Abstract
Anomalous origin of the left coronary artery from the pulmonary artery (ALCAPA), or Bland-White-Garland syndrome, is a rare congenital malformation described in children and adults. In this condition, the left coronary artery, which normally originates from the left coronary sinus in the aorta, instead originates from the pulmonary trunk, which results in retrograde flow of blood away from the myocardium into the lower-pressure pulmonary artery. Myocardial hypoxic-ischemic injury results in cardiac dysfunction, failure, and eventually in patient death if not surgically repaired. This report describes gross and microscopic findings in 4 beef calves with ALCAPA. All the calves had a history of being found dead with few or no premonitory signs, 2 shortly after sudden strenuous exercise. Gross necropsy lesions suggestive of heart failure included cardiomegaly with atrial and ventricular dilation and/or ventricular hypertrophy, and hepatomegaly. Dissection of each heart revealed the origin of the left coronary artery arising in the pulmonary trunk above the anterior cusp of the pulmonic valve. No other cardiac anomalies were identified. Histopathologic changes in the heart included cardiomyocyte degeneration; mineralization; and fiber loss, with replacement by fibrous connective tissue, predominantly in the left ventricular papillary muscle and the interventricular septum. Changes observed in the liver and lungs, including hepatomegaly, sinusoidal congestion, centrilobular fibrosis, and pulmonary congestion, edema, and intra-alveolar pigment-laden macrophages were consistent with heart failure.
Introduction
Anomalous origin of the left coronary artery from the pulmonary artery (ALCAPA), or Bland-White-Garland syndrome, is a rare congenital malformation described in children and adults, with an incidence of about 1 in 300,000 births. 7 Normally, the left coronary artery arises from the left coronary sinus in the aorta, courses under the left auricle, then branches into the descending (paraconal) and circumflex branches. The left coronary artery provides two thirds of the cardiac blood supply, including that of the entire left atrium and ventricle. The remainder of the cardiac blood supply is provided by the right coronary artery, which originates from the right coronary sinus in the aorta, courses under the right auricle, then continues into the right coronary groove. 9 In ALCAPA, the left coronary artery originates from the pulmonary trunk, usually just above the pulmonic valve cusps. The misplaced left coronary artery contains well-oxygenated blood (measured in human studies with oxygen saturation studies) because of its normally present but limited intra- and extramyocardial anastomoses with the right coronary artery; however, in ALCAPA, blood does not perfuse the myocardium normally (antegrade flow) but instead is shunted away from the higher-pressure myocardium and flows retrograde into the lower-pressure pulmonary artery (coronary steal phenomenon). 6, 19, 20 This leads to myocardial hypoxic-ischemic injury and results in compromised conductivity, decreased contractility, and, eventually, death. Patients usually present with clinical signs of congestive heart failure secondary to myocardial infarction and ischemic cardiomyopathy. Infants with this condition present with dyspnea and difficulty nursing, and patients that survive into adulthood may present with exercise intolerance, dyspnea, limb edema, and/or chest pain. 6 Clinical signs are exacerbated by physical activity. This anomaly is diagnosed with echocardiography or coronary angiography, and, if left untreated, the mortality rate is greater than 90% in the first year of life. 13 Patient survival into adulthood is associated with the development of increased numbers of anastomoses between the right and left coronary arteries, with the right coronary artery having or developing a greater percentage of cardiac perfusion via an increase in size and number of branches.
