Abstract
Simmental thrombopathia is an inherited platelet disorder that closely resembles the platelet disorders described in Basset Hounds and Eskimo Spitz dogs. Recently, two different mutations in the gene encoding calcium diacylglycerol guanine nucleotide exchange factor I (CalDAG-GEFI) were described to be associated with the Basset Hound and Spitz thrombopathia disorders, and a third distinct mutation was identified in CalDAG-GEFI in thrombopathic Landseers of European Continental Type. The gene encoding CalDAG-GEFI was sequenced using DNA obtained from normal cattle and from a thrombopathic calf studied in Canada. The affected calf was found to have a nucleotide change (c.701 T°C), which would result in the substitution of a proline for a leucine within structurally conserved region two (SCR2) of the catalytic domain of the protein. This change is likely responsible for the thrombopathic phenotype observed in Simmental cattle and underscores the critical nature of this signal transduction protein in platelets.
An inherited intrinsic platelet disorder was first described in Simmental cattle in 1980 and was subsequently described and studied by several investigators in Canada and in the United States between 1980 and 2000. 9, 10, 12, 13, 15– 18 Affected cattle experienced mild to severe bleeding episodes, including epistaxis, gingival bleeding, and hematuria. Bleeding became severe in situations of trauma or following surgical procedures. The platelet disorder closely resembles the platelet disorders described in Basset Hounds and Eskimo Spitz dogs at the biochemical and functional levels. 4, 5, 11 The disorder in both species is characterized by impaired platelet aggregation responses to adenosine diphosphate and collagen and a delayed but full response to thrombin. Thrombopathic platelets release serotonin normally in response to thrombin and support normal clot retraction. Platelet membrane concentrations of glycoproteins IIb and IIIa (integrin alphaIIb-beta 3) are normal, as is platelet morphology at the electron microscopic level. Recently, mutations in the gene that encodes calcium diacylglycerol guanine nucleotide exchange factor I (CalDAG-GEFI), were identified and associated with thrombopathias in Basset Hounds, Spitz dogs, and Landseers of European Continental Type (ECT). 3 All of the mutations were located in areas of the gene encoding for structurally conserved regions (SCRs) of the catalytic domain. CalDAG-GEFI, also known as Ras guanyl-releasing protein 2 (RasGRP2), is a critical signal transduction protein that functions as a guanine nucleotide exchange factor (GEF) in platelets, leading to the activation of the guanosine triphosphate-binding protein (GTPase) Rap1b. 7 CalDAG-GEFI contains four major domain structures, including the Ras exchanger motif (REM), a catalytic domain, two EF hand domains for interaction with calcium, and a C1 domain for interaction with diacylglycerol (DAG) and phorbol esters. 8 The catalytic domain of CalDAG-GEFI contains five structurally conserved regions (SCRs) that are critically important for interaction of GEF with Rap1b. The SCRs within CalDAG-GEFI are not only conserved across species lines but are also highly conserved across multiple types of GEFs in different cell types, thus emphasizing the importance of maintaining a particular structure for proper function. Guanine nucleotide exchange factors facilitate the exchange of GTP for GDP in their target GTPases. GTP-bound GTPases in turn become activated and engage their effector proteins. 2 Rap1b is present in high concentrations in platelets and is thought to modulate the affinity and avidity of integrin alphaIIb-beta 3, leading to fibrinogen binding and platelet aggregation. 1 Most of the platelet agonists that ultimately activate Rap1b do so either directly or indirectly through stimulation of Gi-coupled receptors, leading to inhibition of adenylate cyclase, lowering of cyclic adenosine monophosphate (cAMP), and generation of second messengers, including calcium and DAG. Calcium and DAG bind to the EF hand and C1 domains of CalDAG-GEFI, resulting in activation of the GEF, which then mediates the activation of Rap1b. The cycle is completed when GTPase-activating proteins (GAPs) catalyze the conversion of Rap1b-GTP to inactive Rap1b-GDP forms.
The platelet disorders described in dogs and CalDAG-GEFI knockout mice are strikingly similar to the platelet disorder described in Simmental cattle. The purpose of this study was to evaluate the gene encoding CalDAG-GEFI in a thrombopathic Simmental calf and compare the DNA sequence obtained to DNA sequences obtained in its full siblings, as well as other cattle without a bleeding diathesis due to thrombopathia. Particular attention would be paid to areas of the gene encoding for SCRs of the catalytic domain.
