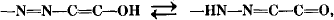Abstract
Raman spectra of a number of azo dye structures have been analyzed. In azonaphthol and azopyrazol dyes, hydroxyazo to keto-hydrazone tautomerizations,
were observed in aqueous basic and acidic media. Observations of azo and ring vibrations, as well as hydrazone (C=N plus C=0) bands, were made. It was noted that the azo-anions (examined at pH 12) convert to the protonated neutral hydrazone (keto) form in aqueous acid (run at pH 2). Infrared spectra were used to confirm the presence of the C=0 group in keto-tautomeric forms.
Get full access to this article
View all access options for this article.