A study by van Nie 16 in 1966 reported cardiac anomalies in 88 of 50,742 bovine hearts examined at routine meat inspections over 6 years, for an prevalence of 0.17%. The predominant abnormality was ventricular septal defect, alone or in combination with other malformations. 16 In veterinary literature, single case reports of ALCAPA have been described in a dog 14 and 2 separate cases in calves. 11, 12, 15 One additional case, in an adult cow, was suspected, but the basal portion of the heart was missing so a definitive diagnosis could not be made. 2 Anomalous origin of the right coronary artery from the pulmonary trunk (ARCAPA) appears to have a higher incidence than ALCAPA in cattle, with 11 total cases reported, but, because the right coronary artery supplies only a portion of the right side of the heart and none of the left side, the effects on cardiac function are not as severe nor have such dire consequences for survival. 5, 17, 18
This report describes gross and microscopic findings in 4 beef calves presented to the University of Kentucky Livestock Disease Diagnostic Center (LDDC) in April 2006 (one case), May 2007 (two cases), and February 2008 (one case) and diagnosed at necropsy with ALCAPA. All calves had a history of being found dead, with few or no premonitory signs, two shortly after sudden strenuous exercise. Gross necropsy lesions were suggestive of heart failure and included cardiomegaly, with atrial and ventricular dilatation and/or ventricular hypertrophy, and hepatomegaly. Sections of heart from the right and left atria, right and left ventricular papillary muscles, and interventricular septum, along with cerebrum, cerebellum, brainstem, lung, liver, spleen, kidney, thymus, rumen, reticulum, omasum, abomasum, small intestine, and colon, were collected and fixed in 10% neutral buffered formalin. Paraffin-embedded tissues were sectioned, mounted, routinely stained with HE and examined under light microscopy. Masson's trichrome was used to confirm fibrosis in the liver.
Calf No. 1
A 9-week-old male mixed-breed beef calf was submitted in good postmortem preservation and good body condition. The calf had been chased by dogs and within hours developed stiffness and difficulty breathing. Treatment with antibiotics resulted in some improvement, but the calf subsequently died the day after treatment was instituted.
At necropsy, the liver was moderately enlarged, with rounded edges and diffuse mottling. The left and right cardiac ventricles were moderately dilated with hypertrophy of the free wall musculature to two or three times normal thickness, with the left ventricle more severely affected than the right. The origin of the right coronary artery was located in the right coronary sinus in the aorta (normal location). The left coronary artery measured 1 cm in diameter and was located above the middle cusp of the right pulmonic valve. The course of the artery was otherwise normal. The endocardium was moderately thickened and opaque, especially in the left ventricle. No discernible evidence of external trauma was detected. No other significant gross lesions were observed. Routine aerobic culture of the lung, kidney, and liver resulted in no growth, and a culture of the intestines yielded nonpathogenic bacteria. Fluorescent antibody testing was negative for infectious bovine rhinotracheitis (IBR), bovine viral diarrhea (BVD), bovine respiratory syncytial virus (BRSV), and parainfluenza-3 (PI-3).
Microscopic findings in the heart consisted of multifocal to coalescing areas of myofiber loss and replacement with fibrous connective tissue, with regions of myofiber degeneration characterized by fiber pallor, loss of cross striations, variation in fiber size, and anisokaryosis. The endocardium was moderately thickened by fibrosis, and vasculature throughout the myocardium, but most evident in fibrotic areas, was congested and dilated. Lesions were most prominent or severe in sections from the left ventricular papillary muscle, mild to moderate in the interventricular septum and left atrium, and minimal to mild in the right atrium and right ventricular papillary muscle. Changes in the lung included mild perivascular and peribronchiolar fibrosis, mild fibrous thickening of interlobular septa, and small numbers of intra-alveolar foamy macrophages, many containing intracytoplasmic globular to granular golden-brown pigment (heart failure cells). In the liver, multiple centrilobular regions were thickened by mild, occasionally bridging, fibrosis, and there was mild sinusoidal and venous congestion. There were no significant microscopic lesions in other tissues examined.
Calf No. 2
A 5-week-old female Angus calf presented in good body condition and good postmortem preservation. The herd had been moved to a new field the day before, and this calf appeared sick the next morning and was separated from the rest of the cattle. The calf was found dead at midday. On gross examination, the thoracic and abdominal cavities contained a moderate amount of serosanguineous fluid. The surface of the liver was dark gray, with an irregular capsular surface. The heart was globally enlarged with a rounded profile. The right coronary artery originated from the right coronary sinus in the aorta (normal location) (Fig. 1). The left coronary artery was 1 cm in diameter and originated approximately 1 cm above the middle cusp of the pulmonic valve (Fig. 2). The left coronary artery otherwise followed a normal course.
