P1NP at 6 weeks after fracture is consistent with that of our previous study, where we reported that SDCP enhanced the promotion of osteoblast differentiation and the increased synthesis of prostaglandin E2 (Sun et al. 2003a). In another study by Sun et al. (2003b), when bone cells were cultured with SDCP particles smaller than 53 microns, both the osteoblast and osteoclast cell counts decreased significantly. When the SDCP particles were larger than 177 microns, the osteoblast population increased significantly and the osteoclast population decreased significantly. Coulibaly et al. (2010) suggested that serum P1NP has potential to become a blood serum marker for bone healing, and concluded that higher concentrations of P1NP reflected collagen type I synthesis and callus formation in mice at a time point later after fracture. In this study, at 16 weeks after fracture, the level of P1NP in the SDCP group was less than that in the ALN and CN groups (Fig. 5B). This result suggests that fracture healing in the SDCP group was similar to that in the sham group.
Our findings in the OVx groups (Fig. 5) are consistent with those of Yang et al. (2011). The increase in serum bone formation markers in OVx rats may be attributed to attempts by the activated osteoblasts to compensate for the bone loss caused by the estrogen depletion; however, the rate of bone formation was still slower than that of bone resorption resulting in severe osteoporosis in OVx rats. SDCP may either influence bone resorption by osteoclasts or bone formation by osteoblasts. Yang et al. (2011) showed that SDCP decreased bone turnover markers pertaining to both systems, with a significant decrease in the serum bone formation marker and inhibition of serum resorption markers. In this study, the bone resorption marker was significant lower for rats in the SDCP group at 16 weeks than for the other OVx rats, and the bone formation marker was also lower for rats in the SDCP group at 16 weeks than those in the OVx, ALN and CN groups.
This study has some limitations. First, a conventional rat fracture model was used in this study. However, this model is different from common clinical fractures because there are no Haversian systems in rodent bone. The Haversian system and remodeling are important aspects of the fracture healing process. However, the rat fracture model has been highly useful in elucidating the process of fracture repair and the possible effects of pharmacologic agents on healing (Saito et al. 2010). Therefore, this enabled the observation of the typical healing process and the effects of drugs on this process. Second, the rats used were 8 weeks old, and thus had relatively immature skeletons. The effect of bone growth cannot be clearly separated from the effects of SDCP, ALN, and CN in the fracture healing process. However, by the end of the study, the rats were 28 weeks of age. Third, the number of animals in each of the groups was relatively small and the study duration was relatively short. Nonetheless, the current study serves as a proof-of-concept study; the exact mechanisms examined over a longer time period should be the subject of future investigations. A histochemical analysis was not performed, blood samples prior to treatment (baseline) were not obtained, and we did not directly measure cellular activity. Lastly, the treatments were not tested in the absence of OVx, and therefore may not be specific to the treatment of osteoporotic bone healing; i.e., they might work as well or better in normal fracture healing.
In conclusion, as the number of pharmacological treatment options for osteoporosis and the number of patients requiring such treatment is increasing, it is evident that the potential interaction between osteoporotic treatment regimens and fracture healing must be addressed. There is experimental evidence for a beneficial effect of SDCP currently being developed for treatment of osteoporosis. The results of this study show that SDCP can induce the formation of a large fracture callus in the early fracture healing phase, and does not delay woven bone remodeling into lamellar bone at later stages. The healing of osteoporotic bone with SDCP more closely resembles that of normal bone than treatment with ALN or CN. However, these findings must be verified by further experimental and human studies.
Footnotes
Acknowledgements
We thank the Taiwan Mouse Clinic, which is funded by the National Research Program for Biopharmaceuticals (NRPB) at the National Science Council (NSC) of Taiwan, for technical support in this experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by