Genomic DNA was harvested from ethylenediaminetetraacetic acid (EDTA)-anticoagulated blood obtained from normal cattle using the QIAamp DNA Blood Mini Kit (Qiagen Inc., Valencia, CA). Primers were designed within introns flanking coding regions of the gene to assure assessment of all exon-intron splice sites. Primers were designed using sequence obtained from the bovine genome available on GenBank. The correct area of the bovine genome was located by comparing the human CalDAG-GEFI cDNA sequence available on GenBank (gi:24797102). The coding region sequence information obtained in this study for bovine CalDAG-GEFI was submitted to GenBank (accession no. EF633475). DNA was isolated from blood obtained from 20 cattle of varying breeds, 18 Simmental cattle, and 2 Simmental-crosses not related to the thrombopathic calf. The Simmental-cross cattle included one calf with von Willebrand's disease (vWD; diagnosis was based on the presence of a bleeding diathesis combined with normal coagulation screening assays, normal platelet numbers, and a von Willebrand factor antigen concentration = 8%; analysis performed by the Animal Health Diagnostic Center, College of Veterinary Medicine, Cornell University, Ithaca, NY) and the dam of that calf. Platelet aggregation studies performed on the obligate carrier and her vWD-affected calf were normal (data not shown). DNA samples from 3 Simmental cattle that were related to a thrombopathic calf studied at Michigan State University during the 1990s, a thrombopathic calf (No. 145) studied by Mapletoft et al. 12 at the University of Saskatchewan, 22 of its embryo transfer (ET) full siblings, and 1 ET half-sibling, as well as 10 cows related to the affected embryo donor were also evaluated. The gene sequence encoding Rap1b was also evaluated in the thrombopathic calf to rule out the possibility of a mutation in the target protein of CalDAG-GEFI. The coding region of Rap1b was found to be identical to cow genome sequence available on GenBank (data not shown).
Polymorphisms were found within several of the intronic sequences of the gene encoding CalDAG-GEFI. Most of the intronic polymorphisms constituted single-nucleotide changes and were not considered to be of significance, since they were found in the affected calf as well as within normal cattle. A 177-base pair deletion in the intron between exons 3 and 4 was noted in both normal cows and in the thrombopathic calf when sequences were compared to the bovine genome. An 18-base pair deletion in the intron between exons 12 and 13 was noted in the DNA of some normal cattle but was not present in the thrombopathic calf. Within exon 4 of the coding region, a single nucleotide change was noted in both affected and normal cattle (c.281A>C) compared with the bovine genome sequence. This change (CAT to CCT) would result in the substitution of a proline for a histidine. When dog, equine, and human sequences were compared to bovine sequence for this region it was noted that dog, human, and equine sequences encoded for a proline in this location; thus, this may constitute either a variant or an error in the bovine sequence entered on GenBank.
Two single nucleotide changes that altered amino acids were documented within the coding region of the CalDAG-GEFI gene in the affected calf in a homozygous state. One change was documented within exon 6 (c.653C>T), which would be predicted to result in the substitution of a leucine for a proline (CCG to CTG). Although seemingly significant, this homozygous change was also documented in one related Canadian cow and in an unrelated Simmental-cross who was also an obligate vWD carrier. Neither cow had a bleeding diathesis. This change occurred within an area of the gene encoding for a segment between SCR1 and SCR2. The predicted substitution apparently does not impact significantly on the function of the enzyme. Several other cattle, both Simmentals and non-Simmentals, were found to be heterozygous for this mutation.
The other homozygous mutation in the thrombopathic calf was a single-nucleotide change in exon 7 (c.701T>C). This change would be predicted to result in the substitution of a proline for a leucine (CTC to CCC). This change was considered highly significant, since it was located in a region of the gene encoding for SCR2 (Table 1). Proline is highly conserved in this segment of the catalytic domain across species lines, and a change at this location would likely have an impact on the function of the protein. This change was not documented in the DNA samples of any of the nonrelated cattle (Simmentals or non-Simmentals) or in the DNA samples of 3 clinically normal Simmental cattle related to the affected calf studied at Michigan State. However, 17 of the 23 siblings of the thrombopathic Canadian calf and 6 of the 10 related cows were found to be heterozygous for this mutation. As expected, none of these siblings or cows had a bleeding diathesis, since this disorder is inherited as an autosomal recessive condition. Although this nucleotide change is considered highly significant in this location of the gene, evaluation of DNA obtained from other affected calves will be necessary to completely confirm the significance of this mutation.