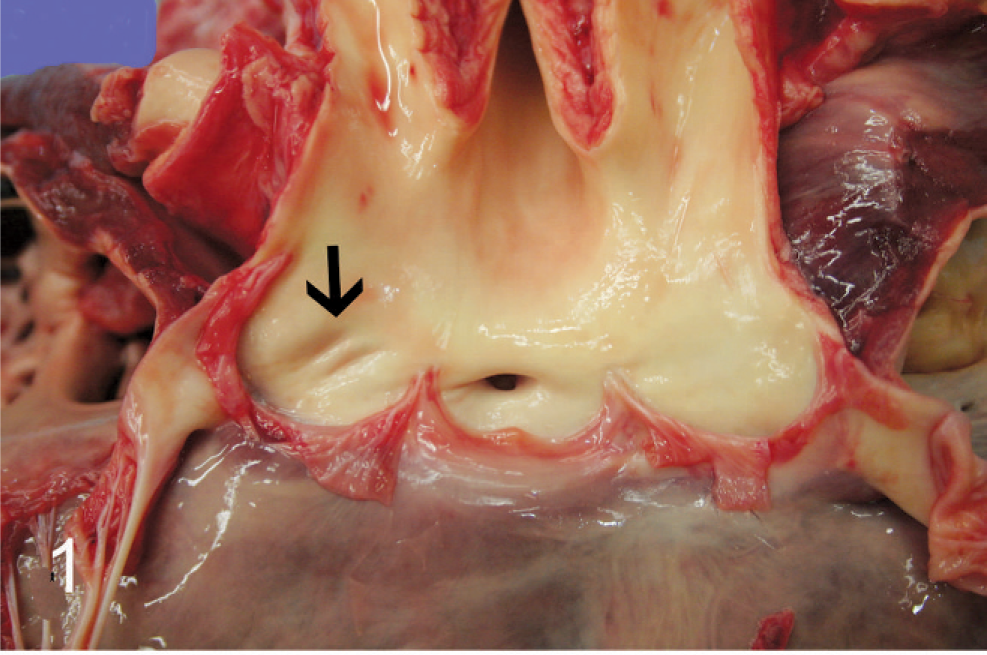
Aorta and left ventricle; calf No. 2. The opening for the right coronary artery is present in the right coronary sinus; however, the opening for the left coronary artery is absent and there is a slight depression in the left coronary sinus where normally the opening would be (arrow).
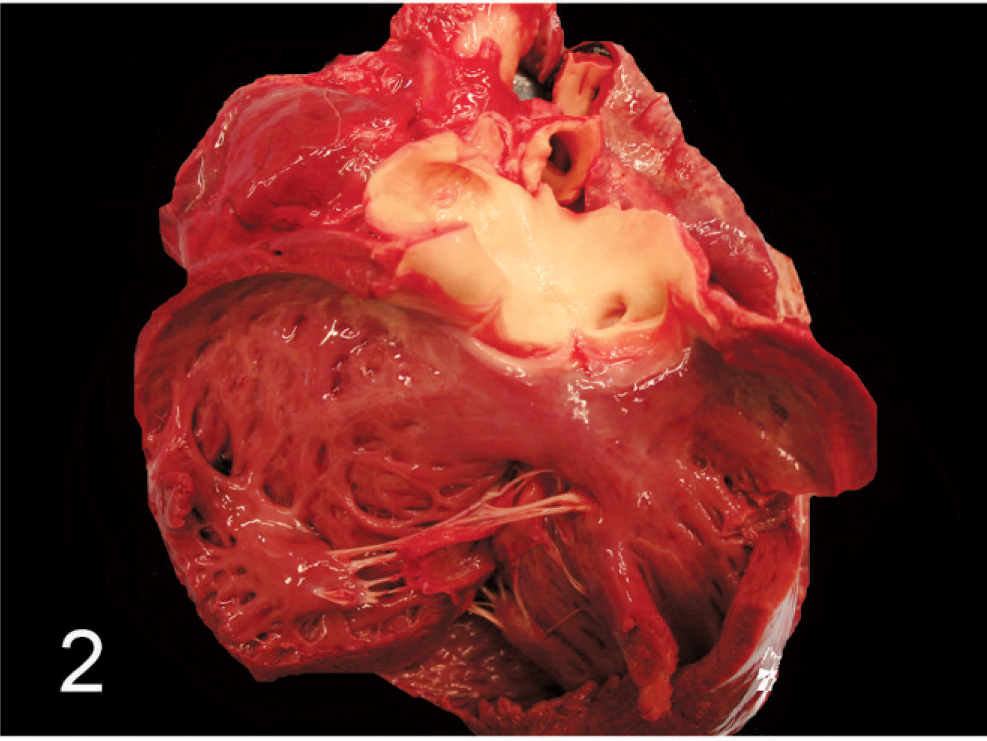
Pulmonary artery and right ventricle; calf No. 2. The opening for the left coronary artery arises above the pulmonic valve cusp within the pulmonary trunk.
No significant bacteria were isolated in routine culture of lungs, liver, and intestines. Fluorescent antibody testing was negative for IBR, BVD, BRSV, and PI-3. Microscopic changes in the heart were similar to those described in calf No. 1, with moderate multifocal myocardial degeneration and fibrosis, increased prominence of vasculature (predominantly in fibrotic areas), multifocal variably sized areas of myofiber mineralization, hypertrophy of medium and large vessel walls, mild epicardial fibrosis, and minimal endocardial fibrosis (Fig. 3. Alterations were most evident in sections from the left ventricular papillary muscle and interventricular septum (fibrosis and mineralization), and the left atrium (vascular-wall thickening). Changes in the liver included mild centrilobular fibrosis, mild hepatocellular cytoplasmic vacuolization, and moderate sinusoidal and central venous congestion. Within the lung, alveolar spaces contained low-to-moderate numbers of alveolar macrophages with intracytoplasmic golden-brown pigment, and rare neutrophils. No additional significant microscopic abnormalities were observed in other tissues.
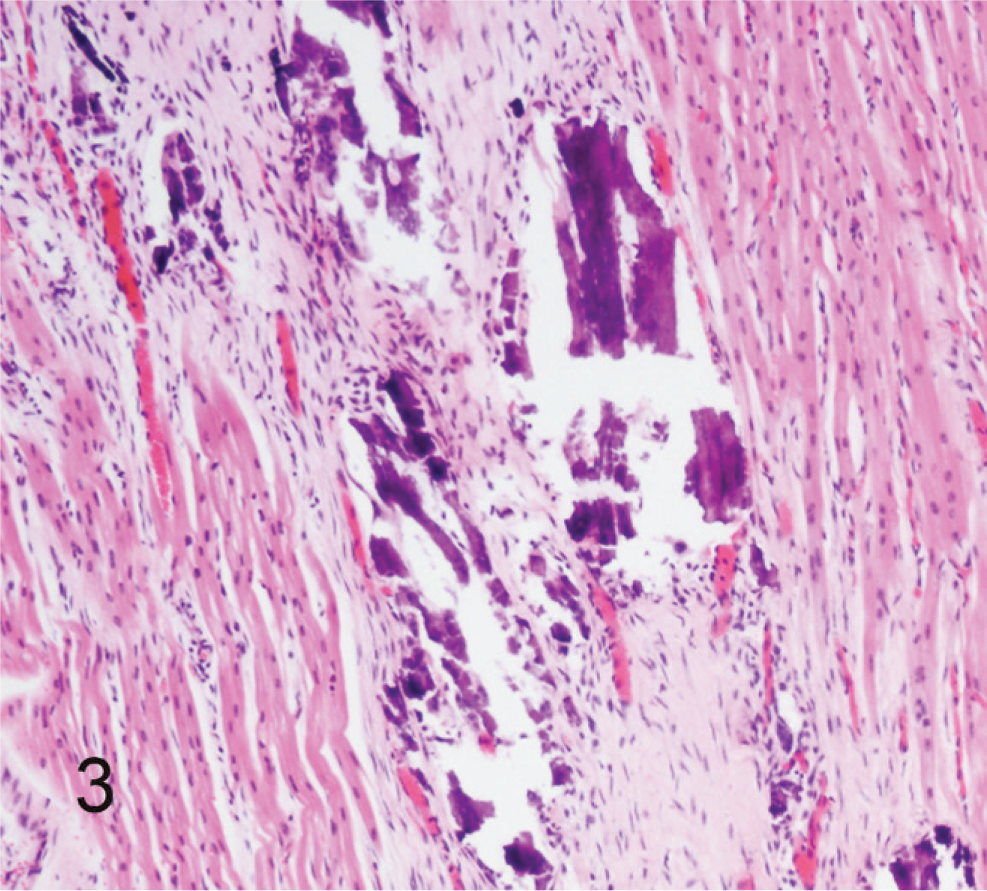
Left ventricular papillary muscle; calf No. 2. The myocardium contains extensive areas of mineralization and fibrous replacement of myocytes. HE.