DNA sequences representing the end of intron 6 and the beginning of exon 7(underlined) for CalDAG-GEFI in a normal cow and a thrombopathic calf.∗
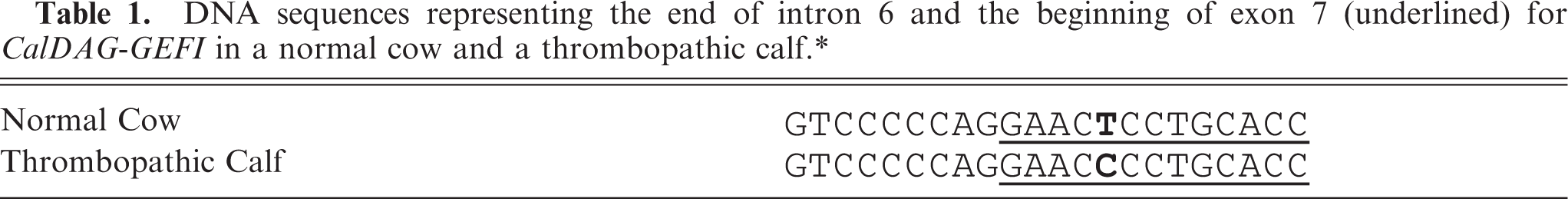
∗The single-nucleotide change is in boldface. The codon change of CTC to CCC would be predicted to result in the substitution of a proline for a leucine within structurally conserved region two (SCR2) of CalDAG-GEFI.
CalDAG-GEFI is critically important in mediating the effects of agonists, such as ADP and collagen, that signal either directly or indirectly through Gi. The ability of thrombin to activate thrombopathic platelets and induce clot retraction implies that at least one of the thrombin receptors is able to bypass CalDAG-GEFI and activate Rap1b directly or mediate activation via a Rap1b-independent pathway. Human platelets have at least two thrombin receptors, protease-activated receptor 1 (PAR1) and protease-activated receptor 4 (PAR4). PAR1 and PAR4 signaling are biphasic, with PAR1 activation resulting in a rapid spike in calcium influx, followed by a slower and more prolonged calcium influx mediated by PAR4. 6 PAR4 generates a more sustained calcium signal and is considered to be more effective than PAR1 in generating secondary signals necessary for complete platelet activation. Based on platelet cDNA sequence findings, it is likely that canine platelets are similar to human platelets with regard to platelet PAR receptors. Conceivably, the delayed thrombin response observed in thrombopathic canine platelets is mediated via PAR4, which may not require CalDAG-GEFI in the signaling pathway leading to activation of Rap1b. PAR receptor types have not been determined for bovine platelets; however, based on similar impaired platelet function phenotypes in thrombopathic cattle it is likely that bovine platelets also possess PAR1 and PAR4 thrombin receptors.
This is the second species with a documented inherited intrinsic platelet disorder related to impaired signal transduction and inability to change the affinity/avidity of the alphaIIb-beta 3 receptor to be identified with a spontaneous mutation in the gene encoding CalDAG-GEFI. Thus far, all of the spontaneous mutations have been detected within areas of the gene encoding structurally conserved regions of the catalytic domain of the GEF (Table 2). The phenotypes of cattle and dogs with these mutations are strikingly similar to each other and to the phenotype observed in CalDAG-GEFI knockouts. Signal transduction platelet disorders are the most commonly observed inherited intrinsic platelet disorders in people, and in most cases the cause is unknown. 14 It is likely that mutations in the CalDAG-GEFI gene result in similar platelet disorders in people.
Amino acid sequences of the SCRs within the catalytic unit of CalDAG-GEFI.∗

∗Simmental cattle with thrombopathia have substitution of a C for a T at position 701(c.701T>C) in exon 7 (C
Platelets from a thrombopathic calf were not available for this study, negating the ability to perform further specific biochemical studies. Western blots and functional studies, including Rap1 activation pull-down assays, would be useful in determining the effects of this mutation on CalDAG-GEFI at the quantitative and qualitative level.
This disorder in the homozygous state is considered to be rare at the present time. However, the identification of this mutation provides a means to test potential carriers and eliminate this disease from breeding animals.
Footnotes
Acknowledgements
We thank Tom Berryere for obtaining the DNA sequence from the Canadian cattle, Dr. Dwight Wolfe for obtaining blood samples from Simmental cattle, and Kevin King for technical assistance. We are grateful to the ranchers who have provided us with the samples from their cattle.