Calf No. 3
An 8-week-old male mixed-breed beef calf in good body condition and good postmortem preservation was submitted for necropsy. The calf was found dead in the field, with no previous signs of illness. At necropsy, the liver was enlarged with rounded edges and irregular capsular surface. The heart was markedly enlarged with a globoid profile due to right and left ventricular and atrial enlargement. Upon opening the heart, the left coronary artery was identified as arising from approximately 1 cm above the middle cusp of the pulmonary valve but otherwise followed a normal course. The right coronary artery originated from the right coronary sinus (normal). Both atria were dilated, and the left ventricle was also dilated. The right ventricular free wall was moderately thickened, with hypertrophy of the moderator band. Bilateral ventral lung fields were firm and dark purple. The lateral ventricles of the cerebrum were moderately dilated. Aerobic culture of the lung resulted in growth of Pasteurella multocida. No significant bacteria were isolated from a culture of the liver or intestines. Fluorescent antibody testing was negative for IBR, BVD, BRSV, and PI-3.
Microscopic changes in the heart were similar to those previously described in calf No. 1 and calf No. 2, with moderate myocardial and epicardial fibrosis; mild myofiber degeneration, with scant mineralization; and increased prominence of vasculature, predominantly in fibrotic areas. Changes were most severe in sections from the left ventricular papillary muscle and the interventricular septum. The proliferative fibrous connective tissue of the epicardium contained small numbers of neutrophils, macrophages, and lymphocytes. In the lung, numerous bronchioles and alveoli contained abundant neutrophils and macrophages mixed with acidophilic amorphous to fibrillar proteinaceous material. Some macrophages contained intracytoplasmic golden-brown pigment. Liver changes were characterized by marked centrilobular hepatocellular necrosis, moderate midzonal and centrilobular sinusoidal congestion, mild centrilobular fibrosis, and mild diffuse hepatocellular cytoplasmic vacuolization. No significant microscopic changes were observed in other tissues.
Calf No. 4
A 6-week-old female mixed-breed beef calf presented for necropsy in good postmortem preservation, with a body condition score of 4/9. The calf was found dead in the field and was previously observed eating and moving around normally. On gross examination, the liver was severely enlarged and congested, with blue-purple parenchyma and rounded edges. The vena cava was dilated. The lungs were mottled red-tan and mildly firm. The left coronary artery was observed to originate from the pulmonary artery, just above the anterior cusp of the pulmonic valve but otherwise followed a normal course. The right coronary artery was in the normal location in the aorta. The right ventricle had a moderately dilated lumen and moderate thickening of the free wall and interventricular septum. No further significant gross findings were observed.
Aerobic culture of the lung and intestines resulted in growth of Escherichia coli. No significant bacteria were isolated from a culture of the liver or kidney. Microscopic changes in the heart were similar to those previously described in other calves, with moderate myocardial, epicardial, and endocardial fibrosis; mild-to-moderate myofiber degeneration, with scattered mineralized foci; and dilated prominent vasculature, predominantly in fibrotic areas. Changes were most severe in sections from the left ventricular papillary muscle and the interventricular septum. In the lung, vasculature was dilated and congested, with numerous small and medium vessels encircled by mild fibrosis. The interstitium was mildly expanded by spindle cells and low numbers of macrophages. Alveolar lumens contained small numbers of macrophages with intracytoplasmic golden-brown pigment. Liver changes were characterized by moderate-to-severe diffuse sinusoidal congestion and minimal centrilobular fibrosis. No significant microscopic lesions were observed in the remainder of the tissues.
Discussion
Numerous cases of abnormal origin of coronary arteries have been published in human medicine and fewer in veterinary literature. Variations include anomalous origination of the right or left coronary artery from a location other than the respective coronary sinus but still originating in the aorta; origination of both arteries from a single common sinus within the aorta; or origination of the right coronary artery from the pulmonary artery (ARCAPA). 1, 3, 15 These cases are generally discovered as incidental findings at an autopsy or necropsy, or in human patients who underwent an angiography. Abnormal origination of the left coronary artery at a location in the aorta other than the left coronary sinus is not associated with clinical signs of heart disease, unless the course of the artery travels between the pulmonary artery and the aorta, causing constriction of the artery between the two great vessels. 4, 8 Origination of the right coronary artery from the pulmonary artery results in compromised perfusion to a portion of the right atrium and ventricle, but, because this portion of the heart also receives blood from the left coronary artery, and the workload of the right side of the heart is not as great as the left side, these cases may have no or only mild clinical manifestations of cardiac disease. In veterinary medicine, abnormal origination of the left coronary artery from the pulmonary artery has rarely been reported, and only as single case reports in one dog and two calves. 11, 12, 14, 15
During cardiogenesis, coronary arteries form from a coronary capillary plexus that contacts and penetrates the aorta, then differentiates into the mature arterial system. 3 The left coronary artery is the predominant source of blood for the left atrial and left ventricular myocardium. Anastomoses between the right and left coronary arteries in a normal heart are minimal but may increase in response to myocardial hypoxia. 6, 13 Wesselhoeft et al. 19 reported right coronary artery to left coronary artery anastomoses are present, even when not grossly visible, based on postmortem perfusion studies in humans. Increased diameter and prominence of intramyocardial vessels microscopically observed in the 3 calves may represent anastomoses forming between the right and left coronary arteries. Without angiography or perfusion studies of normal versus affected hearts, this is only speculation. Other causes may include proliferation of existing vessels in an attempt to better perfuse oxygen-deprived myocardium or increased tortuosity of vessels in response to increased resistance that results from myocardial fibrosis (similar to the tortuous vasculature that develops with hepatic shunts or vascular occlusion).
Microscopic findings in the hearts from the 4 calves were consistent with hypoxic-ischemic injury and were characterized by myofiber degeneration, mineralization, loss, and replacement by fibrous connective tissue. These changes were most severe in the left ventricular papillary muscle and the interventricular septum, which are the areas of the heart with the greatest workload and would suffer most from a compromised blood supply. Changes observed in the liver and lungs, including hepatomegaly, sinusoidal congestion, centrilobular fibrosis, and pulmonary congestion, edema, and intra-alveolar pigment-laden macrophages were consistent with heart failure.
ALCAPA has grave consequences for heart function and ultimately survival. Although the anomaly is present at birth, the effects do not develop for weeks or months afterward. Before birth, pulmonary resistance is high, hence pulmonary arterial pressure is high. Blood from the anomalous left coronary artery is better able to perfuse the myocardium, because the difference in pressure gradient between the myocardium and pulmonary artery is not as great as after birth when pulmonary resistance and hence pressure in the pulmonary artery decreases. Also, before and shortly after birth (days to 2 weeks), fetal blood has a greater concentration of hemoglobin than in juvenile or adult animals, and this hemoglobin has a higher oxygen carrying capacity, so effects of compromised perfusion are decreased. As animals age, the concentration of hemoglobin, as well as the oxygen-carrying capacity decreases, so abnormalities in perfusion have greater deleterious effects. As an animal grows, cardiac output demands increase. These factors, along with limited collateral circulation normally present between right and left coronary arteries, are the primary reasons why clinical signs are not apparent until after a few weeks after birth. Differences in activity level and the amount of perfusion provided by the right coronary artery are also factors in the age and rate at which clinical signs develop. The age range of the calves presented here was 5 to 9 weeks old, although the myocardial lesions ranged from subacute (loss of cross striations, variability in fiber size, fiber dissolution) to chronic (fibrosis) within the same heart, indicating an ongoing process. Lesions were consistently most severe in histologic sections from the left ventricular papillary muscle and interventricular septum, the areas of the heart with the greatest workload. Two of the 3 calves were reported as clinically normal, then they died after sudden induced exercise. This most likely caused acute decompensation of the damaged heart, which was unable to compensate for the sudden demand in increased cardiac output. No information was available on the third calf, and a history on the fourth calf did not include information on exercise other than the calf had been observed walking around normally before death.
Damage to myofibers and replacement by fibrous connective tissue may result in decreased conductivity of electrical impulses through the myocardium, which impairs contractility and hence cardiac output. Papillary muscle fibrosis or fibrosis of the mitral valve ring may also result in compromised mitral valve function. 19 Over time, because the ventricle is unable to empty completely and blood leaks back into the left atrium, a volume overload of the left ventricle occurs, resulting in ventricular dilatation and hypertrophy and increased resistance to blood entering the left atrium from the pulmonary vein. Pulmonary vascular hypertension ensues, increasing the right ventricular workload. Right ventricular dilatation and hypertrophy result, and eventually hepatic congestion and centrilobular fibrosis develop. All 4 calves reported here had gross and microscopic changes in the heart, liver, and lungs consistent with chronic heart failure.
Vitamin E and selenium deficiency is a common cause of myocardial necrosis in young calves. Necrosis and mineralization are usually grossly observed as distinct chalky or pale streaks or patches in the myocardium, predominantly in the left ventricle and interventricular septum. Microscopic changes are similar to those observed in calves with ALCAPA. Soil in the southeastern United States is selenium deficient, so calves raised on Kentucky-grown forages are at higher risk of developing vitamin E and/or selenium deficiency than calves that consume forages grown in the Midwest. Most cows and calves are supplemented with selenized trace mineral mixes or with intramuscular injection. Vitamin E and selenium concentrations were not measured in these calves. Typically, at this laboratory, chemical analysis of the liver for selenium concentration is chosen if gross and microscopic evidence of heart disease is observed with no gross evidence of a congenital or developmental cardiac anomaly.
Calf No. 3 had concurrent bacterial pneumonia not related to its cardiac anomaly, and moderate hydrocephalus, another congenital defect. Animals with a congenital anomaly in one organ system often have concurrent anomalies in other systems, so this finding is not unexpected. The breeding of these calves is unknown; a common ancestor with a genetic abnormality that causes coronary artery defects is unlikely but cannot be ruled out. Although unknown in animals, it is estimated that 8% of cardiac malformations in people are because of genetic factors, 2% are related to environmental agents (viruses, teratogens, etc.), but the majority are because of environmental-genetic interactions. 10
From January 2006 through December 2007, 1549 calves aged 3 months or younger (including fetuses) were submitted for necropsy to the University of Kentucky LDDC. Of these cases, 23 were diagnosed with congenital cardiac malformations, a prevalence of approximately 1.5%. This is higher than the prevalence of congenital cardiac abnormalities reported by van Nie 16 in an abattoir study (0.17%). Differences may be because of population sampling differences; cattle examined in the van Nie study were presented for slaughter, not necropsy, and calves with severe cardiac abnormalities do not usually make it to slaughter age. 16 The most common malformation reported at the LDDC was ventricular septal defect (VSD) (9/23 [39%]), alone or in conjunction with other malformations, such as patent ductus arteriosus. This is in agreement with the van Nie study that reported VSD as the most common congenital cardiac abnormality. 16 The second most common congenital defect reported at the LDDC was atrial septal defect (5/23 [22%]). All calves except one were beef calves (pure and crossbred), but this is because dairy herds are uncommon in eastern Kentucky, so submission of dairy calves is limited. The diagnosis of 4 calves with ALCAPA in 2 years' time is unusual, especially because previously only 2 single reports in calves were published. These cases represented approximately 2.5% of calf necropsy submissions (4/1549) and accounted for 17% of congenital cardiac abnormalities diagnosed (4/23).
In conclusion, ALCAPA should be included as a differential diagnosis of sudden death in young animals with gross evidence of heart failure.
